Abstract
Poly(methyl methacrylate-co-ethylene glycol dimethacrylate-co-vinylbenzyl chloride) microsphere was prepared by dispersion polymerization, initiated by light using water/ethanol mixture. The sulfonated hypercrosslinked (HXL) polymer resin was synthesized via three stages, namely, precursor synthesis (photoinitiated dispersion polymerization), post-polymerization after hypercrosslinking reaction, and post-polymerization before hypercrosslinking reaction. The optimized precursor was functionalized before the hypercrosslinking reaction to investigate its ion-exchange capacity, specific surface area (SSA), morphology, and thermal stability. Post-polymerization after hypercrosslinking reaction showed that the chlorine content and SSA are higher than post-polymerization before hypercrosslinking reaction. HXL reaction took place using Friedel–Crafts alkylation with the aid of FeCl3 catalyst. Lewis acid (FeCl3) also played a significant role which contributed to high SSA of the HXL polymer. The particles produced from photoinitiated dispersion polymerization have advantages and could be used in solid-phase extraction, drug delivery, and high-performance liquid chromatography.
Keywords
Introduction
Monodisperse polymeric particles are widely used in numerous applications, such as standard calibration, 1 -3 biomedical and biochemical analysis, 4 -6 and column packing materials for chromatography. 5 Generally, these microsphere polymers are synthesized by precipitation polymerization where a large amount of crosslinker is required, 7 -11 and they are sensitive toward the addition of comonomer. 12,13 However, the higher amount of crosslinker has disadvantages which are not applicable for some applications due to rigidities limitation. The addition of comonomer into the system can be countered through the technique proposed by Winnik’s group through dispersion polymerization technique.
Dispersion polymerization is an attractive method to prepare monodisperse polymeric microspheres which has become interesting to the researchers over the past two decades. 14 -16 In dispersion polymerization, the synthesis of cross-linked polymer microspheres is the challenge where the nucleation stage can be easily disturbed with the presence of functional reagents, such as crosslinker and comonomer. 17 Winnik’s group developed a two-stage dispersion polymerization method to synthesize monodisperse cross-linked polymer particles where functional reagents were added after the nucleation stage is finished to avoid the disturbance of functional reagents to the nucleation stage. 18,19 The two-stage dispersion polymerization is a complicated procedure, the particles do not homogeneously cross-linked, and longer reaction period is required to obtain monodisperse cross-linked microspheres.
To overcome the drawbacks, the photoinitiated dispersion polymerization technique was introduced to control nucleation stage and monodisperse microspheres. 20,21 The advantage of this approach is that the nucleation stage becomes robust where functional reagents can be added at the beginning of reaction without disturbing the nucleation. Thus, the photoinitiated dispersion polymerization is a possible approach to overcome the problems of traditional dispersion polymerization to synthesize cross-linked microsphere polymer. It is a very rapid procedure where photo initiator (PI) decomposes and generates free radicals quickly under the ultraviolet or visible light irradiation at room temperature. In addition, this method is capable of achieving a high monomer conversion of poly(methyl methacrylate). 14,15
Experimental design acts as a statistical tool of fundamental importance to improve a process where higher reliability conditions and lower costs can be achieved. 22 Besides, it is one of the best approaches instead of classical experimental method where the data are collected and analyzed in a systematic way and can be represented in a mathematical term. Hence, this technique has been implemented for the study of polymerization reactions. 23 -27 Moreover, the great interest of this approach instead of traditional methods would be expected to improve validation of the experiment and consumed time.
In this study, cross-linked poly(methyl methacrylate-co-ethylene glycol dimethacrylate-co-vinylbenzyl chloride) (poly(MMA-co-EGDMA-co-VBC)) microsphere was successfully synthesized via photoinitiated dispersion polymerization. The effect of post-polymerization chemical modification was discussed to investigate the performance of the resin. The resins were modified by chemical modifications on pendent functional groups through sulfonation reaction. The precursors were synthesized via photoinitiated dispersion polymerization and then went through a post-polymerization chemical modification after (route A) and before the hypercrosslinked (HXL) reaction (route B) using lauroylchlorosulfonic acid as sulfonating reagent. The performance of the modified resins was evaluated in terms of thermal, structural, morphological, compositional, ion-exchange, and surface properties, and after that, the results were compared.
