Abstract
Hybrid materials composed of semiconductor oxide metals and conducting polymers have been highlighted as a new class of materials, with superior properties compared to their pure constituents. Among the studied composites to photocatalytic applications, the hybrids of titanium dioxide (TiO2) and polypyrrole (PPy) are promising due to several advantages over the pure TiO2 nanoparticles. The PPy/TiO2 composite has been effectively synthetized by chemical polymerization methods as in situ polymerization, photopolymerization, electrochemical polymerization, and molecular imprinting polymerization (MIP). All the cited methods appear to be effective in reducing the band gap energy, which suggests an increase in the formation of photoexcited electron-hole pairs and, consequently, an improvement of the light absorption in the visible region (400–700 nm). In addition, the doping of PPy/TiO2 with noble metals improves the separation of charges in the semiconductor particle, inhibiting the recombination of photogenerated electron-hole pairs. All advantages are evidenced by the characterization results of SEM, TEM, HRTEM, UV-vis DRS, FTIR, XRD, PL, TGA and electrical properties. Finally, results from literature present that PPy/TiO2 composites have better photocatalytic activity than the pure TiO2, being an alternative photocatalyst promising for visible light applications. Thus, this work presents a review of the synthesis, characterization, and application of PPy/TiO2 composites in the photocatalytic processes.
Introduction
The heterogeneous photocatalysis experienced a fast expansion in the last few decades, particularly in terms of environmental remediation and energy generation. Among the most studied fields, attention has been driven to the mineralization of organic contaminants 1-3 and reduction of inorganic contaminants,4-7 disinfection of water and air with different concentrations of pollutants,8-13 production of hydrogen from water and energy production from photovoltaic cells.14-21
The process of heterogeneous photocatalysis is characterized by the formation of free radicals, especially hydroxyl (•OH). These radicals are highly oxidant, non-selective, and capable of reacting with a variety of substances to produce deep modifications in their chemical structure. The process revealed a high efficiency concerning the mineralization of organic compounds and is capable of transforming most of the contaminants into carbon dioxide, water, and inorganic anions, which are far less harmful to the environment.22,23 In general, the process is carried out using a semiconductor material, highly photoactive under visible light and or UV light, biological and chemically inert, photostable and of low cost. 24 The most used inorganic semiconductors include titanium dioxide (TiO2), zinc oxide (ZnO), cadmium oxide (CdO), cadmium selenide (CdSe), among others.25,26
Semiconductors are efficient photocatalysts due to the suitable energy (band gap) between their valence bands (VB) and conduction bands (CB) in the fundamental state. The mechanism that usually explains the reactions that occur on the surface of the photocatalyst comprises the reactions of reduction and oxidation with molecules of water and oxygen and pollutants within the system. The irradiation of the semiconductor with photon energy (hv) higher or equal to the band gap energy, promotes the excitation of an electron from the VB to CB, and creates a highly reactive electron-hole pair. In the hole, the oxidation reactions take place, while the electron in the CB is responsible for the reduction reactions. In case these reactions do not continue, the electron-hole pair may recombine, occurring the release of the initial absorbed energy in the form of heat.
Titanium dioxide (TiO2) is the most studied photocatalyst because of the unique properties as non-toxicity, excellent photoelectric response, low cost, high chemical stability and thermal stability, and others. 25 However, the TiO2 holds a wide band gap (e.g., 3,2 eV for anatase phase), which restricts the utilization of solar radiation as an energy source. The TiO2 activates under ultraviolet radiation, which accounts for less than 4% of the solar spectrum. 27 Different studies have focused on enhancing the TiO2 photocatalyst efficiency through the shift of its optical response from the UV to the visible range. This process can be made by doping the semiconductor with metals and non-metals, via sensitization of the surface with dyes, or even, using composites with other semiconductors.28,29 The use of hybrid composites in photocatalytic processes emerged as a great alternative to overcome disadvantages of pure oxides, as low response to visible light, high rate of recombination of the electron-hole pairs, leaching, and thermal decomposition. The Intrinsically Conducting Polymers (ICPs) have appeared recently as stable materials able to modify the TiO2 nanoparticles positively due to their notable physical and electrical properties.
The ICPs, also known as “synthetic metals,” can act as conductors, semiconductors, superconductors, and magnetic materials. 30 The most widely studied ICPs as polyaniline (PAni), polyacetylene (PAc), polypyrrole (PPy), and polythiophene (PTh), have functioned as dopants that shift the border of the TiO2 particles to longer wavelengths, thus improving the optical absorption in the visible region. The hybrid ICP/TiO2 composites exhibit excellent properties comparing to the individual materials, with the capacity to adsorb a higher amount of pollutants, absorb more light, as well as to remove the electrons from the CB of the semiconductor, preventing the recombination and enhancing the photocatalytic activity.30-32
Polypyrrole (PPy) is one of the most promising ICPs due to several advantages as unique electrical conductivity (from 2 to 100 S cm−1 and controlled by the doping degree), excellent chemical stability (even in acid and basic solutions), thermal stability, biocompatibility, high absorption coefficient in the visible part of the spectra, and direct synthesis by chemical oxidation and electrochemical methods.32,33 This polymer is also an efficient electron donor and a great hole transporter by excitation on visible light. 27
Different research groups reported that the PPy/TiO2 composites have higher adsorption capacity and better photocatalytic activity under visible light irradiation than the neat TiO2 nanoparticles.28,31,32,34,35 The composites present a higher degradation capacity of organic compounds in aqueous solutions using a clean and renewable source of energy—the sun—with great potential for scaling up and applications in new technologies, such as photovoltaic cells.28,35 Hence, this work aimed to review the synthesis, characterization, and application of the hybrid PPy/TiO2 composites in photocatalytic processes.
Synthesis of the PPy/TiO2 composites
Novel technologies are searching for materials capable of combining properties that the called conventional technologies do not have since new and improved characteristics can be created from the union of very different materials at a molecular level.36,37 The hybrid composites produced from inorganic semiconductors and conducting polymers are part of this class of materials because of the synergy created from the union of these two components.
PPy/TiO2 composites can be synthetized by distinct methods, including the synthesis by chemical routes that are most used. The benefits of chemical polymerization include the low cost and easy processing, which provide different possible routes for the synthesis of metal/metalloid conducting polymers. 30 In addition, the advantages of chemical polymerization of pyrrole (Py) comprise the higher solid stability and structural crystallinity, the bigger productivity (is the most used method industrially) and lower environmental impact, since the process can be conducted in water. 38
Recent studies showed that the chemical synthesis by in situ polymerization, photopolymerization, electrochemical polymerization and molecular imprinting polymerization (MIP) is effective, and reduces the recombination rate and the band gap simultaneously.27-29 In all methods, the PPy/TiO2 composites showed enhanced photocatalytic activity in comparison to pure nanoparticles of TiO2.
In situ polymerization
The in situ polymerization has advantages due to the simplicity, good reproducibility, and easier scale-up of the process. Two different routes are generally applied to perform the in situ polymerization (Figure 1):
Route 1 (Figure 1 (a)): Py and acid are added into an aqueous solution, followed by the addition of TiO2, and the oxidant agent that starts the polymerization reaction;32,36,39
Route 2 (Figure 1 (b)): TiO2 and acid are dispersed into an aqueous solution, followed by the addition of Py, and the oxidant agent that also starts the polymerization reaction.28,34,35,40,41

