Abstract
Multi-walled carbon nanotubes (MWCNTs)/high density polyethylene (HDPE) composites were prepared by a masterbatch method (mPEC) in which a commercial HDPE was blended with a MWCNTs/HDPE masterbatch obtained from in situ polymerization. Owing to the interfacial interaction, a 13 cm−1 up-shift of the G band for the MWCNTs was observed in the Raman spectrum of the MWCNTs/HDPE masterbatch and the homogeneous distribution of MWCNTs in the mPEC was realized. Compared to the pure HDPE and the MWCNTs/HDPE composites prepared by a direct melt-blending method (dPEC), the mPEC had better electrical, mechanical and rheological properties, suggesting that the in situ polymerized HDPE covering on the MWCNTs surfaces played an important role in the reinforcing effects as an interfacial modifier. The tensile yield strength and the Young’s modulus of the mPEC containing 3 wt% MWCNTs (mPEC3), and the flexural strength and the flexural modulus of the mPEC containing 1 wt% MWCNTs were improved by 38.3%, 41.7%, 24.4%, and 42.9%, respectively, compared to those of the pure HDPE. For, the electrical resistivity of mPEC3 was decreased by about three orders of magnitude relative to that of the pure HDPE. The |η*|, G′, and G″ of the mPEC were obviously higher than those of pure HDPE. Moreover, the polyethylene-modified MWCNTs obtained from in situ polymerization could facilitate the crystallization of the HDPE macromolecular chains more effectively compared to the unmodified MWCNTs.
Keywords
Introduction
Carbon nanotubes (CNTs) have been used as ideal nanofillers to prepare high-performance polymer-based composites due to their excellent mechanical, electrical, and thermal properties.1–10 However, it is difficult to fully realize the advantages of CNTs because they are very easy to aggregate into bundles. 1 It is well known that the homogeneous distribution of CNTs in the polymer matrix and the strong interfacial interaction between CNTs and polymer molecular chains are two key factors to achieve enhanced properties of the resultant CNTs/polymer composites.2–10 Therefore, lots of researches have been focused on how to improve the compatibility between CNTs and polymer matrix.
Polyethylene (PE) is one of the most commonly used thermoplastics consisting of long hydrocarbon chains which don’t contain any functional groups on the backbone. PEs have been widely utilized as packaging materials, underground gas and water pipes, thin films, electrical insulation for power cables, building materials, etc. The incorporation of CNTs, especially the cost-effective multi-walled carbon nanotubes (MWCNTs), into PE to form composites can further extend the applications. However, it is very necessary to find an effective way to improve the interfacial interaction between the CNTs and the PE matrix because the inertness of PE makes it incompatible with CNTs. Recently, it has been reported that the immobilization of a coordinative catalyst on fillers followed by in situ ethylene polymerization can produce filler/PE composites.11–28 More importantly, such composites showed better dispersion of the fillers and stronger interfacial interaction between fillers and PE matrix than those prepared by melt-blending or solution-blending. Vega et al. reported that the utilization of a masterbatch obtaned from a MWCNTs-supported metallocene catalyst could prepare MWCNTs/high density polyethylene (HDPE) composites with improved properties.7,8 This combined the advantages of the in situ preparation method yielding high interfacial interaction and the melt-blending preparation method with high efficiency. It seems that the masterbatch method probably will be an effective way to solve the problems mentioned above.29–33 However, up to now, few works have reported on the supporting of the most commonly used TiCl4 catalyst on the MWCNT to prepare the masterbatch for the MWCNTs/HDPE composites.
In this paper, HDPE/MWCNTs composites were prepared by a masterbatch method. This process included three steps: (1) a TiCl4 catalyst was first anchored on the HNO3-treated MWCNTs/MgCl2 bisupport. The MWCNTs/MgCl2 bisupport was an effective carrier for the catalyst, just like SWCNTs/MgCl2 bisupport. 28 The possible reactions are schematically shown in Figure 1; (2) the polymerization of ethylene was initiated after adequate activation by the AlEt3 cocatalyst, and the growing HDPE molecular chains attached onto the MWCNTs surface directly and firmly to prepare the MWCNTs/HDPE masterbatch; (3) the MWCNTs/HDPE composites were prepared by blending the masterbatch with the HDPE matrix using an internal mixer at 170°C. Furthermore, the mechanical, electrical and rheological properties as well as the melting and crystallization behaviors of the MWCNTs/HDPE composites prepared by the masterbatch method (mPEC) were investigated and compared with those of the pure HDPE and MWCNTs/HDPE composites prepared by a direct melt-blending method (dPEC).
