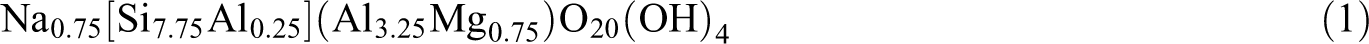Abstract
In the present study, we performed a molecular dynamics simulation of the intercalation of poly(N-isopropyl acrylamide) (NIPAM)3 and poly(N-vinyl caprolactam) (NVCL)3 trimers into Na-montmorillonite (Na-Mt) to evaluate their effects on the interlayer structure and the stability of hydrated Na-Mt. The impact of both trimers on the interlayer species and their dynamics properties at different temperatures in a canonical ensemble (NVT) were investigated. The results showed that the electrostatic forces exerted by Na cations on H2O molecules and the interlayer H2O molecular arrangement are not affected by the rise in temperature after adding both trimers. Trimer addition reinforced the structure of interlayer H2O molecules so that the effect of temperature increase on them became negligible. The structural dynamics evolution of the radius of gyration of both trimers showed the existence of conformation changes when temperature increased. These conformational changes are more complex in the case of (NVCL)3 than (NIPAM)3 due to its large monomers. Both trimers reduced the mobility of interlayer particles with a better inhibition effect obtained for (NVCL)3 compared to (NIPAM)3. The concentration profile of interlayers’ species showed the affinity of Na cations for clay mineral surfaces while H2O molecules moved away. Compared these two trimers, the most stable state of Na-Mt is achieved with (NVCL)3. These results could help highlight the inhibition properties of (NIPAM)3 and (NVCL)3 on hydrated Na-Mt and to predict its stability against changes in environmental conditions.
Keywords
Introduction
Polymer–clay nanocomposites (PCNs) are materials composed of a polymer matrix and nanometer-size clay particles. Their production is an active area of research that benefits from the high surface area ratio of nanoclay particles to great improvement of chemical and physical properties of the polymer matrix. 1 -3 Through nanocomposite formulations, an increase of the elastic moduli, better mechanical properties, improve thermal stabilities, desirable barrier properties, and an increase of biodegradability can be achieved. 4,5 These features make them future candidates for applications in oil and gas production such as drilling fluids and flow assurance. 6 The goal of the preparation of nanocomposites is to achieve a perfect exfoliation of the aggregates of layered silicates which are not easy to be achieved. 3 The discovery of various clays and their wide use in many applications resulted in continuous developments in polymer sciences and nanotechnology. Important progress was made through the intercalation of guest organic molecules such as polymers in the interlayer space of clay mineral to obtain new nanostructure composites with desirable properties to meet the expansion of industrial and economic activities. 7,8 PCN is synthesized using in situ polymerization, intercalation of the polymer from a solution, melts intercalation of the polymer, and sol–gel technique. 7 Poly(N-isopropyl acrylamide) (NIPAM) and poly(N-vinyl caprolactam) (NVCL) are two well-known “smart polymers” belonging to the group of thermosensitive polymers. In aqueous solution, they are sensitive to the environmental conditions (pH, temperature, ionic strength, and electric field, and so on) 9,10 and to chemical changes such as concentration changes, molecular weight, hydrophobic and hydrophilic components, and salt concentration. 11 -16 PNIPAM and PNVCL have different structures of the monomeric units but the same reversible low critical solution temperature (LCST) near 32°C. Thermosensitive polymers, which become insoluble or soluble upon heating, possess an LCST or an upper critical solution temperature, respectively. 