Abstract
Vegetable oils are a promising class of bioresources for producing green polymeric materials to reduce the dependence on petro-based polymers. In this study, a green copolymer prepared by thermal copolymerization corn oil with sulphur at its molten state is reported for the first time. The proportions of sulphur to corn oil (w/w%) in the reaction mixture were varied in the range of 50/50 to 80/20 and the reactions were carried out at 170°C for 1 h. The obtained copolymers were characterized using Fourier transform infrared (FTIR), thermal gravimetric analysis (TGA), differential scanning calorimetry (DSC), scanning electron microscopy (SEM), and powder X-ray diffraction (PXRD). The percentage of the unsaturated fatty acid portion was found to act as a multifunctional monomer stabilizing polysulphide forming crosslinked structures that vary depending on reactant sulphur content. The obtained copolymers were found to be amorphous thermosets with heavily crosslinked structures and composite morphologies. The copolymers also showed high thermal stability under nitrogen atmosphere. The new copolymers are environmentally friendly hybrid material promoting green chemistry with a potential added value to abundantly available sulphur and corn oil.
Keywords
Introduction
The interest in using bio-resources for producing renewable polymeric materials is fast growing. This is not only to overcome fossil resource depletion concerns and meet new governmental policies but also to reduce the dependency on the petro-based polymers, which has been increased heavily over the past few decades leading to huge environmental disposal problems.1–3 Of all bio-resources, vegetable oils (triglycerides) are of particular significance for the development of bio-plastics of biodegradable nature.4–6 Thus, a number of vegetable oils have been used to prepare different classes of biopolymers including but not limited to epoxies, polyesters, polyethers, and polyurethanes, which have served in several applications such as medical devices, food processing, polymeric composites, insulators, plasticizers.6–10
Sulphur-containing polymers are a new class of hybrid materials that have been recently emerged after realizing that inert Sulphur present in an octet ring structure can act as a bi-radical monomer with eight sulphur atoms chain (thiyl radical) to initiate polymerization when heated to its molten state with a vinylic hydrocarbon source using a mechanism called inverse vulcanization.11–13 The latter is the opposite of vulcanization, which is the most famous and widely used process for crosslinking natural rubber medical gloves.11,13 The key features of inverse vulcanization are in its simplicity in direct polymerization of sulphur with vinylic monomers without the need for any additional initiators or organic solvents. The obtained copolymers have enhanced properties that can be used directly or served as intermediates for further functionalization leading to new polymeric materials for various applications.14–16 The production of new sulphur-containing polymers provides a new application to the huge amount of sulphur (>70 million Tonnes per year) present as a by-product recovered from the crude oil and natural gas refineries in forms of mega-tonne stockpiles accumulating across various open locations in the world refineries.13,17,18 Moreover, it can effectively contribute not only to the elimination of environmental problems and adding value to this cheap by-product but also to develop new engineering materials.6,19
Most of the sulphur-containing polymers were prepared by reacting sulphur with petro-based monomers such as 1,3-diisopropenylbenzene (DIB), 11 divinylbenzene (DVB), 20 1,4-diphenylbutadiyne, 14 vinylaniline, 21 methylmethacrylate, 22 and styrene. 23 Nonetheless, other renewable monomers such as farnesol and myrcene,15,17 limonene, 24 perillyl alcohol and squalene, 25 eugenol allylether, 18 and diallyl disulfide, 15 have been also investigated. However, such renewable monomers are rather expensive, and their reactions were not well established.
Vegetable oils draw interest for the preparation of sulphur-containing polymers by copolymerization with sulphur yielding green materials with desired properties. Particularly, vegetable oils such as sunflower, olive and linseed, 26 soybean, 27 and algae oil 28 were used for the preparation of sulphur-containing polymers. The presence of unsaturated fatty acids in the vegetable oils structures imparts them a monomer-like function for copolymerization with molten sulphur. The higher degree of unsaturation (C=C bonds) in the fatty acids, the more preferable is the oil for the production of green polymers as illustrated in Figure 1.5,8,29

Corn oil is one of the cheapest vegetable oils that is extracted from the germ of corn and widely produced in various parts of the world.32,33 It has a typical triglyceride structure having (C=C=C=C=C= per fatty acids molecules.6,19,34 The level of unsaturation in the corn oil can be as high as 84% giving it a strong potential for copolymerization with other monomers.35,36 Few vegetable oils such as sunflower, olive and linseed, 26 soybean, 27 and algae oil 28 were copolymerized with sulphur. However, copolymerization of corn oil with sulphur has not been reported to date.
