Abstract
In this study, polyurethane (TPU) scaffolds were fabricated using freeze-drying technique and gelatin macromolecules immobilized on the activated surface by oxygen plasma treatment. Scanning electron microscopy (SEM) micrographs indicated an interconnected porous microstructure with randomly oriented pores. According to the results, the diameter of pores increased after plasma treatment and gelatin grafting. Fourier transform infrared spectroscopy illustrated that there is no inappropriate interaction between materials during processing; furthermore, attenuated total reflection Fourier transform infrared spectroscopy confirmed the immobilization of gelatin molecules on the surface of the plasma-treated polymeric scaffolds. Waterdrop contact angle analysis presented that wettability and hydrophilicity of constructs increased after grafting gelatin on the activated surface. Phosphate-buffered saline absorption and hydrolytic biodegradation enhanced after surface modification of the polymeric samples. Cellular behavior demonstrated better adhesion and spreading after grafting gelatin of oxygen plasma-treated constructs. No evidence of toxicity was observed for 7 days. DNA content determined that the number of viable cells increased in TPU-gelatin matrixes after 1 day in contrast with TPU scaffolds. Based on results, oxygen plasma treatment can create an activated surface to graft gelatin macromolecules and achieve optimum physicochemical, mechanical, and biological features for the neo-tissue formation.
Introduction
Interconnected porous scaffolds provide a 3D structure to improve cell–biomaterials interactions and induce regeneration of defects.
1
Tissue engineering constructs can support cellular processes such as migration, proliferation, and differentiation by simulating extracellular matrix (ECM).
2,3
According to the requirement of target tissue such as physicochemical, mechanical, or biological needs, different types of materials are applicable in reconstruction issues. Besides, the balance between the biodegradation rate of scaffolds and the regeneration rate arise is a critical topic in the fabrication of engineered substrates.
4,5
In this regard, material type can be an effective factor. The used materials in soft tissue engineering are categorized in natural and synthetic polymers such as gelatin,
6
chitosan,
7
poly(lactic-
Blood compatible materials have developed owing to the formation of the intima in the lumen after the application of biomaterials in contact with blood. TPU has been extensively used as hemocompatible 10 material in vascular grafts, 11 myocardium tissue engineering, 12 catheters, 13 and so on. In this regard, firstly, endothelial layer will be formed after interaction between biomaterials and cells to gain hemocompatibility. Nevertheless, because of weak cell–TPU interactions and inert properties of this material, 14 surface modification can improve cytocompatibility 9 and its interaction with the surrounding tissue. Collagen is the major component of ECM, but due to its antigenicity, gelatin was replaced. Gelatin is the main product of collagen hydrolysis has developed, and its sequence of Arginylglycylaspartic acid (RGD) can enhance cell adhesion to biomaterials. 15 So it is expected that the cellular interactions can be improved after modification of the TPU surface via gelatin.
Besides the used materials, the scaffolding technique is an essential issue in providing physicochemical, mechanical, and biological properties. There are several methods to fabricate tissue engineering scaffolds, such as freeze-casting, 16 freeze-drying, 17 3D printing, 18 electrospinning, 19 and phase separation. 20 In this study, the freeze-drying technique was utilized to achieve bio-originated and interconnected porous structures, the same as other investigation. 21
Gelatin was immobilized on the surface after plasma treatment to modify the surface of freeze-dried scaffolds composed of TPU. Plasma technology is a cost-effective approach for chemical surface modification of scaffolds without any effect on bulk characteristics. Therefore, it can promote biocompatibility, cellular adhesion, wettability, or decrease surface friction. 22
Guan et al. 23 described that polyester urethane urea/gelatin scaffolds have desirable mechanical properties for cardiovascular treatment. However, results demonstrated that loading basic fibroblast growth factor on gelatin can enhance the degradation rate of polymeric scaffolds fabricated by thermally induced phase separation. Rockwood et al., 24 studies indicated the production of elastomeric TPU nanofibrous scaffold with a range of fiber diameter form several hundred nanometers to tens of microns. Designed scaffolds enhanced degradation rate compared to TPU films that originate from the increased surface area. In a novel study by Stankus et al., 25 infiltration and high cellular density of smooth muscle cells were obtained by cellular electrospray during scaffold formation. Results indicated that the electrospraying technique never affects cellular viability in the presence or absence of TPU nanofibers; furthermore, perfusion bioreactor improves cellular elongation and distribution compared with static culture. A new treatment procedure investigated by Liu et al. 26 to understand the effect of surface modification of polymeric scaffolds. Their experiment demonstrated that gelatin macromolecules entrapped well on the surface of poly(α-hydroxyl acid) films, so hydrophilicity of the constructs increased. Additionally, cellular attachment and proliferation were supported by modified samples.
