Abstract
The purpose of the study was to investigate the presence of mascara deposits on polymeric siloxane-hydrogel contact lenses (CLs) after in vivo 8 h of wear and to compare the affinity to mascara of two siloxane-hydrogels through in vitro contamination tests. The siloxane-hydrogel CLs were the Filcon V monthly replacement OPEN30 and the Delefilcon A daily disposable DAILIES TOTAL1 showing a water gradient from the core to the outermost hydrophilic surface layer. The investigated cosmetic was a blue mascara. Scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDX) were carried out on new CLs, CLs exposed in vitro to mascara, and CLs worn for 8 h by mascara wearers (after rinsing the CLs in sodium chloride solution). Images by an optical microscope were acquired and processed on new CLs and CLs treated in vitro. EDX analyses on mascara allowed to characterize its elemental composition including aluminum (Al), which is neither a tear nor a CL component, thus being a marker of the presence of mascara deposits on polymeric siloxane-hydrogels. Adsorbed deposits containing Al were observed by SEM/EDX on all worn CLs of both materials, which showed a special affinity for the mascara components, more marked than for the tear components. By processing the images taken by optical microscopy after in vitro tests, the presence of mascara deposits was found to be more than two times higher in Delefilcon A compared to Filcon V CLs.
Introduction
Eye cosmetics are mainly used by women and two-thirds of the wearers of contact lenses (CLs) are women, 1 who start to wear CLs quite early from teenage. 2,3 This is one of the reasons why there is a certain interest in the study of the interaction between these products and the polymers of which the CLs are made. Understanding how CLs interact to these eye cosmetics products is expected to provide further understanding of patient comfort and safety.
Since siloxane-hydrogels are more lipophilic than conventional hydrogels, 4 -7 it is more likely they could absorb also cosmetics. Indeed, Tsukiyama et al. 8 reported deformation and swelling of siloxane-hydrogel CLs without plasma polymerization coating when cosmetics and cleansing oil were directly applied on the lenses. The impact in vitro of cosmetics on the shape, power, and optical performance of siloxane-hydrogel CLs was investigated by Luensmann et al. 9 Diffusivity of the dye component of a purple eye shadow was also found to be typically higher in siloxane-hydrogels than in hydrogels. 10 In general, the siloxane moieties promote the transport of oxygen (O) and carbon dioxide across the lens. Therefore, the advent of siloxane-hydrogel materials for CL use shifted the main topics of research from the transport of O and from hypoxic complication to surface interactions with contaminants and tear residues.
This work concerns CL wearers and the interaction between mascara and soft siloxane-hydrogels. Mascara is a cosmetic commonly used to enhance the eyelashes by lengthening, curling, coloring, and thickening them. It is believed that ancient Egyptians started using it. Nowadays, mascara is one of the most used cosmetics around the world, often used on daily basis.
Mascaras available on the market, as all commercial cosmetics, undergo safety testing for human use. However, some ingredients could be accidentally absorbed into the eye and, potentially, could have negative impact on the ocular health. Specific ocular changes associated with this eye cosmetic have been described in the literature. Ciolino et al. reported three cases of anterior eye complications secondary to long-term mascara use. 11 Two patients had multiple pigmented conjunctival lesions represented by nonmelanocytic pigment granules within conjunctival stroma cells and pigment clumping around a punctal plug. The third patient had canalicular obstruction from a mascara-laden dacryolith (defined as “dacryomascaralith”). In 2011, Clifford et al. published a case of exogenous nasolacrimal sac pigmentation secondary to mascara use due to mascara deposits passing down the nasolacrimal duct. 12 Another condition, described as a conjunctival mass (called “mascaroma”) that clinically simulated a melanoma, was described by Shields et al. 13 This lesion consisted of a portion of conjunctival epithelium with a keratin plaque that contained multiple dark particles exhibiting birefringence with polarized light. The final diagnosis was conjunctival hyperkeratosis containing foreign bodies compatible with mascara deposition. Moreover, cases of allergic contact dermatitis of the eyelids due to mascara were discussed by Gallo et al. 14 and, more importantly, also corneal ulcers associated with the use of mascara contaminated with Pseudomonas aeruginosa were reported. 15 In general, either ocular adverse reactions or discomfort symptoms, such as stinging and burning, secondary to eye cosmetics, can be found in the literature. 