Abstract
The present work focuses on comparative properties of β-naphthol based trifunctional epoxy resin and commercial epoxy resin. Reaction of β-naphthol, formaldehyde and epichlorohydrin forms trifunctional epoxy resin. β-Naphthol based trifunctional epoxy resin characterized by FT-IR, elemental analysis (C, H, N, O analyzer), epoxy equivalent weight (EEW), weight average molecular weight
Introduction
Epoxy resins are widely used in composites, electronics & electrical, adhesives & coatings, aerospace, building, automobile and many other areas due to their excellent mechanical and electrical properties, high bonding strength, excellent dimensional stability and good processing abilities.1–3 Nowadays, epoxy resins are becoming more popular in fiber reinforce composite field due to superior heat resistance, better adhesive properties and good mechanical properties of epoxy resin.4–6 The properties of cured epoxy resins are highly related to their network structure, which is critically influenced by the functionality of the epoxy resin. One effective method to achieve excellent epoxy resin systems with high strength and modulus is to increase the functionality of the epoxy resin.7–11 Multifunctional epoxy resin is a way to enhance heat-resistance because of higher curing density.12,13 Multifunctional epoxy resins are well known for their improved mechanical, chemical, thermodynamic and electrical properties.14–17 Other advantages of multifunctional epoxy resins are their high glass transition temperature, high decomposition temperature, long term high temperature performance and good wet strength performance. Multifunctional epoxy resins have two important limitations due to intrinsic brittle nature and considerable moisture absorption tendency from environment. So, it adversely affects most of the physico-mechanical properties of the fabricated articles. These drawbacks increase by enhancing the cross-link density of the network. Hourston et al. studied the dynamic, mechanical and fracture properties of three types of epoxy resins with different functionalities and concluded that glass transition temperature (Tg) varied with functionality, whereas the strain energy release rate and the stress intensity factor varied insignificantly with functionality. 10 Epoxy based fiber composites have become more commonly used in automobile, electronic devices, construction and aerospace industries. This is attributed to the attractive mechanical properties, dimensional stability and corrosion resistance of the composites.18–21 Recently epoxy based hybrid composites have been extensively used in many engineering and industrial applications. Load bearing engineering applications, superior adhesive properties and mechanical strength are the key features of the composite materials. In comparison with the natural fibers composites; synthetic fibers are advantageous due to their attractive price, high density and higher abrasion resistance. The energy consumption needed for the production of synthetic fibers is much more than that needed for a similar quantity of natural fibers. 22 The synthetic fibers have a wide variation in diameter and length, which in turn affects on composites expected mechanical behavior. The variation in dimensions is contributed to fiber type, fiber maturity as well as processing methods adopted for the extraction of fibers, which all affects the diameter and stability of the fiber. Source, age, separating techniques, moisture content and the history of fiber also play an important role in the filament and individual fiber properties. The implementation of synthetic fibers in composites is attractive in industrial sectors like automobiles and construction.23,24 Miguel et al. investigated non-linear behavior of ply carbon fiber-reinforced epoxy laminates and results showed that non-linear behavior is desirable and it can help composite structures to maintain functionality even when they are overloaded to improve safety and reduce the applied safety factors. 25 Patel and Parsania synthesized bisphenol-C based hexafunctional resin and used as matrix material for jute and glass fiber reinforce composites and result showed that this composites had higher water resistance than commercial epoxy resin based composites. 26 Liu et al. investigated interlaminar shear strength of E graded woven glass fiber (GF) reinforced epoxy composites with unmodified and modified matrix. The results showed that modified resin based composites have 25.4% higher interlaminar shear strength than unmodified resin based composites. 27 Beibei et al. done Surface modification of carbon fabrics by nitric acid and multiwall carbon nanotube incorporation into the resin matrix were simultaneously applied for carbon fabrics/resin composites to improve their tribological properties. 28 Ilkay et al. functionalized multi walled carbon nanotubes and boron nitride with cetyltrimethylammonium bromide and prepared composites dispersed in DGEBA and results showed that thermal resistance of composites increased. 29
The main aim of the present work is to synthesize β-naphthol based trifunctional epoxy resin and to determine properties of resin such as epoxy equivalent weight, hydrolysable chlorine content, viscosity, volatile content, FT-IR and average molecular weight, elemental analysis and compare with commercial epoxy resin. Curing behavior of trifunctional epoxy resin and commercial epoxy resin were studied by various hardeners. Mechanical, chemical and thermal properties of carbon fiber based composites were also studied. The resultant data were compared with composites based on epoxy resin.
