Abstract
In the present work, barium ferrite in constant loading was dosed to the rubber matrices based on NR, SBR and NBR. Sulfur, peroxide and mixed sulfur and peroxide curing systems were applied for cross-linking of rubber magnetic composites. The application of sulfur or peroxide curing system leads to the formation of different types of linkages between rubber chain segments. As the structure of the formed cross-links plays a significant role in determining the final properties of rubber articles, the main aim of the work was to use the combination of curing systems in order to suppress the disadvantages of both systems and possibly to highlight their benefits. The results showed that composition of curing system has considerable influence on cross-link density of composites, which was subsequently reflected in typical change of physical-mechanical properties and glass transition temperature. The tensile strength was improved with increasing amount of peroxide curing system. The reason can be attributed to the presence of co-agent zinc methacrylate, which exhibits strong adhesion to magnetic filler and thus it contributes to the improvement of compatibility and homogeneity on the interphase filler–rubber. On the hand, there was observed no influence of curing system composition or type of rubber matrix on magnetic characteristics of composites.
Introduction
Rubber magnetic composites are classified as a group of smart materials, which can be prepared by incorporation of magnetic fillers into rubber matrices. The final properties of composites are strongly dependent on the characteristics of rubber matrix. However, by application of magnetic materials into rubber compounds by processing technologies generally used in rubber industry, new properties and technological abilities can be provided. The advantage of such type of materials is that their properties can be modified for the requirements of specific applications. Rubber magnets combine a unique pattern of suitable magnetic and elastic properties; they are characterized by easy adaptability, excellent flexibility and corrosion resistance. They are able to sensitively respond to vibrations and absorb oscillations of various wavelengths. They can be coiled, curved and shaped with the retention of their magnetic properties. Rubber magnetic composites have already been extensively used as sensors of magnetic and electromagnetic fields, in magnetic imaging, new types of recording media, but also in microwave and radar technology, biotechnology and others.1,2
The optimal elastic and tensile properties of engineering rubber materials are achieved during the process of vulcanization, simply termed as curing. Vulcanization occurs by chemical agents, such as sulfur or peroxides. The sulfur vulcanization is the oldest and the most frequent curing method used for cross-linking of unsaturated rubbers. In order to optimize the curing process, sulfur is always used in combination with activators and accelerators, sometimes also with other additives. 3 During the sulfur vulcanization, different types of sulfidic cross-links between elastomer chains (monososulfidic C–S–C, disulfidic C–S2–C and polysulfidic cross-links C–Sx–C, where x can vary from 3 to 6), are formed.4,5
Peroxides can be used to vulcanize both, unsaturated and saturated rubbers. 6 Cross-linking of elastomers with organic peroxides is radical process, which results in formation of carbon-carbon bonds between macromolecular chains. The main benefit of C–C bonds is their high thermal stability; therefore peroxide cured vulcanizates exhibit high-temperature aging resistance and low compression set at elevated temperatures. Simple formulation of rubber compounding, good electrical properties of vulcanizates, good shelf life stability and no staining of the finished parts belong to other advantages of peroxide vulcanization.7,8 However, there are also some drawbacks when compared to sulfur cured systems, as low scorch safety, worse tensile and tear strength, lower elastic and dynamic properties of vulcanizates. 9
Cross-linking of elastomers with peroxides can be effectively improved by using of co-agents.10,11 Co-agents are multifunctional low molecular weight organic molecules with high reactivity toward free radicals. They are used to increase the cross-linking efficiency of the vulcanization process, but to increase the cross-link density of final vulcanizates as well.12,13 As a result, physical properties and treatability of peroxide-cured elastomers are improved.
When combining vulcanization systems, both sulfidic and carbon-carbon bonds are formed between rubber chains and this gives the real presumption to influence the properties of the final materials in order to suppress the drawbacks of particular vulcanization systems and to highlight their benefits.
A lot of scientific works have been devoted to the topics related to the rubber magnetic composites, in which the content and the type of magnetic fillers were usually changed. The results showed that the incorporation of magnetic fillers leads to the increase of magnetic characteristics of the prepared composites, but the physical-mechanical properties were usually deteriorated.14–16 The reason can be attributed to weak adhesion and compatibility between the rubber and the filler. Therefore, in the present work, not only sulfur curing system, but also peroxide curing system and combined sulfur and peroxide curing systems were used for cross-linking of rubber matrices. The content of magnetic filler was kept constant in all composites and the main focus was aimed at the investigation of the curing system composition on physical-mechanical, dynamic and magnetic properties of the composites. The effect of curing system composition on cross-link density and structure of the formed cross-links were under consideration in relation to the properties of rubber magnets.
