Abstract
The composition of dental resin-based composite (RBC) matrix is partly responsible for many clinical failures in restorations, which may come from dimensional variation or instability in a wet environment. The objective of this study is therefore to evaluate the thermal stability over time of four dental RBC with different matrices. Silicone cylinders were filled with four different materials and then photopolymerized. To simulate ageing in the buccal environment, half of the samples were placed in a dark place at 37°C for 45 days in sealed compartments containing 2 ml of water. All the RBC produced were subjected to thermogravimetric analysis to measure the loss of mass as a function of temperature. Bis-GMA-based resins and Ormocer materials have similar curves before and after soaking in humid atmosphere. The curves of the UDMA resin are different before and after water ageing, meaning that water imbibition has modified the structure of the composite and its degradation. Even if many curves are similar within the same RBC at different polymerization times or at different pre- and post-ageing times, it is rare to observe a common kinetics between two different composites. Our results show good wet stability of polymerized dental RBC according to the manufacturer’s instructions, although the UDMA-based materials show more variation. It therefore seems that Ormocer resin composite with mass placement have ageing properties that can compete with those of conventional composites whereas those incremented on 2 mm layers are more sensitive to the time necessary for polymerization.
Introduction
The composition of resin-based composites (RBC) for direct dental restorations has considerably evolved since their origin. These materials are by definition derived from a mixture of two phases: an organic and an inorganic period that are linked together by a coupling agent.1,2 The desire to improve its mechanical strength and dimensional stability has led over time to several changes in their composition. 2 These changes have long focused on the shape, size, nature and percentage of fillers in order to improve mechanical and optical properties, but the organic fraction of the matrix is now considered to be the main cause of dimensional variations over time. 3 This fraction represents 25 to 50% of the volume of the composite; it includes the matrix resin and its diluents, a priming system, pigments and polymerization stabilizers. 4 The composition of the matrix is partly responsible for many clinical failures in restorations, which may come from dimensional variation or instability in a wet environment.5,6 These materials, which are plastic when inserted into a tooth, are photopolymerized in the oral cavity. Their establishment also requires an optimization of photopolymerization conditions.
The most commonly used base monomer in dental RBC is bis-GMA (bisphenylglycidyl dimethacrylate) which, due to its high viscosity, is mixed with other dimethacrylates such as TEGDMA (triethylene glycol dimethacrylate), UDMA (urethane dimethacrylate) or other monomers.4,7 UDMA was first introduced as a dental resin by Foster and Walker in 1974 as an alternative monomer to bis-GMA. It was obtained by the reaction between an isocyanate and an alcohol and it has been shown that UDMA-based polymers would show similar or very slightly lower water absorption than bis-GMA-based polymers.8,9 These resins modified with urethane dimethacrylate also have a further reduced water absorption when several radicals are added to the base molecule.10,11 Absorption phenomena are correlated with the solubility of the material, and UDMA-based materials would appear to behave correctly.8,9
New Ormocer-based matrices have been introduced. This is an acronym for ‘Organically Modified Ceramics’. They constitute a range of dental RBC which have been available on the market for the past 10 years. 12 They were developed after a collaboration between the Fraunhofer® Institute and the Medical Polytechnic School of Hanover in Germany. While conventional RBC are made of a purely organic matrix, Ormocer-based materials contain polymers at the matrix level, resulting from the addition of small monomer units whose nucleus is essentially composed of silicon atoms combined with silanized inorganic fillers. 13 The Si-O-Si inorganic network is produced by a sol–gel process to produce an inorganic polymer by simple chemical reactions and at a temperature close to room temperature.14-16 The polysiloxane chains found in Ormocers, made up of a succession of silicon and oxygen molecules, combine with other chains. These chains react during the polymerization process and thus contribute to an organic functionalization of the inorganic network initially formed. 13 In dental RBC, for example, the additional polymer-organic structure is established in the cavity by light-induced radical-based polymerization. 17 They therefore form cross-linked copolymers in a three-dimensional network. The Ormocer matrix already contains a polymer even before the final polymerization. This skeleton formed by silicon dioxide (SiO2) would give the material a low shrinkage compared to dimethacrylate matrices. 18
To explain this property, it should be noted that in ceramics, the bonds between the atoms adopt a character qualified as ionocovalent. 19 The ionic or covalent tendency of an interatomic bond is related to the electronegativity gradient, 20 given that the link will be more ionic if the difference in electronegativity is higher. 20 Silicon dioxide SiO2 is considered ionic at about 51% so its links have the double ionic-covalent character. 21 In addition, the abundance of polymerization sites allows Ormocers to crosslink without leaving any residual monomer, thus providing better biocompatibility. 13 Both organic and inorganic fillers are incorporated into this three-dimensional network. In the end, the silicon dioxide skeleton of the Ormocers lends the product its name and explains their relationship to ceramics.
