Abstract
Polyaniline (PANI)/poly(4-styrene sulfonic acid) (PSS) has been prepared for various molar ratios of hydrochloric acid (HCl) (1, 2, 3 and 4 M, respectively) through in situ chemical polymerization method. The reaction temperature was 25°C for 3 h. Fourier transform infrared spectroscopy spectrum confirms the attachment of the structure to functional groups. The intensity of PANI is increased with increasing HCl concentration. Scanning electron microscopic graphs reveal the irregular morphology. The complex impedance of the PANI/PSS shows two distinguished semicircles and the diameter of the arcs decreased with an increase in ammonia concentration at room temperature. The sensitivity of the sensor increased with time, and the maximum sensitivity is 7.4 at 3 M of HCl for 500 ppm. The results are discussed and reported.
Keywords
Introduction
Ammonia (NH3) exists widely in our daily life both in nature and in industrial areas such as animals and plants rotting, industrial nitrogen fixation and concrete filler. Exposure to NH3 is harmful to human body; when people are exposed to low concentrations of NH3, it harms the nasal cavity and skin, while exposure to high concentrations can even lead to death. 1,2 On the other side, NH3 can react with •OH and •NO3 radicals and ozone which exist widely in the atmosphere in a complex way to generate some harmful and toxic products, such as formaldehyde, formic acid and hydrogen cyanide 3,4 affecting human health and environment. So it is quite important to realize the rapid, trace detection of NH3. The semiconductor gas sensor provides a good choice to detect NH3 with several advantages in terms of high sensitivity, fast response, low cost, flexibility and simplicity in fabrication. 5 An overview of the diverse NH3-sensing technologies under development was detailed by Timmer et al. 6 In brief, other NH3-sensing devices comprises of those based on solid-state sensors, 7 spectroscopic techniques 8 and conducting polymers. 9 Conducting polymer-based sensors have been progressively employed over the last few years with detection limits of the parts per million (ppm) region being demonstrated. However, these sensors can disppear through irreversible reactions leading to a reduction in response. 10 At present, most of the NH3 sensors included inorganic, inorganic oxide/dioxide and usually be expensive and exhibited low response as well as operated at high temperature, which leads to high power consumption and low lifetime. 11 -19 Therefore, fabricating the NH3 sensor which is reliable, inexpensive and operable at room temperature is significant. Nowadays, conducting polymers such as polyaniline (PANI), polypyrrole, polythiophene and their derivatives have been widely investigated as chemical sensors due to fast response, low cost and operation at room temperature. Among them, PANI is widely applied as a sensing material because of its ease of synthesis, low production costs and environmental stability as well as it has excellent sensing function to NH3 gas. NH3 sensor based on a PANI/dodecylbenzene sulfonate emulsion (in chloroform) is described by Wu et al. 20 and more recently, Dutta et al. reported the use of a PANI-coated filter paper for a number of applications including the colorimetric detection of gaseous and aqueous NH3. 21 Yun et al. 22 prepared the PANI-multi-walled carbon nanotube composite by applying a simple oxyfluorination method to control the morphology and discussed its effect on NH3 sensing of composite pellet sensors. Venkatesan and Cindrella 23 reported the study of conducting polymer-based nanomaterial pelletized sensors for NH3 sensing.
Anil G Sonkusare et al. 24 fabricated room temperature NH3 gas sensor based on PANI nanoparticles. The gas sensors developed so far were in thin films and pellet forms, irrespective of the material used. Thin-film gas sensors required the use of substrate for deposition of sensing material with some particular technique. However, the pellet form of a gas sensor does not require any substrate and time-consuming deposition technique. Therefore, pelletized gas sensors are cost-effective, robust and reasonably sensitive. The gas sensors were prepared in pellet form, which would be robust, cheap and reasonably sensitive to NH3 vapour sensing. However, to the best of our knowledge, few works have been reported for PANI and its composite for NH3 sensing using pellets.
In the present work, the preparation and characterization of PANI/poly(4-styrene sulfonic acid) (PSS) composite were done by chemical oxidation method. The prepared PANI/PSS composite was fabricated as the solid-state sensor device for NH3 sensor applications. The samples were characterized by scanning electron microscopy (SEM), Fourier transform infrared spectroscopy (FTIR), photoluminescence (PL) spectroscopy, particle size, NH3 gas sensor and impedance spectroscopy. The results are discussed and reported.