Synthesis of PPy/TiO2 composite by in situ polymerization via (a) route 1 and (b) route 2.
Experimental conditions found in the literature for both routes are presented in Table 1. In the chemical polymerization process, the oxidizing agents are responsible for the oxidation of monomers in the presence of titanium dioxide, forming the polymer with the desired conductive characteristic coupled to the TiO2 particles. 42 The oxidant agents mostly used for polymerization are ferric chloride (FeCl3), ammonium persulfate ((NH4)2S2O8), hydrogen peroxide (H2O2) and potassium persulfate (K2S2O8). In addition, the acids used are HCl and H2SO4 at different concentrations. Reactions are carried out at low temperatures and constant stirring, and reagents are added slowly. At the end of reaction for both routes, a dark precipitated PPy is separated using filtration or centrifugation, with subsequent washing with copious amounts of acid and or distillate water. Finally, the solid obtained is dried (40–100 ºC) and stored for later use.
The optimum experimental parameters for different polymerization approaches may vary and have a strong influence on the morphology and properties of the resulting composite system. 41 For instance, Wei et al. 40 showed that PPy/TiO2 nanocomposites prepared by oxidative polymerization with FeCl3 (at room temperature) presented a higher electrical conductivity than that with (NH4)2S2O8. Still, Villora-Picó et al. 6 proposed a new methodology of in situ polymerization, without the addition of acid, and showed that the oxidizing agent FeCl3 remains more effective to obtain composites with high conductivity than K2S2O8. Liang et al. 43 found that the PPy synthesized at lower temperatures has a higher doping level, fewer structural defects, and longer conjugation lengths. According to these authors, the electrical conductivity is also higher, probably due to the formation of a chain more asymmetric at lower temperatures, improving the energy flux.
Synthesis conditions for in situ polymerization of PPy/TiO2 composites.

One of the main synthesis parameters that shows a considerable effect on the photocatalytic activity of PPy/ TiO2 composite is the molar or mass ratio of the monomer to titanium dioxide. 48 There is a consensus that the chemical combination of these two substances (here PPy and TiO2) has a synergistic effect with the improvement of light absorption in the visible region and separation of charges in the final compound. For synthesis methodologies that use larger amounts of monomer compared to semiconductor, the increase of TiO2 quantity in the composite enhances the photocatalytic activity.32,46,47,49 On the other hand, for hybrid materials with higher amounts of TiO2, the addition of PPy generally increases the photoactivity of the composite up to an optimal concentration.34,35,50 When the presence of polymer becomes excessive in the composite, it can interfere in the absorption of light and, consequently, decrease the amount of irradiation passing through the reaction system.44,50
Photopolymerization
The polymerization can also be photocatalyzed by semiconductor particles, which, when photoexcited, can accelerate the formation of free radicals and consequently catalyze the polymer formation.51-53 Therefore, chemical photooxidation is an alternative and simple strategy to synthesize the heterojunction composites based on PPy and TiO2. Weng and Ni 51 successfully initiated the photooxidative polymerization of polypyrrole using TiO2 nanoparticles in aqueous suspension without adding any extra reagent and generated a hybrid material with strong interaction between the components and good conductivity. Oliveira et al. 54 tested different synthesis methodologies (chemical oxidation and photopolymerization method) and found that the addition of AgNO3 as a dopant in the monomer photopolymerization using TiO2 particles resulted in a composite with greater photocatalytic activity.
Electrochemical polymerization
The electrochemical method involves the direct formation of conductive polymers with better control of morphology and with the possibility of forming thin films.55-57 The reaction, in this case, proceeds by adding the pyrrole monomer, the TiO2 nanoparticles, a surfactant, and by the presence of direct current in the system. Macedo et al. 56 used the electrochemical method to evaluate the influence of different parameters (surfactant, electric field, monomer concentration, and reaction time) in the synthesis of a PPy/TiO2 composite. For these authors, the combination of two different surfactants during electrochemical polymerization generates a highly branched composite of PPy/TiO2 with superior electrical properties. Electrochemical polymerization was also conducted by Jia et al. 57 using the potentiostatic method to obtain a hybrid material with better absorption intensity and shift in the visible range.
Molecular imprinting polymerization (MIP)
The low selectivity of photocatalysts based on TiO2 is not attractive for the degradation of pollutants in complex systems. One of the proposed approaches to increase the selectivity of the photocatalyst is the use of the molecular imprinting polymerization (MIP) for selective removal of the organic pollutant. 58 This technique is used for the preparation of polymeric materials that contain highly specific recognition sites for a model molecule (such as dye, for example). 27 Molecular imprinting polymers (MIPs) have chemical/mechanical stability, high selectivity for a model molecule, low cost, and simplicity of preparation. 27 More recently, this molecular imprinting technique has improved, and polymers obtained from surface molecularly imprinted (SMIPs) not only have a strong affinity and higher adsorption capacity with the target contaminant but also avoid problems with the mass transfer because of inaccessible locations.27,29
Figure 2 shows a representative diagram of the synthesis route of a composite based on TiO2 and molecularly imprinted polymer for use as a photocatalyst of the methyl orange dye (template molecule). When TiO2 nanoparticles are coated with SMIP, the hybrid material (MIP-PPy/TiO2) obtained has a strong affinity for target contaminants, and also has high photocatalytic activity. 29 Highly selective surface molecularly imprinted composites have been successfully prepared with rhodamine B and methyl orange as model molecules.27,29 In addition to high selectivity, the composites showed obvious absorption under the visible light, efficient separation of the electron-hole pairs, high adsorption capacity, and improved photocatalytic activity compared to composites synthesized without the MIP technique.27,29