9,11,17 Recently, thermoresponsive composite materials were synthesized by incorporating montmorillonite (Mt) into PNIPAM gels to improve their mechanical properties. 18 Autieri et al. 19 performed molecular dynamics (MD) and metadynamics simulation to investigate the conformational feature, structure, and dynamics properties of PNIPAM trimer in aqueous solution. They found that the experimentally observed lower hydrophilicity of isotactic PNIPAM in comparison with the syndiotactic one is related to the conformational entropy. Katsumoto et al. 20 investigated PNIPAM dimer model compounds with computational and experimental methods to understand the effect of tacticity on the polymer hydrophilicity in H2O. Their results showed that the dimer model (DNIPA) compounds with the racemo configuration (r-DNIPA) is more soluble in H2O than DNIPA with the meso configuration (m-DNIPA). Tucker and Stevens 21 studied the effect of PNIPAM length on the LCST in H2O using MD simulations. Their results showed that no coil-to-globule transition for shorter oligomers less than 8 repeating units occurred but the conformation transitions were observed for polymers with more than 11 repeating units. The coil-to-globule behavior of poly{γ-2-[2-(2methoxyethoxy)ethoxy]ethoxy-3-caprolactone} (PMEEECL) was investigated by means of MD simulation in H2O. 22 Results showed that the LCST of PMEEECL occurred at 320 K, in a good agreement with previous experimental results. Deshmukh et al. 23 compared the effect of temperature on the hydration and conformational transition of PNIPAM, poly(acrylamide) (PAA), and poly(ethylene glycol) (PEG). They observed that PAA and PEG do not exhibit a coil-to-globule transition due to existing strong H-bonds of their hydrophilic groups. MD simulations were used to investigate the effect of stereochemistry on the LCST. 24 De Oliveira et al. 25 used MD simulations to study the LCST of different molecular architectures of PNIPAM-based (co)polymers. They found that the collapse of PNIPAM chain upon heating is dependent on the hydration structure around the monomers, the tacticity, and the presence of more hydrophilic acrylamide monomers. Several experimental studies have synthesized and characterized PNVCL for subsequent use as nanofiller, nanocomposite hydrogels, biocompatible thermosensitive PNVCL-clay nanocomposite hydrogels, and drug delivery. 26 -29 Here, we extended the analysis with a molecular approach to the (NIPAM)3 and (NVCL)3 to relate the effect of trimers on the stability of hydrated Na-Mt. Mt is widely used in polymer nanocomposites because of its easy availability, well-known intercalation/exfoliation chemistry, high surface area, and high surface reactivity. The swelling properties of clay minerals are influenced by several factors such as thermodynamic variables (temperature, external pressure, H2O chemical potential, and osmotic pressure) and structural variables (layer charge, charge location, and interlayer cations). 30 In contact with H2O and polar solvent, Na-Mt has a high ability to swell and exhibits two different regimes which are crystalline and osmotic swellings. 30 Clay swelling benefits many commercial applications including drilling mud, catalyst in petroleum engineering, PCNs, and landfill lines in environmental engineering. 31 Many properties of polymer-layered clay have been investigated at macroscopic and microscopic levels, while there still remain significant questions regarding their structure and effect on the interlayer of clay minerals. 