Materials and methods
Raw materials
Solvents and chemicals used were of laboratory grade and purified prior to their use. β-naphthol and epichlorhydrin purchased from Sigma-Aldrich. Formaldehyde, diethylene triamine, triethylene tetraamine, methyl ethyl ketone, perchloric acid, acetic acid, tetraethylammonium bromide and sodium hydroxide were purchased from S. D. Fine-Chem Ltd. Aromatic hardeners phenalkamine, polyamido amines and polyamides were obtained from Admark Polycoats Pvt. Ltd., Vadodara. Carbon fiber was purchased from Composites Tomorrow, Vadodara and commercial epoxy resin supplied by Atul Ltd., Atul, India.
Methods
Synthesis of trifunctional epoxy resin based on β-naphthol
2 liter three neck flask equipped with a mechanical stirrer and condenser was placed into a thermostat heating unit. To this flask 1.0 mole of β-naphthol and 2.0 mole of formaldehyde were added in the presence of alkaline medium. Stirred continues for 1.5 hours at 55°C temperature then add 3.0 mole epichlorhydrin in a reaction flask and add aqueous NaOH solution drop wise (2.2 mole and 40% concentrated solution). After completion of addition, temperature was increased to 80°C and the reaction mixture was stirred for 3 hours then 50 ml methyl ethyl ketone was added as a solvent to maintain the viscosity of resin. Vacuum distillation was used to removal of salt and then resin was distillate at 118°C temperature to remove excess water, methyl ethyl ketone and epichlorhydrin. Resin was cooled at room temperature. Resin had dark yellow color with no specific odor. The product yield was 87%. Probable reaction scheme is given in Figure 1.
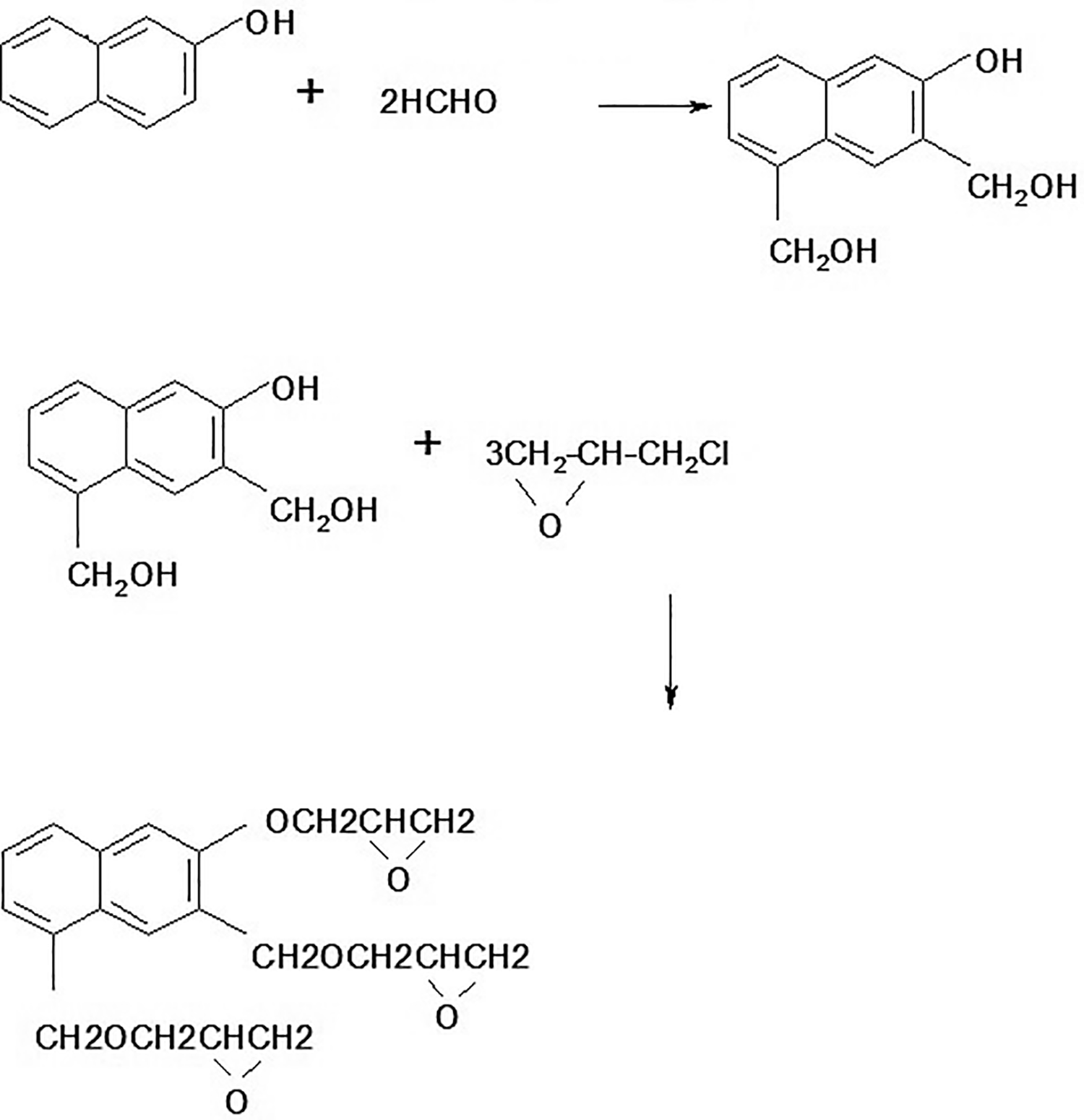
Probable reaction reaction of trifunctional resin.
Fabrication of carbon fiber based composites
