Abstract
With excellent damping capacity, growing interest has been focused on polymer concrete (PC) as a novel machine tool bed material in the field of ultraprecision machining. It is widely acknowledged that the fly ash (FA) is an essential component material in reducing the curing shrinkage of PC, which can significantly affect the curing characteristic of PC. However, the effect of FA on the curing characteristic of epoxy resin is not studied in detail. In this article, the effect of FA on the curing characteristic of epoxy resin was examined by differential scanning calorimetry. Experimental results show that the peak temperature and curing rates of epoxy resin/curing agent and epoxy resin/curing agent/FA increase with the increasing heating rates, and the peak temperature and curing rates of epoxy resin/curing agent are greater than that of the epoxy resin/curing agent/FA with the same heating rates. In addition, the difference between the maximum curing rates of epoxy resin/curing agent and epoxy resin/curing agent/FA decreased with the increasing heating rates. This article can provide technical reference for curing process of PC for machine tool beds and further improve the machining accuracy.
Introduction
Coal resource was the second largest energy source in the world, which offered almost 30% of the world’s nonrenewable energy resource. 1 In China, more than 80% of power comes from coal power plants. 1 However, the large consumption of coal resources brings a large amount of serious problems. For example, the sulfur dioxide and nitrogen dioxide generated from coal combustion can damage the human respiratory system, and the acid rain generated by the reaction of water and sulfur dioxide (or nitrogen dioxide) destroys the natural environment. 2 In addition, fly ash (FA), another product of coal combustion, even brings more serious pollution of the environment.
FA is a farinose solid waste produced during coal combustion, which is the major industrial waste of the thermal power plant. With the rapid development of energy demand, the output of FA exhibited a trend of increasing year by year. However, the problems caused by FA are becoming ever more prominent. In the past 50 years, the FA generated during coal combustion was usually discharged without any treatment, resulting in the pollution of the surrounding environment and further endangering human health. 2 Especially in China, air pollution caused by smog has increased the number of people suffering from lung cancer. 3 Therefore, the effective treatment of FA will become a new research focus, and the efforts for solving the pollution caused by FA will directly or indirectly generate enormous economic and social value.
A convenient and viable way to deal with the pollution caused by FA is improving the current efficiency of FA use. The utilization of FA has varied widely from a minimum of 3% to a maximum of 90%. 1 Yao et al. 4 estimated that the FA utilization factors of 50% for the United States, over 90% for the European Union, and about 60% for India. But in China, the utilization of FA has increased year by year, which has remained around 67% in recent years. FA has been applied successfully in many industries such as agriculture, 5 civil construction industry, 1,6 sewage treatment industry, 1,7 and machine tools. 8,9 Especially in the field of mechanical engineering, FA has attracted wide attention by the machine tool researcher. 9
In the field of machine tool industries, machining precision is one of the most important parameters, which can determine the operational performance of machine parts. However, the machining precision is easily vulnerable to a wide variety of factors in a complex cutting process. 10 These factors including properties of the machined material, relative chatter vibration between the tool and workpiece, the vibration of the tool itself, vibration of machine tool spindle, geometrical parameter of cutter, cutter material, and cutting conditions. 11 The vibration produced during the whole process of machining is a crucial factor, which directly determines the machining accuracy and the structural stability of machine tool. 12 Therefore, material of machine tool bed must exhibit a favorable damping property aim at obtaining high machining precision. With inferior damping properties, the traditional materials of machine tool bed, that is, cast iron and welded steel, can no longer satisfy the requirement of superior machine accuracy. 13 -15 Accordingly, future research of machine tool structures aims at developing an alternate material, which exhibits perfect damping property along with its excellent mechanical properties. 13 Interestingly, the appearance of polymer concrete (PC, also named as FA-reinforced polymer composite) can compensate for the above deficiency by luck coincidence.
Unlike traditional cement concrete, PC 16,17 is a new composite that used FA as filler and organic resin as binder, which exhibits prominent damping properties due to the presence of resin matrix, FA, 9 and porosity of material. 18 PC has huge application prospects in machine tool beds because of it displays a multitude of unique merits compared with other material of machine too bed, for instance, superior adhesion properties, higher impact toughness, lower curing shrinkage, positive heat insulation properties, low cost, excellent waterproofness, shorter curing time, and so on. 17
In the past two decades, a considerable amount of research concerning the effect of FA on the performance of the PC was performed. 9,18 -22 Compared with metal materials in machine tool bed, PC exhibits inferior mechanical properties. Therefore, previous study regarding the effect of FA on the performance of PC mainly focused on the mechanical properties of PC. 19 -22 It is widely acknowledged that the mechanical properties of PC have a close relationship with the curing characteristic of PC. Numerous methods can be implemented to investigate the cure kinetics of epoxy resin/curing agents, 23 such as differential scanning calorimetry (DSC), infrared spectroscopy and dielectric spectroscopy. In this article, the DSC technique was applied to investigate the kinetics characteristics of epoxy resin and its additives. Harsch et al. 23 investigated the cure kinetics of a cycloaliphatic epoxy resin with and without additives and cured with an anhydride hardener by the method of DSC. De Schoenmaker et al. 24 investigated the effect of nanofibres on the curing characteristic of an epoxy matrix by the method of DSC. The results showed that the polyamide nanofibres have a catalytic effect on the diglycidyl ether of bisphenol-A (DGEBA)-4,4’-methylene dianiline (MDA) cure. Vijayan et al. 25 studied the effects of nanoclay, carboxyl-terminated (butadiene-co-acrylonitrile) liquid rubber, and the combination of both on the cure kinetics of DGEBA-based epoxy resin/nadic methyl anhydride. However, to the best of our knowledge, no information has been focused on the effect of FA on the curing characteristic of epoxy resin. Namely, previous studies ignored the importance of the curing characteristic of the PC. Therefore, the purpose of this article is to discuss the curing characteristic of epoxy resin when the FA was added into epoxy resin. Moreover, the effect of heating rate on the curing characteristic of epoxy resin and PC was further investigated systematically.
Experimental procedures
Materials
Resin matrix
Epoxy resin 618A was used in this study, which was supplied by Yi Sheng Resin Co. Ltd (Jinan, China). The curing agents used in this investigation were 593, which were produced by Wuxi Guangming Huagong Equipment Co. Ltd (Wuxi, China), and the flexibilizer dibutyl phthalate (DBP) was purchased from Jing Hao Chemical Co. Ltd (Jinan, China). Acetone was used as the diluent to reduce the viscosity of epoxy resin, which was provided by Highly Chemical New Materials Co. Ltd (Qingdao, China). Mass content of various components in the resin system is listed in Table 1.
Mass fraction of the resin system.