7,18,26,32 -35 Despite their contributions to our understanding of PCN and shale inhibitors, there are relatively few comparable investigations involving interaction between Na-Mt, H2O molecules, and trimers. Understanding the impact of (NVCL)3 and (NIPAM)3 addition on hydrated Na-Mt can elucidate the mechanisms underlining the interaction between the small thermosensitive oligomers and clay mineral for the improvement of drilling fluids performance in the hostile environments and assist the design of shale inhibitor and materials with improved properties. Therefore, in this study, we used MD simulations to analyze the interlayer structure and dynamics properties of Na-Mt with (NIPAM)3 and (NVCL)3 intercalation. The effect of both trimers’ addition on the structure of Na-Mt, the interlayer species structures, and dynamics properties is investigated. This study is the first approach to a simulation activity that compares the inhibition effect of (NIPAM)3 and (NVCL)3 into hydrated Na-Mt and their effects on its stability.
Model setup
(NIPAM)3 and (NVCL)3 models
The molecular architecture of (NIPAM)3 and (NVCL)3 consists of a hydrophobic backbone and hydrophilic amide group capped by hydrophobic isopropyl and lactam moieties, as shown in Figure 1. The initial isotactic conformation of the oligomers with a polymerization degree of 3 was constructed using the “Build polymer” module in Material Studio 7.0 software and optimized using the Smart minimizer method.
Snapshot of (NIPAM)3 (a) and (NVCL)3 (b) trimers; the backbone chain is shown in purple, the O atom in red, H atom in white, and N atom in blue.
Na-Mt model
The clay mineral used in this study was taken from the model derived by skipper and coworkers.
36
The unit cell formula is as follows:
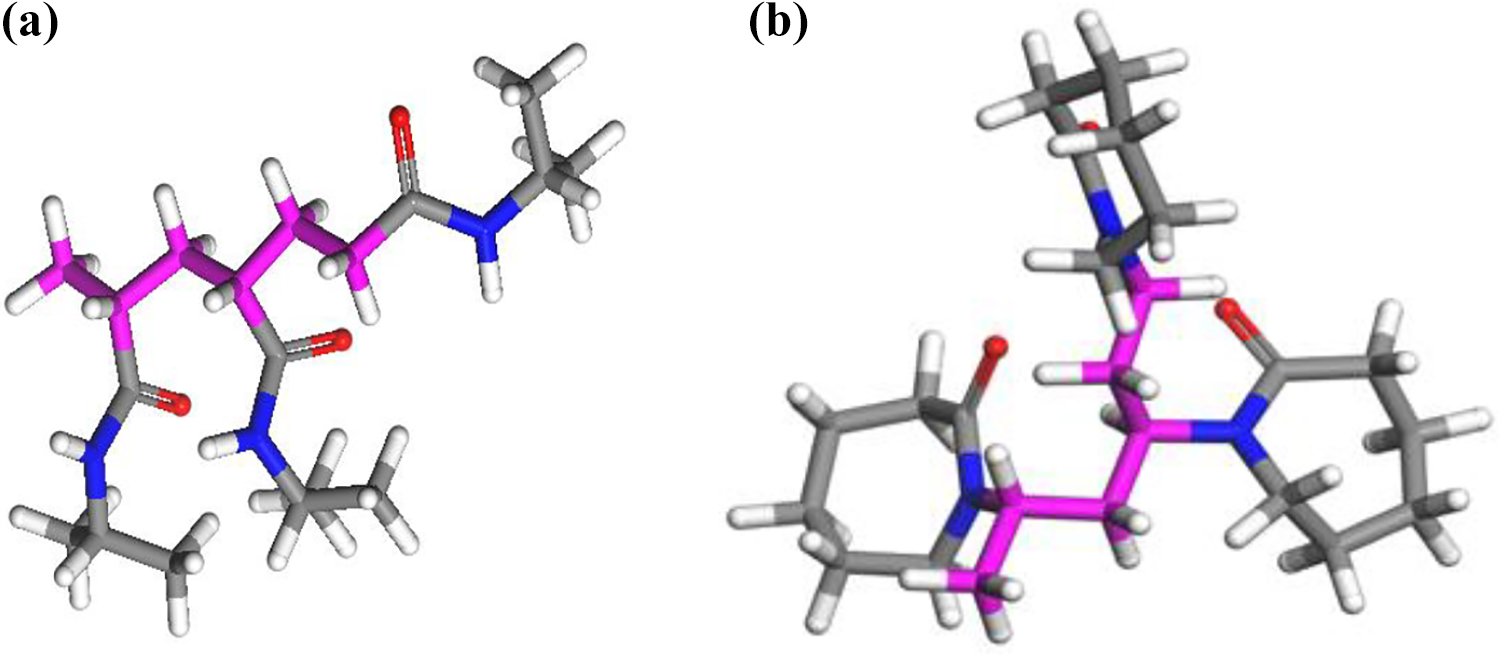
Na-Mt model was constructed, and the atomic coordinates were derived from the space group of monoclinic C2/m with a = 5.23 Å, b = 9.06 Å, c = 9.60 Å (with no H2O in interlayer spaces), c value changed with H2O molecule content between the layers, α = 90°, β = 99.00°, γ = 90°, and the symmetrical L2PC structure. The clay mineral layers were considered as rigid molecules and mirror image. The atomic charges assigned to each atom sites in the clay were given by Smith. 37 Based on the primitive unit cell, the supercell of the model (8a × 4b × 1c) was built (Figure 2). The measure of the three-dimensional, periodic boundary conditions applied to the system was 41.84 × 36.24 Å2, which consists of 1280 atoms in the clay mineral layers equivalent to 32 unit cells and 24 atoms of sodium. Based on this formula, our clay sheets with 32 unit cells have 16 isomorphic substitutions of Al by Mg ion in the octahedral sheets, 8 isomorphic substitutions of Si by Al ion in the tetrahedral sheets, and 24 compensating Na cations in the interlayer region.

A snapshot of Na-Mt 32 unit cells, tetrahedral Si (yellow), O (red), H (white), octahedral Al (pink), octahedral Mg (green), interlayer Na cations (purple).
Computational details
MD simulations were performed under the module “Forcite” of Materials Studio 7.0 software to investigate the effect of (NIPAM)3 and (NVCL)3 addition on the hydration process of Na-Mt at temperature changing from 300, 305, 310, 325 to 335 K. The interaction between the atoms was described by Universal Force Field (UFF) which is a wider applicable force field, and its parameters spread all over the entire element of the periodic table.
38
-40
Metal–oxygen interactions were based on a Lennard-Jones potential combined with electrostatics. For H2O and trimer behavior, the SPC/E model of Berendsen et al.