Experimental section
Materials
Methyl methacrylate (MMA) (99%), ethylene glycol dimethacrylate (EGDMA) (98%) with 90–110 ppm of monomethyl ether hydroquinone, 4-vinylbenzyl chloride (VBC) (97%), poly(N-vinylpyrrolidone) (PVP) (MW∼55,000 kg/kmol), Triton X-305 (70% in H2O), and ethanol (90%) were purchased from Sigma-Aldrich, St. Louis, Missouri, USA. Irgacure 819 was obtained from BASF, Ludwigshafen, Germany. All chemicals were used in this study as they were received.
Synthesis of precursors
The precursor particles were synthesized via photoinitiated dispersion polymerization. The dispersion medium was prepared by dissolving the PVP in the distilled water. Ethanol was poured into the MMA, PVP, Irgacure 819, Triton X-305, EGDMA, and VBC which then dissolved in the dispersion medium. The amount of the chemicals required for the experiment is listed in Table 1. The mixture was placed in a three-necked, round-bottomed flask irradiated by the 3-W 365-nm LED lamp and stirred at 300 r min−1 using a magnetic stirrer. The mixture was purged with nitrogen gas for 15 min before being irradiated by the LED lamp. The polymerization process was conducted at room temperature for the whole duration of experiment. After the polymerization, the precursor polymers were washed by distilled water and ethanol before being dried up in the oven for 24 h. The polymerization process is illustrated in Figure 1.
Formulation of precursor synthesis.

PI: photo initiator; PVP: poly(N-vinylpyrrolidone); EGDMA: ethylene glycol dimethacrylate; VBC: 4-vinylbenzyl chloride.

Schematic diagram of the precursor synthesis process. MMA: methyl methacrylate; EGDMA: ethylene glycol dimethacrylate; VBC: 4-vinylbenzyl chloride.
Synthesis of poly(MMA-co-EGDMA-co-VBC) microsphere
The polymerization process was carried out in a ratio of 3:2 of water and ethanol solvent in the reaction medium of 15-wt% poly(N-vinylpyrolidone) solution. Different compositions of EGDMA (20, 35, and 50 wt%), VBC (20, 30, and 40 wt%), PI (2, 3, and 4 wt%), and Triton X-305 (5.5 wt%) with respect to the weight of MMA monomer were separately mixed with the monomer. The maximum yield was obtained as 87.29% for the setting of 50 wt% of EGDMA, 20 wt% of VBC, 2 wt% of PI, and 12 h of reaction time. The inert nitrogen gas was bubbled in the reactant mixer for 15 min and stirred continuously at 200 r min−1. The reactant mixer was exposed to the 3-W 365-nm LED lamp and irradiated separately for 6, 9, and 12 h to complete the reaction process. After completion of polymerization, the polymer precursors were washed repeatedly three times with distilled water and ethanol and were vacuum filtered using 0.22-µm nylon membrane filter. The polymer particles were dried in an oven at 40°C for 24 h. 28 Finally, the samples were subjected for the analysis.
Synthesis of HXL polymers
This section describes the chemical and HXL reaction process used for synthesizing HXL polymers. During the synthesis, the reagents used for HXL reaction were 1,2-dichloroethane (DCE; 99% purity) and iron (III) chloride (FeCl3), obtained from Sigma Aldrich. Methanol with 99.98% purity was supplied by HmbG Chemicals, Kuala Lumpur, Malaysia, whereas nitric acid (HNO3) with 65% concentration was supplied by the R&M Chemicals, Loughborough, UK.
HXL reaction
In this reaction, 10 g of precursor particles were added into 100 mL of DCE in a five-necked, round-bottomed flask (250 mL) and were left to swell fully under nitrogen atmosphere at room temperature for 1 h. FeCl3: Cl (1:1 ratio) was added into the DCE and the mixture was heated at 80°C for 18 h. After 18 h, the particles were filtered and washed with methanol and diluted HNO3. Then, the particles were extracted for 24 h with acetone in a Soxhlet extractor and washed again with methanol and diethyl ether. The particles were dried in an oven at 50°C.
Synthesis of functionalization HXL polymer
The chemicals and lauroyl sulfate preparations were used to synthesize sulfonate HXL polymer. The reagents used to synthesize a strong cation exchange resin polymer were lauric acid (99%), cyclohexane (99%), chlorosulfonic acid (97%), and petroleum ether. These were supplied by R&M Chemicals. All of the chemicals were used as received.