In this study, bio-originated TPU matrixes were fabricated by the freeze-drying method. Created active surface via oxygen plasma treatment provides a condition to immobilize gelatin macromolecules on the surface of scaffolds that lead to the improvement of surface hydrophilicity. Finally, biological, mechanical, and physicochemical features of the scaffolds evaluated to determine the effect of surface treatment on properties, and finally introducing the most suitable microstructure for further studies in the tissue regeneration area.
Materials and methods
Materials
TPU (Estane 5701) was purchased from Lubrizol Specialty Chemicals Manufacturing (Shanghai) Co Ltd (China). Gelatin (9000-70-8, weight-average molecular weight (
Preparation of scaffolds
TPU solutions with concentrations of 5% (w/v) were prepared by dissolving TPU in 1,4-Dioxane at 50°C for more than 10 h. Gelatin solutions with concentrations of 2% (w/v) were prepared by dissolving gelatin in PBS for 1 h. Then, the TPU solutions were transferred into a cylindrical mold. All of the samples were frozen at −20°C for 24 h. Frozen scaffolds were freeze-dried (Pishtaz Engineering Co., Iran) at a temperature of −55°C and pressure of 0.6 mbar for 48 h.
After the freeze-drying operation, the scaffolds were prepared for plasma-oxygen treatment. Plasma technique was done about 2 min with an oxygen injection speed of 15 ml min−1, pressure about 8 mbar and power of 60 W. Thirty minutes after plasma treatment, all the samples were put in gelatin solution for 1 h; then, were transferred to an oven at 37°C for 48 h.
Characterization of freeze-drying scaffolds
Microstructure observation
The microstructure of freeze-dried TPU scaffolds before and after gelatin grafting was evaluated by scanning electron microscopy (SEM, Stereoscan S 360-Leica, UK) that operated at the acceleration voltage of 20 kV. All samples were coated with a thin layer of gold to produce a conductive surface.
The diameters of the pores were measured by image analyzer software (KLONK Image Measurement Light, Edition 11.2.0.0). Twenty-five measurements in each micrograph were measured to find the correct average and standard deviation.
The density of the TPU scaffolds was identically computed by using equation 1, where

Finally, the porosity of scaffolds was calculated by using equation (2).

FTIR and attenuated total reflection FTIR spectra
TPU and TPU-gelatin scaffolds were assessed by both Fourier transform infrared spectrophotometer (FTIR, Nicolet Is10, Thermo Fisher Scientific, Waltham, Massachusetts, USA) and attenuated total reflection Fourier transform infrared (ATR-FTIR, Nexus, Nicolet) spectrophotometer. The spectra were obtained in the range of 400–4000 cm−1 with a resolution of 4 cm−1 and 120 scans.
Mechanical behavior
Mechanical stability of the final scaffolds in dry condition was determined by a universal tensile strength test machine (Santam, STM 20, Iran) with a crosshead speed of 250 mm min−1. All the samples were cut into 20 cm in depth. The results of five experiments were reported as a mean ± standard deviation.
Hydrophilicity behavior
Contact angles were measured with deionized water using a sessile drop method (Kruss, G10, Germany) at room temperature. All the contact angles were determined by averaging the values measured at five different locations on each sample surface.