41
and CVFF force fields were used, respectively. The simulation was performed for two systems (with and without trimer addition) in the canonical ensemble (NVT) in which the volume was constant and temperature changing. In order to analyze the complex system which combines the effect of the confinement (due to the clay) and the effect of interaction (due to the trimers), we built another system (water box) with pure H2O, containing 24 Na cations and trimers (without Na-Mt) in the similar simulation conditions for comparison (Figure 3). We chose to output the atomic positions every 1.0 fs during production runs, the total simulation time was set to 2000 ps, the number of steps was 2,000,000, and the frame output was obtained every 5000 steps. The short-range van der Waals forces were applied with a 15.5 and 12 Å cutoff distance for Na-Mt and the water box, respectively. To calculate the long-range electrostatics, the Ewald summation method was used. The total potential energy was given by the summation of all interaction sites of the system:
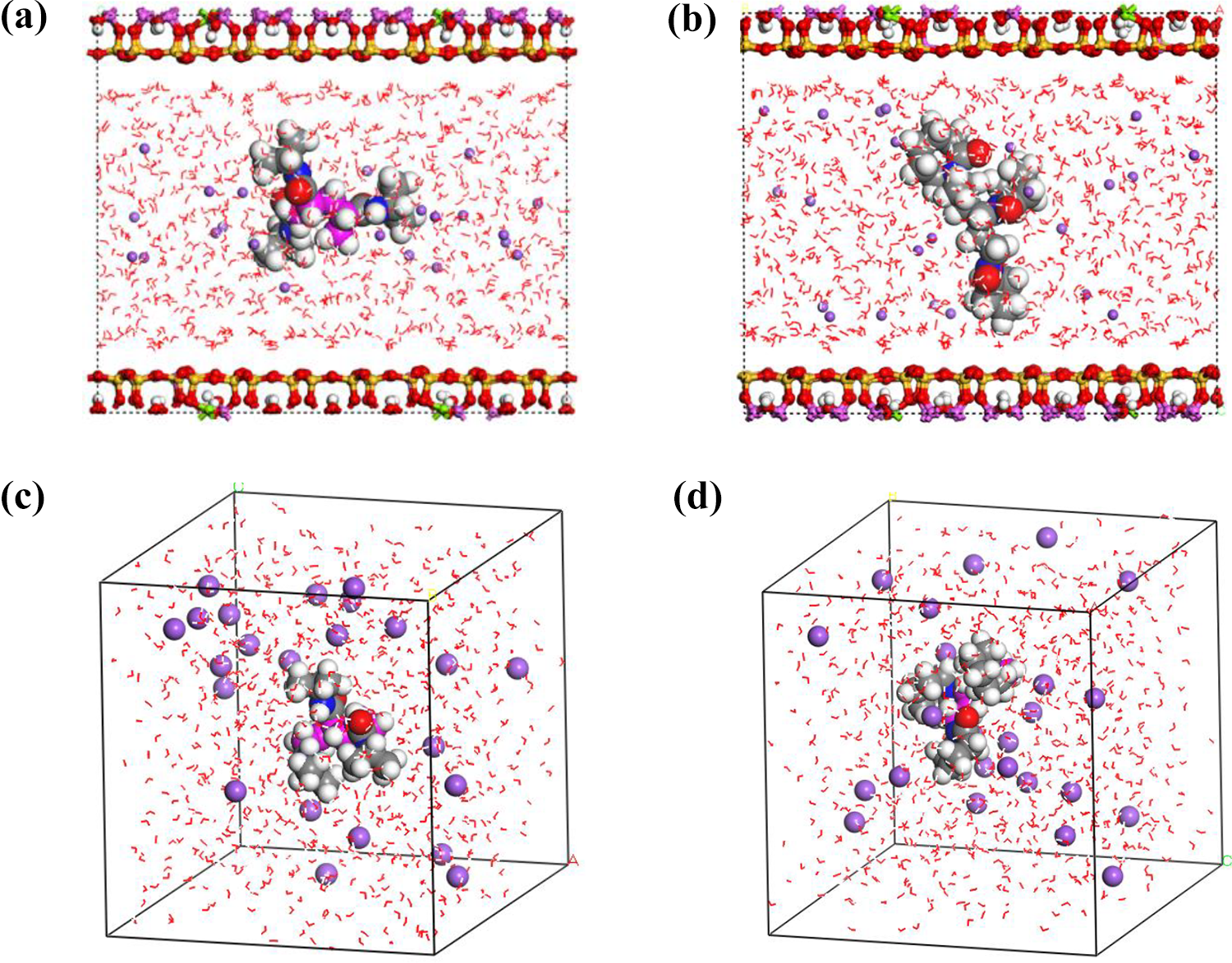
Snapshot of Na-Mt and water box with H2O molecules (red dotted lines), Na cations (purple), (NVCL)3 and (NIPAM)3 trimers are in the center of Na-Mt and the cubic box; (a) and (c) with (NIPAM)3 and (b) and (d) with (NVCL)3.
where qi and qj are the charges on atoms
The potential parameters used for oxygen and hydrogen were those used in H2O. 42 The method of velocity initialization used for the dynamics calculation was Random. The H2O contents considered in this study were 800 H2O molecules for both Na-Mt and the water box. The initial basal spacing was 40 Å for Na-Mt and the size of the cubic box was 30 Å (Figure 3). From the output data obtained, the radial distribution functions (RDFs), radius of gyration, concentration profile, coordination number, hydration radius, and diffusion coefficient profiles were calculated.
Trajectories analysis
The RDF of H2O molecules around the interlayer cations was calculated as follows 43 :
where
where
where mi is the mass of atom i and ri is the position of atom i with respect to the center of mass of the molecule.
The ion hydration number, ion coordination number, and hydration radius were defined as follows 46,47 :
Results and discussion
Interaction between Na cations and H2O molecules after trimers addition
The effect of (NIPAM)3 and (NVCL)3 addition on the interaction between Na cations and interlayer H2O molecules was investigated by analyzing the RDF between Na cations and H2O molecules’ oxygen (

RDF curves of Na-Ow before and after (NIPAM)3 and (NVCL)3 addition at different temperatures into Na-Mt.

RDF curves comparative chart of Na-Ow after (NIPAM)3 and (NVCL)3 addition at different temperatures into Na-Mt.
