Abstract
The heat-resistant agent N-(4-carboxyphenyl) maleimide–alt–styrene (PCS) was used to modify polylactic acid (PLA) in order to improve the thermostability of PLA. The heat deflection temperature (HDT), thermodegradation, crystallization behavior and thermomechanical property were analyzed. The HDT of PLA added with PCS increased by about 10°C, and after heat treatment, the improvement of HDT was more significant. The thermal degradation behavior of PLA before and after modification hardly changed. The crystallinity (Xc), crystallization rate (1/t1/2) and crystallization temperature (Tc) of the composite material were significantly improved, but the crystalline structure did not change. The crystallinity of the composite material was twice that of pure PLA, the crystallization rate increased by about 30% and the crystallization temperature went up more than 10°C. The storage modulus (E’) of PLA was improved by PCS due to enhancing its crystallinity. The thermostability of PLA was significant improved after PCS is added.
Keywords
Introduction
Polylactic acid (PLA), a light yellow or transparent solid also known as polylactide, is a class of non-toxic, non-irritable, and biodegradable green polymer material synthesized from lactic acid. It is widely used because of its wide source of raw materials, good biocompatibility, and mechanical properties, as well as its environment-friendly attributes.1-7 With an increase in pressure on the environment caused by the refractory characteristics of plastics, the distinct biodegradability of PLA has gradually drawn attention. However, the poor heat deflection temperature (HDT) caused by its chemical structure severely restricts its development and limits its application.8-10 Accordingly, the prospect of improving the thermostability of PLA has prompted researchers’ interests.
Physical modification is a widely used method to enhance the thermostability of PLA because of its simple operation and convenience. It can be divided into organic filling modification and inorganic filling modification. A large number of literatures reported the research progress of heat-resistant modification of PLA by physical modification.
Masud Huda et al. 11 prepared a kind of glass fiber (GF) and regenerated newspaper cellulose fiber (RNCF) reinforced polylactic acid (PLA) composite material. The HDTs of pure PLA, PLA/GF (70/30), and PLA/RNCF (70/30) were 64.5°C, 73.9°C, and 80.2°C. The addition of the two fibers increased the HDT of PLA. Li Mengying et al. 12 investigated the effect of organic bentonite (OBT) and annealing on the heat resistance of PLA. The results showed that when the amount of OBT was 0.5%, the HDT could be as high as 109°C after annealing at 120°C for 10 min. Jing Liu et al. 13 used PLA as a matrix material and added polycarbonate (PC) and other additives to it. It was found that the HDT of the PLA/PC composites prepared by melt extrusion reached 108°C. Shi Qingfeng et al. 14 added natural bamboo fiber (BF) and talcum powder (talc) to PLA to prepare the composite material. The analysis results indicated that after heat treatment, the modified effect of the composite material was the best when the dosages of both BF and talc were 20%. Heat resistance temperature increased by nearly 40°C relative to that of pure PLA.
Although the methods used in these literatures can effectively improve the thermostability of polylactic acid, they have the disadvantages of too large addition amount, too large fiber size, the improvement effect is not obvious, etc. In order to overcome these shortcomings, this paper proposes a solution: using an organic polymer materials to blend with PLA to improve its thermostability. Organic polymer materials can be dispersed in PLA in a micro-nano scale, and the modified PLA can be used in products such as films. The organic polymer materials, N-(4-carboxyphenyl) maleimide–alt–styrene (PCS), we used, is a kind of heat-resistant agent which is multi-component copolymer composed of maleic anhydride, imide and other aromatic heterocycles. Its molecular chain has extremely strong rigid structure and high thermal stability.
A five-membered-ring imide is a typical heat-resistant rigid structure that can hinder the rotation of polymer chains and impede the movement of polymer chains. The heat resistance of polymer can be effectively enhanced by introducing an imide ring into the matrix material15-20. N-(4-carboxyphenyl) maleimide (CPMI) is an important new resin modified monomer, which can significantly increase the glass transition temperature (Tg) and HDT of polymers and improve the technological and mechanical properties of materials.21,22 Maleimide is an electron-withdrawing monomer, which can be polymerized with an electron-donating monomer vinyl monomer (such as styrene, methylstyrene, isobutylene, vinylpyridine, vinyl ether, etc.) via the free radical mechanism, electron transfer complex mechanism, or free-complexation mechanism.23-25 The heat-resistant agent N-(4-carboxyphenyl) maleimide–alt–styrene (PCS) was synthesized from CPMI and styrene by free radical copolymerization. Compared with CPMI, PCS exhibits better capability of modifying heat resistance and processability. 23
In the present study, PLA was modified by PCS. The effects of PCS on the heat resistance, crystallization behavior and thermomechanical property of PLA were evaluated by heat deflection testing (HDT), thermal gravimetric analyzer (TGA), differential scanning calorimetry (DSC), X-ray diffraction (XRD), polarized optical microscopy (POM) and dynamic thermomechanical analysis (DMA) to study the effect of PCS on the thermostability of PLA.