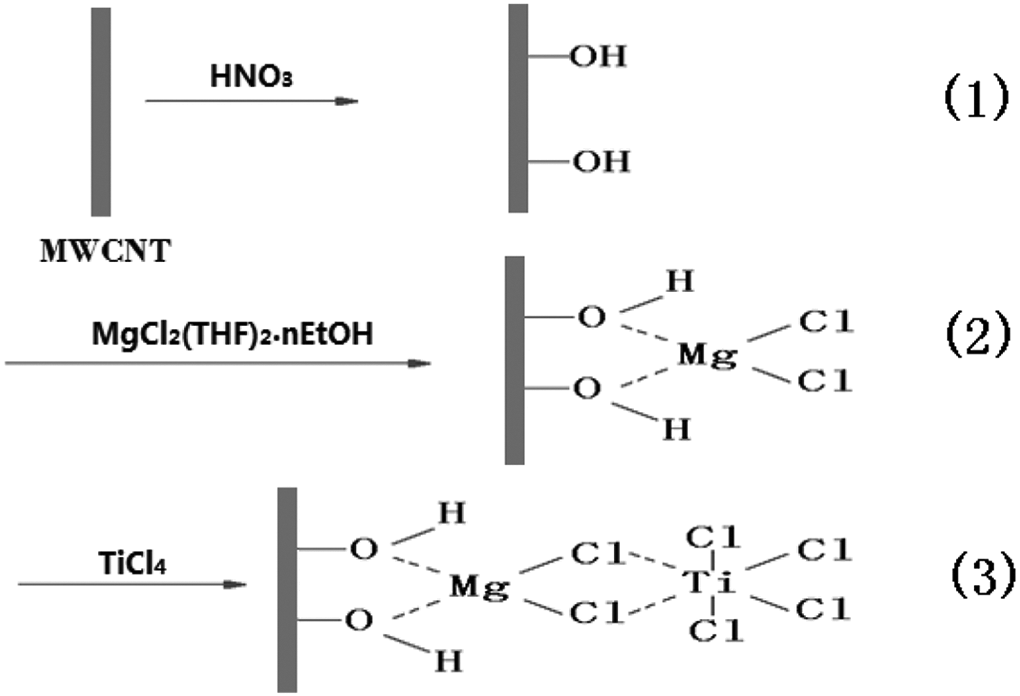
Schematic illustration of preparation of MWCNTs-supported Ziegler-Natta catalyst.
Experimental
Materials
5 g MWCNTs (diameter: 20–50 nm, length: 1–2 μm) purchased from Carbon Nanotech Port Co. Ltd. (China) were refluxed in 30 mL HNO3 (30 wt.%) at 120°C for 4 h to introduce some hydroxyl groups onto the surface of the MWCNTs. The HNO3-treated MWCNTs were washed several times with water to remove any impurities. The HDPE (melt index = 3.0g/10 min, density = 950 kg·m−3) was supplied by LG Chemical Corp. (South Korea). Ethylene was provided by the Beijing Qianjing Chemical Factory (China) and used after treatment with molecular sieves for the removal of water. Triethylaluminum (AlEt3) purchased from Shanghai Petrochemical Corp. (China) was used as a 400 g/L solution in n-heptane. Ethanol, tetrahydrofuran (THF), n-hexane and n-heptane were distilled and dried by using molecular sieves. TiCl4 and MgCl2 were obtained from the Xingta Chemical Factory (China) and Aluminum Smelting Factory (China), respectively, and used as-received without further purification.
Preparation of MWCNTs/MgCl2/TiCl4 Catalyst
5 g MgCl2 was added into a glass reactor equipped with a magnetic stirrer, and then 40 mL tetrahydrofuran (THF) and 3 mL absolute ethanol were added to form a mixture. The mixture was heated to 45°C and stirred overnight until the MgCl2 was completely dissolved to form a mixed solution. Next, 5 g HNO3-treated MWCNTs were dispersed in the mixed solution under magnetic stirring. After being stirred at 45°C for 2 h, the agglomerated black solid was separated by decantation, washed with hexane and dried under vacuum at 60°C for 2 h to obtain the MWCNTs/MgCl2 bi-support. 1 mL TiCl4 was then added to the MWCNTs/MgCl2 bi-support suspended in 60 mL hexane. The suspension was stirred at 45°C for 2 h. After decantation, the solid was washed with hexane several times, and then dried under vacuum to obtain the supported catalyst as a solid black powder.