Sulfonation reaction
For the sulfonation reaction, 5 g of HXL polymer and 100 mL of cyclohexane were placed in a round-bottomed flask (250 mL), while the mixture was left under nitrogen atmosphere. Chlorosulfonic acid was added into the mixture and stirred at room temperature for 1 h under nitrogen atmosphere. The mixture was heated at 50°C with stirring for 24 h. After 24 h, the particles were filtered with the 0.2-µm nylon filter membrane and washed with petroleum ether. The particles were also extracted in a Soxhlet extractor with petroleum ether. The product formed was a brown powder and was washed with petroleum ether and dried in an oven at 40°C for 24 h. The process for the chemical modification using route A and route B is illustrated in Figure 2.

Schematic diagram for post-polymerization chemical modification after hypercrosslinked reaction and post-polymerization chemical modification before hypercrosslinked reaction.
Characterizations
Elemental analysis
The amount of carbon, hydrogen, nitrogen, and sulfur in the polymeric particles was determined with an elemental analysis from Elementar, Germany. For the sample preparation, the particles were burnt in pure oxygen using tin as the catalyst. Sulfur, phosphorus, or halogens were removed, and excess oxygen was reduced leaving CO2, H2O, and N2 as the combustion products. The respective gases were dried using the magnesium perchlorate trap before being separated by a pack of gas chromatography column, and the elements were detected by a thermal conductivity detector.
Fourier transform infrared spectroscopy analysis
The functional group present in the polymeric particles was determined using the Spherical Diamond ATR Nicolet iS50 (Thermo Scientific, Waltham, MA, USA) Fourier transform infrared (FTIR) spectroscopy analysis. In this method, the polymer beads were brought into contact with the diamond window. The spectrum was scanned with a resolution of 4 cm−1, and an average of 32 scans was taken for each sample.
Surface morphology
The morphology of the polymer precursors was examined using scanning electron microscopy (SEM) (model—UNIT JOEL.JSM-7800F). Different magnifications and 1.32 × 10−2 Pa operating conditions were used to observe the surfaces of the samples. The samples were coated with platinum at 40 mA for 70 s before the analysis was performed.
Thermogravimetric analysis
Thermal stability of the polymeric particles was studied through thermogravimetric analysis (TGA) using a TA instruments (TGA Q500), New Castle, DE, USA. The thermal stability in terms of thermal decomposition and weight loss of the polymeric particles were examined. The ramping was used at 10°C min−1 starting from 0°C to 900°C. Nitrogen gas was purged throughout the entire analysis.
Ion-exchange capacity
Ion-exchange capacity (IEC) of the resins was determined by immersing sulfonated resin in 45-mL 0.2-mol L−1 NaOH solution for 24 h. The hydroxide depleted from the solution during the ion exchange process was determined by titration against 0.2-mol L−1Na2SO4 solution using a burette and a pH meter. The IEC was calculated according to the following equation:

where VNaOH is the volume of NaOH solution (mL), MNaOH is the molarity of NaOH (mol), and Wdry is the mass of the polymer resin (g).
X-ray fluorescence analysis
The amount of chlorine present in the polymeric particles was determined using Bruker (Shah Alam, Selangor, Malaysia), S8 Tiger X-ray Fluorescence. The particles were directly struck by the X-ray photons without any coating.
Brunauer–Emmett–Teller analysis
The HXL polymer and functionalized HXL materials were characterized using the Brunauer–Emmett–Teller (BET) treatment. The N2 sorption isotherm data were generated to measure the specific surface area (SSA). The particles were degassed at 150°C for overnight to remove the moisture.
Results and discussion
Post-polymerization chemical modification after hypercrosslinking reaction
In this study, chemical modification was conducted after HXL reaction. In post-polymerization chemical modification, lauroylchlorosulfonic acid was used as a sulfonation reagent to introduce sulfur attachment in the backbone. As the sulfonation reaction conducted, strong cation exchange resins were successfully synthesized. Several analyses were conducted to investigate the properties of the strong cation exchange resins produced via this route. The elemental analysis for this route is presented in Table 2. It can be seen that the precursor polymer has a sulfur content of 0.45% which was reduced by 50% (0.23%) after the HXL reaction. However, the amount of sulfur content increased to 3.01% after sulfonation reaction. It was happened so due to the element of sulfur added during the sulfonation reaction. It proved that sulfur was successfully incorporated into the sulfonated resin after the chemical modification. For the presence of nitrogen, it can be considered to be negligible. It might be happened due to the contamination occurred during the analysis through the combustion process. The chlorine content for the three samples throughout the respective reaction experienced a decrement. The evidence can be seen based on the formation of methylene bridge (hypercrosslinking) obtained from the FTIR spectra as shown in Figure 3. It showed that chloromethyl group at peak 1265 cm−1 were significantly less intense in the spectra of the HXL materials compared to precursor polymer. The similar finding was also reported by Cormack et al. 29
Elemental analysis for precursor polymer, hypercrosslinked polymer, and sulfonated resin via route A and route B.