DBP: dibutyl phthalate.
Filler
FA was a by-product from Jinan power plants, and the grain diameter of FA is in the range of 0.1–10 μm.
As a kind of concrete composite for the precision machine tool bed, the compressive strength of PC is the main loading to be considered. Based on the maximum compressive of PC, the mass content of FA in 618A/593/FA is 33.3%.
Sample preparation
Sample preparation of 618A/593
In 618A/593, the epoxy resin refers to 618A, and the curing agent refers to 593. The preparation process is divided into three steps. Firstly, the acetone was added into epoxy resin and stirring with the JJ-1 precision booster electric mixer for 2 min. Additionally, the stabilizer was added into the previous mixture and stirring for 2 min. At last, DBP was added into the previous mixture for 2 min.
Sample preparation of 618A/593/FA
618A/593/FA refers to the epoxy resin is 618A, the curing agent is 593, and the filler is FA. The preparation process is divided into four steps. Firstly, the acetone was added into epoxy resin and stirring with the JJ-1 precision booster electric mixer for 2 min. Additionally, the stabilizer was added into the previous mixture and stirring for 2 min. Thirdly, DBP was added into the previous mixture for 2 min. At last, the FA was added into the previous mixture and stirring for 2 min.
Differential scanning calorimetry
To investigate the initial curing temperature of the resin system, the heat flow at different heating rates of 3, 8, and 12°C min−1 were measured. The DSC instrument used was NETZSCH DSC 200F3 (Germany). Aluminum crucibles 40 μl were used, being filled with approximately 10 mg of 618A/593 and 618A/593/FA for each test. Nitrogen (70 ml min−1) was used as pure gas.
Curing kinetics
Curing process of epoxy resin is an exothermic process, and the curing kinetic parameters were most frequently obtained by a DSC technique in isothermal and non-isothermal modes. In this article, the registered DSC heat flow characterizing the reaction exothermic rate is directly proportional to the reaction rate, and thus the fractional conversion, α, can be determined by the following equation 26 :