Experimental
Materials
The 4032 D-grade PLA used in this study, with 1.24 g/cm3 density and 1.40% of D-lactide content, was purchased from Nature Works LLC (Minnetonka, USA). The melting flow index was 4–8 g/10 min, the glass transition temperature was 57.8°C, and the melting temperature was 160°C–170°C.
The PCS used in this study was synthesized as described in a previous study. 24
Preparation of samples
PLA and PCS were dried in a vacuum drying oven (DZ-1A) at 60°C for 9 h. Subsequently, 10 wt% PCS was added into PLA, and the mixture was evenly stirred. PLA and PCS were melt-blended and extruded using a micro twin screw extruder (LHFD1-130718) at 170°C–210°C with a speed of 240 r/s. Water cooling and granulation were then conducted. The blends were injected into standard splines on an injection molding machine (CJ80M3 V) at 210°C. A portion of the samples was heated in a vacuum drying oven at 80°C for 24 h. The samples were labeled for ease in distinguishing them, as follows: heat-treated pure PLA, PLA-80; modified PLA without heat treatment, PLA/PCS; and heat-treated modified PLA, PLA/PCS-80.
Testing and analysis methods
The effect of PCS on the HDT of PLA was determined using an MTS ZWK1000 heat deflection temperature tester. The measurements were conducted in accordance with GB/T1634.1-2004, with a constant load of 0.45 MPa and a temperature rate of 120°C/h−1.
The crystallization behavior of the samples was examined by a TA Q20 differential scanning calorimetry system under nitrogen purge. About 2–4 mg sample was cut directly and then encapsulated in aluminum pans. The samples were heated to 210°C at 10°C/min, held for 3 min to eliminate previous thermal history, and cooled to 40°C at 2°C/min.
The formula for calculating the relative crystallinity of the material at time t is:
Where: t∞ is the end time of crystallization; Xt is the relative crystallinity, and Xt can be obtained from the exothermic peak area shown in the figure.
Where: To is the temperature at which crystallization begins and T is the temperature corresponding to time t; Φ is the cooling rate.
The influence of PCS on the crystalline structure of PLA was performed on a Rigaku (Ultima-IV) diffractometer equipped with a Cu Kα radiation source with a scan angle from 5° to 80° at a scan rate of 1°/min.
The evolution of the crystalline structure of the samples was observed via an Axio Scope A1 polarized optical microscope equipped with a digital camera. The samples were placed between two glass plates, melted at 210°C for 3 min to eliminate the thermal history on a Linkam THMS 600 hot stage, and quickly cooled to room temperature. Portions of the samples were heat treated in a vacuum drying oven at 80°C for 5 min after cooling to room temperature.
The dynamic mechanical property and thermal behavior of the samples were studied by a TA Q800 dynamic thermomechanical analyzer in a three-point bending mode at a frequency of 1 Hz. The size of the sample is 15 × 10 × 4 mm3.The temperature range is 30°C to 120°C and the heating rate is 2°C/min.
The thermal degradation behavior of the samples were detected by a NETZSCH TG 209 F1 thermal gravimetric analyzer under nitrogen purge. The samples were heated to 600°C at 10°C/min.
Results and discussion
Influence of PCS on the heat resistance of PLA
The effect of N-(4-carboxyphenyl) maleimide–alt–styrene (PCS) and heat treatment on the heat deflection temperature (HDT) of polylactic acid (PLA) is presented in Figure 1. After PCS was melt-blended with PLA at a certain proportion, the HDT of PLA/PCS was nearly 10°C higher than that of PLA—that is, the HDTs were 62.8°C and 53.3°C, respectively. After heat treatment at 80°C, the HDT of PLA/PCS-80 was about 15°C higher than that of PLA-80—that is, the HDTs were 118.7°C and 103.2°C, respectively. Compared with that of untreated pure PLA, the HDT of PLA/PCS-80 increased by about 65°C. To sum up, when PCS was melt-blended into PLA, the effect of PCS on the heat resistance of PLA was significant; meanwhile, after heat treatment, the effect became more significant. It is well known, the crystallinity of PLA will increase after heat treatment. So we suspected that the results of this experiment could be related to the crystallization of PLA—that is, the addition of PCS could affected the crystallinity or crystalline structure of PLA, considering that the effect of PCS on the heat resistance of PLA was more evident after heat treatment at 80°C.