Effect of TiO2 particle structure
One way to increase the photocatalytic effect of TiO2 and, consequently, the final composite is to modify the semiconductor particle size and morphology. 59 The various TiO2 morphologies used in the synthesis of the composite with the polymer are commercial6,29,45,51,54,56 or synthesized27,28,34,35,60-63 nanoparticles, nanotubes,57,64 nanofibers,50,65,66 nanorod 53 and microbelts.46,47
Nanostructured materials are attractive candidates for the synthesis of semiconductor-based composites for photocatalytic applications. 64 Semiconductors with organized structures such as nanotubes, nanofibers, and nanorods are candidates that can particularly form hybrid materials with large areas of organic/inorganic heterojunctions and better contact with each other.53,64 One-dimensional materials provide not only greater surface area but also take advantage of the fast electron transfer and efficient charge separation.53,57 Meanwhile, Li et al. 47 and Zhang et al. 46 highlight that TiO2 microbelts have low density, high specific surface area and superior delivery capacity.
Nanoparticles of TiO2 with no well-defined structure are widely used for PPy-TiO2 composites due to the ease of obtaining them commercially or through synthesis. However, a disadvantage of using these nanoparticles is their aggregation capacity resulting from the high surface energy of TiO2, which makes the synthesis of a homogeneous composite somewhat difficult and weakens the photocatalytic efficiency. 66 Besides that, the application of nanoparticles in a large-scale photocatalytic system is limited by the challenge of separating them for later reuse. 67 An alternative to the use of suspended photocatalyst composites involves the formation of TiO2-PPy films.54,61 Immobilization is advantageous as it makes the photocatalyst separation process unnecessary. However, reducing the contact area of the photocatalyst with the pollutants can lead to mass transfer limitations.
Still, TiO2 with macro 34 and mesoporous 52 structures has also been studied due to the high photocatalytic performance. 68 The use of mesoporous TiO2 is an alternative for the synthesis of hybrid materials based on polymers due to its structure with ordered pores and large surface area.34,52 However, the pore size of mesoporous TiO2 in the order of nanometers (2–50 nm) can cause rapid blockage and resistance to diffusion, preventing efficient monomer infiltration and the manufacture of composite materials with a stabilized interface, thus excluding a large amount of the internal surface. 34 On the other hand, titanium dioxide with a macroporous structure has more accessible pore openings (>50 nm), improving molecular diffusion and increasing the availability of its internal surface. According to Li et al., 34 the growth of the polymer cores in the cavities should favor the formation of a good stabilized interface between the inorganic and organic phases. As the reaction takes place, it leads to a concentration gradient, which can act as a driving force for the transfer of the pyrrole from the solution to the pore space, and the polymer layer gradually grows on the surface of the nuclei 34 (Figure 3).
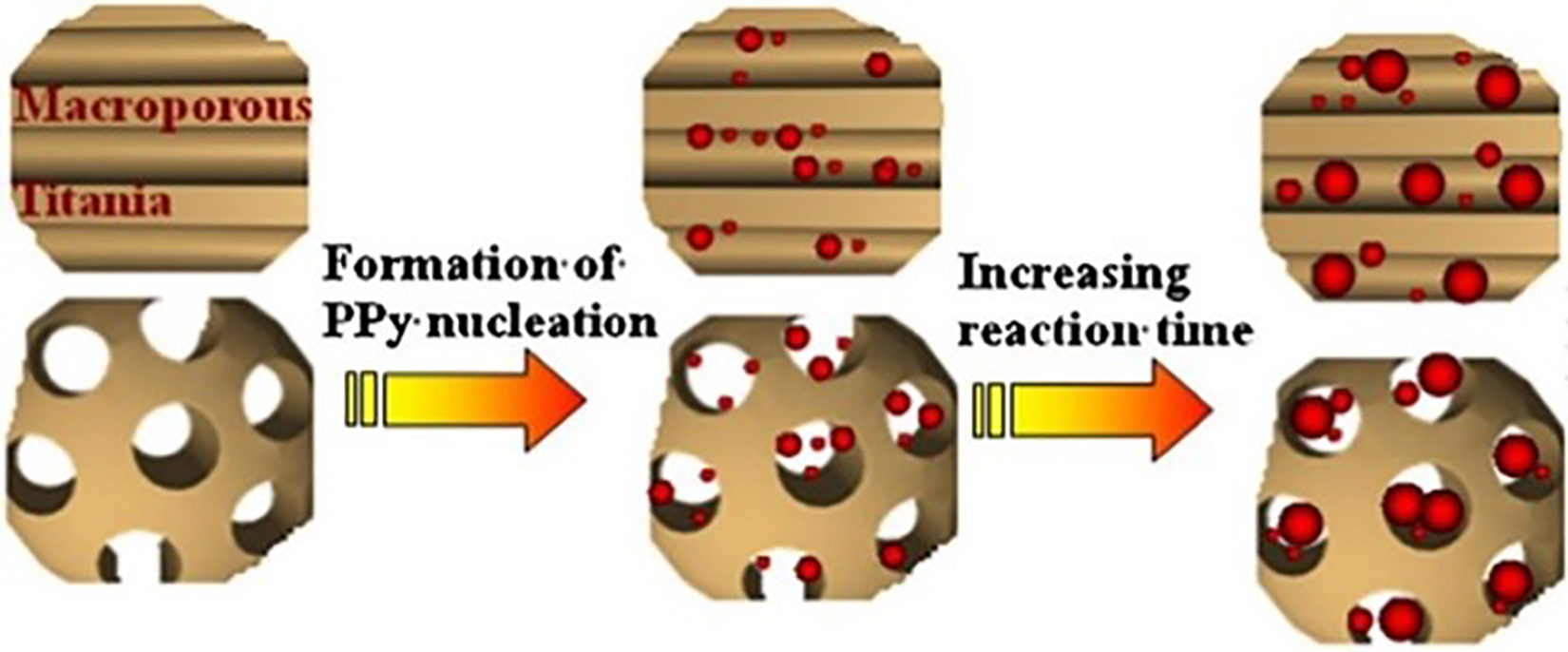
Effect of doping metals
Another way to improve the properties of composites based on polypyrrole and titanium dioxide is the addition of doping metals or oxide metals such as Ag,50,65,69 Pt,6,16,63 or Fe3O4.47,58 Noble metals act by inhibiting the recombination of the photogenerated electron-hole pairs and improving the separation of charges in the semiconductor particle by removing electrons through the metal/semiconductor interface.50,65 Among these noble metals, silver stands out for having a relatively cheap cost and anti-bacterial property. 50 Several authors have shown that, compared to the corresponding samples of one and two components, the three-component PPy/Ag specie/TiO2 system exhibits enhanced photocatalytic activity.50,65,69 The Ag species can be silver nanoparticles, 50 Ag-AgX (X = Cl, Br and I) aggregates 65 and Ag-Ag2O. 69 The use of these last two species is mainly because small silver nanoparticles are very reactive and easily oxidized and lost. 50 In addition, Ag2O is classified as a semiconductor (∼ 1.46 eV) and has been widely used as a photocatalyst. 69 Composites of PPy/TiO2 containing platinum have the same synergistic effect as silver, but they have been used mainly for photocatalytic generation of H2 from H2O.16,63
The incorporation of iron oxide (Fe3O4) particles in the composite structure has been used to improve the photocatalytic activity 47 and also to facilitate the separation of the magnetic particles from the medium for later reuse. 58 The use of a support for the PPy/TiO2 composite particles can also assist in the recovery of the photocatalyst for later reuse. 47 Silica (Si/SiO2) has been used to support the PPy/TiO2 composite due to its chemical inertness, thermal stability and high super artificial area.47,58,66,70 Shi et al. 70 synthesized a hierarchical structure composed of PPy/TiO2/Si (Figure 4) with a greater specific surface area and photocatalytic activity than the PPy/TiO2 composite and the individual components.
The conductive polymers polypyrrole and polyaniline have been used individually to improve the electronic conductivity as well as the solar energy absorption and photocatalytic activities of TiO2. 71 It was reported by Deng et al. 71 that the combination of these two conducting polymers with TiO2 showed a synergistic effect with the considerable improvement in conductivity, thermal stability, and photoactivity compared to TiO2, PPy/TiO2 and PAni/TiO2.