Preparation of MWCNTs/HDPE Masterbatch
200 mL heptane and 5 mL AlEt3 solution were added into a glass reactor equipped with a mechanical stirrer in sequence after the glass reactor was flushed by highly purified nitrogen for three times. Next, the supported catalyst was added into the glass reactor and the [Al]/[Ti] molar ratio was 100. The polymerization was carried out under a constant ethylene pressure of 106.7 kPa at 50°C and then terminated with acidified ethanol. The precipitated products were washed with ethanol for several times and dried under vacuum at 60°C for 24 h to obtain the masterbatch. The MWCNTs content in the MWCNTs/HDPE masterbatch was determined to be 53 wt.% by thermogravimetric analysis.
Preparation of MWCNTs/HDPE Composites
50 g mixture composed of HDPE and the masterbatch (or MWCNTs) was blended at 170°C in an internal mixer (model HL-200, Jilin University Instrument Factory, China) with a mixing speed of 50 rpm and a mixing time of 10 min. The MWCNTs/HDPE composites obtained from the masterbatch method and direct melt-blending method were abbreviated as “mPEC” and “dPEC,” respectively. The weight percentage of MWCNTs for the composites was represented after “mPEC” or “dPEC.” For example, mPEC1 denoted MWCNTs/HDPE composites containing 1 wt.% MWCNTs prepared by the masterbatch method, while dPEC1 denoted MWCNTs/HDPE composites containing 1 wt.% MWCNTs prepared by the direct melt-blending method. Samples containing 0.5, 1, 2, and 3 wt.% MWCNTs were prepared by the two methods mentioned above.
Characterization
A JEM-2010HR (Japan) transmission electron microscope (TEM) was used to observe the distribution of the MWCNTs in the MWCNTs/HDPE composites. Raman spectra were obtained from a Renishaw Via microRaman spectrometer (England) with 514 nm excitation wavelength. The melt rheological properties of HDPE and its composites were investigated at 160°C using an ARES-rheometer (Rheometrics Scientific, USA). The rheological measurements were performed in the dynamic mode by using two 25 mm parallel plates with the gap setting of 2 mm. A fixed strain of 0.2% was used to ensure that the rheological measurements were taken within the linear viscoelastic regime. The dumb-bell-shape samples for mechanical testing were prepared using the HTF60W1 injection molding machine (Ningbo Haitian Corp., China). The injection temperature was 170°C and the dumb-bell-shape samples were cooled to room temperature naturally. The tensile properties and the flexure properties of HDPE and its composites were measured on a Hounsfield THE 10K-S testing machine at a cross-head rate of 10 mm/min and 2 mm/min, respectively, in which five specimens of each sample were measured and an average value was taken. AZC-36 Resistance Test Meter (Shanghai Precision Technology Corp., China) equipped with ring electrodes was used to measure the resistance of the samples. The heating and cooling differential scanning calorimetry (DSC) curves of PE and its composites were obtained with a Perkin-Elmer (USA) DSC-7 equipment in nitrogen at the changing temperature rate of 10oC/min.
Results and discussion
To achieve the high-performance MWCNTs/HDPE composites, two problems must be addressed: (1) the MWCNTs tend to aggregate caused by the van der Waals interactions between them, and (2) the MWCNTs cannot be effectively wetted by the HDPE matrix due to their poor interfacial contact. The preparation process of our MWCNTs/HDPE masterbatch is shown in Figure 2. The TiCl4 catalysts could be easily immobilized onto the MWCNTs surfaces by the coordination bonding with the MgCl2 supported on the HNO3-treated MWCNTs containing hydroxyl groups. After the ethylene polymerization began, the polymerized HDPE macromolecular chains anchored on the MWCNTs surfaces directly and firmly. As the ethylene polymerization proceeded, the continually growing HDPE molecular chains gradually disentangled the aggregates of MWCNTs by destroying the van der Waals interactions between them. Finally, the separated MWCNTs were wrapped by the HDPE covers entirely and tightly to form core-shell structures. These HDPE shells could act as the effective interfacial modifiers to bind the HDPE matrix with MWCNTs cores during the melt-blending process.