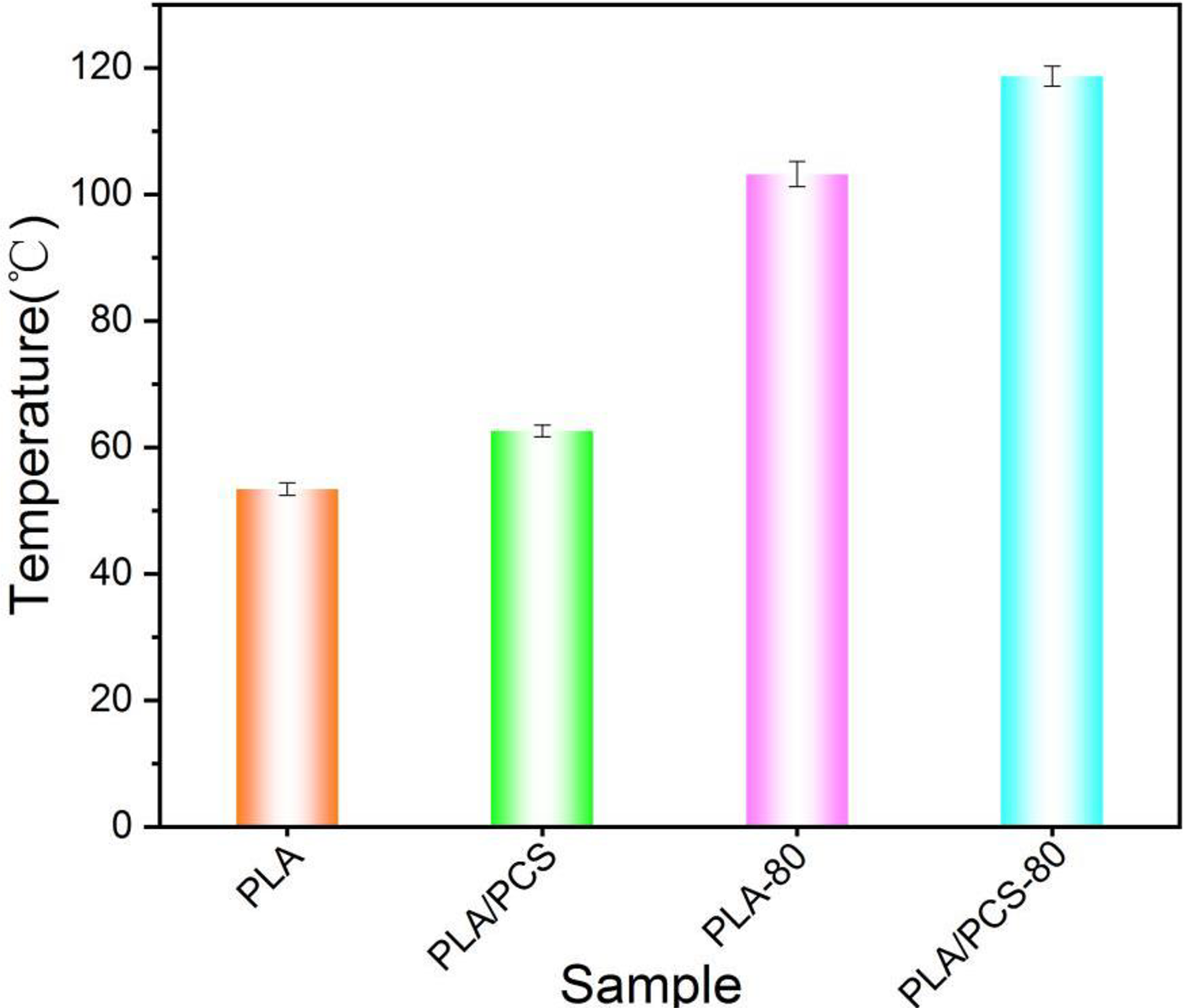
Heat deflection temperature of the samples.