Some dental RBC, including several Ormocers, are more translucent to have the ability to polymerize on the 4 mm layer instead of 2 mm. These composites are described as ‘bulk-fill’. The photopolymerization light diffuses more deeply, but the range of shade is more restricted to preserve the translucency essential for correct photopolymerization.
The new class of materials, led by Ormocers, is expected to combine the properties of silicone, the strength of organic polymers and the hardness and thermal stability of ceramics. These alternatives are even more interesting as doubts are currently emerging as to the toxicity of bisphenol A, found in the composition of bis-GMA. 22 Nevertheless, further studies are needed to assess the chemical and physical properties of these materials.
Thanks to a heating-up process called thermogravimetry, which makes it possible to evaluate the degradation kinetics of composites as it would occur under the effect of ageing but at an accelerated rate, several questions can be resolved. Do these organo-mineral matrices have the same thermal stability after maintenance in a wet environment? Is their degradation kinetics the same as that of other organic matrix composites (Bis-GMA, UDMA)? As all these materials are photopolymerizable, does the curing time play an important role in the stability of the material? Having a degradation profile enables the practitioner to anticipate the phenomena of the ageing of materials and to highlight the importance of such clinical implementation.
The objective of this study is therefore to evaluate the thermal stability over time of four dental RBC with different matrices.
Materials and methods
Materials
Four materials were evaluated: a traditional bis-GMA composite resin (Grandioso®, VOCO, Cuxhaven, Germany), an UDMA composite resin (G-aenial®, GC, Leuven, Belgium), two Ormocers composites resins including a classic one polymerizable per 2 mm layers (Admira Fusion®, VOCO, Cuxhaven, Germany) and the other polymerizable on 4 mm layers (Admira Fusion Xtra®, VOCO, Cuxhaven, Germany).
The samples were constituted under the manufacturer’s recommendations, as duplicates and by the same operator. Briefly, silicone cylinders 4 mm in diameter and 2 or 4 mm in height were filled with conventional composites (2 mm cylinders) and mass placement (4 mm cylinders), and then photopolymerized (1300 mW/cm2, CELALUX 3, VOCO®, Cuxhaven, Germany) at 50 and 100% of the time recommended by the manufacturer (Table 1).
Polymerization times for tested dental resin-based composites.

To simulate ageing in the buccal environment, half of the samples were placed in a dark place at 37°C for 45 days in sealed compartments containing 2 ml of water.
Thermogravimetric analysis (TGA)
All the RBC produced were subjected to thermogravimetric analysis to measure the loss of mass as a function of temperature. The quantitative determination of the organic and inorganic parts of the RBC was performed with a TGAQ50 (TA Instruments, New Castle, DE) with 25 mg of each sample and the analysis was performed at 30 to 1000°C at 10°C/min under synthetic air atmosphere. There was no systematization of a blank measurement before measuring a sample, but a temperature calibration based on nickel’s Curie Temperature was performed every month on the device.
Results
The TGA and derivative thermogravimetric curves are shown in Figure 1. The slope of the derivative objectifies the speed of mass variation.

TGA thermograms and temperature derivative thermograms of (A) bis-GMA-based material, (B) UDMA-based material, (C) Ormocer material and (D) Ormocer bulk-fill material. All have been photopolymerized at the time recommended by their manufacturer, and aged at 37°C during 45 days or not.