The preparation route of the MWCNTs/HDPE masterbatches.
Figure 3 shows the Raman spectra of HDPE, raw MWCNTs, HNO3-treated MWCNTs, and MWCNTs/HDPE masterbatch. Both MWCNTs and HNO3-treated MWCNTs had two adsorption peaks at about 1348 cm−1 for the disordered carbon atoms (D band) and 1572 cm−1 for the crystalline graphitic carbon atoms (G band), respectively. It could be seen that the relative intensity ratio (I[D]/I[G]) of the MWCNTs increased slightly after the HNO3 treatment, indicating the introduction of functional groups, such as carboxylic and hydroxyl groups, onto the MWCNTs surface in the presence of HNO3 at high temperature. 27 The spectrum of MWCNTs/HDPE masterbatch clearly exhibited the characteristic absorption bands attributed to both the HDPE and the MWCNTs. Moreover, an up-shift of wave number by 13 cm−1 for the G band of the MWCNTs in the Raman spectrum of MWCNTs/HDPE masterbatch was observed in comparison with that of the HNO3-treated MWCNTs. This shifting of the G band to a higher wave number could be attributed to: (1) the disentanglement of the MWCNTs and the subsequent dispersion of the separated MWCNTs in the HDPE matrix as results of the penetration of the HDPE macromolecular chains into the MWCNTs bundles, (2) the encapsulation of the HDPE cover onto the MWCNTs surface during the polymerization process,28,34–36 and (3) the strong interfacial interaction between the HDPE molecular chains and the MWCNTs.37,38

Raman spectra for (a) pure HDPE, (b) MWCNTs, (c) HNO3-treated MWCNTs, (d) MWCNTs/HDPE masterbatch.
TEM was used to investigate the distribution state of the MWCNTs in the MWCNTs/HDPE composites for sample mPEC3 and sample dPEC3, as shown in Figure 4. The MWCNTs were distributed quite well in the HDPE matrix and a large number of isolated MWCNTs could be found in the TEM image of sample mPEC3. On the other hand, although the TEM images of sample dPEC3 also showed some individual MWCNTs in the HDPE matrix, most of MWCNTs were still in the form of agglomerates. These results suggest that a better compatibilization effect between the MWCNTs and the HDPE matrix was achieved by the masterbatch method because the polymerized HDPE shells wrapping the MWNTs cores acted as the surface modifiers to aid the melt-blending process.

TEM images of mPEC3 and dPEC3, each of two different magnifications.
As shown in Figure 5(a) and (b), both the tensile yield strength and the Young’s modulus of the MWCNTs/HDPE composite increased with the increase of the MWCNTs content regardless of the preparation method, which were obviously higher than those of the pure HDPE. When the MWCNTs/HDPE composites were subjected to the tensile testing, the external force was transferred from the HDPE matrix to the MWCNTs. Hence, the good tensile properties of the MWCNTs/HDPE composites could be obtained because of the excellent mechanical properties of the MWCNTs. Moreover, the tensile properties of mPEC were better than those of dPEC. For example, the tensile yield strength and Young’s modulus of sample mPEC3 were improved by 38.3% and 41.7% in comparison with those of the pure HDPE, respectively, while for sample dPEC3 the corresponding values were only 25.2% and 26.2%, respectively. This indicated that the masterbatch method enhanced the compatibility between the MWCNTs and the HDPE matrix, leading to the homogeneous distribution of MWCNTs and the strong interfacial interaction. Therefore, better tensile properties were obtained from mPEC owing to the more effective interfacial stress transfer; while for dPEC, the agglomerates of MWCNTs might act as many flaws in the HDPE matrix thus reduced their reinforcing effects.39–41

Tensile yield strengths, Young’s moduli, and elongations at break of PE and its composites. (a) Tensile yield strengths, (b) Young’s moduli, and (c) elongations at break of PE and its composites.
The elongations at break of the pure HDPE and its composites are presented in Figure 5(c). It can be seen that the pure HDPE was quite ductile, with an elongation at break of almost 1000%. On the other hand, the elongation at break of the MWCNTs/HDPE composites continuously decreased with the increasing MWCNTs concentration for both mPEC and dPEC, which implied that the incorporation of MWCNTs into HDPE not only improved the stiffness (as shown by the data in Figure 5(b)) but also reduced the flexibility of the resultant MWCNTs/HDPE composites. This result could be attributed to the interfacial interaction between the HDPE molecular chains and MWCNTs in the MWCNTs/HDPE composites making the HDPE molecular chains difficult to move. Moreover, the elongation at break of mPEC was lower than that of dPEC owing to the stronger interfacial interaction between the HDPE molecular chains and MWCNTs in mPEC compared to that in dPEC. 39 This strong interfacial interaction could result in not only the inhibition of movement of the HDPE molecular chains but also the lower deformation of the MWCNTs/HDPE composites.
