Abstract
Shelled herbal foods are widely consumed. The evaluation of the shells of these foods is important due to their features such as low cost, ease of recycling and environmental friendliness. In this study, hazelnut shell (HS), pistachio shell (PS), and apricot kernel shell (AKS) were brought to powder particles by grinding to dimensions of 300–425 µm. Some of the powder particles were converted into ash at 900°C. The amounts of cellulose, ash, humidity, and metal in these particles via chemical analyses were determined, while their structural properties via X-Ray Diffraction (XRD) and Fourier Transform Infrared Spectroscopy (FT-IR) analyses. Composite materials were produced by adding 15 wt.% to the polyester matrix material from these powder particles and ashes. Compression strength, hardness, specific weight, and thermal conductivity of these composites were analyzed. The lowest and highest humidity, ash, cellulose, hemicellulose, and lignin ratios in powders showed differences depending on the type of powders. The amount of Sn and K in the HS, PS, and AKS powders were close to each other, while the amount of Ca, Na, Mg, Fe, Mn, Cu, Zn and Si was higher in AKS powder. The reinforcement adding to the polyester increased the compression strength, hardness, specific weight and thermal conductivity properties.
Keywords
Introduction
The improvement of the physical, chemical, and mechanical properties of composite materials is a function of the type, size, and shape of the reinforcement to be added into the matrix and its distribution within the matrix. In addition, it depends on how the applied external load will be transmitted to the reinforcement element.1–3
The reinforcement type has a great importance in determining the properties of composites. Reinforcement elements should be non-reactive and stable at operating temperature. 4 Synthetic and natural materials are preferred as reinforcement elements in composites since these reinforcement elements have superiorities among themselves. Synthetic materials are superior to natural materials in terms of physical, chemical and mechanical properties. However, natural materials utilized instead of synthetic materials in composite materials are preferred due to the properties such as the impact of global climate change, the increase in greenhouse gas emissions, the difficulty in biodegradation, the high processing costs, the difficulties in recycling, the high energy consumption, and the health hazards on human and environment.5–7 In the last few years, it has been seen that there has been a significant increase in the production of the environmentally friendly composites in which natural fibers such as flax, jute, hemp, pineapple and sisal are used. The thermal conductivity of the epoxy composite reinforced with 10% bamboo was measured as 0.216 (W/mK). 8 When using an epoxy reinforced composite containing with 70% fly ash, the thermal conductivity of the composite material decreased by 68.2% compared to the neat epoxy. In the optimization studies performed with 0–70% fly ash composition, it was determined that the specific temperature of the composite material increased in parallel with the increase in the fly ash content in the composite material, while it decreased in the thermal diffusivity. 9 The strength and heat conduction capacity of the epoxy composite material reinforced with the 30% cashew nut shell increased significantly. 10 The thermal insulation of the epoxy-based composite, prepared using fiber obtained from sisal, showed a 200% better efficiency than raw fiber. 11 The thermal conductivity of the epoxy composite reinforced with the corn stalk was measured as 0.1999 Wm−1K−1. 12 The thermal resistance of the epoxy matrix composite material reinforced with 80% chicken feather fiber was determined as 0.175 m 2 KW−1. 13
Today, 30% of composites made of plant-based reinforcements in Europe are used in the structure industry; and 25% of them are used in the automotive industry. In addition, approximately 45% of the composites produced are preferred in areas such as electronics, sports, metallurgy and transportation, electronic technology, air and space industries, medicine, and railway. 14 According to the Nova Institute, the use of wood composites in European automotive and construction industry reached 315000 tons in 2010, while the natural composites reached 145,000 tons. In the short term, it is estimated that the demand for wood composites will be about 830,000 tons by increasing 2.5 times, and the natural composites will be approximately 320,000 tons. 15 This market situation results in the replacement of wood biomass from lignocellulose fibers obtained from non-renewable parts of annual plants, which offer an interesting alternative, especially in countries where vegetation is not grown.