16 -21 For example, Platia et al. reported conjunctival pigmentation in a young woman who used eyelid cosmetics: the deposited material consisted of ferritin particles and probably iron oxide and carbon (C). 21 The different mascaras available on the market contain similar basic components (pigments, oils, waxes, and preservatives) with various formulas depending on the color, the form (liquid, cake, or cream), and the manufacturer. Among the possible ingredients, there are some which are considered potentially dangerous for ocular health. For example, it is well known that benzalkonium chloride is a preservative potentially toxic for the epithelial cells of the eye. 18 Parabens are also considered to be dangerous because they are endocrine disruptors. 22 Concerning pigments, cosmetics can contain iron oxides (typically found in red, yellow, brown, and black mascara), titanium dioxide (white), or ultramarine which contains aluminum (Al, blue). Other metals which can be found in mascara are nickel and chromium, which are considered to be neurotoxins and can also have consequences in case of specific allergy. 23 In order to give to eye makeup its hue, Al powder is often used. However, Al is considered to be a neurotoxin and has been linked to organ system toxicity. 24
In the present study, the presence of mascara deposits adsorbed on the surface of two different types of siloxane-hydrogels was investigated through optical transmission microscopy and scanning electron microscopy (SEM) combined with an elemental characterization of the surface through energy-dispersive X-ray spectroscopy (EDX). New CLs, CLs exposed in vitro to mascara during contamination tests, and CLs worn for 8 h together were investigated and compared.
Materials and methods
Mascara
The investigated cosmetic is a non-waterproof blue mascara (color 102 by 3INA Cosmetics, London, UK), whose declared ingredients are reported in Table 1. Besides basic components, the manufacturer declares that it may also contain one or more of the dyes reported in the second part of Table 1. For a preliminary elemental characterization of the mascara, the product was brushed on a plastic substrate and left for 2 h under the laboratory fume hood to dehydrate before EDX analysis.
Ingredients of the blue mascara declared by the manufacturer.

Siloxane-hydrogel CLs and sample preparations
Delefilcon A daily disposable CLs (DAILIES TOTAL1, Alcon Laboratories, Ft Worth, TX, USA) were among the siloxane-hydrogel CLs used in this work. The manufacturer declares that these CLs show a water gradient from a relatively low core water content (33%) to a higher water content (>80%) at the outermost hydrophilic surface layer. This surface layer was reported to have a thickness of a few micrometers and to be made of covalently linked hydrophilic monomers. 25 Filcon V monthly replacement CLs (OPEN30, Safilens, Italy) were also used in this work (equilibrium water content declared by the manufacturer 45%). The presence of a surface layer with different properties than the bulk was reported also for Filcon V CLs. 26 This layer, about 10 µm thick, was found to be porous to the point of absorbing hyaluronic acid molecules with a molecular weight of 1000 kDa, unlike the bulk. 26
Nine new CLs (optical power −3.00 D) for each of the two materials (a total of 18 CLs) were extracted from their blisters and exposed for 5 min, in different glass vials, to a sterile 0.9% sodium chloride (NaCl) solution without any preservatives (Alcon Laboratories). After this rinsing, five of these nine new CLs (five for each material) were prepared for SEM/EDX measurements (see next paragraph). They were gently deposited on SEM graphite adhesive stub substrates with the anterior CL surface exposed to air and were left for 6 h under the laboratory fume hood to dehydrate. After 6 h, a film of gold of about 10 nm was deposited on the samples by sputtering in vacuum conditions. The other four rinsed CLs (four for each of the two materials) were observed under a digital transmission optical microscope (see next paragraph).
Other nine CLs (nine CLs of each of the two materials) were taken from their blisters and were exposed for 5 min to the sterile 0.9% NaCl solution, then for 60 min to a solution of mascara dissolved in the NaCl solution with mascara concentration equal to 3 mg/mL, and finally for another 5 min to the NaCl solution (each CL in a separate vial for each single step). After this rinsing, five CLs of each of the two materials were prepared for SEM/EDX measurements following the same procedure described above. The other four CLs of each of the two materials were observed under the digital transmission optical microscope.
Wear of CLs
SEM/EDX analyses were carried out on 10 worn Delefilcon A CLs (worn by five different women) and 6 worn Filcon V CLs (worn by three different women). Each pair was worn by a woman for 8 h during the day, when wearing also the blue mascara.