The swelling performance of polymeric scaffolds was specified by calculating the swelling ratio. So, the samples weighted (

Hydrolytic biodegradation
The biodegradation study of the freeze-dried matrixes was followed by calculating the biodegradation rate after immersion the samples in the PBS solution. The scaffolds were weighed (

Cells–scaffold interaction
The cell–biomaterials interaction was determined by culturing fibroblast cell lines (L929) on the polymeric scaffolds to evaluate cellular proliferation. In brief, scaffolds were seeded with 6 × 105 cells, covered by Dulbecco’s modified Eagle’s medium (Gibco-BRL, Life Technologies, Grand Island, New York, USA) supplemented with 15% fetal bovine serum (Gibco-BRL, Life Technologies), 100 g mL−1 penicillin–streptomycin (Gibco-BRL, Life Technologies), and incubated in 37°C, 5% CO2, and 95% humidity for 7 days. The cellular morphology was determined by SEM micrographs. Then, all the cell-seeded scaffolds were washed with the PBS solution three times, fixed in 3% glutaraldehyde and 2% osmium tetroxide, and finally washed with the PBS solution. Additionally, fixed samples were dehydrated by exposure to an ethanol solution (50, 70, 85, 90, and 100%), and let dry in air. At last, all the samples were coated with gold to observe with SEM microscopy. The proliferation of L929 cells was determined using the methyl thiazolyl tetrazolium assay. Therefore, 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT): cell culture medium (1:5) solution was added to each well and incubated in the mentioned condition. The precipitates were resuspended in dimethyl sulfoxide. The absorbance was evaluated using a microplate spectrophotometer at a wavelength of 570 nm. 30 It should be noted that polystyrene surfaces on the cell culture dish were selected as the control group in this study. Then, 20,000 cell cm−2 3-day transfer, inoculum 3×105 cells (3T3) fibroblast cells were seeded on both grafted and ungrafted polymeric scaffolds for 1 h and 4 days to evaluate cellular adhesion rate. At these time points, cell-seeded samples were rinsed with PBS to remove non-adherent cells. After that, cells were lysed by mammalian protein extraction reagent solution and the number of adherent cells determined by dsDNA assay (dsDNA Assay Kit; Invitrogen Corp., Carlsbad, California, USA).
Statistical analysis
Data were processed as the mean ± standard deviation of at least five experiments. Statistical analysis was performed using one-way analysis of variance and Tukey’s test with significance reported when
Results and discussion
Microstructure observation
Freeze-drying process is extensively used in the fabrication of polymeric scaffolds and consists of three main steps, freezing operation, sublimation of ice crystals, and desorption of frozen solvent in the depth of samples. 31 In the freezing stage, nucleation of ice crystals and formation of the network microstructure will be observed. This process will be followed by sublimating frozen solvent crystals, and it terminates to formation of interconnected porous scaffold. 31,32 Herein, the pore size distribution and surface morphology of freeze-dried scaffolds determined by SEM micrographs. Besides, the effect of grafting gelatin after oxygen plasma treatment was compared with the unmodified constructs. Figure 1(a) to (d) illustrates SEM micrographs of freeze-dried TPU scaffolds before (Figure 1(a) and (b)) and after (Figure 1(c) and (d)) gelatin immobilization. Results (Figure 1(a) to (d)) show interconnected porous microstructure with randomly orientated pores in both test groups, so that the uniformity of pores increased after oxygen plasma treatment and gelatin grafting. Results of equation (2) indicated 95% pores which provide vascularization, absorption of nutrients, and removing cellular waste products. 33 Moreover, pore size distribution (Figure 1(e) and (f)) demonstrated average diameter of pores was found with a range of 50–130 µm and 50–150 µm for TPU scaffolds before and after plasma treatment and gelatin grafting, respectively, in both horizontal and vertical directions. The similar diameter of pores in horizontal and vertical directions represented that polymeric scaffolds have uniformity in size and shape even though they contain randomly orientation. After plasma treatment, the pore size distribution tended to increase maybe because of activation of the surface by shockwaves. According to the results, scaffolds can achieve suitable pore size to support cellular interactions with the matrix.