β-Naphthol based trifunctional resin-carbon fiber-diethylene triamine (NCD) composite fabricated as under:
Fiber reinforced composite was prepared by using resin to fabric ratio of 60:40. Diethylene triamine (DETA) was used as a hardener according to its amine hydroxide equivalent weight (AHEW). The required quantity of resin and DETA were mixed well and applied on 16 sheets of carbon fiber having size of 15 × 12 cm by hand lay-up technique. All sheets were stacked one over another in between two teflon sheets. All sheets kept between two plates of compression moulding machine at 80°C temperature for 90 minutes for curing and finally 50 psi pressure applied for 2 minutes. Prepared composite cooled at room temperature and remove from two plates of compression moulding machine. After removal of teflon sheet all dimension of composite sheet were measured.
All carbon fiber reinforced composites were prepared by a similar method discussed above namely β-Naphthol based trifunctional resin-carbon fiber-triethylene tetramine (NCT), β-Naphthol based trifunctional resin-carbon fiber-phenalkamine (NCP), β-Naphthol based trifunctional resin-carbon fiber-polyamido amines (NCPA) and β-Naphthol based trifunctional resin-carbon fiber-polyamides (NCPD). Similarly, carbon fiber reinforced composites based on commercial epoxy resin represented as epoxy resin-carbon fiber-diethylene triamine (ECD), epoxy resin-carbon fiber-triethylene tetramine (ECT), epoxy resin-carbon fiber-phenalkamine (ECP), epoxy resin-carbon fiber-polyamido amines (ECPA) and epoxy resin-carbon fiber-polyamides (ECPD).
Characterization
Epoxy equivalent weight (EEW) (ASTM D 1652)
An accurate weight of resin, dissolved in tetraethyleneammonium bromide solution was titrated against 0.5 N perchloric acid solution. A Metrohm Auto titrator was used for measurement.