HXL: hypercrosslinked; FXN: sulfonated poly(MMA-co-EGDMA-co-VBC).
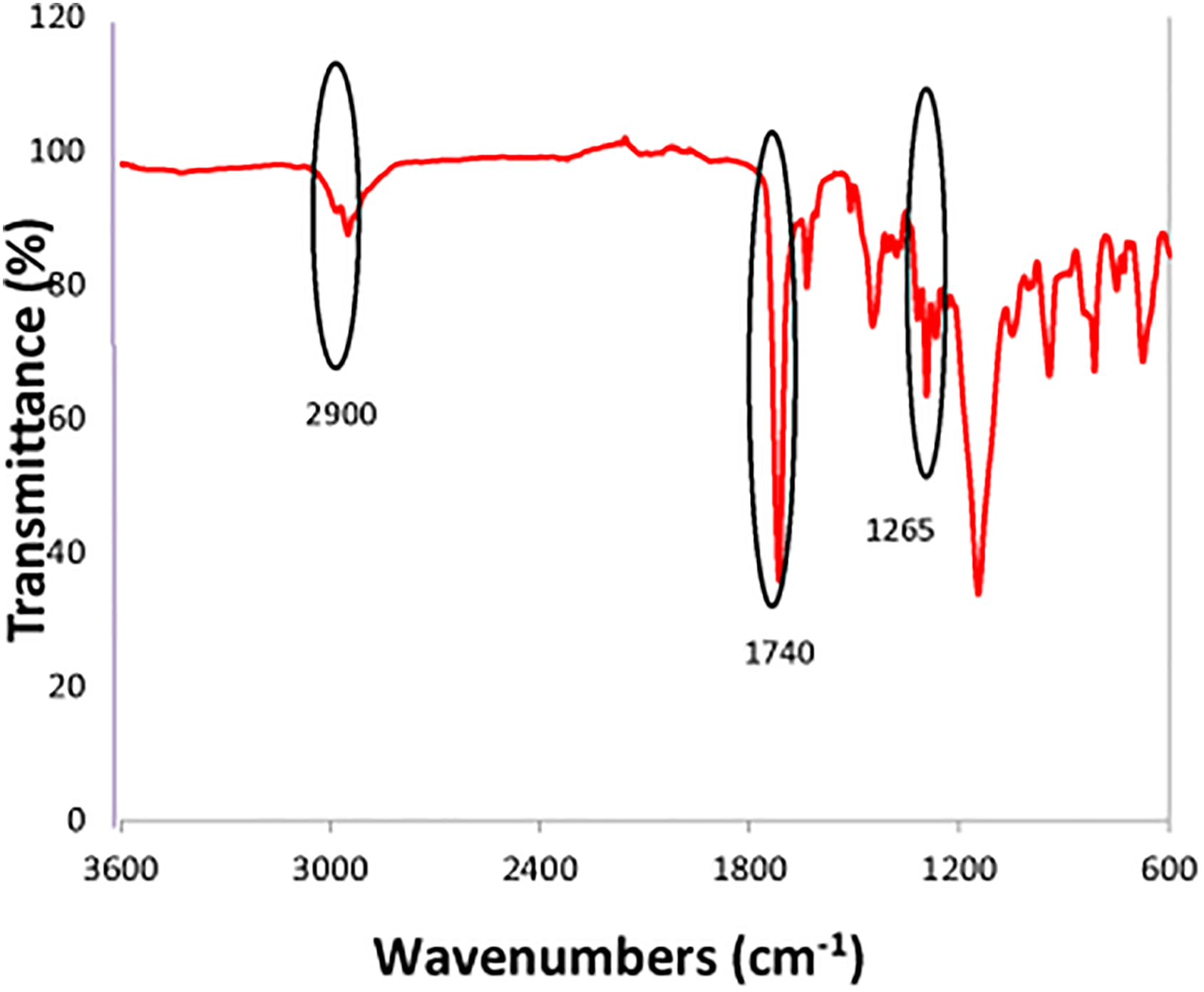
FTIR spectra of optimized poly(MMA-co-EGDMA-co-VBC). FTIR: Fourier transform infrared; MMA: methyl methacrylate; EGDMA: ethylene glycol dimethacrylate; VBC: 4-vinylbenzyl chloride.