The objective of the present study is to report the copolymerization of corn oil and elemental sulphur at its molten state using the inverse vulcanization method. The effect of variation of sulphur to oil proportions on the properties of the copolymers was investigated. The properties of the obtained copolymers were evaluated using Fourier transform infrared (FTIR), thermal gravimetric analysis (TGA), differential scanning calorimetry (DSC), scanning electron microscopy (SEM), and powder X-ray diffraction (PXRD). The amounts of reacted sulphur under different proportions in the reaction mixtures were elucidated.
Experimental
Materials
Elemental sulphur with an assay of 99.9% was purchased from PC Laboratory Reagent, Malaysia. Refined corn oil produced by Yee Lee Corporation Bhd (Malaysia) and sold with a brand named Vecorn® was used as received without any further treatment.
Copolymerization of corn oil with elemental sulphur
The elemental sulphur was placed in a glass vial and heated gradually until it melted and reached 170°C using a thermostatic oil bath. The selection of this temperature was based on previous studies suggesting that reacting vegetable oils with sulphur requires a relatively high temperature to achieve a visually single-phase copolymer product.26–28 The heating process was accompanied by the sulphur colour change from yellow at 120°C to orange at 160°C reflecting the initiation of the ring-opening process of cyclic-S8 elemental sulphur and formation of thiyl bifunctional radicals, respectively. The desired corn oil portion was subsequently added to the melted sulphur gradually under continuous stirring. The sulphur content in the reaction mixture was varied from 50 to 80 w/w% and the reaction was allowed at 170°C for 1 h. After the reaction, the glass vial was removed and held to cool down to room temperature to allow polymer extraction. Figure 2 shows a schematic representation for inverse vulcanization of elemental sulphur with linoleic acid as one of the main unsaturated fatty acid components in corn oil using the technique.

Schematic representation for inverse vulcanization of elemental sulphur with linoleic acid.
Characterization of sulphur-containing polymer
FTIR analysis was carried out using PerkinElmer’s Frontier model spectrometer equipped with attenuated total reflectance (ATR) in a frequency range of 4000–500 cm−1 with 8 scans at a resolution of 4 cm−1. TGA measurements were performed using a PerkinElmer STA 6000 simultaneous thermal analyser. The temperature of the samples was varied in the range of 30–900°C at a heating rate of 10°C/min under a nitrogen atmosphere. DSC heating runs were performed on a TA Instruments Q2000 thermal analyser with a heating rate of 10°C/min in temperatures ranging from −50 to 200°C under a nitrogen atmosphere. Powder X-ray Diffraction (PXRD) measurements were conducted using a Malvern PANalytical X’Pert powder diffractometer equipped with a Cu anode material (Kα1 = 1.540598 Å, Kα2 = 1.544426 Å) at 40 mA in transmission geometry using a capillary spinner. A Zeiss EVO LS 15 microscope was used to investigate the morphology of the samples after dipping in liquid nitrogen, fracturing and gold coating using Emitech K550X sputter coater. Freshly prepared polysulphide samples obtained by heating sulphur to 170°C, casting the melted sulphur in a petri dish and cooling to room temperature. The pure polysulphide was used as a reference in XRD, DSC and SEM measurements.