The study was conducted following the Declaration of Helsinki: after the explanation of the procedures, consent was obtained from all eight women. Inclusion criteria for the wearers were to be already a habitual CL wearer, not to have allergies and ocular pathologies, to be a young woman (age between 18 and 30 years), to be willing to wear blue mascara. Preliminary eye and visual examinations were performed to determine whether each subject was eligible in respect to the inclusion criteria and to find the best optical correction to provide the proper CLs. 27 -29 Non-cycloplegic subjective refraction at distance (4 m) was carried out by a phoropter procedure using high-contrast (97%) early treatment of diabetic retinopathy study (ETDRS) chart displayed on an LCD optotype system (Vision Chart, CSO, Italy). The visual acuity threshold was determined in logarithm of the minimum angle of resolution using a letter-by-letter criterion. Ophthalmoscopy and slit-lamp examination were also carried out to detect any ocular anomaly. Each woman received a pair of CLs of the power needed to correct her ametropia, two empty and sterile glass vials with caps, two closed single doses (15 mL each) of sterile 0.9% NaCl aqueous solution without preservatives (Alcon Laboratories), a closed tube of the blue mascara (color 102 by 3INA Cosmetics), a document with the protocol to be followed for the application of CLs/mascara and for their removal on the day of the CL/mascara wear.
The application protocol was the following: In the morning of the chosen day, open the two CL blisters and open one of the two single doses of sterile NaCl solution; wash your hands with neutral soap (avoid using oily products and/or containing dyes/fragrance); rinse the CLs with the sterile NaCl solution, and apply the CLs with clean fingers. After 5 min, gently apply the mascara. When applying it, keep the product from touching your eyes and your CLs. Avoid any other eye/face cosmetic and the use of eye drops for 8 h. In the event of any eye irritation/reaction/cry during the 8 h, wash the mascara off and remove the CLs. If necessary, contact an eye care practitioner. In this case, it is not necessary to return the CLs to us. After 8 h of CL/mascara wear, wash your hands with neutral soap (avoid using oily products and/or containing dyes/fragrance); remove CLs before removing the mascara, and put each CL in one of the two glass vials. Open the second single dose of sterile NaCl solution and pour about 5 mL into each glass vial. Close each vial with its cap. Finally, remove the mascara with an eye makeup remover of your choice. Return the two vials with the CLs to us the next day.
All eight women worn the CLs for 8 h and completed the protocol. On the day when the worn CLs were returned, they were all prepared for SEM/EDX measurements following the same procedure described above. Worn CLs were not analyzed under the optical microscope because it was not possible to distinguish with this technique deposits of mascara from other deposits deriving from the tear film. On the contrary, it was possible to identify mascara deposits using SEM/EDX analysis thanks to the elementary analysis (EDX) which allowed to detect the Al peak.
SEM/EDX and optical microscopy
All samples (new CLs, worn CLs, and CLs treated in vitro) were characterized by SEM/EDX. SEM analyses were carried out with a Tescan VEGA TS5136XM SEM (Brno – Kohoutovice, Czech Republic), equipped for EDX. The operating voltage used for all the SEM images was 20 kV, while EDX analyses were performed with an EDAX Genesis 4000 XMS Imaging 60 under the following conditions: operating voltage 20 kV, probe current 190 pA, probe size 230 nm, working distance 23 mm, calibration with 53 Minerals Standard Mount MINM25-53. The EDX operating principle of the technique can be briefly described as follows. An incident beam of electrons excites an electron in an inner shell of an atom, ejecting it from the shell while creating a hole. An electron from a higher energy shell fills the hole and the difference in energy is released (X-ray). This energy depends on the atomic structure of the emitting element, thus allowing an elemental analysis. Data are usually reported in graphs where the x-axis shows the energy (keV) of the released photons and the y-axis is proportional to the number of emitted photons (counts) per unit time. The higher the peak corresponding to the energy of a specific element, the higher its presence at that point of the sample under investigation. In this work, the values on the y-axis of EDX profiles were normalized to the number of counts measured at the silicon (Si) peak, that is, at energy equal to 1.739 keV.
New CLs and CLs treated in vitro were also characterized by taking black and white images by a digital transmission optical microscope Leica MZ12 at magnification 1.25×. The ratio N b/N between the number of black pixels (contaminated area) and the total number of pixels was deduced for each image by using the software Image J (developed by National Institutes of Health, USA). Five images were acquired for each sample in different areas and the mean value of the five ratios N b/N was calculated on these five images for each sample.