The annual production of the hazelnut produced in Turkey is about 70% compared to the world market. The amount of total production of hazelnut was 420,000 tons according to the Ministry of Food, Agriculture and Livestock in 2016. 16 The share of pistachios, produced in Turkey, is about 15% compared to the world market. Annual pistachio production, according to 2016 data of Turkey Statistical Institute, was realized as 170,000 tons. 17 In addition, the amount of apricot produced in Turkey was 730,000 tons according to the Ministry of Food, Agriculture and Livestock in 2016. The share of apricot production of Turkey in the world market is approximately 10%. The evaluation of the shells or kernel shells of hazelnuts, pistachios, and apricots, the vast majority of which is grown in the Black Sea, Southeastern Anatolia, and Eastern Anatolia Regions, respectively, has significant economic value. Considering the evaluation of hazelnut, pistachio, and apricot kernel shells, they can be mentioned to be serious natural reinforcement elements. 18
In today’s world of environmental concerns, the use of environmentally damaging and non-recyclable materials should be reduced, and the use of environmentally friendly natural materials should be encouraged. The aim of this study is to bring natural materials such as hazelnut, pistachio, and apricot kernel to the industry and to investigate the usage methods. In this study, the hazelnut, pistachio, and apricot kernel shells were powdered. Some of these powders were converted into ash. Chemical and physical analyses of powders and ashes were carried out and the effects of these analyses on physical, mechanical, and thermal properties such as compression strength, hardness, specific weight, and thermal conductivity of the composite were investigated.
Materials and methods
Determination of physical and chemical properties of powders
HS, PS, and AKS found in the abundant quantity in different regions of Turkey were chosen as natural reinforcement elements to be used in experimental work. These selected shells, respectively, were ground in the ELE brand Los Angeles wear machine. The ground materials were sieved through sieves with dimensions of 300 and 425 µm; and powder particles with dimensions of 300–425 µm were obtained. Humidity, ash, cellulose, hemicellulose, lignin, element, and heavy metal analyses of the natural reinforcement elements, which were brought to the powdered particles, were performed at room temperature in Batman University Research Laboratory. To determine the humidity content, they were dried in the drying containers in a preheated Blulab TT107 brand oven at 105°C. Then, they were cooled until room temperature in the desiccator. The tare of the drying cups was taken by weighing in a Shimadzu ATX224 brand analytical precision scale having a sensitivity of 0.001 g (m1). HS, PS, and AKS powders of approximately 3 g, by weighing, were put into the drying containers, respectively and their weights were calculated (m2). The drying containers were placed in a heated oven at 105°C. Drying process took 1 h 30 min. At the end of this process, the drying containers were placed in the desiccator and they were weighed after reaching room temperature. The process was repeated until the constant weight was obtained (m3). The percentage of humidity content (H) was calculated using equation (1). 19
To determine the amount of ash, the tare of the blank crucible, used for burning the natural particles, was taken. Then, HS, PS, and AKS powders of about 3 g (m0) were taken into the crucible. The crucible was kept in the furnace (Protherm-Furnaces) for 5 h at a temperature of 900°C. Then the crucibles were kept until room temperature by taking to the desiccator; and then they were weighed (m).
The percentage of ash content (A) was calculated using equation (2). 19
In the experiments, in order to investigate the effects of different alkaline solutions on enzymatic hydrolysis, HS, PS, and AKS powders were processed for 30 min with 2.5% (w/v) Ca(OH)2, CaCO3 and NaOH solutions at 60°C with the solid/liquid ratio of 1/10. The experiments later went on with NaOH. The effects of NaOH concentration (0.5, 2.25 and 4.0 (w/v)), pretreatment time (30, 60 and 120 min), temperature (30°C, 60°C and 90°C) and solid-liquid ratio (1/10-1/20-1/40 g/mL) on the chemical composition of pretreated raw materials and enzymatic hydrolyses were investigated. After the pretreatments, the powdered raw materials were filtered through the filter paper and were neutralized with the distilled water. Then, they were dried up to about 50% humidity in the air and were stored in a fridge in plastic bags for enzymatic hydrolysis experiments. The enzymatic hydrolysis experiments were carried out in a shaking incubator. In the enzymatic hydrolysis processes of the pretreated powders, a mixture of 60 FPU/g (dry raw material) cellulose enzyme (Celluclast 