Experiment
Materials and methods
Aniline (Merck, Bangalore, Karnataka, India) and PSS (molecular weight Mw ∼ 75,000; Sigma-Aldrich, Bangalore, Karnataka, India) were purified using a distillation plant. Hydrochloric acid (HCl) and ammonium persulfate (APS) were obtained from Merck (analytical reagent grade, 99.99%; India).
PANI/PSS composites preparation
PANI/PSS composite was prepared for the different molar ratios of HCl from 1 M to 4 M in steps of 1 M. Aniline of 0.3 mol was dissolved in 100 ml of deionized water. After 30 min, 0.06 mol of APS was prepared for 100 ml deionized water and added dropwise into the solution mixture. Aqueous PSS solution of 0.6 g (Mw ∼ 75,000) was mixed with an anilinium ion solution for 1 h. Negatively charged sulphonated groups of PSS are combined with positively charged anilinium ions in aqueous solution subsequently due to the electrostatic interaction (PANI:PSS = 1:12). The polymerization was conducted for 3 h at 25°C. The resultant PANI-PSS was dark green as a conducting emeraldine salt form and well dispersed in aqueous solution without any phase separation even after 6 months. Figure 1 shows the molecular structures of PANI/PSS and the charge–charge interaction of anilinium ion/PSS complex.

Molecular structure of PANI/PSS and charge–charge interaction of anilinium ion/PSS complex.
Characterization techniques
FTIR analysis was conducted in the range of 4000–400 cm− 1 using Thermo Nicolet V-200 (India) FTIR Spectrometer by potassium bromide pellet technique. A JEOL JSM-5610 (India) SEM was used to study the surface morphology of the PANI/PSS composite powder. Impedance measurements were performed using a frequency response, Solartron, Model-1360 (India) coupled with the Solartron Dielectric Interface, 1296. The room temperature PL spectrum was recorded with the spectrofluorometer (Fluorolog, Model FL3-11, India). The particle size distribution PANI/PSS composite was analysed using Malvern Model No. 500999 instrumental analyser (India). Copper (Cu) electrodes were deposited by sputtering method.
Results and discussion
Scanning electron microscopy
SEM images of various molar ratios of HCl-doped PANI/PSS composite is shown in Figure 2(a) to (d), respectively. Figure 2(a) to (d) images exhibit irregular morphology of PANI/PSS. From Figure 2(a) and (c), it is seen that the increase in dopant concentration also increases the diameter of the particle size.

(a to d) SEM micrographs of PANI/PSS composites with various HCl concentrations (1, 2, 3 and 4 M).
FTIR spectroscopy of PANI/PSS composites
The FTIR spectra of various molar ratios of HCl/PSS-doped PANI composites are shown in Figure 3(a) to (e), respectively. C–C stretching appears around 1570 cm−1 is due to an oxidation state of PANI. 25 The band at 800 cm−1 shows C–Cl aromatics out of a plane bend. The peak at 1298 cm−1 is due to C–N stretch of a benzenoid ring. The band at 623 cm−1 relates to C–Cl stretching vibration. The N–H symmetric stretching of PANI appears at 3425 cm−1. The peak at 1020 cm−1 is due to C–H in-plane bending and S=O stretching from the doped PSS, respectively. 26,27 The bands at 635 and 798 cm−1 are the characteristics of C–H out-of-plane ring deformation vibration. The characteristic symmetric- and antisymmetric-stretching peaks of SO3 − were observed at 1185 and 1041 cm−1. It was observed that the increase in dopant concentration of HCl shows less variation in the intensity of peaks and only a slight shift in the peak positions.

FTIR spectra of PANI/PSS composites with various HCl concentrations (1, 2, 3 and 4 M).
PL of PANI/PSS composites
Figure 4 shows the PL spectrum of various molar ratios of HCl/PSS-doped PANI composite. The peak at 490 nm shows for PANI/PSS composites, which is specified in the wavelength of green light. 28 Shimano and MacDiarmid have reported that a fully doped state of PANI, the peak at 409 nm will completely disappear and peak at 494 nm will be highly intense. In our case, the combination of a sharp high intense peak at 490 nm shows that the PANI-doped HCl is partially doped which confirms its semiconducting behaviour. 29 Banerjee et al. studied that the PL peak at 494 nm is assigned to polaron → π transition. 30 When the dopant concentration has increased the density of defect states in the polaronic band also increases which results in more transitions from the polaronic band to the π-band Highest Occupied Molecular Orbital and therefore the increase in the intensity of the PL peaks can also be attributed to variation of the dopant acids. This leads to an increase in the density of charge carriers which increases with the increase in dopant concentration. This gives rise to the higher density of states in the bandgap resulting in increased PL efficiency. PL study shows that an increase in the intensity has increased the dopant concentration.