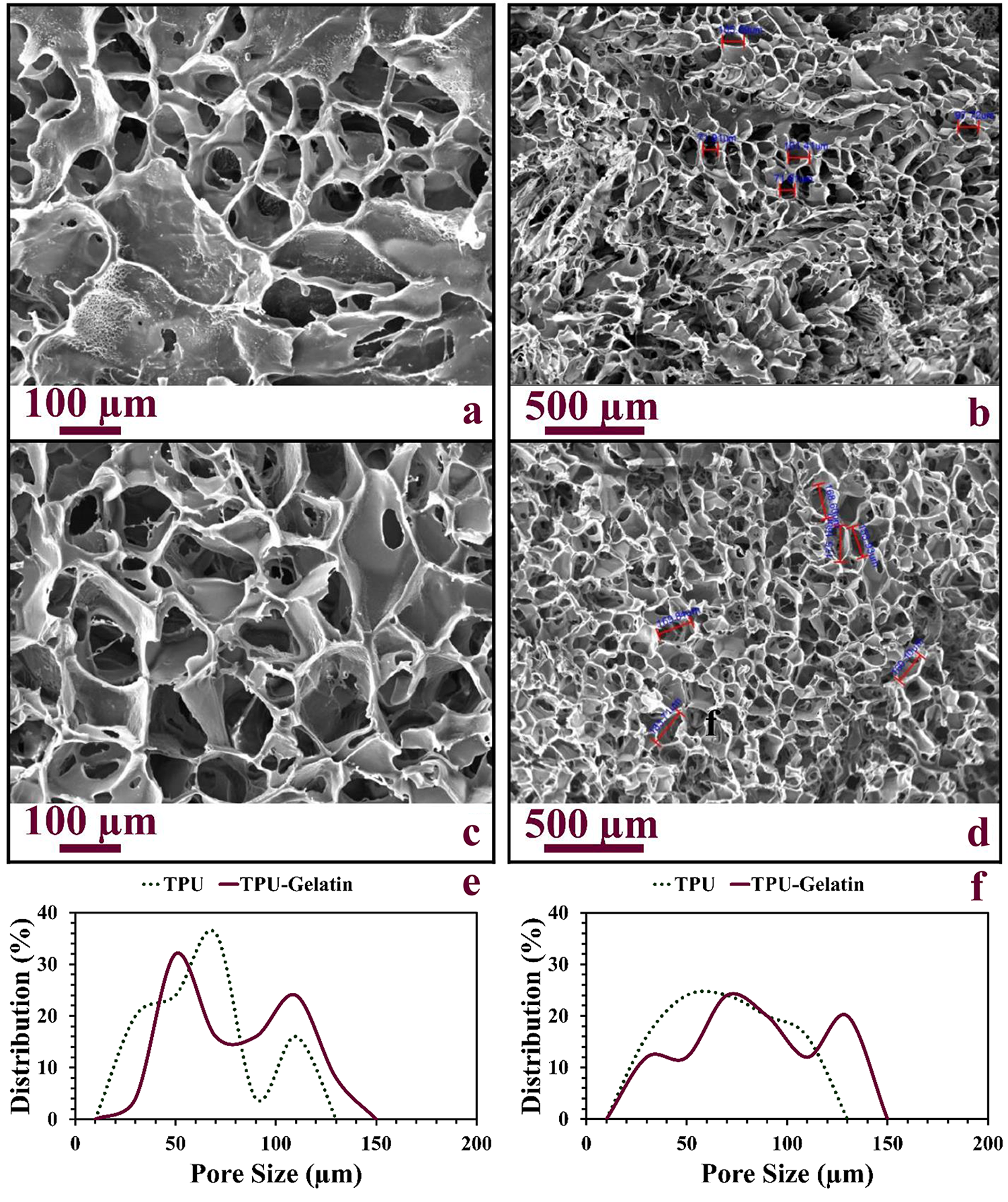
SEM micrographs of freeze-dried TPU before and after plasma treatment: (a and b) TPU scaffolds and (c and d) TPU scaffolds after gelatin grafting. The pore distribution of freeze-dried TPU and TPU-gelatin scaffolds by image measurement software: (e) horizontal measurement and (f) vertical measurement.
FTR and ATR-FTR spectra
FTIR spectroscopy determines chemical groups by radiation of electromagnetic waves in the range of 4000–400 cm−1. Hitting of infrared (IR) in a given sample can excite the proper bonds in defined wave number and different modes such as stretching, contraction, and bending. In contrast, ART-FTIR could be an appropriate method to determine chemical bonds in thin films. 22 Hence, the characterization of modified and unmodified samples followed by both mentioned methods. Figure 2 demonstrated FTIR and ATR-FTIR spectra of TPU scaffolds before and after gelatin grafting and schematic of grafting gelatin on polymeric plasma-modified scaffolds. The characteristic absorption peaks of the TPU are observed at 3306 cm−1 (N–H stretching frequency), 2925–2852 cm−1 (–CH2– and –CH3 stretching frequencies), 1731 cm−1 (carbonyl urethane stretching), 1526 cm−1 (CHN vibration), 1223 cm−1 (coupled C–N and C–O stretching), and 1079 cm−1 (C–O stretching). 34 The amide I and amide II peaks related to gelatin was detected at 1650 and 1538 cm−1. 35

FTIR and ATR-FTIR spectra of TPU, gelatin, and TPU-gelatin (a), schematic of plasma treatment to graft gelatin macromolecules (b).