Viscosity measurement (ASTM D 789)
Brookfield viscometer model number RV digital viscometer used to determine viscosity of resin at 25°C temperature.
Volatile content (ASTM D 1259)
Accurate weight of resin poured in petri dish which was kept in oven at 110°C temperature for 30 minutes. Volatile content was determined by percentage change in initial and final weight.
Hydrolysable chlorine content (HyCl) (ASTM D 1726)
2 g of resin was dissolved in a mixture of 15 ml toluene and 25 ml alcoholic KOH solution. The reaction mixture reflux for 20 minutes at 300°C temperature. Blank reading was taken without sample. Blank and sample titrated against 0.5 N HCl solution. Metroham Auto titrator was used to carry out titration.
Gel Permeation Chromatograph (GPC)
The weight average molecular weight
Fourier transform infrared spectroscopy (FT-IR)
Spectrum GX (Perkin Elmer, U.S.A.) spectrophotometer was used to carried out FT-IR. The range of FT-IR is 10,000 cm−1 to 370 cm−1 in presence of KBr pellet. The scanning speed of FT-IR was 0.2 cm/s and signal average over 20 scans was applied to measure the absorbance spectra.
C, H, N, O analyzer (ASTM D 5373)
Carbon, hydrogen and oxygen elements of β-naphthol based trifunctional epoxy resin measured using 2400 series II, Perkin Elmer, U.S.A.
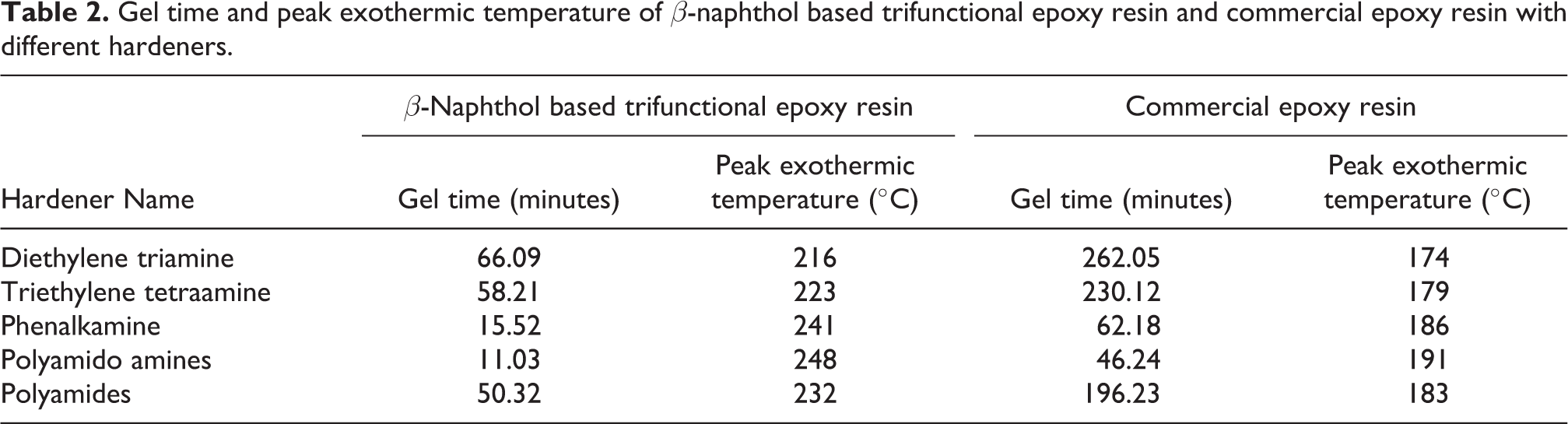
Gel time and peak exothermic temperature (ASTM D 2471-99)
Gel time and peak exothermic temperature of β-naphthol based trifunctional epoxy resin and commercial epoxy resin measured by Brookfield DV2 T instrument.