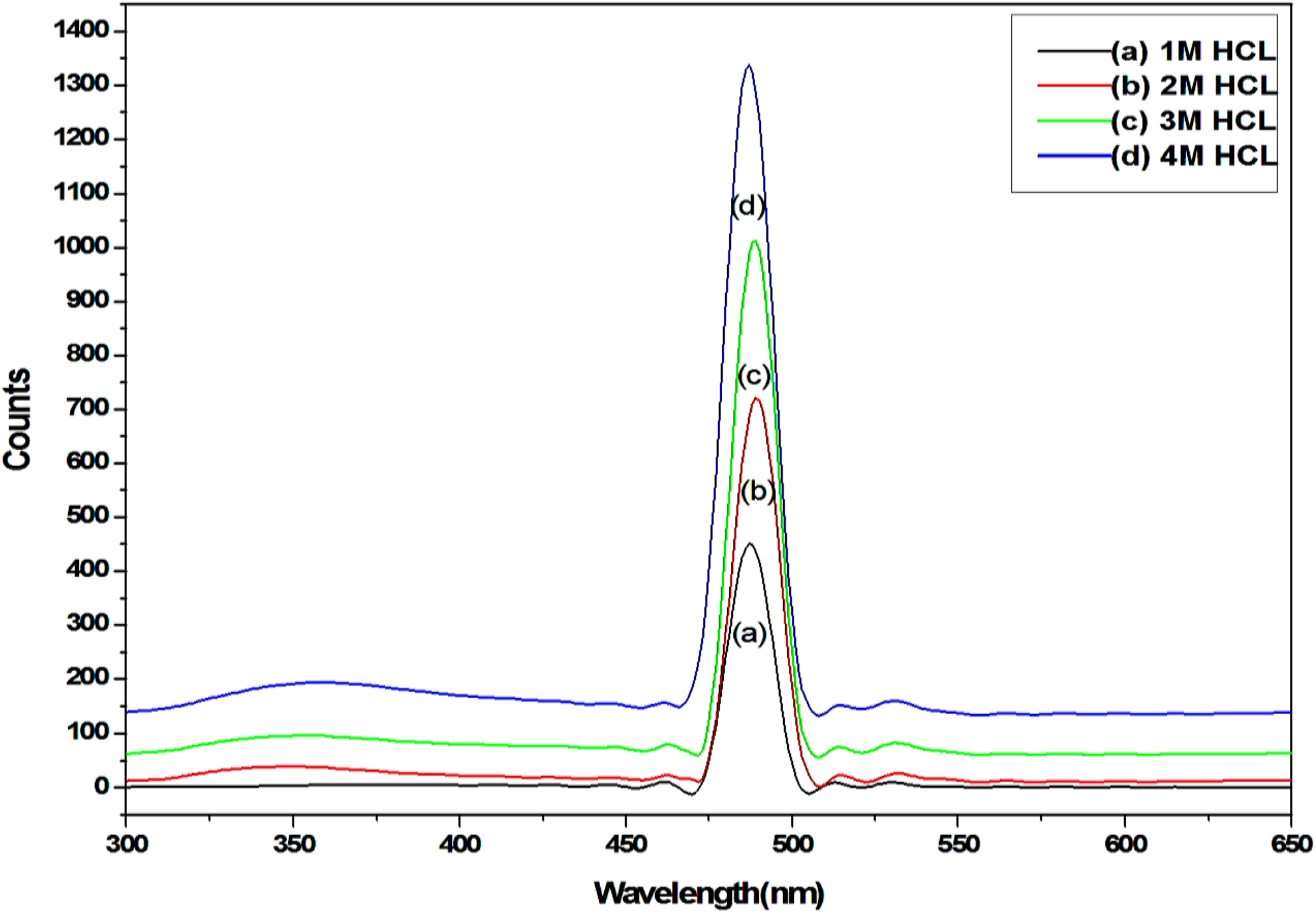
PL spectra of PANI/PSS composites with various HCl concentrations (1, 2, 3 and 4 M).
Particle size analysis of PANI/PSS composites
The particle size distribution of PANI/PSS composites is shown in Figure 5. PANI/PSS was dispersed in distilled water. The size of the composites was found to be 151, 180, 205 and 375 nm for different molar ratios (1, 2, 3 and 4 M) of HCl, respectively. In this case, the intensity of PANI/PSS composite increases with increasing HCl molar ratio. It was also observed that the PANI in the presence of a higher ratio of HCl increases particle size.