Figure 6 shows the flexural strength and flexural modulus of the pure HDPE and its composites. With the increase of the MWCNTs concentration, the flexural properties of the mPEC increased and reached a maximum value at the MWCNTs concentration of 1.0 wt.%. When MWCNTs concentration exceeded 1.0 wt.%, the flexural properties of mPEC fell but still remained higher than that of the pure HDPE. Similar to the tensile testing result, the flexural properties of mPEC were better than those of dPEC. For instance, the flexural strength and the flexural modulus of mPEC1 were improved by 24.4% and 42.9% in comparison with those of pure HDPE, respectively; but for dPEC1, the corresponding values were only 16.3% and 12.1%, respectively. We believed that this phenomenon also could be explained by the stronger interfacial interaction between the HDPE molecular chains and MWCNTs in mPEC compared to that in dPEC.
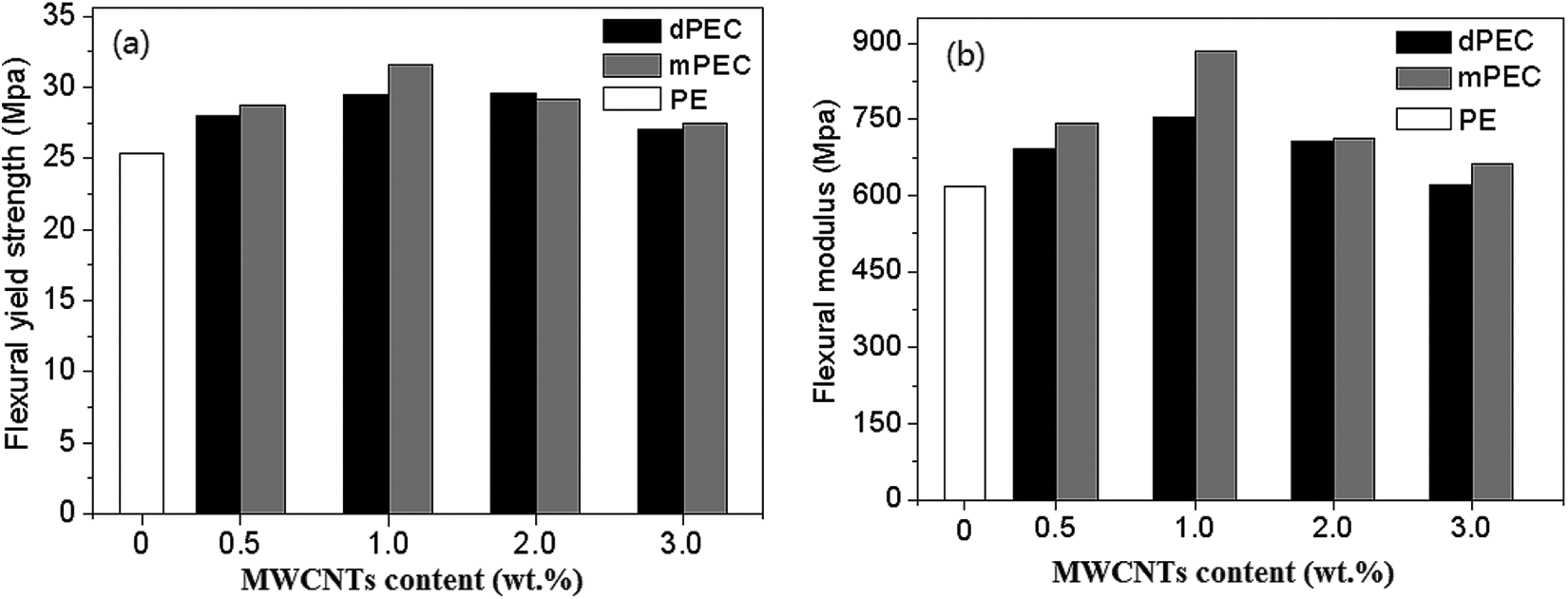
Flexural yield strengths and flexural moduli of PE and its composites.