Mechanical properties of carbon reinforce composites
Tensile strength (ASTM D 638)
Tensile strength measures material’s ability to withstand the forces that tend to pull it apart and determines to what extent material stretches before breaking. Test was performed on UTM (universal testing machine) Shimadzu AG 100 at 100% strain rate, and crosshead speed of 50 mm/min was maintained in test. 30
Flexural strength (ASTM D 790)
Flexural strength is the ability of the material to withstand bending forces applied perpendicular to its longitudinal axis. The stresses induced by the flexural load are a combination of compressive and tensile stresses. Flexural properties were reported and calculated in terms of the maximum stress and strain that occur at the outside surface of the test bar. Test was performed on UTM Shimadzu AG 100, at a crosshead speed of 1.2 mm/min. 30
Izod impact strength (ASTM D 256)
Izod impact test results are expressed in terms of kinetic energy consumed by the pendulum in order to break the specimen. The specimen used for the test was notched. Izod impact strength was measured on CEAST Izod tester. 30
Rockwell hardness (ASTM D 785)
The information from a hardness test can be used to provide critical material performance information and insight into the durability, strength, flexibility and capabilities of a variety of component types from raw materials to the prepared specimens and finished goods. Digital Rockwell hardness tester with HRL indenter was used to measure the hardness value of the test specimen. 30
All mechanical properties were measured at room temperature. Three samples of each test were analyzed, and the average of the results were taken into an account.
Thermogravimetric analysis (TGA)
Resistance against weight loss at various temperature of all composites were studied on Perkin-Elmer Pyris-1. The composites samples contain 5–10 mg were scanned at temperature range of 50–1000°C at a heating rate of 10°C/min in nitrogen environment.
Chemical resistance test (ASTM D 543-87)
The chemical resistance of the composites was studied as per ASTM D 543-87 method. This method covers the chemical resistance of all composites for change in weight, dimensions, appearance and strength properties by the action of different chemical reagents. Test samples have dimension of 1.5 × 1.5 cm immersed in containers in presence of 250 ml chemical reagents such as concentrated sulfuric acid (10% wt/wt), aqueous sodium hydroxide (10% wt/wt), sodium chloride (10% wt/wt), methanol and tetrahydrofuran. Water absorption of all composites test samples was studied. All samples were observed after exposer of 7 days in presence of chemical reagents. After 7 days samples were washed by distilled water and dried by pressing them both sides with filter paper at room temperature. Before and after test cycle all specimens were weigh in a précised electronic balance and percentage weight loss/gain were measured. Percentage change in thickness was determine with the use of digital micro meter. In each case two test specimens were used and their average value reported as a results.
Results and discussion
β-Naphthol based trifunctional epoxy resin
The epoxy equivalent weight of β-naphthol based trifunctional epoxy resin was 397 g and weight average molecular weight
Hydrolysable chlorine content of β-naphthol based trifunctional epoxy resin was 0.91% while commercial epoxy resin has 0.63% which clearly indicate that reactivity of β-naphthol based trifunctional epoxy resin was similar with commercial epoxy resin. C, H, N, O analyzer used to determine percentage of carbon, hydrogen and oxygen. Results of C, H, N, O gave the confirmation of polymerization reaction. Calculated data and obtain data were shown in Table 1.
C–H–O measurement of β-naphthol based trifunctional epoxy resin.

Gel time and peak exothermic temperature of commercial epoxy resin and β-naphthol based trifunctional epoxy resin in presence of different hardeners was given in Table 2. For β-naphthol based trifunctional epoxy resin diethylene triamine required 66 minutes to convert resin into gel form meanwhile polyamido amines work six times faster and converted resin into gel form in 11 minutes. Commercial epoxy resin converted into gel form by various hardeners was almost four times slower than β-naphthol based trifunctional epoxy resin. Peak exothermic temperature of β-naphthol based trifunctional epoxy resin observed 1.5 time higher than commercial epoxy resin for all hardener system.
Gel time and peak exothermic temperature of β-naphthol based trifunctional epoxy resin and commercial epoxy resin with different hardeners.