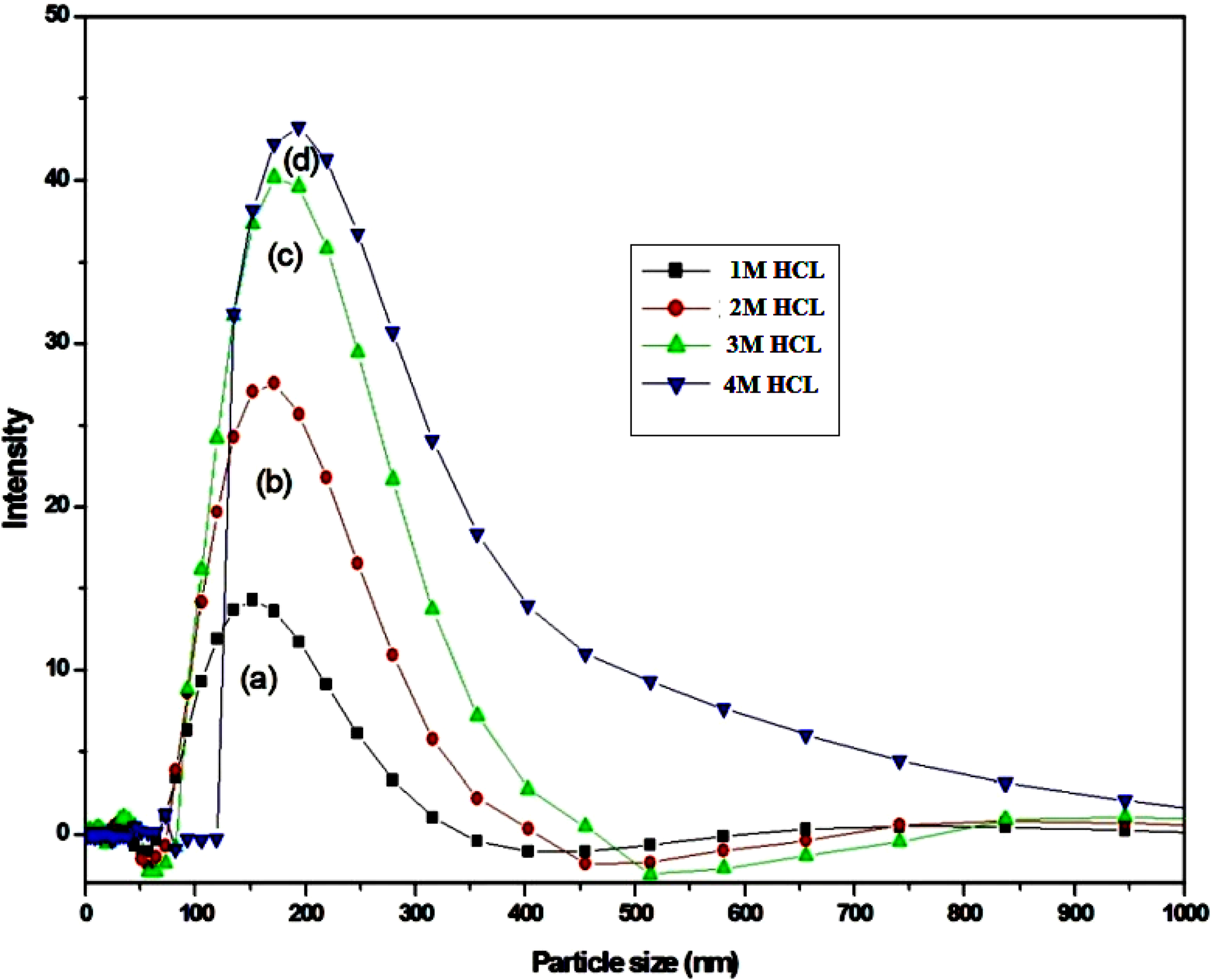
Particle size analysis of PANI/PSS composites with various HCl concentrations (1, 2, 3 and 4 M).
NH3 gas-sensing properties of PANI/PSS composite device
Figure 6 shows the schematic diagram of the sensor device developed for the study. The sensor consists of PANI/PSS composite pellet on top of the Cu electrode. The contacts were made by sputtering of an electrode of Cu through a shadow mask. The distance between the Cu electrodes was about 2 mm with a thickness of approximately 250 nm. Prior to deposition, the surface was cleaned in acetone for 15 min, followed by 15 min of ultrasonic treatment to remove organic contaminants.