Properties of PPy/TiO2 composites
The synthesized solids are usually characterized by Scanning Electron Microscopy (SEM), Transmission Electron Microscopy (TEM), High-Resolution Transmission Electron Microscopy (HRTEM), Diffuse Reflectance Spectra (UV-vis DRS), Fourier-Transform Infrared Spectra (FTIR), X-Ray Diffraction (XRD), Thermogravimetric Analysis (TGA), Photoluminescence Spectra (PL) and electrical properties.
Microscopy analysis
Sangareswari and Sundaram, 32 performed the oxidative polymerization of Py in the presence of negatively charged TiO2 nanoparticles, using (NH4)2S2O8 as oxidant (via route 1). This synthesis mechanism (Figure 1 (a)) includes a sequence of reactions between Py, acid and TiO2, and the subsequent chemical oxidation of Py to PPy, with the production of PPy/ TiO2 composite. As shown in the SEM image for this solid (Figure 5(a) and (b)), the TiO2 nanoparticles (∼ 21 nm according to manufacturer) coated the outer surface of the polymer. The nanoparticles of TiO2 are distributed uniformly on the PPy surface, and an increase in the size of the spheres is observed from 0.3 µm (Figure 5 (a)) to 3.0 µm (Figure 5 (b)). The morphology of PPy presents visible spheres (Figure 5 (a)), typical of this material.34,42

SEM images of (a) pure PPy and (b) PPy/TiO2 composite; 32 (c) macroporous TiO2 and (d) PPy/macroporous TiO2 composite; 34 (e) TiO2 nanofibers and (f) TiO2/Ag-AgCl@PPy composite. 65 “Reprinted from Sangareswari and Sundaram, 32 Copyright © 2015 at Springer Nature Switzerland AG, distributed under the terms of the Creative Commons Attribution License (open access). Reprinted from Li et al., 34 Copyright © 2013, with permission from Elsevier B.V. Reprinted from Yao et al., 65 Copyright © 2016, with permission from John Wiley & Sons Inc.”
The use of TiO2 particles with different morphologies has been done as a way to improve the final composite properties. Li et al. 34 used a macroporous titanium dioxide (Figure 5 (c)) to obtain a composite with smaller size PPy particles (∼ 100 nm) inserted in the semiconductor pores (Figure 5 (d)). The average size of the PPy particles synthesized by this methodology and without TiO2 is approximately 250 nm.32,34,42 According to Li et al., 34 the higher the reaction time, as well as the higher the PPy/TiO2 ratio, the more similar is the morphology of the composite surface to pure PPy. Yao et al. 65 synthesized TiO2 nanofibers with average diameters of 100–200 nm and length of dozens of microns (Figure 5 (e)). These nanofibers were later doped with Ag-AgCl and coated with PPy by in situ polymerization. The TiO2/Ag-AgCl@PPy composite (Figure 5 (f)) presents a similar size to pure TiO2, but the surface is much rougher.
Gao et al. 28 verified by TEM analysis that the PPy/TiO2 nanocomposites have formed relatively lose aggregates of nanoparticles, ranging from 14 to 17 nm (Figure 6 (b)). In addition, the authors observed a film coating the TiO2 surface (Figure 6 (c)). Like Gao et al., 28 Wang et al. 35 described that the morphology and size of the hybrid nanocomposite (Figure 6 (e)) were very similar to the neat TiO2 particles (Figure 6 (d)). Still, TEM images (Figure 6 (d) and (e)) showed aggregated spherical nanoparticles, and the aggregation was slightly more significant than that of the neat TiO2 particles, implying a strong interaction between the components.35,41 The EDS elemental mapping by Silvestri et al. 39 with the composite sample (Figure 6 (f)) showed that the main characteristic elements of polypyrrole (C and N) and titanium dioxide (Ti and O) were detected.

TEM images of (a) TiO2 nanoparticles, (b) PPy/TiO2 nanocomposite and (c) HRTEM image of PPy/TiO2 nanocomposite; 28 TEM images of (d) TiO2 and (e) PPy/TiO2; 35 EDS analysis of (f) PPy/TiO2. 39 “Reprinted from Gao et al., 28 Copyright © 2016, with permission from Elsevier B.V. Reprinted from Wang et al., 35 Copyright © 2018, with permission from Elsevier BV. Reprinted from Silvestri et al., 39 Copyright © 2020, with permission from Elsevier B.V.”
FTIR spectra and XRD analysis
The molecular structure and functional groups of the PPy/TiO2 composites have been investigated using the FTIR technique.28,42,46,57 The analysis of pure PPy conducted by Li et al. 60 showed absorption bands characteristic of the polymer (Figure 7 (a)). The peaks at 3400, 1548, 1450, 1045 and 790 cm−1 correspond to N–H, C=C (in pyrrole ring), C–N, C–O–C, and C–C stretching vibrations, respectively.57,60 The absorption bands at 1652 and 1324 cm−1 are assigned to C–N in-plane bending vibration and C–N bending vibration. Additionally, the band at 1175 is associated with C–H in-plane bending vibration, and the bands at 690 and 910 cm−1 can be attributed to C–H out-of-plane bending vibration.57,60 In the spectrum of pure TiO2 (Figure 7 (a)) the vibrations of Ti–O–Ti and O–Ti–O bands are present at 1250–400 cm− 1 region. 72 The maximum peak characteristic of the TiO2 spectrum at 585 cm−1 appears in the spectrum of the PPy/TiO2 composite at 571 cm−1. The absorption band at 3400 cm−1 of the PPy spectrum that corresponds to the stretching vibration of the N–H disappears in the composite, indicating that there is an interaction between the components. 60 As titanium is a transition metal, there is a tendency to form a coordination compound with the nitrogen atom of the PPy molecule. 60 All characteristic PPy bands (1700–600 cm−1) are also present in the PPy/TiO2 composite spectrum. Still, an attenuation in the intensity was noted, and even a slight displacement on the peaks, in comparison with the pure compounds, resulting from the chemical interaction between them.42,57,60 The nature and concentration of surface groups found in the different synthesized photocatalysts may be responsible for the interaction with the pollutants, and, thus, to the variation in their adsorption capacity. 31

From the XRD analysis, all authors highlighted the crystallography construction of the obtained composites. XRD patterns obtained by Baig et al. 73 (Figure 7 (b)), showed that the PPy/TiO2 composite presents characteristic peaks of both components. The pure polymer (Figure 7(b)) has an amorphous structure characterized by the presence of a broad peak in the region of 10–35º.28,39,42,73 The pure TiO2 (Figure 7 (b)) has main diffraction peaks at 25°, 38º and 48°, corresponding to the anatase phase.42,73,74 In this case, the synthesized composite contains a larger amount of PPy in its structure than TiO2; thus, the characteristic peak of PPy appears, even if subtly, in the final composite. The same behavior was observed by other authors.17,39,42,74 On the other hand, the XRD analysis of composites containing more TiO2 indicated that the oxidative polymerization of PPy on TiO2 did not change its crystalline phase.28,34,49,62,75 However, an increase in the amount of PPy decreased the intensity of the diffraction peaks of the pure TiO2, possibly due to the chemical interactions between PPy and TiO2.
UV–Vis DRS and PL spectra
The optical properties of composites synthesized in different works from literature are summarized in Table 2. The band gap energies of the samples were estimated according to the Kulbeka-Munk model28,32 or determined from the cut-off wavelength values of the UV–Vis diffuse reflectance spectra and using the following equation:35,41

where Eg is the band gap energy (eV), h is Planks constant (J/s), C is the speed of light (m/s) and λ is the cut-off wavelength (nm).
Band gap of PPy/TiO2 composites.