Results and discussion
Physical properties of poly(S/corn oil)
The reaction of corn oil with sulphur yielded brown coloured copolymers having apparent single phases. The colour of the copolymer became lighter with the increase in the initial sulphur content until 80 wt% as shown in Figure 3, which depicts photos of the prepared copolymers. As the sulphur concentration further increased to 90 wt%, a two-phase product started to appear. This justifies the use of 80 wt % as the upper limit for sulphur percentage in the reaction mixtures and allows having a single-phase structure in the obtained polymers. Similar brown colour was observed for copolymers obtained from reaction sulphur with sunflower, olive, and linseed oils unlike poly(S/algae oil), which showed black coloured copolymer for sulphur content in reaction mixture in the range of 50–90%.26,28,37
The obtained poly(S/corn oil) of different sulphur contents were tested for solubility in some solvents including toluene, chloroform, methanol, acetonitrile, and acetone without showing any degree of solubility. Thus, it was impossible to conduct solution-based tests such as proton nuclear magnetic resonance (1H NMR), light scattering and gel permeation chromatography (GPC).

Photos of copolymers prepared from inverse vulcanization of various proportions of S to corn oil (w/w%): (a) 50/50, (b) 60/40, (c) 70/30, (d) 80/20 and (e) 90/10.
Chemical structure of poly(S/corn oil)
Figure 4 shows FTIR spectra for the sulphur-containing polymers made by the reaction of sulphur and corn oil at various proportions. The copolymers demonstrated a similar trend to that of the pure unreacted corn oil with slight differences. For instance, pure corn oil showed a small peak at 1655 cm−1, which is due to vinylic C=C double bonds stretching vibration representing the unsaturated structure of the corn oil. This was coupled with the peak at 3009 cm−1 resembling the vinylic C–H stretching vibration in the corn oil. The peaks at 1655 and 3009 cm−1 almost vanished in all the spectra of the copolymers.26–28 A new peak could be seen for the copolymers at 797 cm−1 which represents the rocking vibrations of C–H bonds in the vicinity of C–S bonds confirming the formation of C–S bonds between sulphur and corn oil. 26 These results confirm the formation of sulphur-containing polymers and there is no visible peak regarding the presence of S–H bonds at around 2550 to 2600 cm−1 which indicates that no thiol groups are available inside the obtained polymers.26,27 These observations are going along previous studies reporting copolymers made by inverse vulcanization of sulphur with sunflower, olive and linseed, 26 soybean, 27 and algae oil. 28

FTIR spectra of: (a) corn oil and copolymers prepared from various proportions of S to corn oil (w/w%): (b) 50/50, (c) 60/40, (d) 70/30, and (e) 80/20.
Morphology of structure of poly(S/corn oil)
SEM images for the obtained sulphur-containing polymers in comparison with fresh polysulphide are presented in Figure 5. Fresh polysulphide demonstrates a uniform homogenous morphology showing stable sulphur polymerized chains without depolymerization back to elemental sulphur. The surface morphology of the copolymers begins to change showing rougher texture with the formation of composite-like structures, which started to have more densely distributed particles with the increase in the sulphur content in the reaction mixtures from 50 to 80 wt%. For instance, the copolymers from 50/50 and 60/40 S/corn oil showed homogenous sulphur particle distribution, which is denser in the latter composition. This suggests that sulphur did not go under complete conversion, i.e. some element sulphur remained trapped in the formed poly(S/corn oil) confirming the formation of composite structure. Unlikely, copolymers from 70/30 and 80/20 (S wt%/oil wt%) showed porous composite structures and this could possibly be due to the release of H2S gas associated with the higher sulphur contents. The evolved H2S gas was detected by blackening of the lower part of filter paper strip wetted with lead acetate solution. Similar trend was observed in similar sulphur-based polymers.26,38 The average pore size was found to decrease from 25 to 12 µm with the increase of S/oil (w/w%) proportions in the reaction mixture from 70/30 to 80/20, respectively. 37 This suggests that copolymers with high sulphur content have potential for application as adsorbents. Similar composite structures containing isolated sulphur particles that were increased in number by increasing the initial sulphur content were also reported for sulphur-containing copolymers made from sunflower, olive and linseed, 26 soybean, 27 and recycling cooking oil. 37

SEM images of: (a) fresh polysulphide and copolymers from various proportions of S to corn oil (w/w%): (b) 50/50, (c) 60/40, (d) 70/30, and (e) 80/20.