Results
Different EDX profiles were acquired on dehydrated blue mascara. At different points in the sample and in different measures, the relative intensities of the observed EDX peaks were variable. Table 2 indicates the energies of the observed peaks in the range 0.2–5.0 keV corresponding to the characteristic X-rays emitted by each element. On average, the most intense ones were those of C, Si, and Al.
Energies of the peaks observed in measured EDX profiles of blue mascara in the range 0.2–5.0 keV corresponding to the characteristic X-rays emitted by each element.a
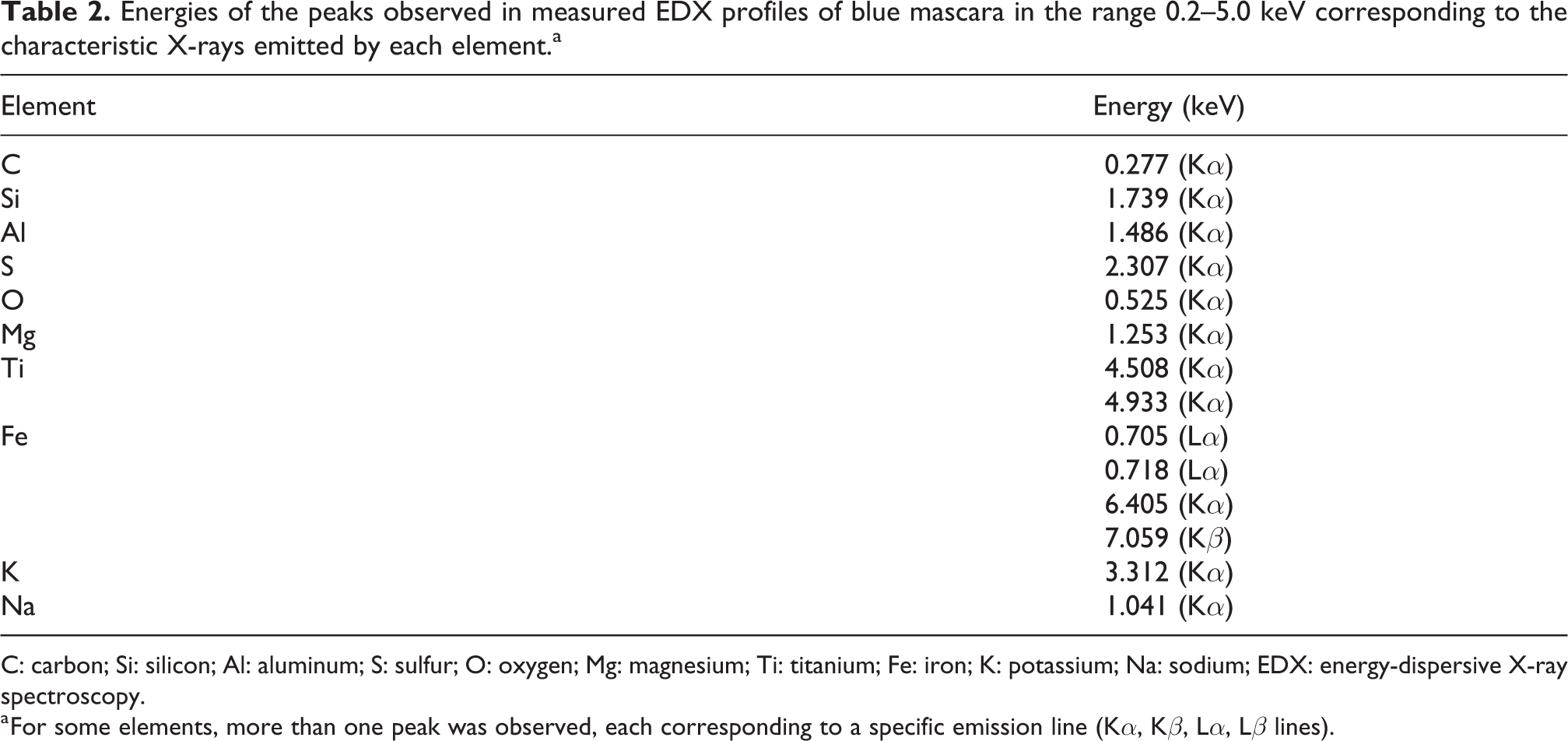
C: carbon; Si: silicon; Al: aluminum; S: sulfur; O: oxygen; Mg: magnesium; Ti: titanium; Fe: iron; K: potassium; Na: sodium; EDX: energy-dispersive X-ray spectroscopy.
a For some elements, more than one peak was observed, each corresponding to a specific emission line (Kα, Kβ, Lα, Lβ lines).