For all the above cases (Table 2), regardless of the synthesis route and amount of PPy and TiO2 used, the composites presented a smaller band gap value in comparison to pure TiO2. Besides, an ideal PPy/TiO2 molar ratio was observed, which leads to a reduction in the band gap. Usually, the higher the concentration of TiO2, the more similar is the band gap to the pure solid. Gao et al., 28 for instance, found a reduction in the band gap for nanocomposites prepared with a PPy/TiO2 molar ratio up to 1/100. According to Luo et al., 44 the UV-vis diffuse reflectance spectra shows that the absorbance of PPy/TiO2 enhances within the whole range of visible light (Figure 8). With the extension of the polymerization time (2–10 h), the visible light absorbance increases, which might be associated with the content of PPy on the surface of the TiO2 nanoparticles. These results suggest the formation of more photoexcited electron-hole pairs by the nanocomposites that can result in greater photocatalytic activity. Figure 8 presents the UV-vis DRS results of PPy-TiO2 with different molar ratios obtained by Wang et al. 35 The band gap energies (Eg) of pure TiO2, PPy-TiO2(1:60), PPy-TiO2(1:80), PPy-TiO2(1:100), PPy-TiO2(1:120), PPy-TiO2(1:140) are respectively 3.12, 2.78, 2.82, 2.84, 2.92, and 2.95 eV. Therefore, greater amounts of PPy lead to lower band-gap energies. It is worth mentioning again that in composites with a higher amount of TiO2, the addition of PPy improves the photocatalytic properties until an optimal composition.

The photoluminescence spectra of different hybrid materials obtained by Yang et al. 50 (Figure 9) showed a strong peak at 380 nm, which can be associated with the recombination of excited electrons. The peak intensity of the composite (PPy/TiO2) PL spectrum is reduced in comparison to pure TiO2. It shows a decrease in the recombination rates of electron-hole pairs by introducing the PPy and the potential to improve the photocatalytic activity of the solid.28,32,34 Still, doping titanium dioxide with silver nanoparticles (Ag/TiO2) produces a semiconductor with a lower electron-hole recombination rate than pure TiO2. The TiO2-based composite containing both silver and PPy unites the best of the three substances, generating a photocatalyst with the lowest PL intensity. In this case, silver nanoparticles act as electron acceptors, improving separation efficiency in the photocatalyst. 50 Still, PPy greatly reduces the TiO2 band gap (Table 2) and consequently improves the light absorption in the visible region (400–700 nm) (Figure 8).

TGA analysis
Finally, the TGA analysis (Figure 10) showed that the thermal stability of the composite is much higher in comparison to the pure PPy, at the same temperature range of 30°C to 800°C, even using high quantities of the monomer (e.g., composite materials with a PPy content of 98.3 mol.%). 32 The TiO2, applied in photocatalysis, is a crystalline solid with good stability, and its TGA analysis shows a small mass variation. The PPy, in turn, is an organic polymer that degrades more easily, losing moisture and solvent until it decomposes at temperatures higher than 220°C. 41

Electrical properties
The electrical properties of the PPy/TiO2 composites have been evaluated in terms of photocurrent response, electrical conductivity, dielectric constant, and dielectric loss. The photocurrent test is commonly applied to evidence the presence of charge transfer on the composite surface during the activation and deactivation under a light source,50,53,57,71 as occurs in photocatalysis. Typical photocurrent response curves of TiO2, Ag/TiO2, PPy/TiO2 and PPy/Ag/TiO2 samples as a function of time for several on-off cycles of intermittently visible light irradiation are shown in Figure 11. The incidence of visible light in the different samples promotes an initial anodic peak, i.e. an increase in current that can be attributed to the separation of the electron-hole pairs.50,53,57,71.After reaching the initial anodic peak during irradiation, the current continuously decreases until it reaches a photocurrent in steady-state. When the irradiation is abruptly removed, a current decay occurs, indicating the complete recombination of charges on the material surface.50,53,57,71

The photocurrent of the pure TiO2, PPy/TiO2, Ag/TiO2 and PPy/Ag/TiO2 electrodes at steady-state was 0.18, 0.43, 0.54 and 0.83 mA cm-2, respectively. 50 The combination of the polymer and titanium dioxide caused an increase in photocurrent that was clearly accentuated by the addition of silver. This increase indicates less recombination and more efficient separation of the photogenerated electron gap pairs, improving the photoelectrochemical properties of the composite materials concerning pure TiO2. Li et al. 53 evaluated the photocurrent response of PPy/TiO2 polymerized at different times. The results showed that the longer the polymerization time, the greater the photocurrent response under visible light. The explanation for this behavior is related to the greater amount of polymer in the material and, consequently, the greater light uptake at the interface. 53
The doping of PPy with TiO2 nanoparticles promotes significant changes in the electrical properties of composites. 76 The variation in electrical conductivity as a function of the TiO2 amount in the composite was assessed by Babazadeh et al. 42 using the four-point probe technique, and the results are shown in Figure 12. The electrical conductivity increases with an increasing amount of TiO2 in the composite, up to an optimum value at which it starts to decrease. The addition of TiO2 nanoparticles can generate a more efficient network for transporting loads in the polymer chains, leading to higher conductivity.42,76,77 On the other hand, the excess of TiO2 results in the dominance of the insulating properties of the semiconductor, thus reducing the conductivity of the composites. 77