Hydration parameters of interlayer Na cations before and after trimers addition.

Mt: montmorillonite; NIPAM: poly(N-isopropyl acrylamide); NVCL: poly(N-vinyl caprolactam).
Interaction between interlayer H2O molecules after trimers addition
The structure of interlayer H2O molecules is described by the RDF of
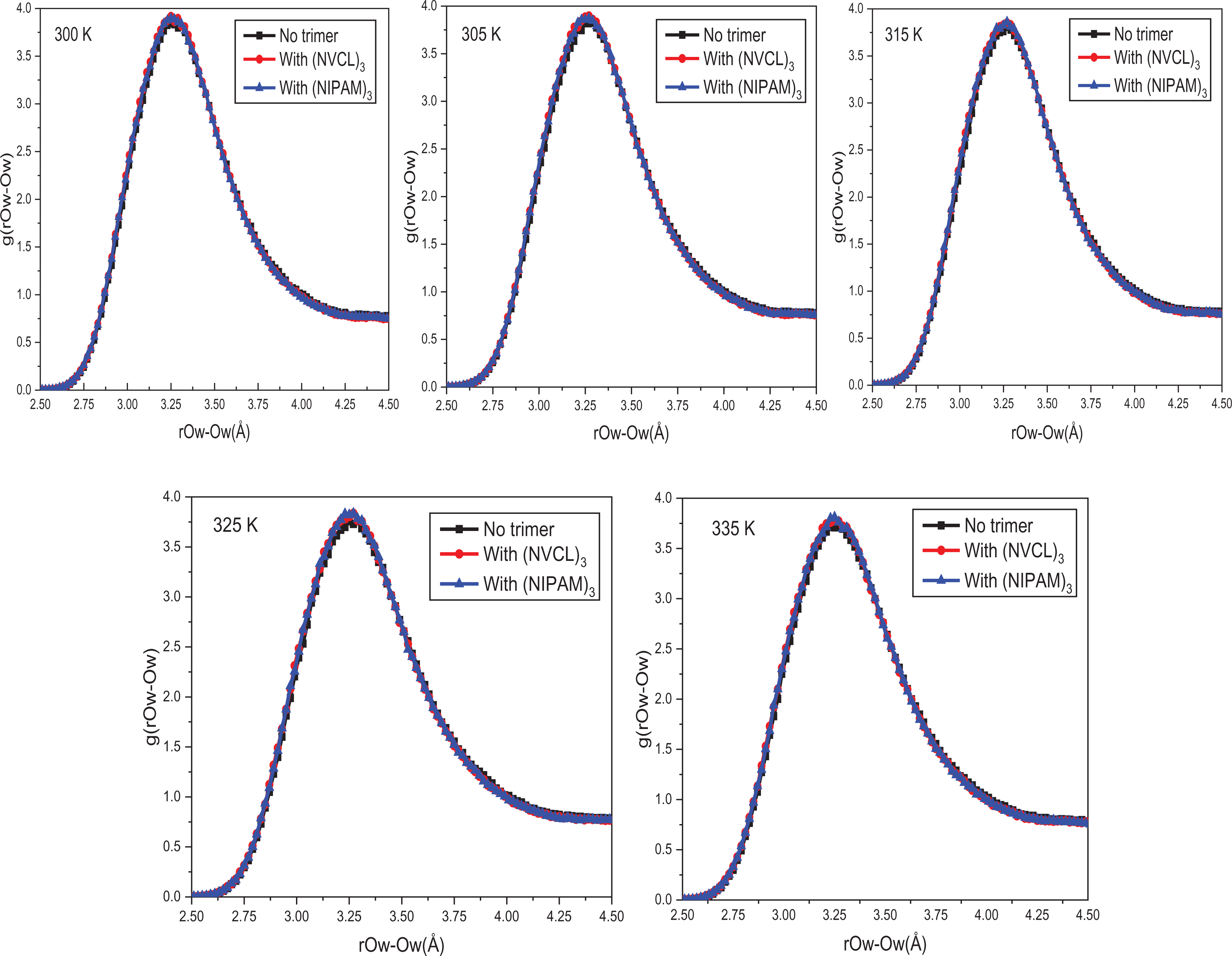
RDF curves of Ow-Ow before and after (NIPAM)3 and (NVCL)3 addition at different temperatures into Na-Mt.