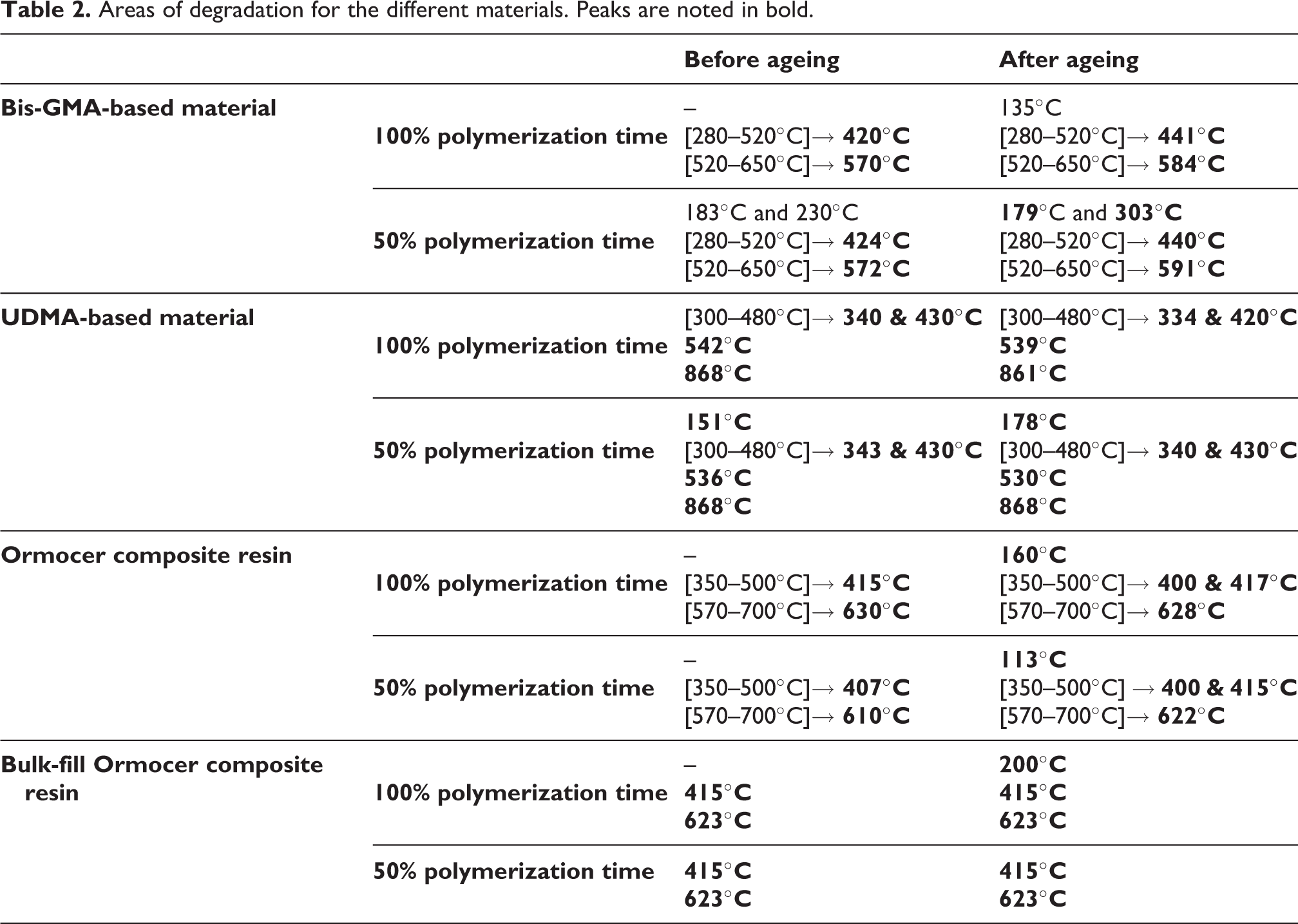
Two regions can be clearly identified for the samples of the Bis-GMA-based material, also for those of the Ormocer material, even with or without the ageing process, indicating a two-step degradation process (Table 2). It does not differ if the polymerization time is changed for the Bis-GMA-based material. The aged sample polymerized as recommended by the manufacturer has an offset of the order of +20°C on the abscissa axis compared to the unaged samples. The weight loss after each step represents the organic part (≈15%), whereas the inert residues (≈85%) corresponds to the inorganic part (Table 3).
Areas of degradation for the different materials. Peaks are noted in bold.
Relative proportions of organic and inorganic phases of materials.