Thermal stability of poly(S/corn oil)
TGA thermograms and derivative thermogravimetric (DTG) curves of the copolymers made from elemental sulphur and corn oil are shown in Figure 6 and Figure 7, respectively. TGA thermogram of elemental sulphur shows a single-degradation step with a decomposition temperature of 327°C as revealed by the corresponding DTG curve. On the other hand, all TGA thermograms of the copolymers with different sulphur contents display a two-step degradation pattern starting by the weight loss taking place due to the decomposition of polysulphide components followed by the weight loss caused by the thermal decomposition of the polymerized corn oil fraction of the copolymer. The first weight loss increases with the rise in the sulphur content in the reaction mixture and the decomposition temperature of sulphur in the copolymer becomes lower than elemental sulphur. The sulphur decomposition temperature starts to increase from 253 to 263°C with the increase in sulphur portion from 50 to 80 wt%. The two-step thermal degradation behaviour of the sulphur-containing copolymers is in line with the literature.14,18,26,39 Coincidently, the weight loss and the thermal decomposition temperatures of the polymerized corn oil in the copolymers decrease with the increase in the sulphur portion in the reaction mixture. For instance, this temperature decreases from 385 to 371°C when sulphur increases from 50 to 80 wt%. Similar observations were also reported for thermal degradation behaviour of sulphur-based polymers that originated from sulphur with canola and soybean oils.27,37 It is noteworthy mentioning that S-corn oil copolymers did not undergo completed volatilization at the end of heating runs. Instead, residues of S and C of different weights were left. The source of S is most likely resulted from the unreacted sulphur embedded inside the copolymers. Thus, it can be concluded that the volatilization of sulphur from S/corn oil copolymers takes place at temperatures lower than the volatilization temperature of elemental sulphur, i.e. the thermal degradation of S/corn oil copolymers takes place at faster rates than elemental sulphur confirming the formation of polymeric structures, which have high thermal stability.

TGA thermograms of: (a) elemental sulphur and copolymers prepared from various proportions of S to corn oil (w/w%): (b) 50/50, (c) 60/40, (d) 70/30, and (e) 80/20.

DTG curves for: (a) elemental sulphur and copolymers prepared from various proportions of S to corn oil (w/w%): (b) 50/50, (c) 60/40, (d) 70/30, and (e) 80/20.
To further illustrate the thermal stability behaviour of S/corn oil copolymers, TGA data were also used to determine the exact percentage of sulphur reacted with corn oil to form the copolymer. Table 1 presents the variation of sulphur contents in copolymers, unreacted sulphur fractions and their ratios with different proportions of sulphur and corn oil in the reaction mixtures. As can be seen, the temperatures of the complete sulphur volatilization increase with the presence of more sulphur in the reaction mixture. A similar increasing trend was observed for the fractions of the reacted sulphur and ratios of reacted sulphur to unreacted sulphur. These results confirm that the sulphur does not go undergo a complete reaction (conversion) and a maximum of about 11 wt% or less remains embedded in the formed copolymers causing the formation of ash residues with carbon at the end of TGA heating runs.
Variation of sulphur content in copolymers with the content of sulphur and corn oil in the reaction mixture.