Experimental
Materials
Natural rubber (SVR 3 L, Thuyuyen Trading Production Co. Ltd., Vietnam), Styrene-butadiene rubber (Kralex 1502, content of styrene 23.5%, Synthos, Kralupy, Czech Republic) and acrylonitrile-butadiene rubber NBR (SKN 3345, content of acrylonitrile 31–35%, Sibur International, Russia) were used as rubber matrices. Barium ferrite BaFe12O19 (Magnety, Světlá Hora, Czech Republic) was used as magnetic filler. The structural and magnetic characteristics of ferrite are listed in Table 1. Peroxide curing system consisted of dicumyl peroxide DCP (Merck Schuchardt OHG, Germany) as peroxide curing agent and methacrylic acid zinc salt ZDMA (zinc methacrylate, Sigma-Aldrich, USA) as co-agent. Sulfur curing system consisted of zinc oxide (Slovlak, Košeca, Slovakia) and stearic acid (Setuza, Ústí nad Labem, Czech Republic) as activators, N-cyclohexyl-2-benzothiazole sulfenamide CBS (Duslo, Šaľa, Slovakia) as accelerator and sulfur (Siarkopol, Tarnobrzeg, Poland) as curing agent.
Characteristics of barium ferrite.

Methods
Preparation and curing of rubber compounds
Compounding of rubber formulations was performed in the chamber of a laboratory equipment Brabender at 90°C and rotor speed of 50 rpm. The rubber and the filler were compounded in the first step for 9 min, while the curing system was added in the second step (4 min, 90°C). Finally, the rubber compounds were treated into thin sheets by using the two roll calender. Five types of rubber magnetic composites with different composition of vulcanization system were prepared. The first composite was prepared only by application of peroxide curing system and the last one only with sulfur curing system. In the rest three type rubber magnets, the mutual ratio of peroxide and sulfur system was equally changed, but the total amount of ingredients of vulcanization system was kept on constant level in all prepared composites. The amount of accelerator CBS in sulfur curing system and co-agent ZDMA in peroxide curing system were also proportionally changed with the change in vulcanization systems. The composites were specified according to content and ratio of peroxide and sulfur system. The composite cured only with peroxide system is designated as S0–P2 and the composite cured only with sulfur system is identified as S2–P0. The composite cured with equivalent ratio of peroxide and sulfur is designated as S1–P1. The content of barium ferrite was kept constant in all composites −50 phr. Table 2 summarizes the composition of composite materials and their designation. Phr stands for parts per hundred rubber.
Composition of composite materials in phr and their designation.
The curing process of the prepared rubber compounds was performed by using a hydraulic press Fontijne at 160°C based upon previously determined optimum curing time under a pressure of 15 MPa. The rubber compounds were cured into thin sheets with dimensions of 15 × 15 cm and thickness of 2 mm.
Determination of cross-link density
The cross-link density ν was determined based on equilibrium swelling of the composite samples in suitable solvent. Composites based on NR and SBR were immersed and swelled in non-polar xylene, while acetone as polar solvent was used for the same procedure for composites based on NBR. The weight of samples was measured every hour until the equilibrium swelling was reached and the weight of samples was kept constant. The experiments were performed at a room temperature and the swelling time was equal to 30 h. To calculate the cross-link density of composites, the well-known Flory-Rehner equation modified by Krause 17 was introduced.
Evaluation of physical-mechanical properties
Zwick Roell/Z 2.5 appliance was used to evaluate tensile properties of composite materials. The tests were performed in accordance with the valid technical standards and the cross-head speed of the measuring device was set up to 500 mm/min. Dumbbell-shaped test samples (width 6.4 mm, length 80 mm, thickness 2 mm) were used for measurements. The hardness was measured by using durometer and was expressed in Shore A.
Determination of dynamical-mechanical properties
Dynamical-mechanical performances of composites were obtained by using a dynamical-mechanical analyzer DMTA MkIII, fy Rheometric Scientific. The samples were analyzed in tensile mode at a frequency 10 Hz, amplitude of dynamic deformation 64 µm and static force 0.2 N in temperature range from −100°C to 100°C.
Determination of magnetic characteristics
Magnetic characteristics of composites were evaluated at a laboratory temperature and maximum coercivity of Hm = 750 kA/m. Magnetometer TVM−1 (Vúzort, Praha, Czech Republic) equipped with Weiss electromagnet was used. The basic principle of measurement is induction method of scanning of scattering magnetic flux Φ induced by magnetic vibrating sample. The samples for the determination of magnetic characteristics were of prism shape (8 × 4 × 2 mm).
Microscopic analysis
The microstructure and surface morphology of composites were observed using the scanning electron microscope JEOL JSM-7500F at different accelerating strains. The samples were first cooled down in liquid nitrogen and subsequently fractured into small pieces with surface area of 3 × 2 mm. Each surface was coated with a thin layer of gold and placed into the SEM.