For the UDMA-based material, samples polymerized at 100% of the time indicated by the manufacturer have a common general profile before and after ageing (Table 3). Only the peaks’ amplitudes differ, with the peak at 340°C being less pronounced and the peak at 430°C being more pronounced (Figure 1). The differences are even more important when the material is poorly polymerized with a shift in the first peak from 151°C to 178°C and the amplitude inversion between the peaks at 340°C and 430°C.
In the case of the Ormocer material, after ageing, the thermogram shows a peak at 112°C, a double peak close to 400°C, and a shift of about +10°C on the abscissa axis. With a polymerization time halved, the degradation kinetics of unaged samples show similar areas of stability. After ageing, a peak at 113°C is again found for samples polymerized at 50% of the recommended time.
Finally, all the samples of bulk-fill Ormocer composite resin, polymerized at 100% or 50% of the recommended time, have similar degradation kinetics before or after ageing with peaks at 415°C and 623°C (Figure 1, Table 3). However, there is additional degradation after ageing, meaning that some species degrade before the main degradation for 14% of the material.
Discussion
Our results show good wet stability of polymerized dental RBC according to the manufacturer’s instructions, although the UDMA-based materials show more variation.
Thermogravimetric analysis is used to measure the change in sample mass as the temperature rises. The use of TGA enables the researcher to monitor the evolution of the processes over time which occurred during the temperature rise and to evaluate whether monomers remain in the polymer. Additionally, it also defines which types of bonds have been established in the sample. 23 Thermal degradation concerns the organic part of the RBC so the technique also allows one to determine the proportions of the organic and inorganic parts inside the structure.
In the case of dental RBC, mass loss peaks occur during decomposition,
Indeed, the application of thermogravimetric analysis to dental RBC, that are more or less polymerized and have undergone artificial ageing or not, enables the researcher to compare the differences in their structural stability and to relate them to their internal arrangement. In 2011, Sideridou et al. established a link between the high aromatic ring content of a bis-GMA-based material and its one-peak degradation curve, compared to a two-step degradation of a composite containing UDMA and fewer cycles. 5 Recently, Nascimento et al. have also described the organic and inorganic parts of several dental RBC. 25 Our results are concordant with theirs for the same Ormocer bulk-fill composite as they have obtained 87% of residues after the thermogravimetric analysis. Concerning the other types of matrices, they have shown a range of residues comprised between 60.1% (for a UDMA-based biomaterial) and 84.2% (for a Bis-GMA-based biomaterial). 25
Behaviour in a wet environment
Bis-GMA-based composite resins and Ormocer materials have similar curves before and after soaking in a humid atmosphere. On the other hand, the curves of the UDMA composite resin are different before and after water ageing, meaning that water imbibition has modified the structure of the RBC and its degradation. It has already been shown that there are variations in absorption between the different polymers by differential penetration of water into the polymer network through porosities and intermolecular spaces,3,26 this phenomenon being classically measured higher for bis-GMA-based resins than for urethane-based resins. 11 However, our results show an opposite effect with less degradation of bis-GMA materials in the presence of water.
The analysis of the results obtained for Ormocer composite resins before ageing, for which the polymerization time was respected, gives an overview of the expected profile of the thermograms for the two Ormocers materials. For the one incremented on 2 mm layers as was the case for bulk, a main peak at 430°C is observed, corresponding (according to some researchers) to the breaking of siloxane links.
27
Once maintained at 37°C for 45 days, the same behaviour is observed in both types of composite,
Changes in polymerization time and wet atmosphere
After ageing, the Ormocer material photopolymerized on 2 mm layers with a polymerization time reduced by half presents a new peak of 113°C compared to the reference time, which indicates that when polymerized at a duration below optimal, it has a higher tendency toward degradation. This structural difference is also found in well polymerized but aged samples of this material: at Day 0 (D-0) a single peak is present at 400°C, while a second peak is noted for samples that have been stored in a humid atmosphere at 37°C for 45 days. When the difference between these two peaks is increased for aged samples, the first peak recorded between 78°C and 160°C occurs at a higher temperature (113°C compared to 160°C for polymerization times halved and complete respectively). This RBC thus acquires different conformations depending on the duration of polymerization and this considerably influences the structure it adopts during its ageing. It is therefore sensitive to complete respect of the optimal curing time.