The FT-IR spectrum of the β-naphthol based trifunctional epoxy resin and commercial epoxy resin was shown in Figure 2. In FT-IR spectra peak observed at 3415.03 cm−1 due to the stretching of –OH groups but percentage transmission of this peak is lower than commercial epoxy resin which indicate that more –OH groups used in reaction. The sharp peak at 2967.12 cm−1 indicate C–H stretching vibration. The peak at 1611.25 cm−1 indicate stretching of C=C of aromatic ring while peak at 1515.05 cm−1 indicate stretching of C–C of aromatic ring. The characteristic and strong peak of C–O–C of oxirane ring observed at 845.12 cm−1 which confirmed the polymerization reaction.

FT-IR of (A) trifunctional epoxy resin & (B) commercial epoxy resin.
Carbon fiber reinforced composites based on trifunctional epoxy resin
Mechanical properties (Tensile strength, Flexural strength, Izod impact strength and Rockwell hardness) data given in Table 3. Tensile strength of composites ranges from 81.5-89.3 MPa. Increasing order of tensile strength of composites were NCPA > NCP > NCPD > NCD > NCT. Polymeric hardeners has higher molecular weight so it provides additional strength to composites and so NCPA composite has higher tensile strength than NCT composite. Comparison with commercial epoxy resin based composites tensile strength were decreases around 25%.
Mechanical properties of β-naphthol based trifunctional epoxy resin and commercial epoxy resin.

Flexural strength of composites found almost 2 to 2.5 times higher than their tensile strength results. NCPA composite have higher flexural strength at 211.2 MPa while NCT composite have lowest flexural strength at 199.5 MPa. Polymeric hardeners provides strength to composites due to their higher molecular weight as well as their adhesion property was superior than aliphatic hardeners so NCPA have higher flexural strength than NCD and NCT. The range of flexural strength of carbon fiber composites based on commercial epoxy resin was 139.8 to 155.2 MPa almost 30% lower than carbon fiber reinforce composite based on β-naphthol based trifunctional epoxy resin.
Izod impact strength of all the particulate composites was measured to study the resistance of specimen against the sudden impact force applied on it from the specific direction. Aliphatic hardeners based composites have lower Izod impact strength than aromatic hardeners based composites. NCT composite have 26.5% lower Izod impact strength than NCPA composite. Impact strength of all reinforce composites based on commercial epoxy resin is around 15% lower than β-naphthol based trifunctional epoxy resin based composites.
Rockwell hardness was analyzed to check rigidity of composites. Increasing order of Rockwell hardness of composites were NCPA > NCP > NCPD > NCD > NCT. NCPA have higher Rockwell hardness at 97.4 which was almost 13% higher than NCT. Rockwell hardness of carbon fiber reinforce composites based on commercial epoxy resin were in the range 69.2 to 77.8. The results shows that composites of commercial epoxy resin have 20% lower than composites of β-naphthol based trifunctional epoxy resin.
All mechanical properties results taken three times and mean of them shown in Table 3. All results of mechanical properties were statistically analyzed with data variance and given in brackets of Table 3.
Chemical resistance of all composites were measured against six different chemical reagents. It was observed that the composites of β-naphthol based trifunctional epoxy resin having no gain or lose weight and therefore no erosion has taken place due to the exposure of chemical reagents. The results of chemical resistance were given in Table 4. All carbon fiber reinforce based composites of β-naphthol based trifunctional epoxy resin showed excellent chemical resistance and there was no change in composites surface finishing against six different chemical reagents. In compare with composites of commercial epoxy resin weight loss observed in 10% aqueous H2SO4, methanol and tetrahydro furan.
Chemical resistance of carbon fiber based composites of β-naphthol based trifunctional epoxy resin and commercial epoxy resin.

%a = % change in weight, %b = % change in thickness, NC = no change.