Results and discussion
Cross-link density
Both, sulfur and peroxide curing systems exhibit different reaction mechanisms and lead to the formation of different types of chemical cross-links between rubber chain segments. Therefore there was real presumption that the change in composition of curing could influence the cross-link density and the structure of the formed cross-links within the rubber matrices. From Figure 1 it can be observed that the highest cross-link density ν exhibited composites based on SBR and NBR cured with peroxide curing system (S0–P2). The cross-linking degree of NBR based composite cured only with peroxide system was almost twofold higher when compared to equivalent composite cured with sulfur system (S2–P0). Much bigger difference was recorded in the case of composites based on SBR. As seen in Figure 1, the cross-link density of the composite S0–P2 was more than fourfold higher than that of the composite S2–P0. The cross-link density of the composite based on NR cured in the presence of peroxide system (S0–P2) was much lower in comparison with corresponding composites based on SBR and NBR and roughly comparable with NR based composite cured with sulfur system (S2–P0).

Influence of curing system composition on cross-link density ν of composites.
The differences in cross-link densities of composites marked as S0–P2 relate to reaction mechanisms or rubber matrices with peroxide curing systems. During the curing process with organic peroxide, thermal dissociation of organic peroxide at vulcanization temperature first leads to the formation of primary radical fragments, which can be subsequently fragmented into secondary radicals. Thermal decomposition of the used dicumyl peroxide yields primary cumyloxy radicals, which can be readily dissociated into secondary methyl radicals while releasing acetophenone as by product.18,19 Peroxide derived radical fragments subsequently react with rubber chains to form carbon-carbon cross-links.20–22 There have been suggested two reaction mechanisms of peroxide free radicals with rubber chains. First mechanism involves abstraction of allylic hydrogens from rubber chains. Second one considers addition of peroxide radicals onto double bonds situated in rubber chains segments. Eventually, both reaction mechanisms may proceed concurrently.9,20 During peroxide curing of natural rubber, peroxide derived radicals abstract allylic hydrogens from rubber chains. The formed macroradicals subsequently mutually recombine to form carbon-carbon cross-links. 23 The addition reaction are of minor importance, arguably due to a sterically hindered the in-chain double bonds by methyl substituents, and of course due to quantum of easily abstractable allylic hydrogens in the structure of NR. The relative efficiency of peroxide cross-linking of NR is thus relatively low.21,24 This was also confirmed from experimentally determined cross-link density.
The amount of addition reactions is much more pronounced during peroxide curing of butadiene type rubbers, such as BR and SBR. Peroxide radicals can abstract allylic hydrogens from rubber chains or they can add onto the double bonds in the structure of elastomers. The elastomer radicals thus formed can participate in addition reactions onto the double bonds in rubber chains in addition to mutual recombination reactions.25,26 The chain character of addition reactions results in high cross-linking efficiency, i.e. 10–50 in dependence on the microstructure of polybutadiene. 7
The cross-linking efficiency of acrylonitrile-butadiene rubber was reported to be lower in comparison with BR and SBR. 26 The prevailing mechanism for peroxide cross-linking of NBR was suggested to be hydrogen abstraction. The addition reactions are much less pronounced due to the electron withdrawing acrylonitrile structural units making double bonds in NBR less reactive. 27 But some authors also suggest that addition reactions should also be under consideration.27,28 Is should be also remarked that NBR contains 33% of acrylonitrile groups, and thus the amount of butadiene units in its structure is lower in comparison with SBR, which contains 23.5% of styrene units. Relative efficiency of peroxide cross-linking of NBR is thus lower when compared to SBR.
It becomes apparent that the experimentally obtained results of cross-link density of composites cured only with peroxide system are in line with available literature sources. The highest cross-link density exhibited the composite based on SBR, while the lowest cross-linking degree was found to have the composite based on NR.
On the other hand, the differences in cross-link density of composites cured with sulfur system (S2–P0) are relatively low. The chemistry of accelerated sulfur vulcanization is very complex and still not fully understood. Sequences of chemical reactions run during the process, which may have substitutive, addition or even elimination character. Not only initial components of curing systems, but also products of their conversion can participate in these reactions. Besides this, functional groups in diene type elastomers can react with sulfur cross-linking systems with different speed and different mechanisms to form various intermediate products or final products. Side reactions as isomerization, dehydrogenation, cyclization, formation of diene and triene structures in rubber chains take also significant part in the process. In generally it is supposed that sulfur vulcanization of diene rubbers proceeds in three stages. In the first stage, the interaction of components of curing system leads to the formation of transition complexes, which together with rubber form active cross-linking agent. The second stage is characterized by formation of primary vulcanizate network with dominance of polysulfidic cross-links. During the third stage, this network is restructured in consequence of modification of cross-links (polysulfidic cross-links are transferred into di- and monosulfidic cross-links) and macromolecules of rubber (isomerization, dehydrogenation, cyclization) and the final spatial network of vulcanizate is formed.9,20,29,30 As already outlined, the functional groups of diene rubbers are double bonds and reactive allylic hydrogens. The highest amount of allylic hydrogens in the structure of NR might be a possible explanation for higher cross-link density of NR based composite cured with sulfur system (S2–P0) in comparison with the equivalent composite based on SBR. However, it does not explain the highest degree of cross-linking of the corresponding composite (S2–P0) based on NBR.