Schematic layout of PANI/PSS composite solid work sensor device.
Figure 7(a) and (b) shows the complex impedance analysis of PANI/PSS composite work sensor device in the ratio of HCl (3 and 4 M) with different NH3 concentrations 100, 250 and 500 ppm. The semicircle arcs were generated with centres lying near the real axis providing a Debye-type of relaxation. It is also obvious that PANI/PSS composite shows a typical negative temperature coefficient of resistance up to 30 min where the absorbed oxygen traps the electrons from the PANI/PSS composite surface resulting in the increase of the carrier concentration that led to the decrease of the sample resistance at 3 M of HCl with increase in NH3 concentration. The plots of Z′′ versus log f for the PANI/PSS sensor are shown in Figure 8(a) and (b). It can be seen that the decrease in the values of Z′′ with the increase of NH3 concentration indicates the decrease in the loss in the system. Besides, the peak frequency shifts towards higher frequencies, and the peak height decreases with the increase of NH3 concentrations. The peak broadening suggests the dependence of the relaxation process on the gas concentration.

(a) Complex impedance analysis of PANI/PSS composite solid work in the ratio 3 M HCl for 100, 250 and 500 ppm ammonia. (b) Complex impedance analysis of PANI/PSS composite solid work in the ratio 4 M HCl for 100, 250 and 500 ppm ammonia.
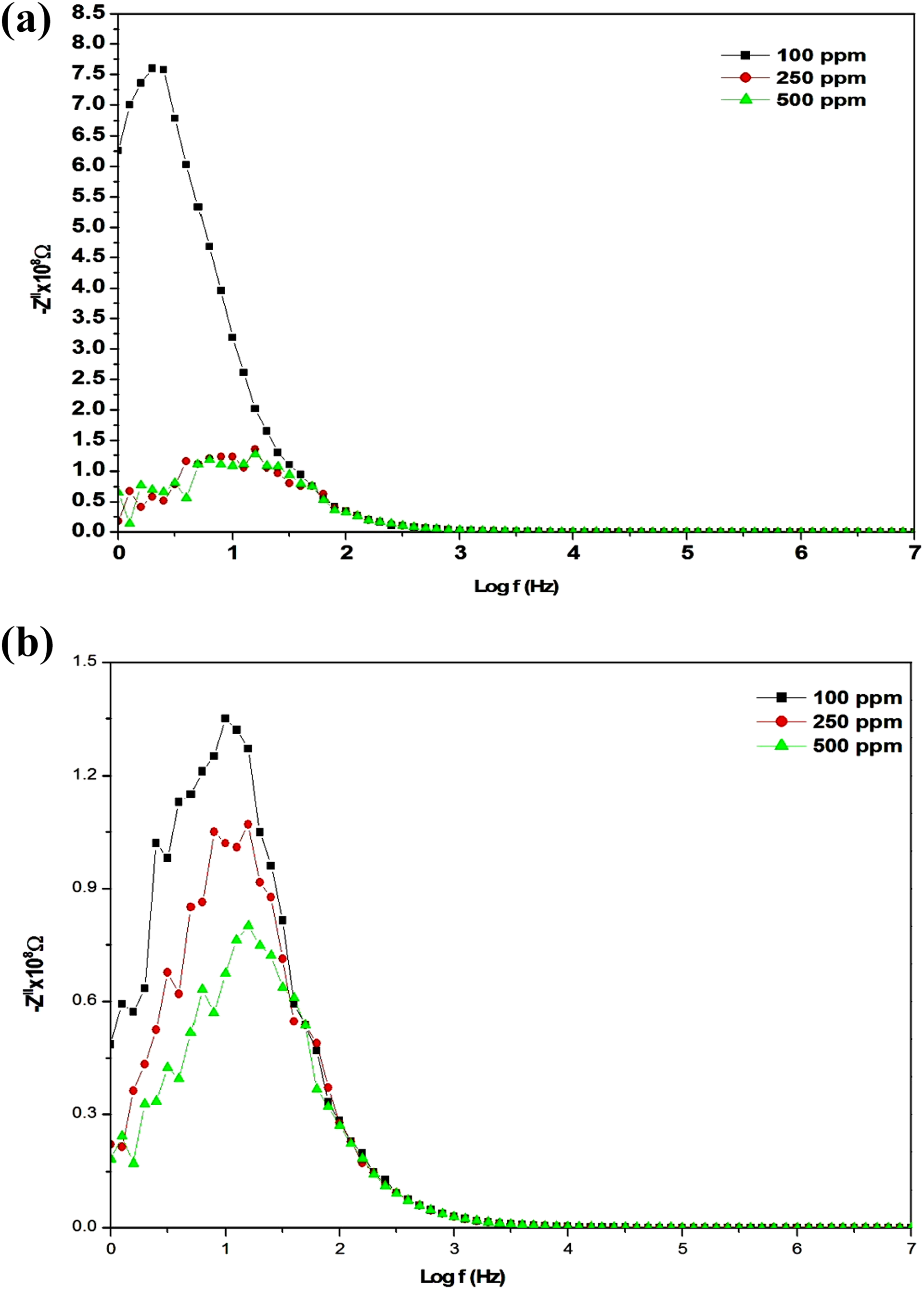
(a) Imaginary part of the impedance versus frequency for PANI/PSS composite solid work in the ratio 3 M HCl for 100, 250 and 500 ppm ammonia. (b) Imaginary part of the impedance versus frequency for PANI/PSS composite solid work in the ratio 4 M HCl for 100, 250 and 500 ppm ammonia.