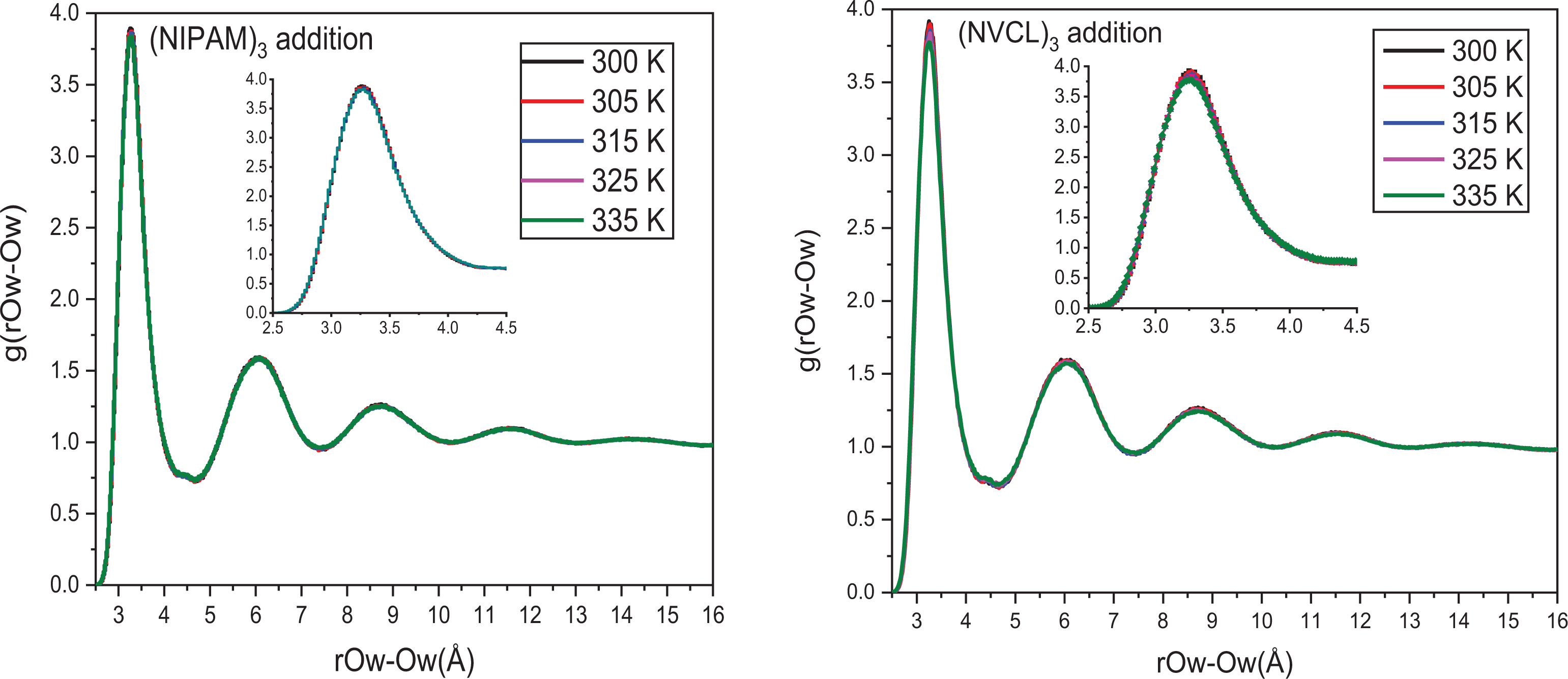
RDF curves comparative chart of Ow-Ow after (NIPAM)3 and (NVCL)3 addition at different temperatures into Na-Mt.

RDF curves comparative chart of Ow-Hw and Hw-Hw after (NIPAM)3 and (NVCL)3 addition at different temperature into Na-Mt.
Ion coordination number of Ow-Ow before and after (NIPAM)3 and (NVCL)3 addition.

Mt: montmorillonite; NIPAM: poly(N-isopropyl acrylamide); NVCL: poly(N-vinyl caprolactam).
Ion coordination number of Ow-Hw and Hw-Hw before and after (NIPAM)3 and (NVCL)3 addition.

Mt: montmorillonite; NIPAM: poly(N-isopropyl acrylamide); NVCL: poly(N-vinyl caprolactam).