The well-defined spherical shape of the precursor and the other resins are shown in Figure 4(a). Upon hypercrosslinking reaction, it maintained its spherical shape (Figure 4(b)). According to the finding of the previous researchers, the particles’ shape of HXL polymer led to more efficient packing in sorbent bed, preventing voids and eventually leading to a more efficient and reproducible extraction procedure. 30 Meanwhile, there were no significant changes in the particles morphology after sulfonation reaction (Figure 4(c)). The particles managed to retain its shape after sulfonation reaction. This might be due to the high amount of cross-linker used.

Scanning electron microscopic images of (a) precursor poly(MMA-co-EGDMA-co-VBC), (b) HXL poly(MMA-co-EGDMA-co-VBC), and (c) sulfonated poly(MMA-co-EGDMA-co-VBC). MMA: methyl methacrylate; EGDMA: ethylene glycol dimethacrylate; VBC: 4-vinylbenzyl chloride; HXL: hypercrosslinked.
Figure 5 shows TGA thermogram of polymer precursor, hypercrosslinked polymer (HXL 1), and sulfonated poly(MMA-co-EGDMA-co-VBC) (FXN 1). Polymer precursor shows the highest decomposition point followed by HXL polymer and sulfonated poly(MMA-co-EGDMA-co-VBC). It can be observed that there are three weight loss stages for those samples. The first stage of decomposition is located below 100°C which attributed to the removal of the adsorbed water on the surface of the particles. This finding is in line with the observation of other researchers. 31,32 The TGA curve showed that FXN 1 experienced more weight loss at first stage than HXL 1 and precursor. This situation takes place when sulfonate functional group absorbed more water from the atmosphere. The same findings were obtained in a different study when the functional group present absorbed more water. 33 Hence, it proved that this situation caused more weight loss for FXN 1 in the first stage. The second stage of decomposition occurred approximately between 100°C and 300°C which probably due to the decomposition of uncrosslinked linear polymers. It could be correlated to the removal of the chloromethyl or hydroxymethyl group derived by side reactions. The next stage of decomposition occurred at 300–450°C due to the decomposition of PI. It can be seen that sulfonated resin decomposes first at 300°C due to the decomposition of sulfonic groups of sulfonated poly(MMA-co-EGDMA-co-VBC). The weight loss from 450°C to 900°C shows the destruction of the polymer. According to the analysis, it shows that poly(MMA-co-EGDMA-co-VBC) has a very good temperature resistance which is up to 300°C. Although FXN 1 had a rapid decomposition at the initial stage, FXN 1 has the highest yield at 900°C among all the samples. It could be seen that FXN 1 is thermally more stable than precursor and HXL 1 which can be used for the further application due to the good temperature resistance up to 900°C. The thermal properties are listed in Table 3.

TGA curves of precursor polymer, hypercrosslinked polymer, and sulfonated resin. TGA: thermogravimetric analysis; HXL: hypercrosslinked; FXN: sulfonated poly(MMA-co-EGDMA-co-VBC).
TGA profile for polymer precursor, hypercrosslinked polymer, and sulfonated resin.

TGA: thermogravimetric analysis; HXL: hypercrosslinked; FXN: sulfonated poly(MMA-co-EGDMA-co-VBC).
Referring to Figure 6 in derivative weight versus temperature analysis, the HXL and sulfonated poly(MMA-co-EGDMA-co-VBC) have the same maximum weight loss which allocated at the same region temperature (200–410°C). However, the precursor undergoes weight loss in the range of 200–380°C. In terms of cumulative weight, the HXL experienced a lower maximum cumulative weight values than the precursor and sulfonated poly(MMA-co-EGDMA-co-VBC). It can be correlated to the degradation of the samples occurred. The HXL poly(MMA-co-EGDMA-co-VBC) rich in volatile matter and degraded at temperature between 200°C and 410°C. The HXL poly(MMA-co-EGDMA-co-VBC) rich in chloromethyl group was more volatile and experienced degradation in the respective temperature. The finding is in agreement with the other researchers’ report. 33 Thus, as the removal of chlorine content in HXL poly(MMA-co-EGDMA-co-VBC) is higher than precursor and sulfonated poly(MMA-co-EGDMA-co-VBC) which forms methylene bridge, HXL polymer had lower weight loss than other two samples.