The dielectric properties of the materials can be altered with the variation of the applied electric field.74,77-79 The dependencies of the dielectric (ε’) and dielectric loss (ε”) constants as a function of frequency at room temperature obtained by Ahmed et al. 79 for composites containing different quantities of TiO2 are shown in Figure 13. Polymers do not normally have high dielectric constants, but their combination with materials that have high dielectric constants can significantly improve this property.74,80
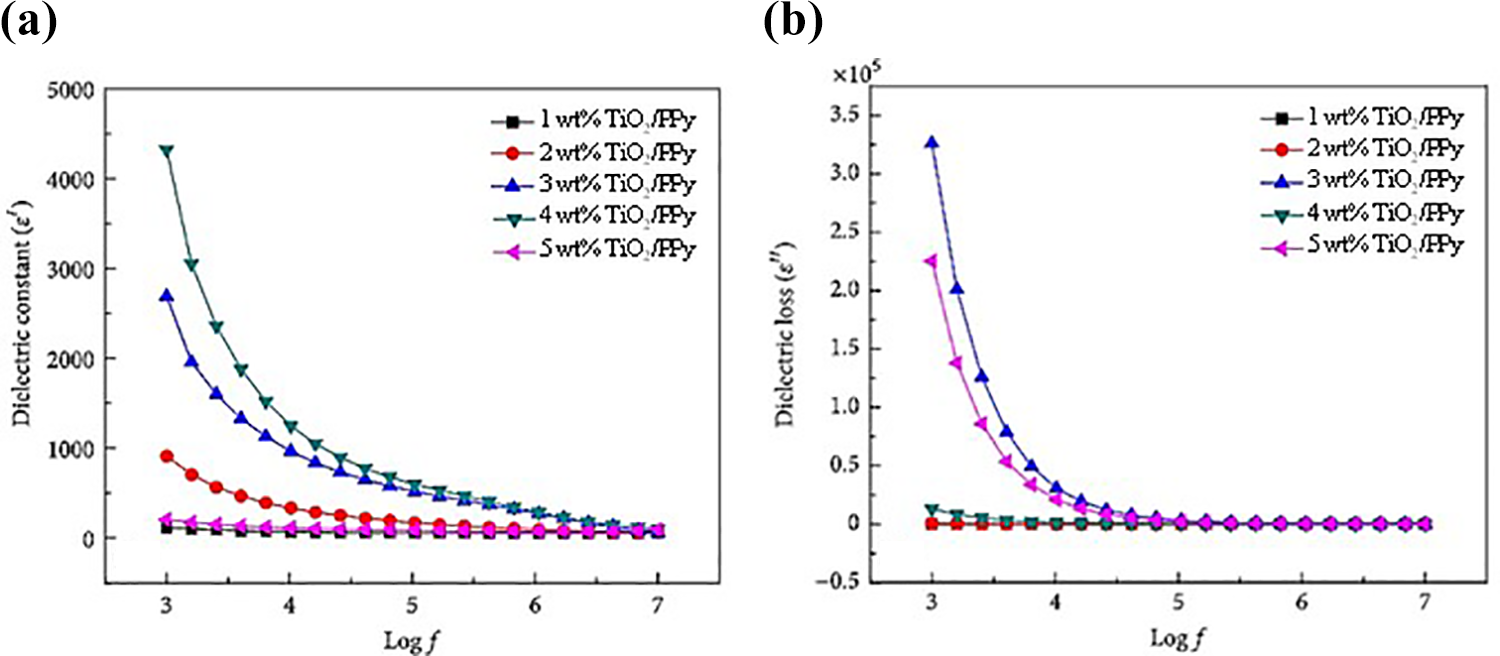
Both the dielectric constant and the loss dielectric decrease with increasing frequency (Figure 13). This behavior is typical for most dielectric materials. 77 At high frequencies, the reduction in the value of ε’ followed by a constant behavior (Figure 13 (a)) can be explained by the orientation of the dipoles, which hinders their rotation, 78 promoting the delay of dipolar oscillations behind those of the applied electrical field. 77 At low frequencies, Ahmed et al. 79 attribute the high values of ε’ to the fact that the polarization mechanisms (displacements of interfacial, dipolar, ionic and electronic charge) follow the applied electric field. Regarding the loss dielectric (Figure 13 (b)), the behavior as a function of frequency can be attributed to the moving charges within the polymer structure.
According to Figure 13, the influence of the composition of the hybrid materials is more evident at low frequencies for both cases. The value of the constants increases with the addition of TiO2 in the PPy up to a maximum value that is attributed to the percolation threshold for these nanocomposites.78,80 A similar trend was obtained by Aaditya et al. 78 for polypyrrole and titanium dioxide composites, i.e. the higher the amount of TiO2, the greater are the values of the dielectric constant and dielectric loss up to a maximum value.
Photocatalytic activity
The photocatalysts based on PPy and TiO2 have been widely applied for degradation of dyes,27,29,34,65,81 nitrates, 6 polyethylene, 62 drugs, and pesticides. 39 In addition, these composites have been tested as photocatalysts for H2 production 63 and CO2 photoreduction with CH4 generation 28 and also as essential constituents for obtaining an intelligent paint (self-cleaning and anti-bacterial). 82
Applications and results from the literature
Table 3 presents the experimental conditions applied during the photocatalytic degradation of dyes, along with the key results from the literature. Sangareswari and Sundaram 32 studied the degradation of methylene blue dye by both pure TiO2 and PPy/TiO2 and found a greater degradation efficiency for the nanocomposites in all studied conditions. The composite has a lower band gap than the pure TiO2 and a lower electron-hole pair recombination rate, facilitating the electron transfer to the conduction band (CB), and increasing the dye degradation. The authors also found that the increase in TiO2 fraction in the composite improves the dye degradation to 99.8% in 90 min of reaction (with PPy/TiO2 of 1/0.017 molar ratio). At last, the discoloration efficiency increased with increasing pH, reaching a maximum degradation between 9 and 11. The zero point charge for TiO2 is 6.25, then, for higher pH values, the catalyst is negatively charged, and the MB (a cationic dye) easily bond on its surface, thus increasing the removal.
Experimental conditions for the photodegradation of dyes with PPy/TiO2 composites.