1,5L®, Novozyme) and 40-CBU/g (dry raw material) β-glycosidase enzyme (Novozyme 188) was used, and the solid-liquid ratio was kept constant at 1/20. All experiments were conducted at a temperature of 50°C, pH of 4.8, 100 rpm stirring speed and for 48 h. For determining lignin, 300 mg of the raw material to be analyzed was put into bottles, and 3 mL 72% sulfuric acid was added onto it, and stirred for 1 min. The H2SO4 concentration in the specimens that were mixed for 60 min in a shaking water bath at 30°C was diluted down to 4% by 84 mL of distilled water, and then subjected to a hydrolysis process for 60 min in an autoclave at the temperature of 121°C. After autoclaving, the raw material was filtered by passing through rough filter paper. After the solid raw material remained on the filter paper was washed with water, it was dried in a drying-oven at 105°C and weighed again. The dried specimen was then fired in a muffle furnace at 900°C for 24 h. The ash remained in the crucible was weighed and recorded. The amount of lignin was calculated according to the dry base. In determining lignin, the cellulose in the biomass was calculated based on the glucose and cellobiose amounts in the filtrate that was separated from the filter paper, and the hemicellulose in the biomass was calculated based on the sum of the amounts of xylose, arabinose, mannose and galactose. The reducing sugar amounts in the enzymatic hydrolysis fluids were calculated as spectrophotometrically with the Dinitrosalicylic acid (DNS) method, while the monomeric sugars were determined by high performance liquid chromatography (HPLC). The experiments were carried out for determining the amounts of monomeric sugar compounds with the HPLC method involved an Agilent 1100 series HPLC system which consisted of a four-piston pump, refractive index detector, automated injection unit and column oven. The separations were carried out at 80°C in an Aminex HPX 87P column (Biorad, Hercules/USA). As the mobile phase, distilled water was used in the flow rate of 0.6 mL/min. This way, percentage of cellulose gain, lignin removal and hemicellulose removal were calculated.
The elemental analyses of specimens belonging to powder and ash were determined by Thermo FLASH 2000 Series CHNS device.
The proportions of metallic materials in the prepared material were determined by using a temperature and pressure controlled analytical microwave oven (Milestone, ETHOS-A). Approximately 0.5 g of powder specimen was taken to the Teflon. Solubilization process was performed by adding HNO3-HCL-HF (5:3:2; v/v/v) of 10 mL to the concentrated mixture. The concentrations of Sn, Ca, K, Na, Mg, Fe, Ni, Mn, Cu, Zn and Si in the clear liquids were measured by Perkin Elmer Optima 7000 DV brand inductively coupled plasma optical emission spectrometry (ICP-OES). Each experiment was performed as three repetitive.
To determine the structural properties of the powders, Rigaku brand Miniflex-2 model X-ray diffractometer device (XRD; X-ray diffraction), which had the filter = Ni, anode = Cu (CuKα = 1.541871 Å), current = 15 mA, voltage = 35 kV, paper speed = 2 cm/min., Goniometer speed = 2°/min., slits = 1° 0.15 mm–1° 0.30 mm, paper spacing = 2θ = 5–35°, and time constant = 1 s, was used.
To examine the surface functional structure of the composite specimens, the infrared spectra of the powdered specimens were recorded by FT-IR (Fourier transform infrared spectroscopy), which is Perkin Elmer Spectrum 100 brand and the ATT (Attenuated total reflection) properties.
Production of composites and determination of its properties
To determine the effects of physical, chemical, and structural properties of HS, PS and AKS on the composite material, the powders were first dehydrated for 3 h in the oven at 105°C and dehumidified. Then, neat polyester was produced with Polipol 320 TA matrix material and naturally reinforced composites were obtained by adding 15 wt.% from HS, PS, and AKS powders and ashes to this matrix material. According to the data sheet obtained from Poliya company, the properties of the matrix material are given in Table 1. The images of the produced composites are given in Figure 1.
Some properties of Polipol 320 TA.


Images of composites.
The matrix material and natural composites were investigated for hardness, compression strength, specific weight, thermal conductivity, and structural properties.
The hardness, compression strength, specific weight, thermal conductivity and structural property of the matrix material and natural composites were investigated.
The hardness measurements were determined with the Shimadzu HMV microhardness measurement instrument depending on the magnitude of trace obtained by applying to the workpiece surface throughout 10 s of a load of 500 mN. Compression tests were carried out in a 5 kN capacity Shimadzu brand AG-IC serial number tensile tester at a pressure velocity of 1 mm/min.