Figure 7 shows the electrical resistivity of the pure HDPE and its composites. The results confirmed that the pure HDPE was an insulator exhibiting a high electrical resistivity. After the incorporation of the electrically-conductive MWCNTs into the HDPE matrix, a progressive decrease in the electrical resistivity with an increase of MWCNTs concentration for both the mPEC and the dPEC was observed. Moreover, the dPEC possessed higher electrical resistivity than that of the mPEC at the same concentration of MWCNTs. This result indicated that the MWCNTs with the HEPE covering obtained from the in situ polymerization was helpful in its homogeneous distribution in the resultant mPEC owing to the good compatibility between the HEPE covering and the HDPE matrix, leading to the easy formation of the electrically-conductive network in comparison with the dPEC. When the MWCNTs concentration was increased to 3 wt.%, the electrical resistivities of mPEC3 and dPEC3 were decreased by about four and three orders of magnitude, respectively, in comparison with that of the pure HDPE. The electrical resistivity of mPEC3 was almost one order of magnitude lower than that of dPEC3. This result suggested that it was easier for the well dispersed MWCNTs in sample mPEC3 to form some conductive paths than the aggregated MWCNTs in sample dPEC3.42,43 Similarly, both the mechanical and electrical properties of the MWCNTs/HDPE composites obtained from the masterbatch method were better than those obtained using the direct melt-blending method, as mentioned above.

Logarithm volume resistivities for PE and its composites.
The linear viscoelastic behaviors of the pure HDPE and its composites in melt state were studied by dynamic oscillatory shear measurements. The plots of the complex viscosity (|η*|) vs frequency for the pure HDPE and its composites with 1 and 3 wt.% filler are shown in Figure 8. The |η*| values of mPEC increased with the increasing MWCNT concentration and were higher than those of the pure HDPE and the dPEC. Especially, the |η*| values of sample dPEC3 were much lower than those of sample mPEC but were slightly higher than those of the pure HDPE. This phenomenon could be explained as follows: (1) MWCNTs in sample mPE3 were homogeneously dispersed so that they could form a network structure more effectively, resulting in higher viscosity, and (2) the stronger interfacial interactions between the MWCNTs and the HDPE matrix in sample mPEC3 could make the movement of HDPE macromolecular chains become more difficult. 44

Sweep frequency dependence of the complex viscosity for PE and its two sets of composites at 160°C.