Thermal properties of poly(S/corn oil)
DSC was used to study the thermal behaviour of the obtained S/corn oil copolymers. Two samples with S wt%/corn oil wt% of 70/30 and 80/20 were chosen for DSC analysis due to their higher sulphur conversion. Figure 8 shows the DSC thermograms of these two copolymers in comparison with freshly prepared pure polysulphide in temperatures ranging from −50 to 200°C. Interestingly, no melting and glass transition temperatures peaks of poly(S/corn oil) could be observed in the investigated temperature range. The absence of glass transition temperature was also reported for few copolymers obtained from reaction of sulphur with type of poly(S/oil) with sulphur contents equal to or higher than 70 wt%. The absence of the former indicates that these copolymers are amorphous in nature, whereas the absence of the latter suggests that they are rather heavily crosslinked thermosets. Instead, three sulphur-representing endothermic peaks with various intensities could be observed in all thermograms in the range 102 to 121°C. The pure polysulphide shows two minor peaks at 102 and 105°C and a strong peak at 117°C. This is different from elemental sulphur, which was reported to have two peaks at 109 and 119°C corresponding to the melting transitions from the orthorhombic and monoclinic phases, respectively.40,41 The thermograms of a copolymer from 70/30 S/oil (w/w%) demonstrated three peaks at 108, 115, and 121°C compared to the three peaks at 109, 114, and 120°C that were also observed for 80/20 S/oil (w/w%) copolymer suggesting the presence of sulphur in three different crystalline phases varying in melting temperatures. This trend could be due to the presence of some unreacted elemental sulphur that was left in the copolymer and depolymerization of some sulphur chains to fragments of different crystallinity. Similar observations were made in the literature for sulphur-containing polymers made from canola oil and soybean oil,27,37 The slight temperature shifts in the melting peaks compared to those of elemental sulphur and polysulphide prove the formation of sulphur-containing polymeric structures.16,17,37 The variation in the sulphur melting peak area in the copolymers reflects the amount of unreacted sulphur left embedded in the obtained copolymers.16,17,27,37 It can be concluded that the obtained copolymer is a thermoset with an amorphous heavily crosslinked structure.
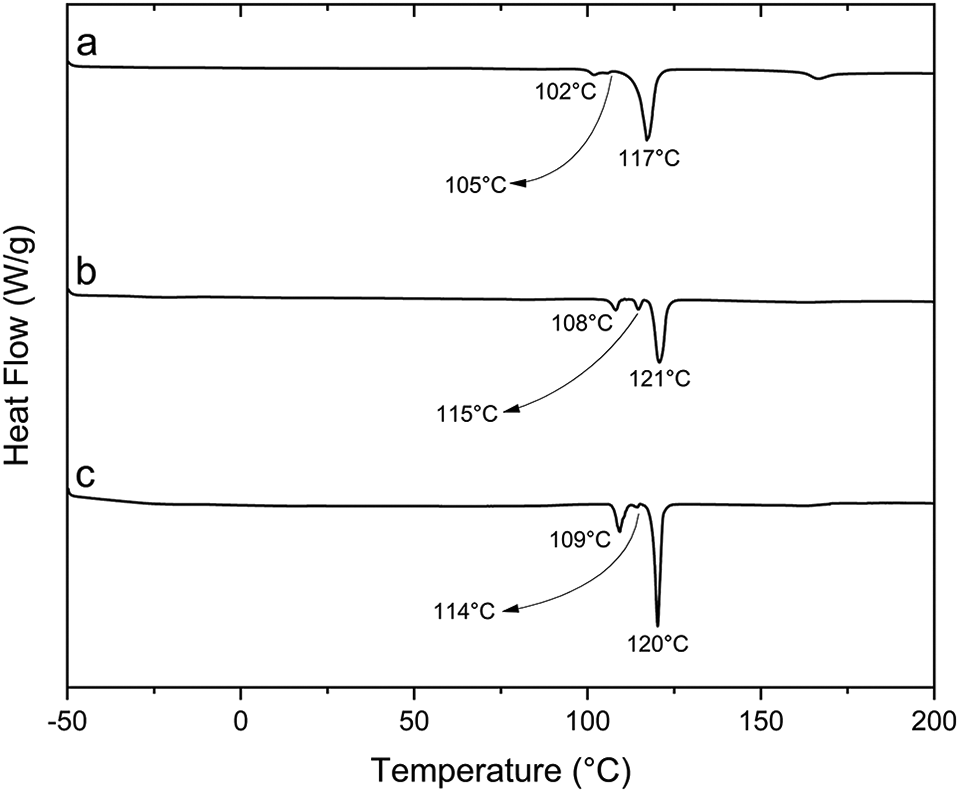
DSC thermograms: (a) fresh polysulphide and copolymers from S/corn oil proportions (w/w%): (b) 70/30 and (c) 80/20.