Specimens were weighed on a precision scale having a sensitivity of 0.001 g (m5) to determine the densities. In addition, volumes depending on the geometry of the specimens were calculated (v). The specific weight (ρ) was calculated by using equation (3).

Heat transfer analyzer, ISOMET model with serial number API 210411 and having the application sensitivity of 0.04–0.30 W/mK, and the application temperature of −5….+50°, were used to determine the thermal conductivity. In addition, the structure of composite was investigated by FT-IR device having Perkin Elmer Spectrum 100 brand and ATR properties.
Experimental results and discussion
Physical and chemical properties of powders
Humidity, cellulose, hemicellulose and lignin ratios of HS, PS and AKS powders and the ash ratios of these powders at 900°C have been determined and given in Table 2.
Humidity, ash, cellulose, hemicellulose, and lignin ratios of powders (%).

When the humidity of the powder was examined, it was seen that while the maximum humidity was preserved with HS powder, and minimum humidity was preserved with AKS powder. When the ash ratio was considered, unlike the humidity content, the least ash content was obtained from HS powder and the most ash content was obtained from AKS powder. This can be thought to be due to the high amount of non-volatile inorganic metals in AKS powder. When the amount of cellulose was ranked according to the ratio higher than the low rate, it was obtained from HS powder with the lowest 15.8%, PS powder with 21.4%, and AKS powder with the highest 28.4%. In terms of hemicellulose, AKS powder had the highest content and PS powder had the lowest content. The change of lignin was the same as the amount of hemicellulose. The lowest and highest humidity, ash, cellulose, hemicellulose and lignin ratios in powders showed differences depending on the type of powder.
The results of elemental analyses of HS, PS, and AKS powders are given in Table 3. The amount of ash obtained from HS was 1.3%; however, it had the highest carbon content with 50.07%. The results in Tables 2 and 3 are consistent with recent studies on HS, PS and AKS.20–22
Elemental analysis results of the natural particulate powders (wt %).
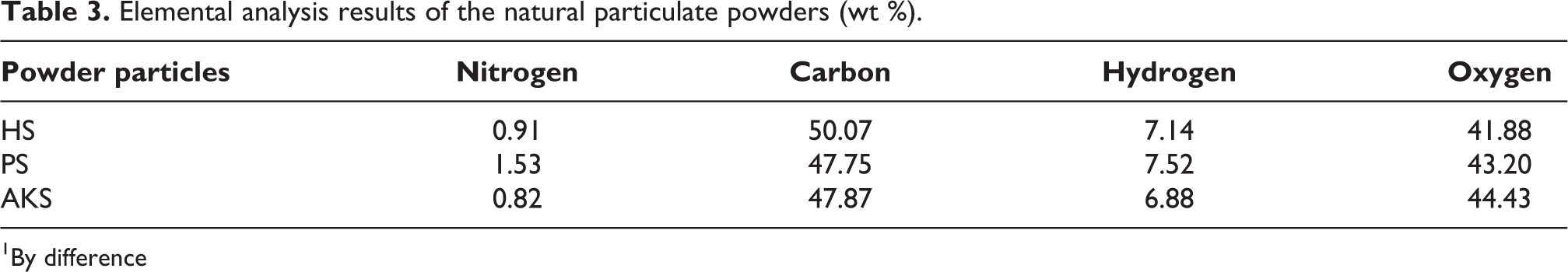
1By difference
According to the results of elemental analyses, it was determined that PS powder had the highest nitrogen and hydrogen content, HS powder had the highest carbon content, and AKS powder had the highest oxygen content.
The results of metal analyses of HS, PS, and AKS powders are given in Table 4. The amounts of Sn and K in the HS, PS and AKS powders were measured to be close to each other. The amount of Ca, Na, Mg, Fe, Mn, Cu, Zn, and Si found in AKS powder was also higher than HS and PS.
Metal analysis results of the natural particulate powders and ashes.