Figure 1 shows some representative SEM micrographs of the surface of CLs exposed to the mascara solution in vitro (Figure 1(a)) and of the surface of CLs worn by different women for 8 h (Figure 1(b) to (e)). In all cases, deposits are clearly observed. Due to the rather high concentration of mascara in in vitro solutions, deposits on the corresponding CLs were typically more frequent than deposits on worn CLs. Neglecting these deposits, the CL surface appears quite smooth. Also, new CLs showed a similar smooth surface (images are here omitted) without, in this case, the typical deposits of treated and worn CLs. Figure 2 shows the representative EDX profiles obtained by directing the electrons toward the smooth surface of new CLs (dotted line) or toward deposits on the surface of CLs exposed to the mascara solution in vitro (continuous line) or worn for 8 h (continuous line with diamonds). Figure 3 shows the same profiles in a restricted energy range, where the intense peak of Al is better displayed.

SEM micrographs of the surface of (a) a Delefilcon A CL exposed to the mascara solution in vitro, (b) a Filcon V CL worn by a woman for 8 h together with the mascara, and (c to e) Delefilcon A CLs worn by different women for 8 h together with the mascara. SEM: scanning electron microscopy; CL: contact lens.
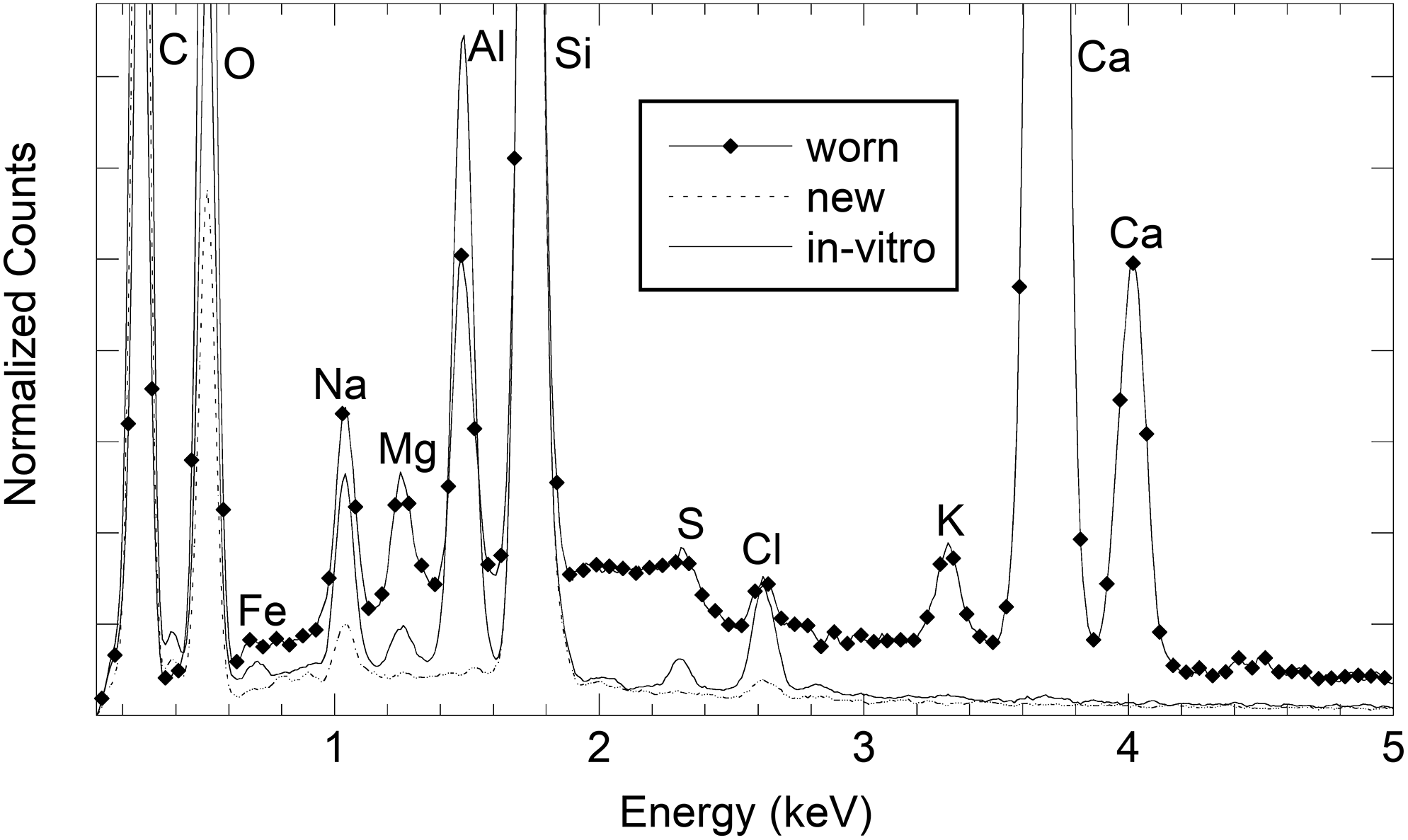
EDX profiles obtained by directing the electrons toward the smooth surface of a new Delefilcon A CL (dotted line), toward deposits observed by SEM on a Delefilcon A CL exposed to the mascara solution in vitro (continuous line), and toward deposits observed by SEM on a worn Delefilcon A CL (continuous line with diamonds).