The lowest cross-link density were found to have composites cured with equivalent ratio of sulfur and peroxide (S1–P1). The possible explanation might be some mutual reactions of peroxide radicals with sulfur fragments, which could lead to their ineffective consumption. Thus the concentration of reactive sites would be then ineffective for cross-links formation. The contribution of the co-agent zinc methacrylate in cross-linking process of rubber magnetic composites is in more detail described in the following section.
Physical-mechanical properties
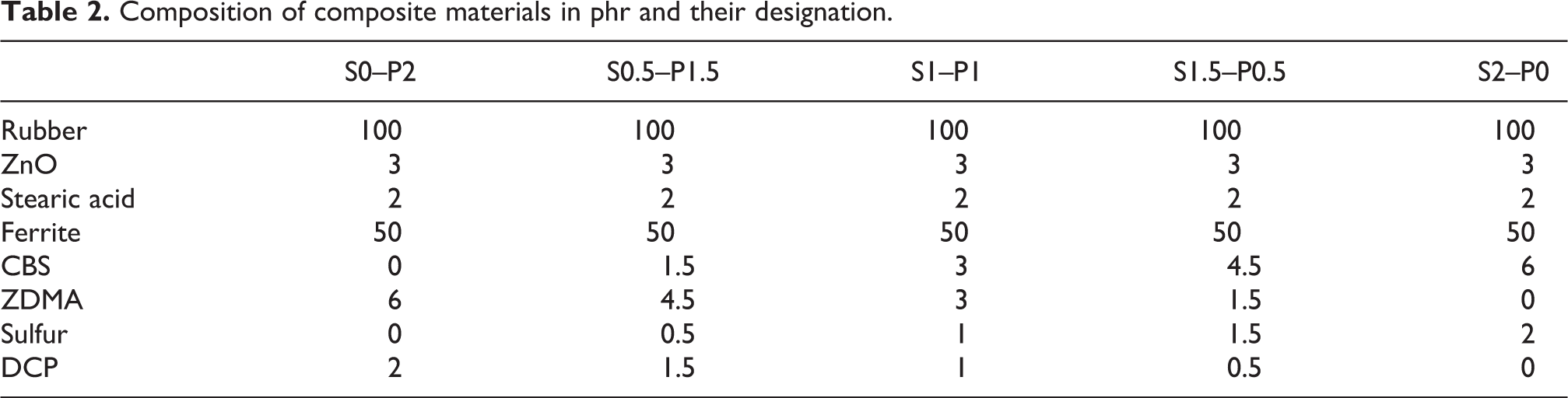
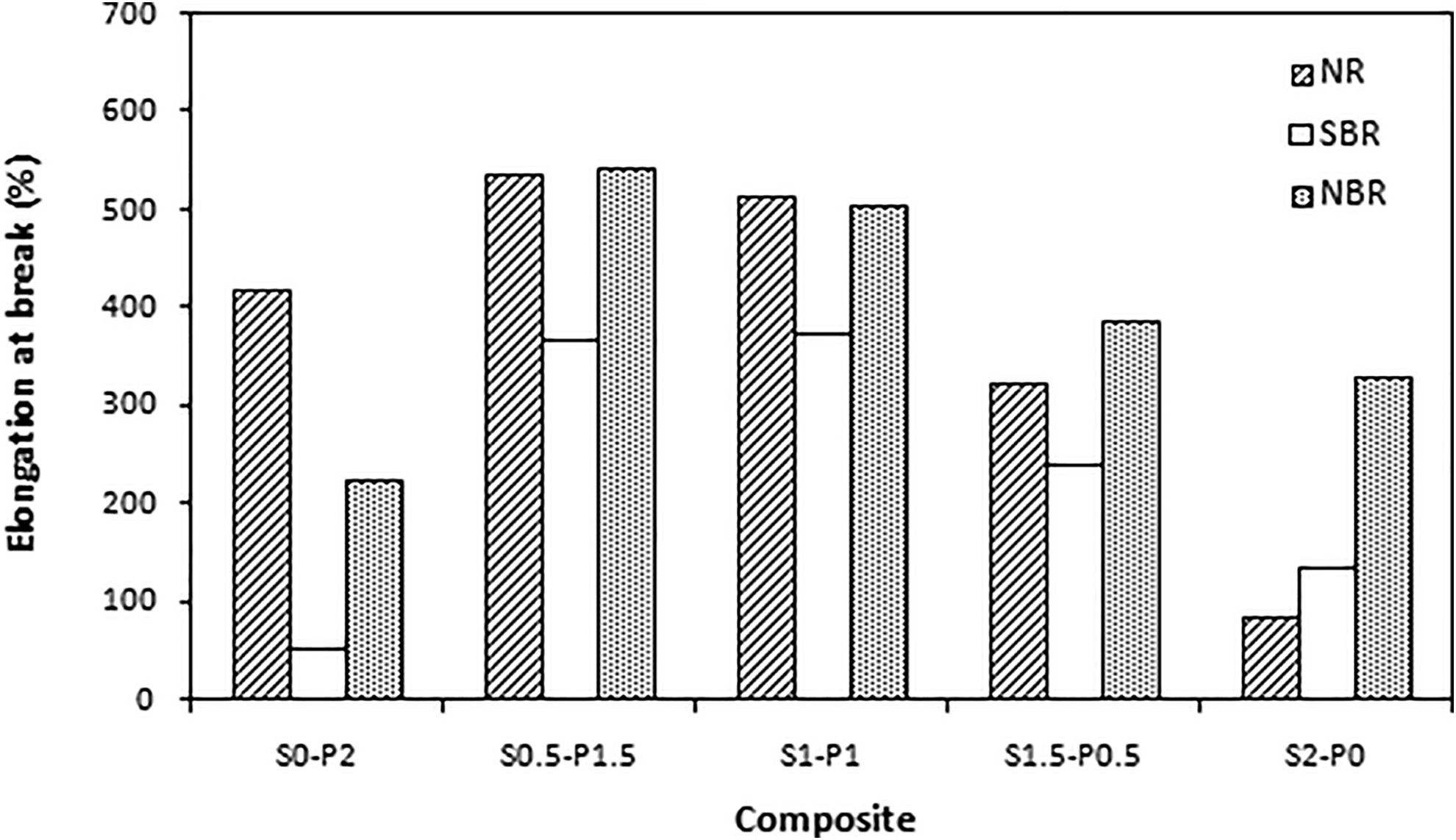
The dependences of physical-mechanical properties of tested composites on curing system composition are presented in Figures 2 to 5. It becomes apparent from Figure 2 and 3 that the highest modulus M100 and hardness exhibited composites cured only with peroxide system (S0–P2) with the highest cross-link density. By contrast, the lowest values of both characteristics were found to have composites cured with equivalent ratio of sulfur and peroxide (S1–P1) with the lowest cross-linking degree. The modulus M100 was not determined for the composite based on SBR cured with peroxide system (S0–P2) as well as for the composite based on NR cured with sulfur system (S2–P0), because the samples of these composites were ruptured before reaching 100% elongation. On the other hand, composites cured with equivalent sulfur to peroxide ratio (S1–P1) showed the highest elongation at break (Figure 4). The increasing degree of cross-linking cause the restriction of elastomer chains mobility and elasticity. As a result the elongation at break tends to decrease. From Figure 5 it can be seen that the tensile strength showed increasing trend with increasing amount of peroxide, or decreasing amount of sulfur in the curing systems applied. The highest tensile strength exhibited composites based on all three type rubbers cured with 0.5 phr of sulfur and 1.5 phr of peroxide (S0.5–P1.5). In generally, higher tensile characteristics exhibit sulfur cured vulcanizates, due to the nature of sulfidic cross-links formed within the rubber matrix. Sulfidic cross-links have lower dissociation energy and higher mobility when compared to carbon-carbon cross-links. When sulfur cured vulcanizates are stretched and deformed, sulfidic cross-links are disrupted earlier than carbon-carbon bonds. This leads to the formation of macroradicals, which may have enough time to form the new cross-links in microregions with lower stress. Such cross-link