The sensitivity (S) of PANI/PSS sensor was calculated as a function of frequency and NH3 concentration according to the following relationship:
where Za and Zg are the real part of the impedance for dry air atmosphere and/or the NH3 atmosphere. The sensitivity (S) of the prepared PANI/PSS composite sample was studied as a function of frequency and NH3 concentration according to equation (1). Figure 9(a) and (b) reveals the effect of both the frequency and NH3 concentrations at an operating temperature of 30°C on the sensitivity of the PANI/PSS composite sample. It can be observed that the sensitivity of the sensor remains constant within the frequency range of 100–1000 Hz, the range where the space charge region rules the conductivity process. The sensitivity decreases sharply as the frequency increases and becomes almost constant at a frequency higher than 100 kHz, in which the conductivity is controlled by the surface charge of the grains. This suggests that the sensor can be tuned to achieve maximum sensitivity be selecting the suitable operating frequency range. It is found that the maximum sensitivity is 7.4 (3 M HCl) and 1.9 (4 M HCl) for 500 ppm. The sensitivity and dopant concentrations are presented in Table 1. Marcelo Eising et al. (2017) studied composites based on carbon nanotubes and PANI films as NH3 gas sensors with three different doping approaches (sulphuric acid, camphor sulphonic acid and m-cresol). The PANI high sensitivity to NH3 combined with the stability of the carbon nanotube with organic acid increased the sensor reproducibility when compared to regular PANI. The sensor in which the doping was performed by camphor sulphonic acid showed the best sensor response for NH3 gas when compared to others. 35 In this present work, PSS-doped PANI shows better sensing response at a 3 M concentration of HCl compared to other doping concentrations.

(a) Response of the PANI/PSS composite solid work in the ratio 3 M HCl for 100, 250 and 500 ppm ammonia. (b) Response of the PANI/PSS composite solid work in the ratio 4 M HCl for 100, 250 and 500 ppm ammonia.
Dopant concentration and sensitivity.

PANI: polyaniline; HCl: hydrochloric acid.
Conclusions
PANI/PSS has been prepared for various molar ratios of HCl (1, 2, 3 and 4 M, respectively) by in situ chemical polymerization method. The reaction temperature was 0–5°C for 24 h. The FTIR spectrum confirms the attachment to functional groups of the structure. SEM graphs reveal the irregular morphology. Impedance spectroscopy analysis indicates that grain boundary resistance decreases with increasing NH3 concentration up to 500 ppm. The complex impedance of the PANI/PSS shows two semicircles and the diameter of the arcs decreased in diameter as the NH3 concentration increases at room temperature. The sensitivity of the sensor was found to be increased from time and maximum sensitivity is 7.4 at 3 M of HCl (500 ppm). The investigation results of the prepared PANI/PSS ensure their stability and suitability for the development of gas sensor devices for room temperature. PL studies reveal that increases in the intensity increase the dopant concentration.
Footnotes
Acknowledgements
The authors are thankful to Sophisticated Analytical Instrument Facility (SAIF), Sophisticated Test and Instrumentation Center (STIC), Cochin, Kerala, and Birla Institute of Technology, Mesra, Ranchi, for providing instrumental facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