Structural properties of poly(S/corn oil)
Figure 9 displays the PXRD diffractograms for S/corn oil copolymers together with freshly prepared polysulphide. All the obtained S/corn oil copolymers show PXRD patterns like freshly prepared polysulphide. These patterns are also identical to that of elemental sulphur reported in the literature.42–44 This indicates that the crystalline reflections visible in the PXRD patterns are not only due to the unreacted elemental sulphur and the sulphur originated from depolymerized polysulphide but also the apparent crystallinity depends on the sulphur content in the reaction mixtures. Therefore, the obtained sulphur-containing polymers demonstrate a fully amorphous structure as reported for similar materials in the literature.20,25,45 It can be further suggested that corn oil in all percent compositions is unable to stabilize the whole sulphur content.17,45
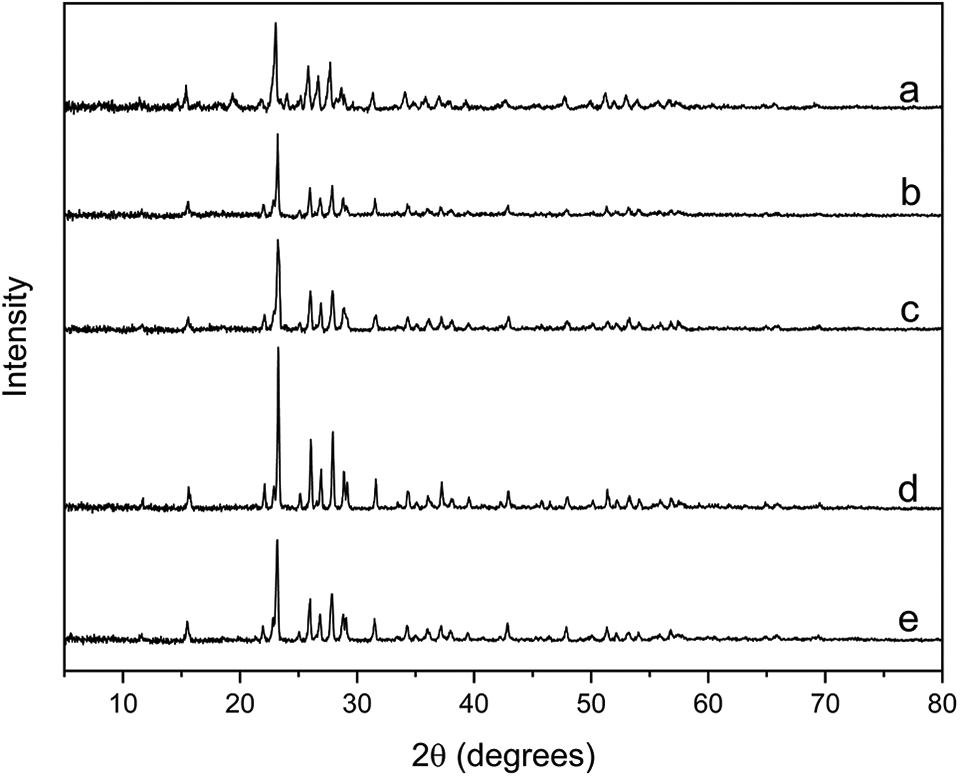
PXRD patterns for: (a) fresh polysulphide and copolymers from various proportions of S to corn oil (w/w%): (b) 50/50, (c) 60/40, (d) 70/30, and (e) 80/20.
Comparison between present copolymer and similar copolymers in previous studies
To understand the similarities and differences between the copolymers obtained by reaction of some vegetables oils and sulphur by inverse vulcanization and the copolymer obtained in the present study, a comparison is made between the properties of sulphur containing copolymers reported in previous studies and the present work as summarized in Table 2.
Summary of properties of sulphur-containing polymers obtained from reaction of various types of vegetable oils and elemental sulphur by inverse vulcanization.