Diffusion coefficient of Na cations and H2O molecules after trimers addition into Na-Mt
Unlike the RDF curves studied above, the mobility of Na cations and H2O molecules is greatly affected by the change in temperature, 23,40 and the type of trimers increased with the rise in temperature in both systems of Na-Mt (Figure 9). The decrease of the diffusion coefficient of Na cations and H2O molecules in the system with trimers addition compared to that without trimers addition was observed due to the inhibition effect of both trimers on their mobility. 48 (NVCL)3 exerted a good inhibition effect on the mobility of Na cations and H2O molecules than (NIPAM)3. Trimers exerted strong attractive forces on interlayer H2O molecules which increased the affinity of Na cations for clay surfaces. In the presence of both trimers, most of Na cations diffused in the inner sphere complexes because of the strong electrostatic forces exerted by the negatively charged layers on them. In this position, they were less hydrated by H2O molecules and coordinated by clay surface’s oxygen and H2O molecules. The interactive forces exerted by both trimers on Na cations reduced their mobility and minimized the solvation of clay surfaces. With the rise in temperature, the attractive forces exerted by H2O molecules and clay surfaces on Na cations became weak. Their affinity to clay surface decreased leading to the rise in their mobilities. The diffusion of H2O molecules into Na-Mt depends on the presence of both trimers, Na cations, and the charges on clay mineral surface. (NIPAM)3 and (NVCL)3 have two stages in aqueous solution which are the hydrogen bonding formation and hydrophobic interaction at low and high temperatures, respectively. 49 At low temperatures, the interlayer H2O solution had a higher viscosity due to the solubility of both trimers. Strong hydrogen bonds were formed between H2O molecules and clay mineral surfaces, between H2O molecules and trimers, and between trimers and clay mineral surfaces. 50 As the temperature increased from 300 to 305 K, the hydrogen bonds formed between trimers and H2O molecules became weak leading to the increase in the mobility of H2O molecules. Above 305 K, the interlayer fluid viscosity became lower, the trimers became insoluble, and attractive forces exerted by clay mineral surface on H2O molecules became weak. Hydrogen bonds between H2O molecules and both trimers are broken causing the release of H2O molecules from them, thus increasing the mobility of H2O molecules. 51 As temperature increased, the mobility of H2O molecules in the system with (NIPAM)3 addition remained higher than that of (NVCL)3, because H2O molecules associated with the side isopropyl moieties are released more easily than that of lactam moieties of (NVCL)3 due to the weaker intramolecular and hydrophobic interaction between (NIPAM)3 residues than those of (NVCL)3.
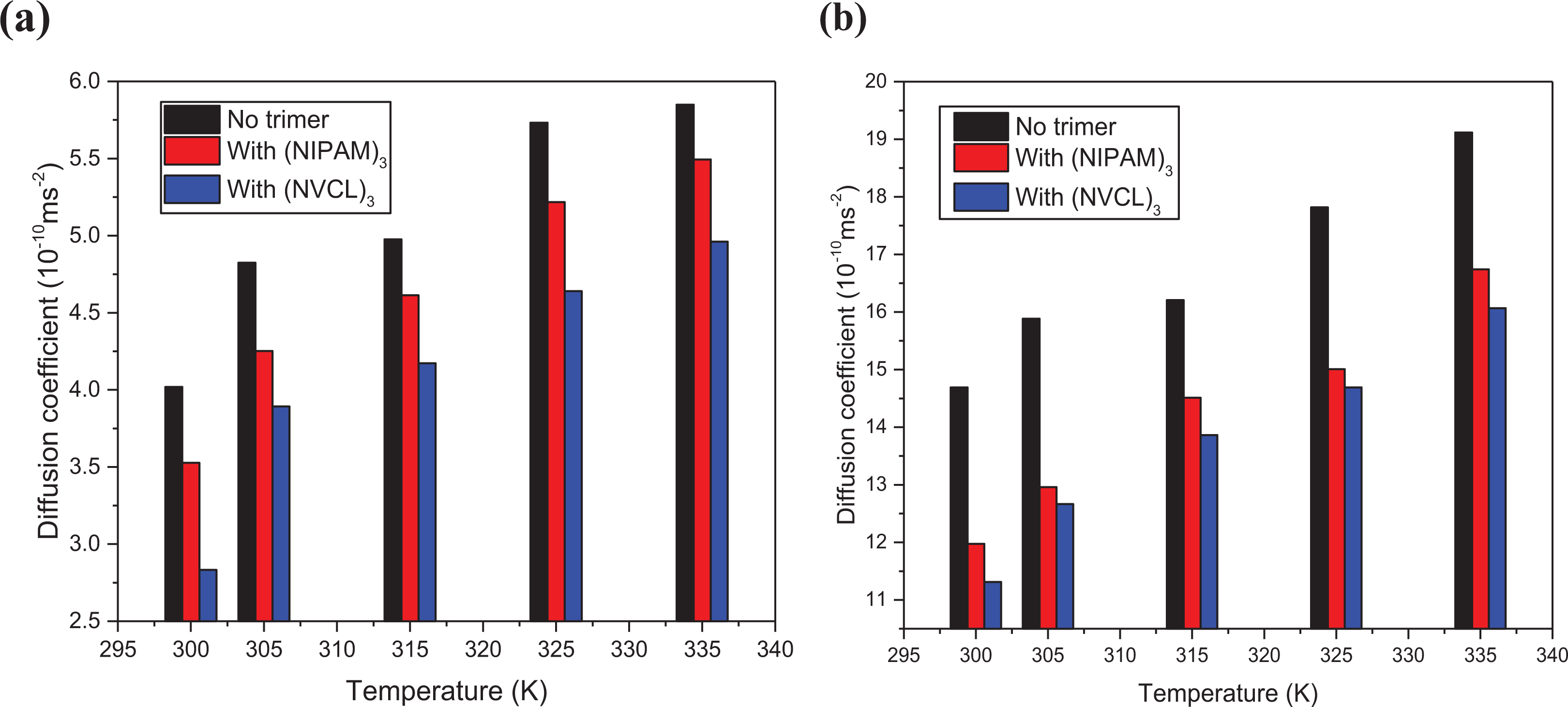
Diffusion coefficient of Na cations (a) and H2O molecules (b) before and after (NIPAM)3 and (NVCL)3 addition at different temperatures.