Figure 2 shows TG and DTG curves of samples in nitrogen atmosphere. The initial degradation temperature (T5%), maximum thermal weight loss rate (WLRmax), corresponding temperature at maximum thermal weight loss rate (Tmax) and residual mass at 600°C (Residues at 600°C) are listed in Table 1. From Figure 2 and Table 1, it can be seen that the TG curve and DTG curve of PLA before and after adding PCS were basically coincident, and the results of thermogravimetric analysis were similar, indicating that PCS has little influence on the thermal degradation of PLA. The final residual mass of the samples added with PCS was larger than that of the samples without PCS, because the decomposition temperature of PCS was much higher than that of PLA and had not completely degrade at 600°C.

TG and DTG curves of samples.
T5%, Tmax, WLRmax, and WLRmax of the samples obtained by TGA.

Influence of PCS on the crystallization behavior of PLA
The DSC heating (at 10°C/min) and subsequent cooling (at 2°C/min) curves of the samples are presented in Figure 3. Some corresponding parameters—that is, melting temperature (Tm), melting enthalpy (ΔHm), crystallization temperature (Tc), and crystallinity (Xc)—are listed in Table 2. As reflected in the DSC curves recorded heated at 10°C/min, the samples without heat treatment displayed an obvious crystallization peak, whereas the samples subjected to heat treatment showed almost no crystallization peak. The reason is that the crystallinity of the samples after heat treatment at 80°C improved, whereas that of the samples without heat treatment was considerably low. The increases in the melting enthalpy of the samples after heat treatment shown in Table 2 was also due to this reason. At the same time, as shown in Table 2, the melting enthalpy of the material also increased after PCS was added, indicating that, similar to heat treatment, the addition of PCS could also expedite the crystallization of PLA. As reflected in the DSC curves recorded cooled at 2°C/min, the samples added with PCS showed an evident crystallization peak, whereas the crystallization peak of PLA without PCS was considerably low. The crystallinity of the PLA samples added with PCS was significantly improved, as determined by calculation, and was more than twice as high as that of the pure PLA samples. Simultaneously, the PLA samples added with PCS had a higher crystallization temperature which was increased by more than 10°C. These results showed that PCS could stimulate the crystallization of PLA. Next, by the cooling curves, the crystallization kinetics methods were used to analyze the effect of PCS on crystallization process of PLA.

DSC curves of the samples: (a) heating at 10°C/min and (b) subsequent cooling at 2°C/min.
Tm, ΔHm, Tc, and Xc of the samples obtained by DSC.
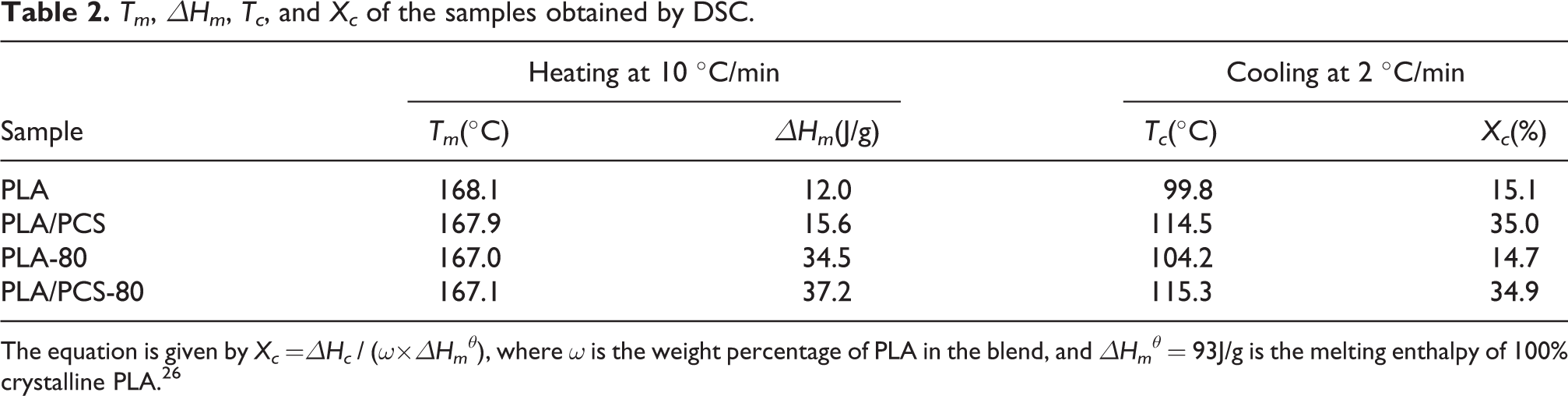
The equation is given by Xc =ΔHc / (ω×ΔHmθ), where ω is the weight percentage of PLA in the blend, and ΔHmθ = 93J/g is the melting enthalpy of 100% crystalline PLA. 26
Figure 4 is the relative crystallinity-time curves of pure PLA and PLA/PCS. As can be seen from the figure, the two curves are typical S-shaped. The tangent slope of each point on the curves is the crystallization rate of the material at that time. The crystallization rates of the materials increased first and then decreased. The reason is that in the early stage of crystallization, there were fewer crystal nuclei, the spacing between crystal nuclei was very large, and there was enough space for the crystals to grow; in the late stage of crystallization, as the number of crystal nuclei increased and the crystals’ volume increased, the space became insufficient, and the crystal nuclei collided with each other, thus the crystals’ growth was hindered and the crystallization rate was reduced. From Figure 4, we calculated that the crystallization rates (1/t1/2) of PLA, PLA/PCS, PLA-80 and PLA/PCS-80 were 0.067 min-1, 0.085 min-1, 0.079 min-1 and 0.106 min-1 respectively, and it was found that PCS accelerated the crystallization and shortened the crystallization time of PLA. This proved that PCS can promote the crystallization of PLA.