where α is the extent of curing reaction, ΔHR is the total heat of reaction, and Q(t) is the heat flow measured by the DSC experiment.
Therefore, the curing rate can be calculated by the following equation 23 :

Because of T = βt, the curing degree and curing rate can also be expressed by the following equation, respectively.


where β is the heating rate and T is the absolute temperature.
Cure kinetic models of 618A/593 and 618A/593/FA can be categorized into two main types, phenomenological and mechanistic. Generally speaking, the curing kinetics can be obtained by two methods: non-isothermal method and isothermal method. In this article, we used the former method to analyze the cure kinetic characteristic.
The apparent activation energy (Ea) was obtained by various computational methods, such as the Friedman method, the Ozawa method, the Kissinger method, and so on.
In the Friedman method, the apparent activation energy (Ea) is assumed to be a variable value. The Friedman method can be expressed as the following equation 24,27,28 :

where A is the pre-exponential factor, Ea is the apparent activation energy, R is the ideal gas constant, and f(α) is a function of conversion which has a close relation with the reaction mechanism and represents the kinetic model.
As during curing of the epoxy resin, the reaction is only dependent on the temperature, both the pre-exponential factor A and the function of conversion f(a) are constant when the conversion is a constant. So, regression analysis between ln(da/dt) and 1/T for a fixed conversion will result in a straight line with a slope.
Based on the assumption that the apparent activation energy is a constant, Ozawa developed an alternative method to determine the apparent activation energy, and this method can be expressed as the following equation 27,28 :
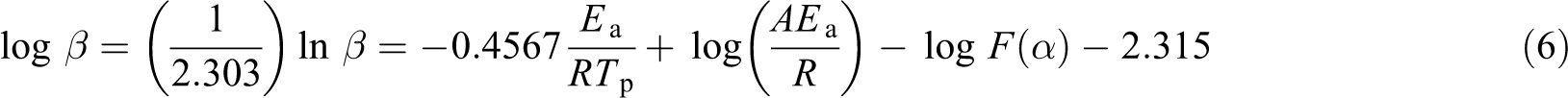
where Tp is the temperature corresponding to the exothermic peak and F(α) is a constant function, described as
The activation energy 27,28 of the curing process can be obtained by the slope of the linear relationship between the logarithm of the heating rate lnβ and the reciprocal of the peak temperature of the curing reaction Tp.

Kissinger 25,27 developed another method to determine the apparent activation energy.

Similarly, the activation energy Ea is obtained by the slope of plots of
The reaction order could be obtained by Crane equation. The Crane equation can be expressed by the following equation when Ea>>2TP:

where n is the reaction order of curing reaction.
Results and discussions
Dynamic DSC curves
To investigate the activation energy and the total heat of curing reaction, the heat flow at different heating rates of 3, 8, and 12°C min−1 was performed. Figure 1 shows the dynamic DSC curves of the non-isothermal curing reactions of 618A/593 and 618A/593/FA with the multiple heating rates. Obviously, the non-isothermal heat flow curves of 618A/593 and 618A/593/FA at different heating rates show a single exothermic peak with good symmetry, and the reason is that the highly exothermic epoxy-amine addition.