When not polymerized to optimal duration, the Ormocer bulk-fill material exhibits an early peak at 74° before ageing. The latter part of its degradation is done in a similar way regardless of the polymerization time. The presence of early peaks during incomplete photopolymerizations indicates that the material still owns non-polymerized monomers in its structure, even if this composite resin presents a structure relatively similar whether it is well polymerized or not. After ageing, the similar appearance of the curves with those obtained on unsoaked samples indicates a similar intrinsic structure between these two samples and reflects a high tolerance of the material for an application under a polymerization time shorter than the optimal time. Indeed, if initially concrete differences are noted for a shorter photopolymerization on the day of the implementation (D-0), the soaking at 37°C during 45 days tends to give the material a conformation similar to that of the complete photopolymerization at D-0 time.
For fully polymerized aged samples, the occurrence of a new early peak at 202°C, which integrates into a curve similar to that observed without ageing, suggests the formation over time of soft silicone bonds, following a reorganization of the siliceous infrastructure. The hypothesis of this restructuring also enables one to explain the plot of the other two peaks. These occur at almost identical temperatures and amplitudes before and after ageing. It would therefore mean that Ormocer bulk-fill material is more permissive with little deviations in the polymerization time, leading to modifications at D-0 but ultimately leading to a comparable conformation after ageing.
The bis-GMA-based composite resin, completely photopolymerized, has a two-phase degradation kinetics (Figure 1), the second phase corresponding to the degradation of acrylate bonds. After ageing, the temperature shift on the curve indicates that the different chains initially noted are still present but that they take longer to degrade (Figure 1). The appearance of a new peak at 135°C reflects a change in the internal structure of the material. Since this peak does not occur when the photopolymerization time is halved, it would not correspond to non-polymerized monomers but rather to low energy bonds. If they were non-polymerized monomers, they would have been detected even at a reduced photopolymerization time. Thus, they are the result of reconfiguration of the links within the matrix, occurring in all samples of this composite during their ageing, as this peak appears at Day 45 between 135°C and 179°C whether the photopolymerization is complete or halved. The variation in the photopolymerization time has no influence on the samples at D-0 but modifies the degradation kinetics after ageing.
For the UDMA material, the degradation of unaged samples correctly photopolymerized has a four-step sequence. This brings it closer to the bis-GMA material, although the UDMA matrix material is distinguished by the occurrence of a peak at a very high temperature (868°C). It could indicate the presence of stronger bonds within its organic network. After ageing, the thermogram amplitude around 430°C is increased while the rest of its curve remains identical. A non-optimal polymerization time results in a new peak at D-0 between 150°C and 175°C and after ageing in an increased peak at 429°C.
Thus, even if many DTG curves are similar within the same composite resin at different polymerization times or at different pre- and post-ageing times, it is rare to observe a common kinetics between two different composites. This highlights the differences between the matrices and therefore the different RBC. Their matrix networks are complex and so do the connections it exists inside. The great heterogeneity of the kinetics observed in the various samples of Ormocer materials confirms a multiplicity of bond types that the material establishes during the ageing process. It also depends on the strict respect of the photopolymerization time. The similar kinetics of the Ormocer bulk-fill material in all tested situations is proof of its stability, whether regarding the photopolymerization time or the ageing process. This may be related to the proportion taken by the polymerization initiators in its composition. Conventional RBC maintain a high degree of structural regularity despite the ageing they undergo or the polymerization variations to which they have been subjected.
Conclusion
Under optimal conditions, the two tested Ormocer materials degrade in two phases during the TGA. The one incremented on 2 mm layers is very sensitive to the respect of the polymerization time, which is not the case for the bulk-fill which also resists ageing very well. The UDMA-based composite tested has a poor initial tolerance to the variation in polymerization time, whereas the bis-GMA-based composite does not. These two show good behaviour after soaking for 45 days.
It therefore seems that Ormocer RBC with mass placement have ageing properties that can compete with those of conventional composites whereas those incremented on 2 mm layers are more sensitive to the time necessary for polymerization.
This study shows a very heterogeneous behaviour between the different materials and therefore the need to follow very precisely the operating procedures.
Footnotes
Acknowledgements
The authors really thank James-Robert Bottriell and Dr. Francis Collins for providing language and content corrections.
Author contributions
TC, PP, JD and SJ contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