EDX profiles in a restricted energy range (0.8–2.0 keV) obtained by directing the electrons toward the smooth surface of a new Delefilcon A CL (dotted line), toward deposits observed by SEM on a Delefilcon A CL exposed to the mascara solution in vitro (continuous line), and toward deposits observed by SEM on a worn Delefilcon A CL (continuous line with diamonds).
Some peaks were detected in the profiles of all samples. They are the peaks of C at 0.277 keV, O at 0.525 keV, Si at 1.739 keV, sodium at 1.041 keV, and chlorine at 2.621 keV. The intensity of the two peaks of sodium and chlorine was larger when the electrons were directed toward some NaCl residues that sometimes form on the CL surface. These residues could be observed both on new CLs and on CLs treated in vitro or after wear, since all samples were rinsed with NaCl aqueous solution before the preparation for SEM/EDX analyses. Probably, they are not adsorbed but are resting on the surface.
The peaks of calcium at 3.692 keV (Kα line) and at 4.013 keV (Kβ line) and the peak of potassium at 3.312 keV were only detected in the profiles of worn CLs and they are reasonably attributable to tear residues.
The most important difference between new CLs on one side and, on the other side, CLs treated in vitro or worn for 8 h is the appearance of the peaks of iron at 0.705 keV, magnesium at 1.253 keV, sulfur at 2.307 keV, and the very intense Al peak at 1.486 keV. Iron, magnesium, sulfur, and Al are attributable to the mascara because they are not present in new CLs, while being detected on both worn CLs and CLs treated in vitro. In Figure 2, a weak structure at about 4.5 keV in the profile of worn CLs is detected, but its weak intensity does not allow a clear attribution. A possible origin is the presence of titanium (line Kα at 4.508 keV). The presence of SEM whitish deposits and the EDX intense peak of Al were identified in all the samples both after in vitro treatment and after 8 h of wear by a woman, both wearing Delefilcon A CLs and wearing Filcon V CLs.
The different affinity of Filcon V and Delefilcon A CLs for the investigated mascara was determined quantitatively by processing the images taken under the optical microscopy in in vitro contamination tests. The ratios N b/N between the number of black pixels and the total number of pixels in the images taken by the optical microscope are reported in Table 3.
Ratios N b/N between the number of black pixels and the total number of pixels in the images taken by the optical microscope for the different CLs (mean values calculated on n samples; for each sample, a mean value was calculated based on five images taken on different areas of the same sample).

CL: contact lens.