Relative crystallinity-time curves of the samples.
The XRD patterns of PLA, PLA/PCS, and the samples heat treated at 80°C, which were obtained by X-ray diffraction experiments, are shown in Figure 5 The differences can be easily discerned. The crystallinity of pure PLA was extremely low, with only one diffraction peak at 2θ = 22.4° corresponding to the 015 plane of the PLA α-crystal. After heat treatment, the diffraction peak of PLA appeared sharper, but no new diffraction peak appeared. For the PLA/PCS samples, two new sharp diffraction peaks appeared at 16.8° and 19.1°. Meanwhile, for the PLA/PCS-80 samples, the two new peaks were located at 16.7° and 19°, which were the characteristic peaks of the 200/110 and 203 planes of α-crystals. This finding indicated that PCS can markedly expedite the formation of α-form PLA crystals with no discernible effect on the crystalline structure.27-29

X-ray diffraction patterns of the samples.
The analysis of XRD confirmed that PCS had a remarkable function in inducing the crystallization of PLA and this was consistent with aforementioned results.
As a visual approach for monitoring the growth and morphology of polymer crystals, POM was used in the present study. Figure 6 shows the POM images of PLA, PLA/PCS, and the samples of PLA and PLA/PCS heat-treated at 80°C for 5 min. For pure PLA (Figure 6a), no visible crystals were observed, indicating its considerably low crystallinity. After PCS was added (Figure 6b), clearly crystal appeared. For the samples heat-treated at 80°C for 5 min, there were more crystals in PLA/PCS (Figure 6d) than PLA (Figure 6c). These observations were in keeping with the previous hypothesis that PCS can promote the crystallization of PLA.

POM photographs of pure PLA and its blends: (a) PLA; (b) PLA/PCS; (c) PLA heat-treated at 80°C for 5 min; (d) PLA/PCS heat-treated at 80°C for 5 min.
Influence of PCS on the thermomechanical property of PLA
Effect of PCS on the thermomechanical property of PLA was studied by DMA. Temperature dependence of the storage modulus (E’) and loss tangent (tan δ) of samples are shown in Figure 7. In Figure 7 (a), the declining of E’ in the temperature range of 55–80°C was caused by the movement of PLA chain segments which started when the temperature reached the glass transition temperature (Tg) of PLA. Due to cold crystallization, the E’ of the sample without heat treatment was increased in the temperature range of 90–105°C. The higher initial values and the vanishing of the “from decline to rise” tendency of E’ for the heat-treated samples were due to more perfect crystallization. The E’ was directly proportional to the maximum elasticity of the sample stored in each cycle, reflecting the elastic components in the viscoelasticity of the material and characterizing the deformation resistance of the material. The greater the modulus, the less likely it was to deform and the greater the rigidity of the material. The E’ of the material increased after PCS was added, which indicated that the rigidity of the material increased and the ability of the material to resist deformation increased.Generally, the temperature corresponding to the maximum loss tangent (tanδmax) is defined as the glass transition temperature (Tg) of the material. As can be seen from Figure 7 (b), the Tg of the material after adding PCS decreased, which indicated that PCS promoted the movement of PLA molecular chains.