Cen et al. 36 expressed ionization of oxygen gas and bombing the surface leading to activation of the polymeric surface. The activated surface provides the proper places for covalent immobilization of gelatin, as represented in other investigation. 9 ATR-FTIR spectra confirmed the existence of all the above-mentioned peaks. Furthermore, based on the observation of Kim et al. 37 and Kang et al., 38 the methyl group of TPU is responsible for covalently coupling with gelatin.
Mechanical behavior
Although biocompatibility and biodegradability of the porous scaffolds are critical to support the cellular process, the mechanical requirements are essential to keep the integrity of the matrix during the regeneration. 39 So the mechanical behavior of the construct is one of the most critical points that could be analyzed. Elongations at break of current TPU scaffolds decreased from approximately 478.2% to 267.3% after surface modification (Figure 3). As a consequence, the elongation of scaffolds declined with the immobilization of gelatin, which was similar to the relevant report. 40 Thus, it could conclude that the grafting of the gelatin macromolecules slightly decreased the mechanical properties of scaffolds with thermoplastic elastomer nature and remarkable flexibility. 41 A slight increase in pore size distribution after immobilization of gelatin can be the other reason for the reduction in toughness and production of the brittle construct, similar to other investigation. 9 The presence of gelatin polymer improved the wettability and absorption behavior of scaffolds to support cellular interaction such as absorption of nutrients and excretion of waste products, 35 so this slightly negative effect on mechanical performance can be ignored.

Load-elongation curves (a) and maximum elongation (b) of freeze-dried TPU and TPU-gelatin scaffolds. Statistical differences are considerable between TPU and TPU-gelatin scaffolds.
Hydrophilicity behavior
Hydrophilicity of tissue engineering scaffolds plays a remarkable role in enhancing cell viability, proliferation, and revascularization. 40,42 The superhydrophobicity of TPU 43 can confront the desirable cellular interaction. Herein, the gelatin macromolecules were grafted onto the surface after oxygen plasma treatment to compensate for this defect. The other studies also demonstrated the influence of gelatin on the improvement of hydrophilicity. Figure 4(a) and (b) indicated that the water drop contact angle of freeze-dried TPU scaffolds before (a) and after (b) gelatin grafting. Based on the obtained results, the contact angle decreased from 103° to 70° after plasma treatment and gelatin grafting. These results illustrated that surface modification induce the hydrophilicity to the scaffold 44,45 owing to the molecular structure of gelatin that contained amine and carboxylic functional groups. 40

Wettability of freeze-dried TPU (a) and TPU-gelatin (b) scaffolds by water drop contact angle measurement. The swelling ratio of freeze-dried TPU and TPU-gelatin scaffolds during 2, 4, 6, and 24 h (c). Significant differences observed all the days between TPU and TPU-gelatin constructs.
The swelling behavior of the freeze-dried scaffolds influences its cellular viability because of nutrient and waste product transfer. 46 The capacity of scaffolds in the absorption of PBS was represented in Figure 4(c) as a function of time of various scaffolds. Based on the results, these values were 271.64% and 645.35% for TPU and modified TPU scaffolds, respectively, after 24 h of incubation. The polymeric samples absorbed a significant amount of PBS within the first 2 h, and then pass a slight increase until the end of the test. The fast swelling was observed because of interconnected pores in polymeric scaffolds that support PBS diffusion. Swelling behavior might be due to hydrophilic functional groups in gelatin structure and their ability to interact with water molecules. 47 Besides, the aminolysis reaction of the modified surface with water can be the other reason for the enhancement of absorption capacity. The same results were observed in Zhu et al. observations. 48
Hydrolytic biodegradation
Figure 5 demonstrated SEM micrographs of scaffolds at 60 days after incubation in PBS (Figure 5(a) and (b)) and the biodegradation profile of scaffolds as a function of incubation time at 37°C (Figure 5(c)), respectively. SEM micrographs of degraded scaffolds indicated that both scaffolds keep their porous structure well, but TPU samples resist degradation owing to weak interaction with water molecules. In contrast, breaking down the pore walls increased in modified scaffolds. Results of the mass loss rate prove the SEM observation in which TPU-gelatin samples degraded rapidly in comparison with TPU one. It could happen because of larger pores in treated constructs, a higher density of hydrophilic functional groups, and lower mechanical behavior. So higher absorption of PBS led to rapid biodegradation; besides, the more bitterness resulted in collapsing the structure sooner. Linhardt et al. 49 presented that covalent immobilization of Heparin on the activated surface of TPU scaffolds improves the stability of constructs compared with the other methods of modification. Accordingly, tissue engineering scaffolds improve cellular adhesion, proliferation, differentiation, and so on by simulating a natural ECM. Moreover, tissue synthesis and formation of new ECM, followed by gathering the cells and degrading the scaffolds with a proportional rate. 50 Therefore, the rate of biodegradation is one of the key factors which is studied in this investigation. Accordingly, biodegradation of the constructs takes place very slowly but proportional to our needs. Because of the sink condition established in the body, the tissue response is expected to be mild. 51

SEM micrographs of biodegraded TPU (a) and TPU-gelatin (b) scaffolds after 8 weeks. Hydrolytic biodegradation profile of freeze-dried TPU and TPU-gelatin scaffolds (c). Except for the first week, considerable statistical differences observed during all the weeks.