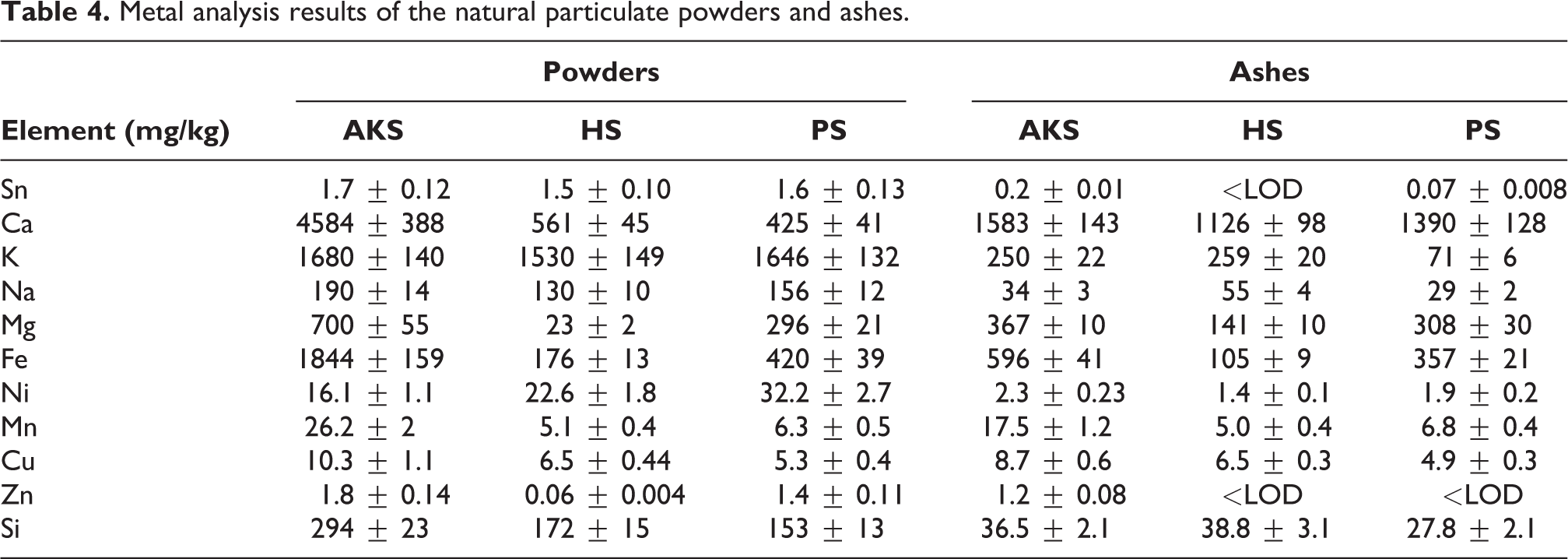
The highest Ca, Mg, Fe, Ni, Mn, Cu, and Zn concentrations in HS, PS, and AKS ashes were measured in AKS ash. Considering the ashing process at 900°C, Na, Ni, and Si can be said to be more volatile in AKS. In high concentrations, the number of elements such as Mn, Sn, Ni, Cu, which are known to be toxic, decreased significantly. In addition, in some specimens, the decrease was found to be below the measurable concentration.
XRD analyses of HS, PS and AKS powders and their ashes at 900°C are given in Figure 2. In considering XRD spectra; the peak observed in the 2θ, 16° and 22° of HS, PS, and AKS powders belonged to cellulose (type I polymorph), which was one of the most important components of cell wall structure of the green plants.23,24 After the ash was heated at 900° C, significant differentiation was observed in each specimen. Cellulite peaks disappeared. The reason for these changes is thought to be due to chemical groups that remain after the breakdown in cellulose structure at high temperature.

XRD analysis results of the powder and ashes belonging to the natural shells.
FT-IR spectra of HS, PS, and AKS powders are shown in Figure 3(a) to (c), respectively. From Figure 3(a), the broad peak at 3287 cm

FT-IR spectra belonging to the natural shells.
The properties of natural reinforced composites
In natural particle reinforced composites, the effects of physical, chemical, and structural properties of the reinforcement elements on compression strength, hardness, specific gravity, and thermal conductivity properties were investigated. The effects of the reinforcing element types on the compression strength are given in Figure 4.