The plots of storage modulus (G′) and loss modulus (G″) vs frequency for the pure HDPE and its composites are shown in Figure 9. The G′ and G″ values of the mPEC increased with the increasing MWCNTs concentration and were obviously higher than those of the pure HDPE and the dPEC. This could be explained by the increase of both the MWCNT-MWCNT interaction and the MWCNT-HDPE molecular chain interaction when the loading amount of MWCNTs was increased, as a result of the increased interfacial interaction between the HDPE-modified MWCNTs and the HDPE matrix. The increment of moduli for the mPEC was higher than that for the dPEC at the same filler concentration. Especially, the G′ and G″ values of mPEC3 had an obvious improvement while sample dPEC3 almost didn’t change at all in comparison with those of the pure HDPE in the low-frequency range. This indicated that mPEC showed solid-like behavior in the low-frequency range owing to the homogeneous dispersion of the MWCNTs and the existence of strong interaction in the three dimensional network structure of MWCNT-MWCNT interactions and the MWCNT-HDPE molecular chain interaction.45–48

Sweep frequency dependence of
The heating and cooling DSC curves of the pure HDPE and its composites are shown in Figure 10. The peak melting temperature (Tm), the peak crystallization temperature (Tc), the enthalpy during crystallization (
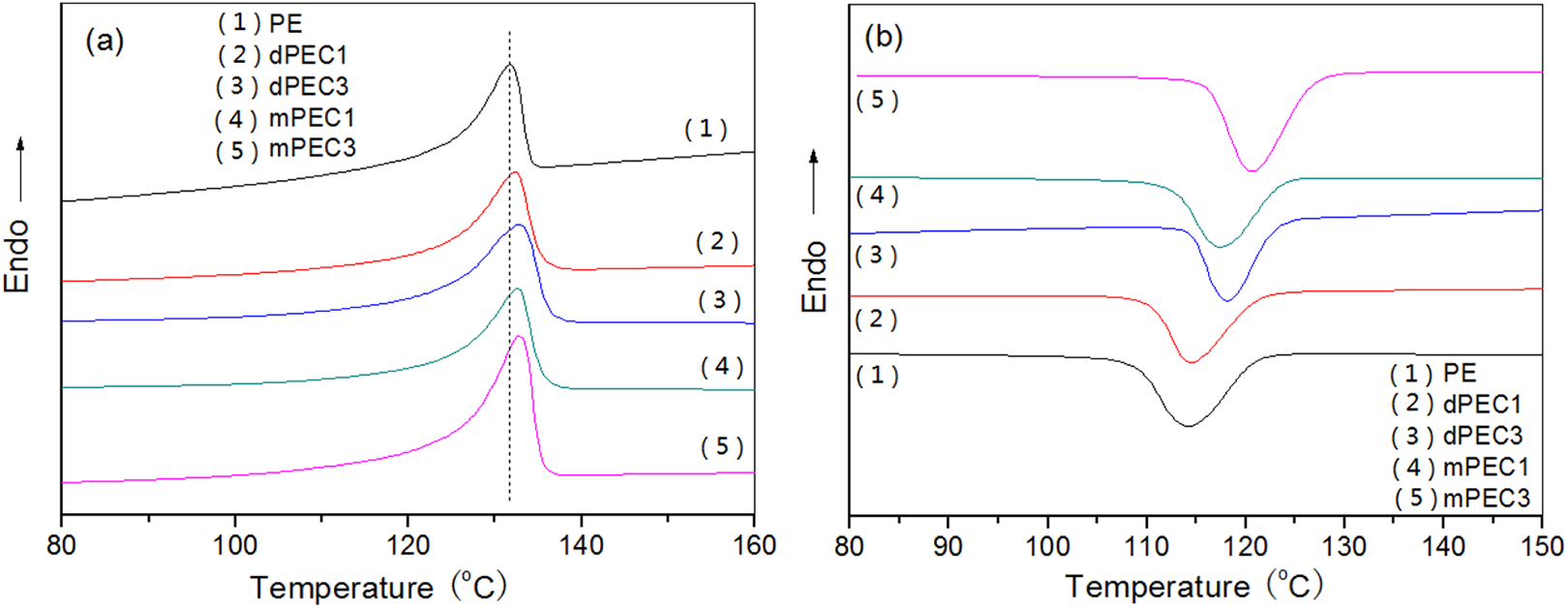
DSC heating (a) and cooling (b) curves for PE and its composites.
Values of Tm, Tc, ΔHc, and Xc for PE and its composites.


where
The values of Xc for the samples were calculated as follows:

where
Conclusions
In this work, the MWCNTs/HDPE composites with good mechanical, electrical and rheological properties were prepared by a masterbatch method. The results implied that the MWCNTs could uniformly distribute in the HDPE matrix and the MWCNTs/HDPE composites prepared by a masterbatch method exhibited better electrical, mechanical and rheological properties than those of the pure HDPE and the MWCNTs/HDPE composites, indicating that the polymerized HDPE cover on the MWCNT surface played an important role as an interfacial modifier. The tensile yield strength and the Young’s modulus of sample mPEC3, and the flexural strength and flexural modulus of sample mPEC1 were improved by 38.3%, 41.7%, 24.4%, and 42.9%, respectively, in comparison with those of the pure HDPE. But for dPEC3 and dPEC1, the corresponding values were only 25.2%, 26.2%, 16.3%, and 12.1%, respectively. An increase of the MWCNTs concentration resulted in a progressive decrease in the volume resistivity of mPEC and dPEC with the volume resistivity of mPEC3 being almost one order of magnitude lower than that of dPEC3 and four orders of magnitude lower than that of the pure HDPE. The |η*|, G′ and G″ values of mPEC were higher than those of the pure HDPE and dPEC with the same filler content. In addition, the HDPE-modified MWCNTs had better crystallization nucleating ability for the HDPE than that of the unmodified MWCNTs. We suggest that the successful preparation of the MWCNTs/HDPE composites with obviously enhanced properties and functionalities by the masterbatch method can serve as a demonstration of an effective way to obtain other advanced polyolefin-based composites.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Guangdong Province in China (No. 039184 and No. 2017A030313268), and the Maoming Public Service Platform for Transformation Upgrading and Technological Innovation of Petrochemical Industry, China (No. 2016B020211001).