Cells–scaffold interaction
Cellular proliferation behavior was evaluated by seeding L929 cells on both TPU and modified TPU constructs. Figure 6 demonstrated the SEM micrographs of scaffolds after cell culture. In TPU scaffolds (Figure 6(a)), no cellular elongation was observed that was affected by the superhydrophobic and hemocompatible nature of this material. In fact, the scaffolds prevent from cellular adhesion and spreading. In contrast, bioactive molecules such as gelatin increase wettability and water absorption of constructs. Therefore, grafting gelatin after plasma treatment influences cellular behavior due to the mentioned features, so cytocompatibility and cellular spreading improved after combined plasma modification and grafting (Figure 6(b)). Besides, Gao et al. 52 indicated that enhancing the level of gelatin increases cell proliferation ratio. Moreover, the bilayered structure of polymeric scaffolds will prevent from cell adhesion in the center and provide interaction of scaffolds with surrounding tissue to improve adhesion if they applied in soft tissue regeneration such as blood vessels. 53 The viability of L929 fibroblast cells on the scaffolds after 1, 3, and 7 days was analyzed by MTT assay and measuring the metabolic reduction of 3-(4,5-dimethylthiazol-2-yl)-2,5 diphenyltetrazolium bromide and its effect on changing the solution color. As shown in Figure 7(a), percent of viable cells due to mitochondrial dehydrogenase activity represented biocompatible microstructure of polymeric samples. In other words, during each time point, more than 85% of cells were alive compared with a control group, which confirms the reality of the previous sentence. This result indicated that although cells could not spread on the surface of TPU scaffolds and secrete their products, these samples are the biocompatible and rounded shape of cells is because of superhydrophobicity and lack of adherent surface. Therefore, lack of adhesion leads to a decrease in the number of viable cells as a function of time since there is no appropriate condition for the formation of new ECM which is the essential mechanism in a cellular process. Moreover, increasing cell viability in TPU-gelatin scaffolds compared with ungrafted ones showed the better cytocompatibility of surface after modification. Accordingly, grafted samples support cellular proliferation, and the number of viable cells increased through passing the time after grafting gelatin. Besides, DNA content determined that the number of viable cells increased in TPU-gelatin samples after 1 day compared with 4 h, while the weak adherent surface on TPU scaffolds decreases the number of live cells (Figure 7(b)) that confirm the above-mentioned events.

SEM micrographs of TPU (a) and TPU-gelatin (b) scaffolds after seeding L929 cells after 7 days.

Cytotoxicity of the scaffolds was determined by MTT assay after 1, 3, and 7 days of cell culture (a). There are no differences between the cellular viability of polymeric scaffolds. Cellular adhesion rate on the polymeric scaffolds was assayed by determining DNA content after 4 and 24 h (b).
Conclusion
In this study, TPU scaffolds were synthesized by freeze-drying technique and surface treated by oxygen plasma to graft gelatin macromolecules. The porous interconnected microstructure was found for all the polymeric scaffolds in which surface treatment increased average pore size distribution slightly. Although grafting gelatin presented predictable adverse effects on mechanical properties due to the increasing average size of pores and poor mechanical characteristics of gelatin, it enhanced hydrophilicity, wettability, water uptake, and hydrolytic biodegradation that arise from hydrophilic nature of gelatin and presence of amine and carboxylic groups. Surface modification of TPU samples provided appropriate properties to support cell adhesion and spreading. In brief, it could be concluded that TPU scaffolds with gelatin grafting showed incipient features to further in vivo studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