Cumulative weight versus temperature plot for precursor, hypercrosslinked, and sulfonated poly(MMA-co-EGDMA-co-VBC). MMA: methyl methacrylate; EGDMA: ethylene glycol dimethacrylate; VBC: 4-vinylbenzyl chloride; HXL: hypercrosslinked; FXN: sulfonated poly(MMA-co-EGDMA-co-VBC).
Post-polymerization chemical modification before HXL reaction
In contrast with the previous part, the post-polymerization chemical modification was carried out before the HXL reaction. It means that sulfonation reaction takes place first and followed by HXL reaction. This approach gives advantages where HXL polymer based on VBC-EGDMA experienced incomplete consumption of VBC residues which takes place in hypercrosslinking reaction. As the residual chloromethyl groups remain in hypercrosslinking processes, it can be used for chemical modification through sulfonation reaction. In this part, poly(MMA-co-EGDMA-co-VBC) undergoes post-hypercrosslinking chemical modifications before HXL to produce strong cation exchange resin. Both routes used lauroylchlorosulfonic as the sulfonation reagent. These two routes were conducted to compare the IEC value, SSA, morphology, and thermal stability.
The elemental analysis for post-polymerization chemical modification before HXL reaction is given in Table 2. It could be compared with the post-polymerization chemical modification after HXL reaction where chlorine content is higher than post-polymerization chemical modification before HXL reaction. The trend is almost the same where chlorine content experienced depletion starting from precursor polymer followed by hypercrosslinking reaction and chemical modification. It can be seen that the amount of chlorine for precursor polymer decreased to 10.81% from 13.26% for HXL polymer and also undergoes decrement for sulfonated resin (7.99%). In this route, chemical modification was conducted first and followed by hypercrosslinking reaction. Chlorine content for sulfonated resin (7.99%) is lower than HXL polymer (10.81%) where chlorine element is not added during sulfonation, whereas element of sulfur was added during sulfonation reaction. Hence, sulfur content showed the highest amount (2.16%) in sulfonated poly(MMA-co-EGDMA-co-VBC) compared to precursor polymer (0.45%) and HXL polymer (0.37%).
The SEM images did not differ much compared to those of post-polymerization chemical modification after HXL reaction which polymeric beads could be observed. Single polymeric beads could be observed in Figure 7(a). As the precursor polymer undergoes sulfonation reaction, the particles tend to agglomerate, however, they were able to maintain its spherical shape (Figure 7(b)). Monodisperse, spherical shapes of particles are ideal for further application such as packing into solid phase extraction cartridges and chromatography columns. Thus, it can be seen that sulfonation reaction does not give effect in terms of shape and size of the particles produced. As for the hypercrosslinking reaction, well-defined spherical shape of particles could be obtained (Figure 7(c)). The particles managed to retain spherical shape in post-polymerization chemical modification before HXL reaction.

Scanning electron microscopic images of (a) precursor poly(MMA-co-EGDMA-co-VBC), (b) sulfonated poly(MMA-co-EGDMA-co-VBC), and (c) HXL poly(MMA-co-EGDMA-co-VBC). MMA: methyl methacrylate; EGDMA: ethylene glycol dimethacrylate; VBC: 4-vinylbenzyl chloride; HXL: hypercrosslinked.