redistribution results in a less stressed, stronger network, in which the stress is uniformly distributed onto higher amount of elastomer chains. On the other hand, shorter and more rigid carbon-carbon linkages restrict mobility and elasticity of rubber chains upon applied deformation forces. In addition, the formed cross-links cause increased deformation stiffness, because of less mobility of elastomer chains, which subsequently leads to the worsening of mechanical properties.9,31 As shown, the achieved results are against the outlined theoretical predictions. The reason for the increase of tensile strength with increasing proportion of peroxide curing system can be attributed to the composition of peroxide curing system and the presence of zinc methacrylate as co-agent. It has been reported that the in situ radical polymerization of zinc methacrylate takes place during the curing process of rubber compounds with organic peroxides.32–34 Polymerized molecules of ZDMA tend to aggregate to form spherical nanoparticles, which can be physically adsorbed or chemically grafted onto rubber chains. Thus they contribute to the formation of physical and chemical couplings in the rubber matrix. Moreover, the formation of ionic clusters with the contribution of zinc ions from co-agent molecules has also been observed.26,35–37 It has also been demonstrated that zinc methacrylate exhibits a strong adhesion to polar materials.38,39 Ferrites are complex compounds of metal oxides. Due to the presence of metal and oxygen ions they belong to the polar materials.40,41 Zinc based co-agent increases the polarity of the rubber matrix by grafting onto polymer chains on one hand, and physically interact with particles of magnetic filler on the other hand. Based upon the outlined aspects it can be stated that by physical adsorption and chemical grafting of co-agent onto rubber chains and strong physical interactions with barium ferrite, ZDMA contributes to the improvement of adhesion and compatibility on the interphase filler–rubber. Subsequently, the overall improvement of physical-mechanical properties of rubber magnets was achieved. This seems to be the most likely explanation for the increase of tensile strength with increasing ratio of peroxide curing system. By contrast, as there are no additives in sulfur curing system needed for improvement of adhesion on the interphase filler–rubber, ferrite behaves as an inactive filler in sulfur cured composites. Thus, as seen in Figure 5, sulfur cured composites exhibited the lowest tensile strength.