For composites with high amounts of TiO2 as those obtained by Li et al., 34 an increase in the quantity of PPy in the composite increases the degradation of MB to an optimum value. When the amount of PPy is excessive, the layer of PPy accumulated on the surface and in the pores of TiO2 can block the penetration of photoenergy into the composites and prevent the diffusion of molecules producing an adverse effect on the activity 34 (Figure 3). As for the catalytic performance of the sample with low PPy content, similar to the performance of pure TiO2, it may be associated with an insufficient conductive layer to promote charge transfer. Thus, the conductive polymer acts on the composite as a photosensitizer improving the efficiency of charge separation at the stable organic-inorganic interface. 34
Gao et al. 28 studied the degradation of rhodamine B dye and also found that the composite is more efficient in the photocatalytic degradation than the pure TiO2. Authors described that the photocatalytic activity increases up to a PPy/TiO2 molar ratio of 1/100, decreasing for higher quantities of the semiconductor, possibly by hindering the absorption of light in the composites. Results suggested that the composite with a PPy/TiO2 molar ratio of 1/100 is the most efficient, with degradation of 97% in 480 min of reaction, or even, 41% greater than the pure TiO2. They also tested the photocatalytic properties of composites in the generation of methanol from CO2. After 8 h of irradiation, the methanol yield obtained with pure TiO2 reached 273.8 μmol/g while the PPy/TiO2 composite (1/100) showed a maximum production of 414.9 μmol/g. 28 In this way, the addition of PPy significantly improved the catalytic efficiency in the photoreduction of CO2. Additionally, cyclic tests were performed to investigate the stability of PPy/TiO2 composite during the catalytic reaction process of methanol production. After each new cycle, the sample was collected from the reaction mixture, dried under vacuum at 70°C for 12 h, and then reused under the same reaction conditions. 28 The yield decreased over five cycles by only 7%, indicating that the composite photoreduction activity was reproducible and stable. 28
A highly selective composite for adsorption and degradation of rhodamine B dye (RhB) was prepared by He et al. 29 using the molecular surface imprinting technique. The structure of the prepared MIPRhB-PPy/TiO2 has cavities imprinted on the surface compatible with the RhB structure. The adsorption and photocatalytic activity of the composites were evaluated for different dyes (rhodamine B, rhodamine 6G and methylene blue) and MIPRhB-PPy/TiO2 showed strong affinity, high adsorption capacity and fast adsorption rate for RhB. 29 Deng et al. 27 also used the surface molecular printing technique to obtain a composite based on PPy and TiO2 using methyl orange as the model molecule, MIPMO-PPy/TiO2 (Figure 2). Likewise, the MIPMO-PPy/TiO2 composite showed greater adsorption capacity and selectivity than the control composites of PPy/TiO2. The photocatalytic activity of MIPMO-PPy/TiO2 being twice as high as PPy/TiO2 27 (Table 3). The molecularly imprinted composites based on PPy and TiO2 are a promising alternative, mainly due to the significant improvement in selectivity, for the treatment of industrial wastewater.27,29
Yao et al. 65 showed that the doping of TiO2 with silver (Ag-AgCl) and subsequent synthesis of the hybrid material with polypyrrole significantly improves the photocatalytic activity of the final compound (TiO2/Ag-AgCl/PPy) under visible light. The superior photocatalytic property is due to the synergistic effect of the combination of these three components: the TiO2 nanofibers, the Ag-AgCl nanoparticles, and the PPy shell. 65 The excited electrons in the PPy/TiO2 composite can be readily injected at the Fermi level of Ag. As a result, there is rapid separation of the charge and slow recombination, increasing photocatalytic activity. 50
Another dye widely applied in photocatalytic activity tests of PPy/TiO2 composites is methyl orange (MO). Wang et al., 35 Li et al. 60 and Sun et al. 49 evaluated the photocatalytic properties of hybrid materials synthesized through the degradation of MO. For all cases, the composites showed greater dye degradation concerning pure TiO2 and pure PPy under visible light and sunlight (Table 3).
Silvestri et al. 39 studied the photocatalytic activity of PPy-TiO2 composite in the conversion of diclofenac (DCF) and 4-chlorophenol (4-CP) under simulated sunlight. Diclofenac is one of the drugs most often found in the aquatic environment, and 4-chlorophenol is a widely used pesticide. The PPy/TiO2 was able to degrade more than 90% of DCF and 40% of 4-CP, in addition to presenting stability and efficiency in five consecutive reuse cycles for both pollutants. These results also show the possibility of using these composites for the degradation of persistent organic compounds such as drugs and pesticides. 39
Composites based on conductive polymer (PPy and PAni) and titanium dioxide were also applied to reduce nitrate from water. Villora-Picó et al. 6 evaluated the ability of hybrid materials (PPy/TiO2 and PAni/TiO2) to reduce aqueous nitrate and compared it to catalytic hydrogenation using platinum and hydrogen gas. The mechanism of nitrate reduction using the synthesized composites involves the adsorption of pollutants on the polymer surface. The transfer of electrons from the semiconductor to the polymer favors the reductive capacity of the polymer chain, where nitrate is selectively reduced with a low generation of undesirable by-products. In this case, as the polymer is the key factor in photoreduction, the oxidant used during polymerization is an important parameter. Thus, for this application, FeCl3 and K2S2O8 are the most suitable oxidants for the in situ polymerization of PPy and PAni monomers, respectively. As a result, the reduction of nitrate produced by the composites TiO2/PPy/FeCl3 and TiO2/PAni/K2S2O8 is considerably more effective than the catalytic hydrogenation produced by platinum nanoparticles. 6 The main advantage of using composites based on conductive polymers and TiO2 to the detriment of metal catalysts for nitrate reduction is that it is not necessary to add hydrogen gas to the system. 6
Li et al. (2010) evaluated the photocatalytic degradation of polyethylene (PE) plastic using a polypyrrole/TiO2 nanocomposite as a photocatalyst. As a result, the photocatalytic degradation process of PE using PPy/TiO2 was much faster than PPy or TiO2 under natural sunlight. The development of this new type of photocatalytic technology can lead to the ecological disposal of polymer waste. The disposal of plastics in the environment, such as polyethylene, is recognized as an environmental problem worldwide mainly because of the time it takes to degrade. 62
Another recent application for PPy/TiO2 composites is the development of self-cleaning/anti-bacterial paints that can be used to reduce the time and costs of maintaining the covered surfaces. The smart paint can be developed by adding suitable additives to a polymeric matrix. Nosrati et al. 82 used TiO2/polypyrrole nanocomposites as an additive in water-based polyacrylic latex, obtaining hydrophilic, water-resistant, anti-bacterial (gram-positive bacteria B. anthracis) and photocatalytic (methylene blue) coatings. According to the results, coatings containing 2% and 3% TiO2/polypyrrole (100/10) nanocomposites are proposed as the best formulations. 82
The modification of PPy/TiO2 composites with platinum has been carried out to develop photocatalysts suitable for the production of H2 from water.16,63 Photocatalytic hydrogen production is usually performed using an aqueous solution containing a sacrificial electron donor such as methanol or triethanolamine. For Kandiel et al. 16 the optimal proportions for the ternary hybrid materials are 0.5 and 1.0 wt.% of Pt and polypyrrole, respectively. The amount of H2 generated by the use of the modified platinum composite is three times greater than that obtained with TiO2 doped with Pt. 16 The improved reactivity of the ternary composite is due to the synergistic effect between Pt nanoparticles and polypyrrole, leading to better separation of cargo carriers.16,63
Reaction mechanism
The widely reported mechanism that explains the reactions on the photocatalyst surface includes reduction and oxidation reactions with water, oxygen, and other substances present in the system. 8 When the semiconductor is irradiated with photon energy (hv) greater than or equal to the band gap energy, an electron (e-) is excited from the valence band (VB) to the conduction band (CB) generating the electron-hole pair. In the hole (h+), oxidation reactions occur while in the conduction band, the electron is responsible for the reduction reactions. Among the reactive oxygen species (ROS) that can be produced by redox reactions are the hydroxyl radical (•OH), the superoxide radical (•O2-), and the hydroperoxide radical (•OOH). However, if there is no reaction, the electron-hole pair may recombine, releasing the energy initially absorbed as heat.
The combination of TiO2 with polypyrrole generally decreases the band gap of the final composite concerning the pure semiconductor, consequently increasing the photocatalytic activity in the visible spectrum. The explanation is that the TiO2/PPy composites form p-n junction heterostructures. 57 In this structure, conduction and valence bands of TiO2 are located in energetically favorable positions for synergistic combination with the highest occupied (HOMO) and lowest unoccupied molecular orbital (LUMO) of PPy. 60 A representative scheme of possible activation of TiO2 and PPy composites is shown in Figure 14.