TGA i.e. weight loss as a function of temperature was done in order to understand the thermal stability of the particulate composites. Mass loss curves for all carbon fiber based composites were shown in Figure 3. Decomposition rate of the composites associated with various temperature intervals calculated and shown in Figure 4. Thermal kinetic parameters like initial system temperature (T0) initial decomposition temperature (Ti), procedural decomposition temperature (PDT) and activation energy (Ea) were derived from TGA and DTGA curve tabulated in Table 5. Activation energy of the particulate composites was measured as per Broido’s method. Thermal stability of composites was found in an order of NCPA > NCP > NCPD > NCD > NCT according to their Ea. Composites of commercial epoxy resin based composites have lower thermal resistance than composites of β-naphthol based trifunctional epoxy resin.

Mass loss curves of (A) trifunctional resin and (B) comercial epoxy resin.
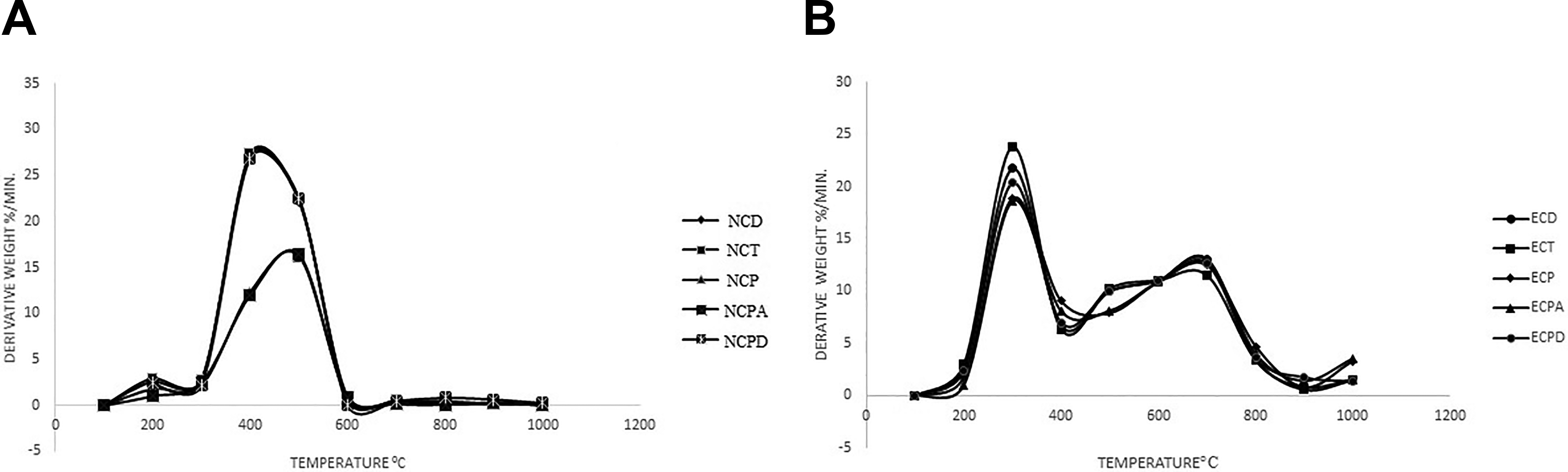
DTGA curves of (A) trifunctional resin and (B) comercial epoxy resin.
Thermal kinetics parameters of carbon based composites of β-naphthol based trifunctional epoxy resin and commercial epoxy resin.

Conclusion
In the present study, β-naphthol based trifunctional epoxy resin was synthesized and resin cured in presence of various curing agent used as a matrix material in carbon fiber reinforce composites. β-Naphthol based trifunctional epoxy resin confirmed by FT-IR and functionality of epoxy resin confirmed by molecular weight and epoxy equivalent weight. Properties of β-naphthol based trifunctional epoxy resin compared against commercial epoxy resin. Results of mechanical properties and thermogravimetric analysis shows that the composite can be used for high performance application. Chemical resistance also supports the bonding of matrix and fiber phase. In comparison with commercial epoxy resin the results shows β-naphthol based trifunctional epoxy resin having superior in all around properties.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