The effects of the reinforcement element types on compression strength
As shown in Figure 4, the addition of both natural particulate powders and the ash of these powders to the polyester material increased the compression strength of the composite material. The compression strength of neat polyester was measured as 81 MPa. The compression strengths of composites reinforced with HS, PS, and AKS were measured as 83, 95 and 106 MPa, respectively. It was observed that the reinforcement type affecting the least compression strength was HS powder. PS, and AKS powders, respectively followed it. This situation was found to be directly proportional to the heavy metal contents of the powders. However, the addition of natural particulate ashes to the polyester matrix material was led to an increase in compression strength more than the addition of powder. This increase was due to the removal of flammable materials from the ashes and the interaction of oxygen with elements such as Fe, Si, and Mg (Figure 2). The highest compression strength was obtained from composite materials produced from AKS ash. PS and HS ashes respectively followed it. The compression strengths of composites reinforced with HS, PS, and AKS ashes were measured as 90, 104 and 112 MPa, respectively.
The effects of the reinforcement element types on the hardness value are given in Figure 5. As with compression strength, the addition of the reinforcing element to the polyester matrix increased the hardness values of the composites. The highest hardness values were obtained from ash reinforced composites. The minimum hardness value was measured as 26 HV from neat polyester. The hardness values of composites reinforced with HS, PS, and AKS were measured as 29, 33 and 34 MPa, respectively. The hardness values of composites reinforced with HS, PS, and AKS ashes were also measured as 36, 37 and 38 MPa, respectively. Maximum hardness value was obtained from AKS ash added composite material.
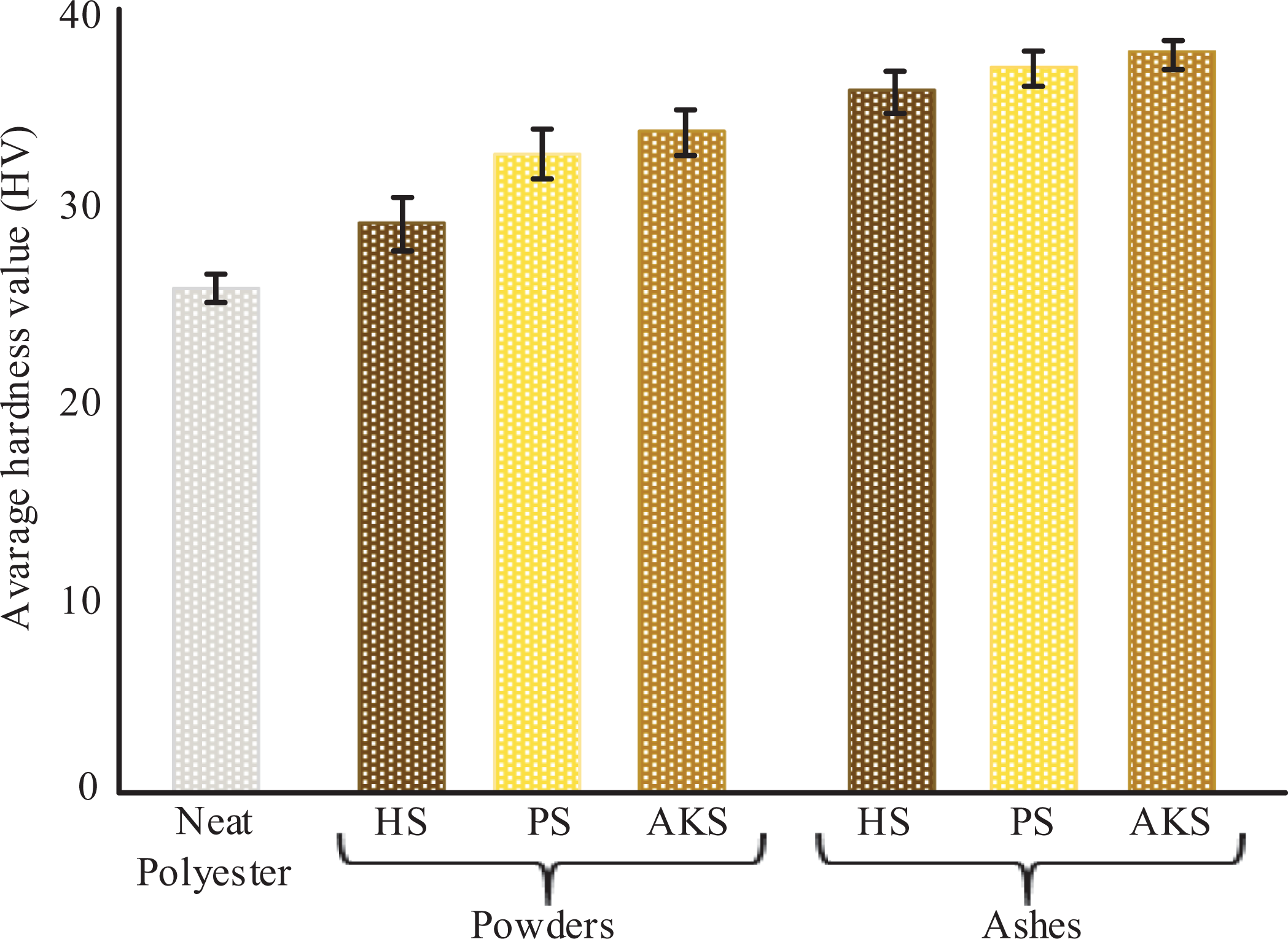
The effects of the reinforcement element types on hardness.