Influence of curing system composition on modulus M100 of composites.

Influence of curing system composition on hardness of composites.
Influence of curing system composition on elongation at break of composites.
Influence of curing system composition on tensile strength of composites.
Microscopic analysis
SEM analysis confirmed the above outlined presumptions and revealed that the homogeneity and compatibility between ferrite and tested rubber matrices were the lowest in the case of composites cured only with sulfur system (S2–P0). From SEM images of sulfur cured composites (Figures 6(b), 7(b), 8(b)) are clearly seen aggregates or agglomerates of magnetic filler. There are also more evidently visible voids and microcavities on the interphase filler–rubber, which are evidently caused by poor adhesion between the two components. The increasing amount of peroxide system leads to the improvement of mutual compatibility between the rubber and the filler, while the most homogenous structure were found to have composites cured only with peroxide system (S0–P2) (Figures 6(a), 7(a), 8(a)). As already outlined, the reason can be attributed to the presence of zinc methacrylate, which exhibits strong adhesion to barium ferrite and contributes to the improvement of adhesion and compatibility on interphase filler–rubber. Surface morphology of composites also revealed that the adhesion between the rubber and the filler is higher in the case of composites based on NBR. This is arguably owing to the polarity of NBR. As barium ferrite and zinc methacrylate are polar materials, their affinity with polar rubber matrix is higher when compared to non-polar elastomers like NR and SBR.

SEM images of composite based on NR cured with peroxide system (S0–P2) (a) and composite based on NR cured with sulfur system (S2–P0) (b).

SEM images of composite based on SBR cured with peroxide system (S0–P2) (a) and composite based on SBR cured with sulfur system (S2–P0) (b).

SEM images of composite based on NBR cured with peroxide system (S0–P2) (a) and composite based on NBR cured with sulfur system (S2–P0) (b).
Dynamical-mechanical analysis
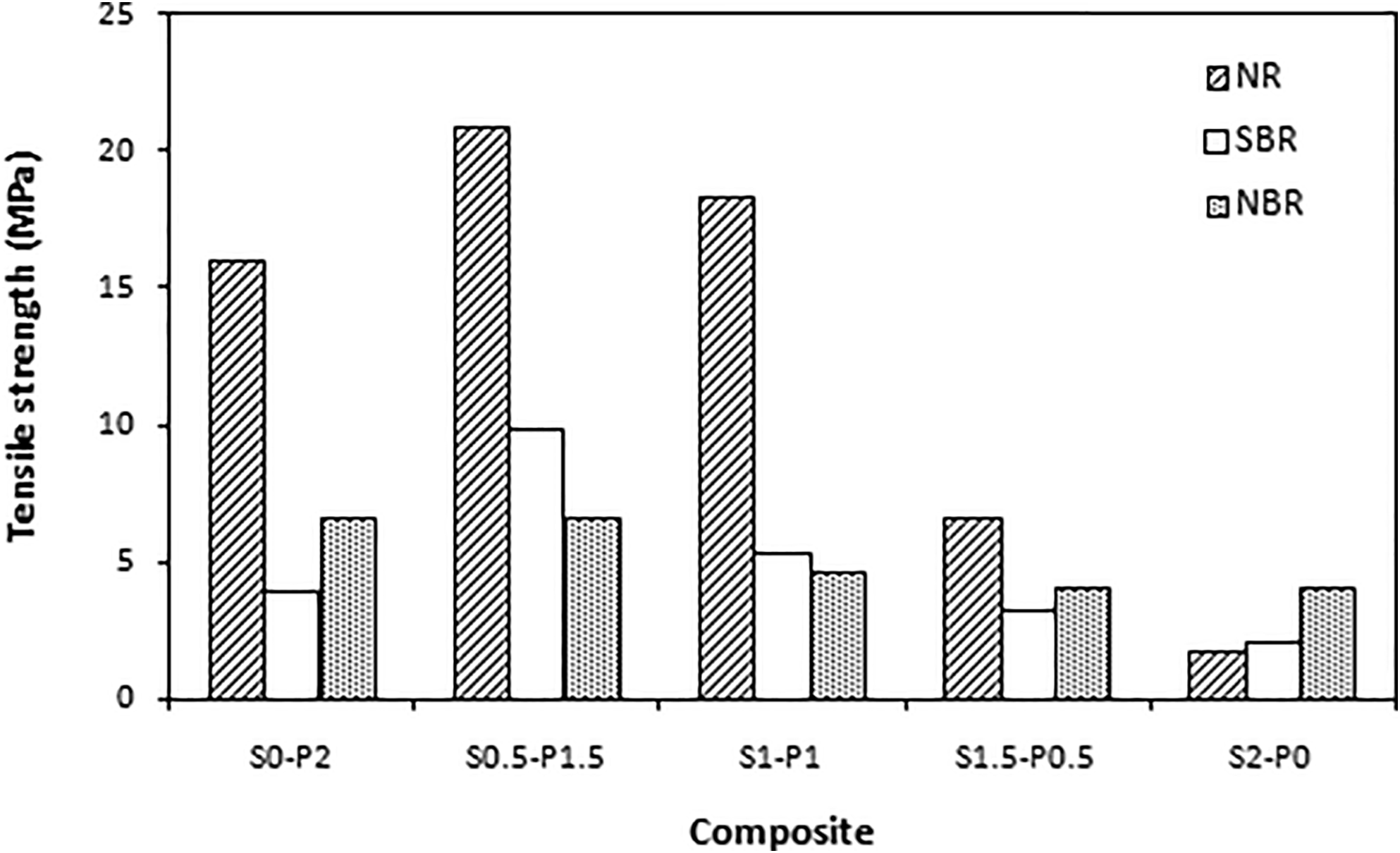
The dependences of tan δ for tested composites on curing system composition are graphically illustrated in Figures 9 to 11. The peak maxima correspond to the glass transition temperature Tg and it becomes evident that Tg of composites is dependent on their cross-link density as also possible to see in Tables 3 to 5. The lowest Tg were found to have composites cured with equivalent sulfur to peroxide ratio with the lowest cross-linking degree. The increasing degree of