Concentration profile of H2O molecules and Na cations into Na-Mt after trimers addition
At different temperatures for both systems with and without trimers addition, it was observed that both particles diffused close to Na-Mt surfaces and in the mid-plane after trimers addition (Figures 10 and 11). The distribution of H2O molecules at each temperature after adding trimers remained the same and that of Na cations undergone some changes near the clay mineral surface. 48 These results indicate that Na cations diffusion was not uniform in the interlayer space of Na-Mt. The highest peaks were observed close to Na-Mt surface, indicating that H2O molecules and Na cations diffused close to Na-Mt surface than the mid-plane. In this position, they were subjected to the attractive forces exerted by Na-Mt surface when the temperature was increased. The hydrophilic and hydrophobic components of both trimers remained surrounded by H2O molecules causing the repulsion of Na cations from the center of the interlayer to the position close to clay mineral surface. For the two trimers at different temperatures, the slight difference between their distribution was observed in both systems when temperature increased. This observation shows that temperature increase has no significant effect on the distribution of the trimers in the interlayer space. However, reduced temperature effect resulted on the distribution of H2O molecules and Na cations between the layers of Na-Mt due to the increased in affinity of Na cations for clay mineral surfaces and the strong hydrogen bond formation in H2O molecules grouped in the interlayer.

Concentration profile of Na cations and H2O molecules before and after (NVCL)3 and (NIPAM)3 addition at different temperatures into Na-Mt.

Concentration profile of Na cations (a) and H2O molecules (b) after (NVCL)3 and (NIPAM)3 addition at different temperatures into Na-Mt.
Dynamic evolution of the radius of gyration of (NVCL)3 and (NIPAM)3 into Na-Mt
Figure 12 shows the radius of gyration (Rg) of both trimers into Na-Mt. The decrease in Rg of both trimers with the rise in temperature was observed. This decrease indicates a distinct conformational transition of both trimers into Na-Mt when the temperature increased. The values of Rg obtained for (NVCL)3 remained larger than those of (NIPAM)3, indicating that (NIPAM)3 was more sensitive to temperature, related to the thermodynamic and molecular mechanisms underlying their phase transition. The large value of Rg of (NVCL)3 was due to its molecular structure which contained NVCL monomers. These monomers make difficult the flexibility of its structure, the hydrophobic interaction between (NVCL)3 residues, and the hydration of its hydrophilic compounds compared to those of (NIPAM)3. Large value of Rg was obtained between 300 and 305 K; at these temperatures, both trimers were soluble in the interlayer H2O solution and existed in a coil conformation. Under this conditions, the trimers were surrounded by H2O molecules to form a homogenous liquid phase, thus contributing to the formation of strong hydrogen bonds between them and H2O molecules and between H2O molecules. These interactions promoted their stretching when they laid into the hydrated Na-Mt. Strong hydrogen bonds were formed between H2O molecules around (NVCL)3 than (NIPAM)3. These hydrogen bonds in their surroundings and the electrostatic forces exerted by Na cations maintained them in the extended form in the interlayer. 27,52 With increasing temperature from 305 to 335 K, the hydrogen bonds formed between the trimers and H2O molecules and the attractive forces in the interlayer became weak. The intramolecular and hydrophobic interactions between monomer units of both trimers increased, resulting in the change of their conformation which was observed by the decrease of the Rg curves. The abrupt change from H2O soluble to the insoluble state was due to a relevant rearrangement of H2O molecules in the proximity of both trimers. 25 Below 305 K, the Rg curve of (NVCL)3 remained higher than that of (NIPAM)3. This result indicates that less conformational changes and more interaction between (NVCL)3 and H2O molecules, and between (NVCL)3 and clay mineral surfaces were observed compared to (NIPAM)3 when temperature increased. The trimer (NVCL)3 remained hydrated than (NIPAM)3 during the increase in temperature. Both trimer chains displayed the conformational change in the interlayer of hydrated Na-Mt, but these transitions are more significant for the polymers containing a large enough chain. 8,25,53

Radius of gyration of (NVCL)3 and (NIPAM)3 at different temperatures into Na-Mt.
Conclusion
We have performed MD simulations to investigate the effect of (NVCL)3 and (NIPAM)3 on the structure of hydrated Na-Mt and to analyze the hydration parameters and the conformation change of both trimers in the interlayer to understand their impacts on Na-Mt structure when the temperature increases. The hydration parameters calculated into Na-Mt were compared with those obtained in the pure H2O for validation. The results obtained show that RDF curves of
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (51874343, U1762212, 51874332), the Natural Science Foundation of Shandong Province (ZR2017MEE027), and the PCSIRT (IRT_14R58).