The specific weight, thermal conductivity, specific heat, thermal diffusivity values of the composites and the neat polyester are given in Table 5.
Specific gravity and thermal properties of reinforcement element types.

The addition of particulate powder and ash to the matrix material showed increase in the specific weight and thermal conductivity of the composite. This increase is due to the fact that the matrix material has a lower specific weight and a thermal conductivity than the reinforcement elements. In addition, the presence of heavy metals such as Fe, Si, and Zn in the natural reinforcing elements increased the specific weight of the composites, while the presence of metals such as Cu caused increase in the thermal conductivity of the composite. On the other hand, the thermal diffusivity increased in direct proportion with the thermal conductivity. The specific heat also increased in inverse proportion with the specific weight. 28
In general, it was seen that combinations of the polyester (Polipol 320 TA) reinforced with ashes could be evaluated from results as the best composite materials. FT-IR spectra of PS ash, polyester, and their composite are comparatively presented in Figure 6. The peaks observed at 1010 and 559 cm

FT-IR spectra of (a) PS ash, (b) neat polyester (Polipol 320 TA), and (c) PS ash/polyester.
Conclusion
The effects of physical, chemical, and structural properties of the reinforcement elements on compression strength, hardness, specific weight, and thermal conductivity properties were analyzed in natural particle reinforced composites. The results are given below: ✓ The amount of lignin, cellulose, and hemicellulose of AKS was higher than HS and PS. ✓ According to the results obtained from ICP-OES, the amount of Ca, Na, Mg, Fe, Mn, Cu, Zn, and Si in AKS was higher than that of HS and the PS. ✓ When the XRD results were evaluated, it was determined that the type I cellulose structure observed in HS, PS, and AKS changed as it was disintegrated after the ashing process. ✓ Surface functional structures of HS, PS, and AKS powders were investigated by FT-IR; and it was determined that observed peaks were mainly caused by cellulose and hemicellulose structures. ✓ When the properties of the composites and neat polyester were examined, it was concluded that the least specific weight, thermal conductivity, specific heat, and thermal diffusivity, respectively were obtained from the neat polyester, AKS powder/polyester composite, HS ash/polyester composite, and AKS powder/polyester composite. ✓ The addition of natural particulate powders to the matrix material increased the compression strength and hardness of the composite. The highest compressive strength and hardness value were obtained as 106 MPa and 34 HV in AKS powder/polyester composite, respectively. Minimum compression strength and hardness value were obtained as 83 MPa and 29 HV from the HS powder/polyester composite, respectively. ✓ The addition of HS, PS, and AKS ashes to the matrix material contributed more than the addition of natural particle powders to the increase of the compression strength and hardness values of the composites. This additive ratio was between about 8–10% for hardness and between about 8–10% for compression strength. ✓ The addition of natural particulate powder and ash to the matrix material increased the specific weight and thermal conductivity of the composite.
Footnotes
Acknowledgments
We would like to thank Batman University and Hasan AKDEMİR for his help and contributions in the analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