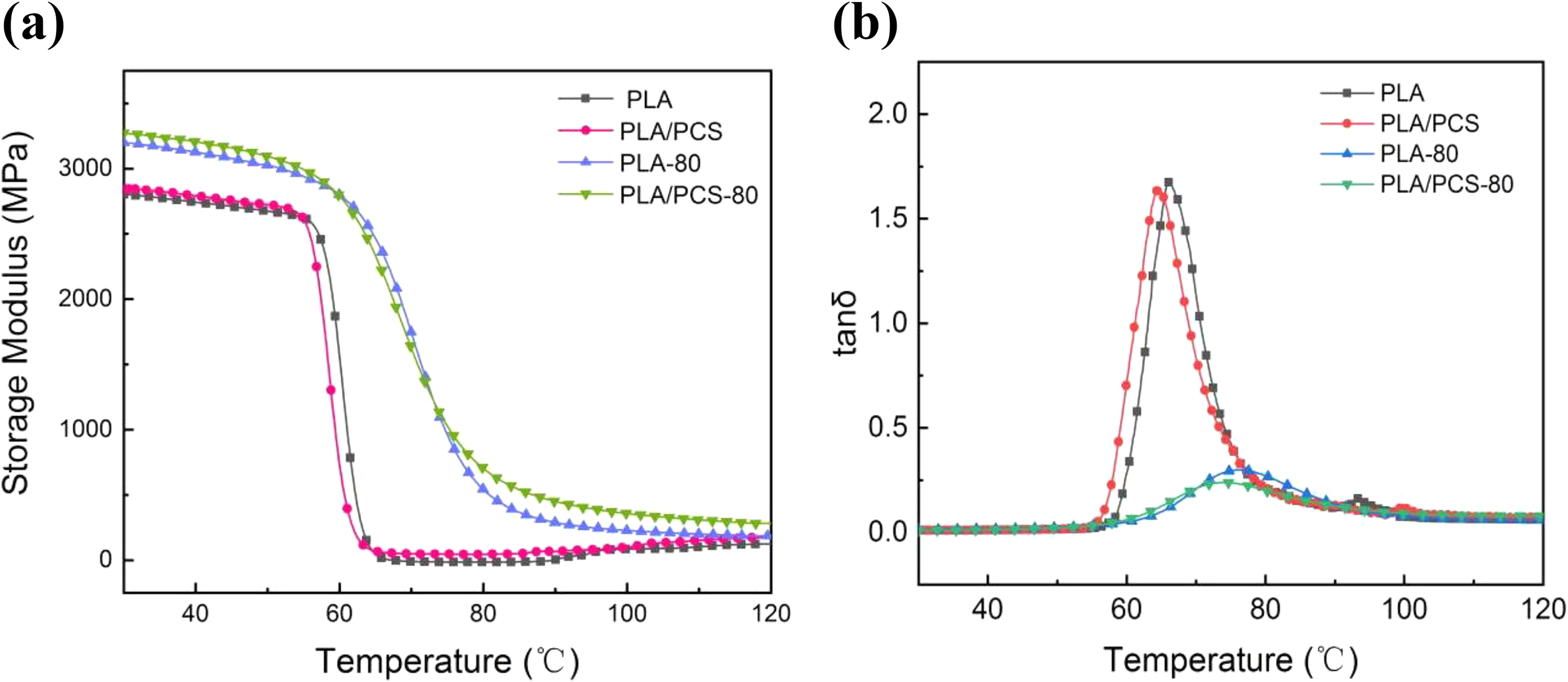
Temperature dependence of (a) storage modulus and (b) loss tangent of samples.
Conclusions
The heat-resistant agent PCS was added to PLA at a dosage of 10 wt%. The mixture was blended using a twin screw extruder, and the splines were prepared using an injection molding machine. The HDT of PLA added with PCS increased by about 10°C relative to that of pure PLA; after heat treatment at 80°C for 24 h, the HDT increased by about 15°C. But PCS had little influence on the thermal degradation of PLA. DSC indicated that after PCS was added, the crystallization temperature and crystallization rate were increased by more than 10°C and 0.018 min-1 respectively. Simultaneously, the crystallinity of the material was markedly improved and more than doubled. XRD and POM showed that PCS can expedite the crystallization of PLA but
Footnotes
Acknowledgments
The authors would like to acknowledge the financial support from Guizhou Postgraduate Research Fund (grant YJSCXJH [2019] 004), the Natural Science Foundation of China (grant 51663006) and Guizhou Province Science and Technology Program Project (grant [2017]5788).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