Dynamic DSC curves of (a) heat flow versus temperature and (b) heat flow versus time at different heating rates.
It can also be observed in Figure 1(a) that the peak temperature of 618A/593 increases with the increasing heating rates and simultaneously exhibits the broader peak area. Similar result was obtained in the non-isothermal heat flow curves of 618A/593/FA.
Figure 1(a) also showed clearly that the peak temperature of 618A/593 is greater than that of the 618A/593/FA with the same heating rates, and the curing reaction starts at lower temperatures when FA is added to 618A/593. Accordingly, it can be concluded that the FA has a catalytic effect on the curing reaction of 618A/593.
To investigate the curing time process of 618A/593 and 618A/593/FA, heat flow of 618A/593 and 618A/593/FA as a function of time for non-isothermal curing with different heating rates of 3, 8, and 12°C min−1 is shown in Figure 1(b). Obviously, the curing time of 618A/593 and 618A/593/FA reduced with the increasing heating rates. Consequently, it can be concluded that the increases in the heating rates accelerate the curing process of 618A/593 and 618A/593/FA. It can also be observed in Figure 1(b) that the total curing time of 618A/593/FA is longer than 618A/593 with the same heating rates. Obviously, results indicate that the curing rate reduced when the FA is incorporated in the 618A/593.
Curing kinetics of 618A/593 and 618A/593/FA
To investigate the activation energy and further obtain the curing kinetics equation of 618A/593 and 618A/593/FA, the relationship between activation energy Ea and curing degree a is depicted in Figure 2 according to equation (5). Obviously, Figure 2 shows that the activation energy of 618A/593 and 618A/593/FA exhibits different levels of decrease with the ascending curing degree when the curing degree increases from 0.1 to 0.9. However, the activation energy of 618A/593 is far lower than the 618A/593/FA. Namely, the activation energy of 618A/593 was reduced due to the absence of FA. In contrast, several studies 24,28,29 showed that the activation energy displayed a slightly decreased when the additives were added into the epoxy resin. Schoenmaker et al. 24 investigated the effect of nanofibers on the activation energy of DGEBA/MDA resin, and the result showed that the activation energy exhibited a slowly reduce owing to the presence of nanofibers. Similar results were obtained by Ghaffari et al. 28 and Zhang et al. 29 when the additives were added into epoxy resin.
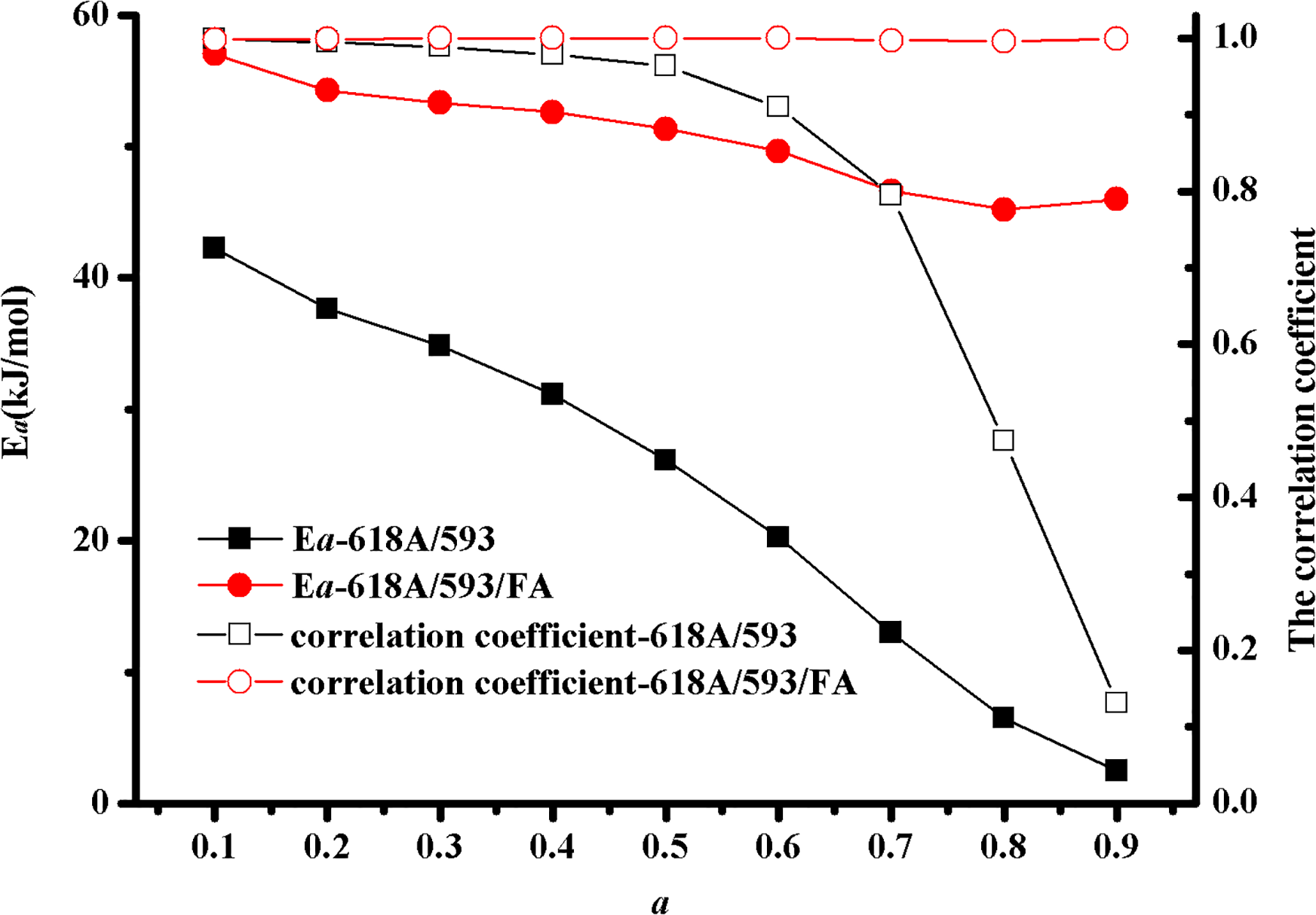
Variation of Ea versus a by the Friedman method.