All the copolymers containing sulphur are brown in colour and the degree of darkness increase with the initial sulphur content and colour of initial oil. For instance, most of investigated oils are yellowish in colour unlike algae oil which is dark greenish that led to a copolymer with black to dark brown colour with higher sulphur content. All copolymer showed visibly homogenous phase below 90 wt% initial sulphur. The present copolymer was found insoluble in conventional solvents such as such as toluene, chloroform, methanol, acetonitrile, hexane and acetone. Solvents such as n-heptane, deuterated chloroform, THF and CHCl3 were able to partially dissolve copolymers from sunflower, canola, soybean and algae oil. The latter was fully soluble in THF and CHCl3. 28
All copolymers including the one from the present study showed a composite morphology above 50 wt% initial sulphur where some elemental sulphur remained unreacted and got embedded in the homogenous copolymer structure. The amount of trapped sulphur particles depends on the initial sulphur content in the reaction as discussed earlier. However, the amount of unreacted sulphur seem to be less affected by the type of oil. For instance, poly(S/corn oil) with 50 wt% reacting sulphur showed ∼10% of unreacted sulphur in the structure compared to 9 and 9.6% for to poly(S/canola oil) and poly(S/soybean oil), respectively.
The thermal stability of the present poly(S/corn oil) showed two-step decomposition pattern similar to poly(S/canola oil) and poly(S/algae oil) unlike copolymers made from olive, linseed, and sunflower oils, all of which demonstrated three-step degradation pattern but similar weight loss trends.26,28,37 The decomposition of polysulphide in the copolymers from corn oil, sunflower oil, algae oil started at about 200°C, which increased to 230°C for canola oil followed by decomposition of the organic components in the copolymers beyond 300°C.
The copolymers based on corn oil, sunflower, olive, linseed and soybean oil showed no Tg within the range of the investigated temperature whereas the corresponding copolymers based canola and algae oils showed Tg at 12.2 and 32°C, respectively.26,28,37 On the other hand, the melting behaviour detected by DSC revealed that there are three melting peaks for all the copolymers resembling the amount of unreacted sulphur trapped inside the copolymers from corn oil, linseed, olive, sunflower, canola and algae oils, which can be extracted by solvents such as n-heptane.26,28,37 The copolymers seem to be amorphous rubbery like materials with crosslinked structure which are likely to be more dense with alga oil, which have multiple double bonds compared to two double bonds in lenoleic acid present in other types of oils.
The XRD patterns for copolymer in this study at different initial sulphur contents are similar to other copolymers based on vegetable oils, which are in turn similar to the patterns reported for the elemental sulphur. On contrary, poly(S/algae oil) showed no peaks for polymers with sulphur contents in the range of 50–70% proving that algae oil has an advantage in stabilizing polysulphide in the polymeric structures at this range of sulphur content.27,28 It can be concluded that the copolymers from corn oil and other reviewed vegetable oils are in forms of brownish amorphous thermoset with composite and heavily crosslinked structures.
Conclusions
Corn oil was successfully copolymerized with elemental sulphur at 170°C using inverse vulcanization technique. The reaction conditions were chosen based on initial monitoring of the parameters (time, temperature, and sulphur content) allowing having visually single-phase copolymer of brownish colour. The properties of the obtained copolymers were evaluated using FTIR, SEM, TGA, DSC, and PXRD. The formation of S/corn oil copolymer was proven by the presence of a peak at 797 cm−1 representing the rocking vibrations of C–H bonds in the vicinity of C–S bonds between sulphur and corn oil as revealed by FTIR analysis. SEM results showed some sulphur particles remained trapped in the formed copolymers from various S/corn oil proportions imparting them composite structures. TGA showed that S/corn oil copolymers were thermally stable to higher temperatures and decomposed in a two-stage pattern faster than elemental sulphur further confirming the formation of polymeric structures. The S/corn oil copolymers were found to be thermosets with amorphous heavily crosslinked structures as suggested by DSC and PXRD. Moreover, the whole added sulphur did not undergo complete reaction leaving some unreacted sulphur and the sulphur originated from depolymerized polysulphide (about 10 wt%) left embedded in the copolymer matrices further confirming the formation of visibly homogenous composite structures. It can be concluded that corn oil can be effectively used as a multiple diene-containing monomer to greatly stabilize the polysulphide leading to the formation of new sulphur-containing copolymers with green nature and interesting properties. More research is needed to further establish the properties of these copolymers and explore their best applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Yayasan Universiti Teknologi PETRONAS (Y-UTP) through cost centre no. 015LC0-065.