Schematic representation of the possible activation mechanism of composites based on PPy and TiO2.
When the composite is irradiated with sunlight (hv), the electrons can be excited from HOMO to LUMO of polypyrrole and valence band to the conduction band of TiO2 (Eq. (2) and Eq. (3)).28,47,60,62


Since the oxidation potential of polypyrrole (−1.15 V versus NHE) 60 is greater than that of titanium dioxide (−0.5 V versus NHE), 62 the excited electrons are injected into the CB of TiO2 leading to the formation of holes in the HOMO of PPy (Eq. (4)).28,47,60,62

Simultaneously, electrons from the VB of titanium dioxide are promoted to the HOMO of polypyrrole recombining with the holes generated (PPy+) and forming new holes in the valence band of TiO2 (Eq. (5)).28,47

The charge separation improvement and consequently, the increase in photocatalytic activity of composites are related to the generation of a greater amount of excited electrons and holes in the CB and VB of TiO2, respectively.28,35,47
The photoactivation of the composite generates powerful oxidizers (h+) in the valence band responsible for the production of the hydroxyl radical (•OH) through the reaction with water (H2O) or the reaction with ions (OH-) on the particle surface. Oxidation reactions with water and ions are represented by Eq. (6) and (7), 8 respectively.


In the absence of electron acceptors, electron-hole pairs of TiO2/PPy composite may recombine. The oxygen present in the medium and adsorbed on composite the surface captures the photogenerated electrons, thus forming the radical superoxide ion (Eq. (8)). 47 Then, the superoxide radical ion is responsible for the formation of other oxidizing species as represented in Eq. (9) to (13). 83






Photocatalytic reactions can occur from the direct oxidation between an organic compound, adsorbed on the photocatalyst surface and the photogenerated hole (h+), or from its reaction with oxidizing radicals formed from the interaction between these holes and the water molecules or hydroxyl ions (OH-) on the semiconductor surface. 84 On the other hand, inorganic substances can be reduced by direct reaction with the photogenerated electrons (e-) present in the conduction band; undergo indirect reduction caused by the intermediates generated during oxidation in the holes or be oxidized. 85
To evaluate the importance of holes and also of reactive oxidizing species (ROS) in the photocatalytic activity of TiO2/PPy composites, different scavengers have been used: disodium ethylenediamine tetraacetate (EDTA), a hole (h+) scavenger, benzoquinone (BQ), superoxide radials (•O2-) scavenger, tert-butyl alcohol (TBA) and isopropyl alcohol (IPA), both hydroxyl radicals (•OH) scavengers.34,39,65 Li et al. 34 and Silvestri et al. 39 studied the effect of adding different scavengers on the photooxidation of methylene blue dye and the 4-chlorophenol organochlorine pollutant using PPy/TiO2-based composites (Figure 15). The authors adjusted a pseudo-first-order model (Eq. (14)) to the experimental data of degradation to obtain the pseudo-first-order rate constant (k).

where C0 is the initial MB concentration after adsorption equilibrium, C is the concentration of MB at time t, and k is the pseudo-first-order rate constant.

Effect of different scavengers: (a) Photocatalytic degradation of methylene blue (10 mg/L) over macroporous PPy /TiO2 composite (150 mg/L) under simulated sunlight using 1.0 mM of EDTA (disodium ethylenediamine tetraacetate) and TBA (tert-butyl alcohol) as scavengers of holes (h+) and hydroxyl radicals (•OH), respectively; 34 and (b) Photoconversion of 4-chlorophenol (10 mg /L) with PPy/TiO2 (500 mg/L) under simulated sunlight using 0.1 mM of EDTA, BQ (benzoquinone) and IPA (isopropyl alcohol) as scavengers of holes, superoxide radials (•O2-) and hydroxyl radicals (•OH), respectively. 39
The pollutant degradation mechanism seems to occur mainly in the presence of hydroxyl radicals because of the addition of TBA and IPA, hydroxyl radicals’ scavengers, significantly reducing the pseudo-first-order rate constant (Figure 15). The generation of this radical with high oxidative capacity occurs from chain reactions involving oxygen as an electron acceptor and through the oxidation reactions of water molecules and hydroxyl ions. The additions of EDTA and BQ have a smaller influence on the photocatalytic activity, which implies that h+ and •O2− are reactive species present in the medium but not the main active species in these cases.
Concluding remarks
Currently, heterogeneous photocatalysis has been commercially applied in a number of sectors, as deodorization of environments, photocatalytic paints, glasses, and anti-embassing mirrors, among others. Present trends comprise the replacement of artificial light sources (UV lamps) by the sunlight, once the larger share of the technology cost includes the energy generation for electron excitation. Hence, it is essential to develop materials that are also activated by visible light. This theme is particularly relevant in Brazil since the geographic location of the country accounts for the advantage of solar energy potential. In this sense, conducting polymers appear as a class of materials with special properties, able to help to overcome the difficulties encountered during the use of the most used semiconductors, such as TiO2.
The PPy/TiO2 composite has been effectively synthetized by chemical polymerization methods as in situ polymerization, photopolymerization, electrochemical polymerization and molecular imprinting polymerization (MIP). All the aforementioned methods appear to be effective to decrease the recombination rates of electron-hole pairs and the band gap value simultaneously, in comparison to pure TiO2 nanoparticles. Consequently, the composite improves the light absorption in the visible region (400– 700 nm) and enhances the photocatalytic activity. The selectivity of the composite can be significantly improved by the MIP technique, which involves the use of a model molecule identical to the pollutant to be treated. In addition, the doping of PPy/TiO2 with noble metals improves the separation of charges in the semiconductor particle, inhibiting the recombination of photogenerated electron-hole pairs. All advantages cited here are evidenced by the characterization results of SEM, TEM, HRTEM, UV-vis DRS, FTIR, XRD, PL, TGA and electrical properties.
The optimum experimental parameters for different polymerization approaches may vary and have a strong influence on the morphology and properties of the resulting composite. The PPy/TiO2 molar or mass ratio presents great influence in the photocatalytic activity. For larger amounts of monomer compared to semiconductor, the increase of TiO2 quantity in the composite enhances the photocatalytic activity. Still, for larger amounts of TiO2, the addition of PPy generally increases the photoactivity of the composite to an optimal concentration, because an excessive amount of polymer can interfere in the absorption of light in the composite.
According to the literature, the PPy/TiO2 composite increases the photodegradation of dyes over a wide range of operating conditions, i.e. type and concentration of dye (5–20 mg L−1), solid concentration (0.4–1.5 g L−1), type of irradiation (artificial sunlight, artificial visible light and natural sunlight), pH (from acid to basic) and time of reaction. It is important to emphasize that the composite can be applied to the degradation of several compounds and the production of fuels as H2. Therefore, in this work, it was demonstrated that the PPy/TiO2 photocatalysts are stable and efficient, showing great potential for practical applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