cross-linking leads to the restriction of rubber chains elasticity. As a consequence, the Tg shifted to higher temperatures. The biggest difference was possible to see in the case of composites based on SBR, where the difference between Tg of the composite cured only with peroxide system (S0–P2) and the composite cured with equivalent ratio of sulfur and peroxide (S1–P1) was almost 10°C (Table 4). These composites showed also the biggest difference in cross-link density. When considering the type of rubber matrix, the lowest Tg were found to have composites based on NR (Table 3), while the highest Tg exhibited composites based on NBR (Table 5). This aspect can be attributed to the microstructure of rubber matrices. Natural rubber has highly stereo-regular structure formed from 99.9% of ci-1.4-isoprene units. A as result, NR shows very good elastics properties even at low temperatures. The Tg is thus low. SBR and SBR are copolymer type rubbers, and besides elastic components—butadiene units, they also contain thermoplastics components—styrene structural units in SBR and acrylonitrile structural units in NBR. But while the content of styrene units in SBR is 23.5%, the amount of acrylonitrile units in NBR is 33%. It becomes clearly apparent that SBR has higher amount of elastic butadiene units and thus composites based on SBR show lower Tg when compared to equivalent composites based on NBR.
Influence of curing system composition on tan δ of composites based on NR.

Influence of curing system composition on tan δ of composites based on SBR.
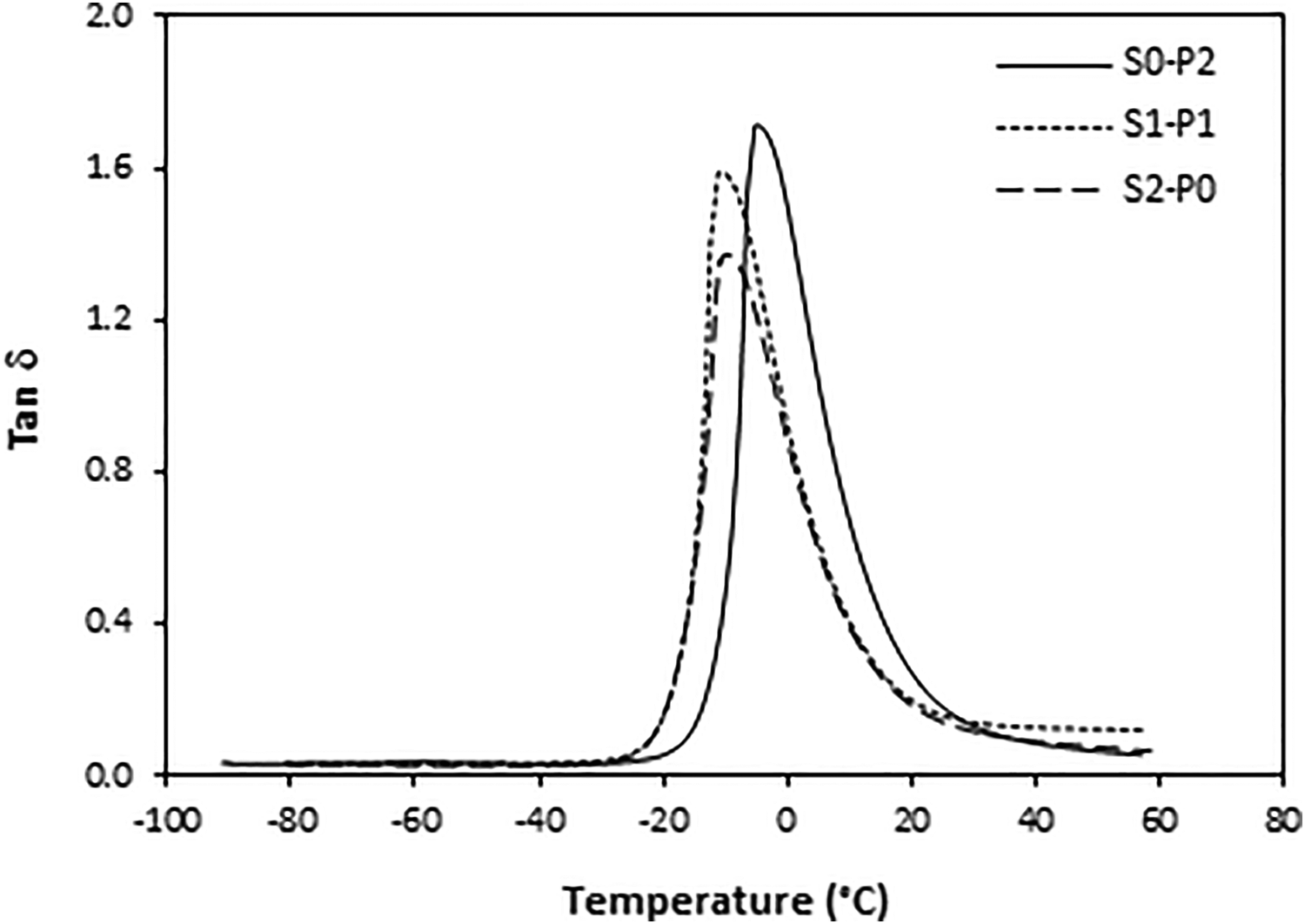
Influence of curing system composition on tan δ of composites based on NBR.
Dependence of glass transition temperature Tg on cross-link density ν of composites based on NR.

Dependence of glass transition temperature Tg on cross-link density ν of composites based on SBR.

Dependence of glass transition temperature Tg on cross-link density ν of composites based on NBR.

Magnetic characteristics
It has been described in many scientific works that the incorporation of magnetic fillers into rubber matrices imparts magnetic properties to the composites significantly.40,42,43 In the current study, the content of magnetic filler was kept on constant level and the goal was to investigate the influence of curing system composition on magnetic characteristics. The dependences of magnetic induction B on intensity of magnetic field H are called magnetizing curves, which provide complete information about technical properties of magnetic substances. The remanent magnetic induction Br, which represents the value of residual magnetism in the material when an external magnetic field is removed and the coercive intensity of magnetic field (coercivity, Hc), which represents the energy needed to abolish the remanent magnetic induction are the most important parameters of all permanents magnets. The higher values of both characteristics magnetic materials have, the better permanent magnets they are.