Figure 8 shows TGA thermogram of polymer precursor, hypercrosslinked polymer (HXL 2), and sulfonated poly(MMA-co-EGDMA-co-VBC) (FXN 2). The polymer precursor shows the highest decomposition point followed by sulfonated resin and HXL particles. It can be observed that there are three weight loss stages for those samples. The first stage of decomposition is located below 200°C which attributed to the removal of the adsorbed water on the surface of the particles. This finding is in line with the observation of other researchers. 31,32 TGA curve showed that FXN 2 experienced more weight loss which occurred at first stage than HXL 2 and precursor. The situation above took place when sulfonate functional group absorbed more water from the atmosphere. The presence of sulfonate functional groups absorbs water and causes more weight loss. 32 Hence, it proved that this situation caused more weight loss for FXN 2 in the first stage. The second stage of decomposition occurred approximately between 100°C and 300°C which probably due to the decomposition of uncrosslinked linear polymer. It can be seen that sulfonated resin decomposes first at 100°C which can be correlated to the removal of the chloromethyl or hydroxymethyl groups derived by the side reactions. The next stage of decomposition occurred approximately between 300°C and 450°C due to the decomposition of PI. It can be seen that sulfonated resin decomposes first at 300°C due to the decomposition of sulfonic groups of sulfonated poly(MMA-co-EGDMA-co-VBC). 32 It was evidenced that the thermal stability of both FXN 1 and FXN 2 was lower than HXL 1, HXL 2, and precursors because of the decomposition mechanism. Precursors, HXL 1, and HXL 2 decompose by the depolymerization mechanism, whereas FXN 1 and FXN 2 decompose by chain scission mechanism followed by depolymerization. 34 The weight loss from 450°C to 900°C shows the destruction of the polymer. The analysis indicated that poly(MMA-co-EGDMA-co-VBC) had a very good temperature resistance which was up to 300°C.

TGA curves of precursor polymer, hypercrosslinked polymer and sulfonated resin. TGA: thermogravimetric analysis; HXL: hypercrosslinked; FXN: sulfonated poly(MMA-co-EGDMA-co-VBC).
Surface area, ion exchange, and BET analysis
These two routes were used to determine the properties of high SSA, IEC value, most stable thermal ability, and good morphology behavior of strong cation exchange resin suitable for desirable applications. IEC value, SSA, and chlorine content for post-polymerization chemical modification after and before HXL reaction is given in Table 4. SCE 1 represented the resins produced via post-polymerization chemical modification after HXL reaction, and SCE 2 represented the resins produced via post-polymerization chemical modification before HXL reaction. The chlorine content of both precursors was 13.26% and decreased to 8.54% for SCE 1 and 4.06% for SCE 2 which was approximately 36% and 70% decrement, respectively. This was due to the formation of multiple bridging from HXL reaction and sulfonation reaction. In terms of IEC value, SCE 1 had 4.5 mmol g−1and 3.7 mmol g−1 for SCE 2. The difference is approximately 17% between the two sulfonated resins. The IEC value for SCE 1 showed the highest value among those two strong cation resins.
SSA, IEC value, and chlorine content for the strong cation resins produced via two different routes.

SSA: specific surface area; IEC: ion-exchange capacity; FXN: sulfonated poly(MMA-co-EGDMA-co-VBC). SCE 1: the resin produced via post-polymerization chemical modification after hypercrosslinked reaction; SCE 2: the resin produced via post-polymerization chemical modification before hypercrosslinked reaction.
Based on BET analysis, the mean size of the microsphere particles was ∼260 µm. The SSA of the resins lowered when post-polymerization chemical modification takes place before HXL process compared to post-polymerization chemical modification takes place after HXL process due to the blockage of the HXL beads which resulted from the formation of sulfur bridge and this situation leading to the decrement of the SSA. Both resins experienced low SSA value ∼4 m2 g−1 which might be due to the incomplete formation of 3-D network within HXL polymer.
Conclusion
In this study, photoinitiated dispersion polymerization technique was chosen in synthesizing precursor polymer particles due to its suitability in producing cross-linked polymer particles. HXL reaction took place using Friedel–Crafts alkylation with the aid of FeCl3 as the catalyst. During HXL reaction, chloromethyl group arising from Cl residue in polymer precursor particles was exploited. Lewis acid (FeCl3) also played a significant role which contributed to high SSA of the HXL polymer. For sulfonation reaction, sulfonation reagent used was lauric chlorosulfonic acid and resulted in the increment of sulfur content. The polymer precursor then subjected to post-polymerization after and before hypercrosslinking reaction to obtain high SSA. For post-polymerization after hypercrosslinking reaction, sulfonation took place after hypercrosslinking reaction, resulting in greater surface area, IEC value, and chlorine content compared to those of the post-polymerization before hypercrosslinking reaction. Post-polymerization after hypercrosslinking reaction showed that the chlorine content and SSA value are higher than post-polymerization before hypercrosslinking reaction. This kind of resin is more applicable for further application, and extensive research is required to obtain higher SSA. This information was important to relate the relationship between the performance of the resin and the properties of the resin.
Footnotes
Acknowledgement
The authors would like to thank Universiti Malaysia Pahang for providing laboratory facilities and financial assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Universiti Malaysia Pahang under UMP Grant Scheme Project No. RDU160331.