Discussion
SEM and EDX, separately or jointly, have already been applied for the study of polymeric soft CLs in previous studies. A few years ago, a SEM analysis allowed to observe globular deposits of fungal spores on soft CLs discarded because of irritation of the cornea. 30 The techniques were also used to evaluate the role of calcium in nodular deposits on the front surface of worn CLs 31 or to compare new and worn siloxane-hydrogel CLs in terms of microscopic structure, surface morphology, and loading of hyaluronan. 26,32 In very recent years, SEM and EDX were used to investigate the surfaces and principal elements of the colorants of cosmetically tinted CLs. 33 Colorants used in the investigated CLs contained chlorine, iron, and titanium, which are elements that have tissue toxicity. In some CLs, colorants were found inside the lens, located to a depth of 8–14 µm from the anterior side, but in other cases colorants were found to be more superficial. In the case of limbal ring CLs, SEM allowed to determine the pigment location on the CL surface. 34
In this study, SEM and EDX were used jointly to detect mascara deposits. A preliminary comment concerns the detectable mascara components based on the measured EDX profile of dehydrated blue mascara (Table 2). C and O are abundant in mascara and are found in many (more or less complex) molecules that are part of its composition. Si is known to be another elemental component, due to the presence of silica, borosilicate, and mica, which are bulking agents. As declared by the manufacturer, mascara contains also Al due to calcium aluminum borosilicate, alumina, mica, and Al powder. Moreover, the mascara can also contain sulfur, magnesium, titanium, iron, potassium, and sodium. Among these components, Al can be considered as a marker of the presence of mascara because it is neither a tear component nor a CL component. Indeed, even if the results are not reported here, Al has never been found in previous analyses performed on CLs worn by subjects without makeup.
Deposits on both CLs exposed to the mascara in vitro and worn CLs were clearly found for both materials (Figure 1). The deposits are typically present in the form of aggregates of variable size ranging from about 1 µm to about 20–30 µm. The deposits are unequivocally due to mascara, since Al was clearly visible in EDX profiles. The deposits containing Al were also the main ones detectable on the surface of the worn CLs. Only a few deposits turned out to be made up of NaCl. Aggregates attributable to tear residues were not substantially observed, probably because the samples were worn only 8 h and because they were rinsed with NaCl solution before the analysis. In general, transport of mascara from an external environment to the ocular surface is of clinical relevance because it can cause adverse effects, 11 -15 as discussed in the “Introduction” section. In this work, mascara residues are not only found in the ocular environment but are also found in aggregates adsorbed on the surface of the CL. Indeed, they could not be removed by rinsing the CLs in the NaCl solution. However, it is not possible to say if the mascara aggregates adsorbed on the CL may cause different adverse reaction respect to what it was found in the literature for mascara directly applied to the ocular environment from makeup.
The CLs used in this work are either daily disposable CLs or monthly CLs (used for 8 h in both cases). Both materials showed mascara deposits adsorbed in the polymeric matrix. However, through SEM/EDX analyses it was not possible to make a reliable quantitative evaluation and comparison between the two materials, except to say that both siloxane-hydrogel materials showed these residues both after 8 h of wear and after exposure to the mascara solution in vitro. The quantitative comparison between the two materials was possible through the analysis of the images taken by optical microscopy. The evaluation was only performed on in vitro treated CLs. Worn CLs were not analyzed under the optical microscope because it was not possible to distinguish with this technique deposits of mascara from NaCl deposits (due to rinsing) or possible other deposits deriving from the tear film. The dark deposits observed in the black and white images were attributed to adsorbed mascara deposits after exposure to the solution in vitro. Since it was not possible to exclude a priori the presence of dark spots due to NaCl crystals or dust residues, new CLs were also analyzed for comparison. As reported in Table 3, a percentage of black pixels was actually found also on new CLs, although much lower than on the treated CLs. There is also a difference between the two materials exposed to the mascara solution. The Delefilcon A CLs showed an affinity for the mascara more than two times higher than the Filcon V CLs. This different affinity is attributable to the different properties of the surface layer of the two types of CLs, rather than to the bulk properties of the silicone-hydrogels. Unfortunately, to the best of our knowledge, the chemical nature of the surface layer is not known. What it is known is that the surface layer of Deleficon A CLs is made of covalently linked hydrophilic monomers. 25 The presence of a surface layer in Filcon V CL was observed by SEM analyses and was confirmed by observing a clear difference in the absorption of hyaluronic acid between the surface layer and the bulk. 26
Conclusions
Al is a marker of the presence of deposits of blue mascara on CLs. Deposits adsorbed on the surface were detected on all investigated worn samples after 8 h of wear, thus showing a special affinity of the surface layer of the two types of CLs under investigation for the mascara components, more marked than for the components of the tear film. In in vitro contamination tests, different affinities were found for the materials taken into consideration. In light of this difference, wearers and manufacturers of both CLs and cosmetics should consider that the combined choice has to be optimized to minimize anterior eye complications secondary to wear.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