Looking at Figures 12 it can be stated that the values of remanent magnetic induction of tested composites were found to be dependent neither on the type of curing system composition, nor on the type of rubber matrix, as its values fluctuated only in the low range of experimental values. The coercivity of composites ranged between 101.5 and 103 kA/m, again, with no dependence on the type of rubber matrix or curing system composition. Thus it can be stated that magnetic characteristics of rubber composites are dependent only on the type and content of magnetic filler.

Influence of curing system composition on remanent magnetic induction Br of composites.
Conclusion
Three types of rubber matrices based on NR, SBR and NBR were filled with barium ferrite in constant level. Sulfur, peroxide and combined sulfur/peroxide curing systems were used for cross-linking of rubber matrices. The work was focused on investigation of curing system composition on cross-link density, morphology and properties of rubber magnetic composites.
The highest cross-link density were found to have composites cured only peroxide system, followed by the composites cured only with sulfur system. The lowest degree of cross-linking exhibited composites cured with equivalent ratio of sulfur and peroxide. The higher was the cross-link density, the higher was hardness and modulus. By contrast the lower was the elongation at break. The dependences of glass transition temperature of composites were also very closely connected with the cross-link density. The increasing degree of cross-linking leads to the restriction of rubber chains mobility, as a results of which the Tg increased. The tensile strength showed increasing tendency with increasing ratio of peroxide curing system, suggesting that zinc methyacrylate as co-agent in peroxide vulcanization contributes to the enhancement of adhesion and compatibility between the rubber and the filler on the filler–rubber interphase. Microscopic analysis confirmed the assumption. On the other side, magnetic characteristics were found to be influenced by neither by the composition of curing system nor by the type of rubber matrix.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Slovak Research and Development Agency under the contract No. APVV-16-0136 and APVV-19-0091.