It can also be seen from Figure 2 that the correlation coefficient of the Friedman method for 618A/593/FA shows not only a higher fit degree but also no obvious change with the increment of curing degree. In addition, the activation energy of 618A/593/FA exhibits a slight reduction with the ascending curing degree. Thus, it can be concluded that the activation energy of 618A/593/FA is a constant. Interestingly, both the correlation coefficient of the Friedman method and the activation energy for 618A/593 presented a sharp decline when the curing degree excess 0.6. Additionally, the correlation coefficient of the Friedman method for 618A/593 shows an excellent fit degree when the curing degree less than 0.6. Accordingly, we can preliminarily deduce that the activation energy of 618A/593 is a constant.
To further verify whether the activation energy is a variable or a constant, the relationship between the curing rate and the curing degree of the 618A/593 system and 618A/593/FA at different heating rates is shown in Figure 3. It seems from Figure 3 that the curing rate first increases and then decreases with the ascending curing degree for both samples, and the maximum curing rate of 618A/593/FA for different heating rates was achieved when the curing degree is nearly 0.47. So, we believed that the activation energy of 618A/593/FA is a constant. However, the maximum curing rate of 618A/593 for 3, 8, and 12°C min−1 was obtained when the curing degree is nearly 0.57, 0.46 and 0.46, respectively. This result indicated that the activation energy of 618A/593 can be considered as a constant.

Plot of curing rate versus curing degree at different heating rates for 618A/593 and 618A/593/FA.
The peak temperatures obtained by non-isothermal measurements for the 618A/593 and 618A/593/FA at different heating rates are transformed according to the equations of Kissinger and Ozawa, respectively. As shown in Figure 4(a), the reciprocal temperature 1/Tp is plotted against the logarithm of the heating rate lnβ for both 618A/593 and 618A/593/FA following the Ozawa equation. It can be seen clearly in Figure 4(a) that the reciprocal temperature 1/Tp has a close relation with the logarithm of the heating rate lnβ, and the correlation coefficient of 618A/593 and 618A/593/FA is on the order of 99.9% and 99.92%, respectively. The activation energy Ea can calculate by the slope of the fitted straight line. The calculated results are 54.18 and 59.85 kJ/mol for 618A/593 and 618A/593/FA, respectively.

Plots of (a) 1/TP versus lnβ(a) and (b)
The peak temperatures of 618A/593 and 618A/593/FA at different heating rates were obtained and
It can also be noted from Figure 4(a) and (b) that the activation energy increased when the FA was added in 618A/593. The values obtained from these two approaches have no noticeable difference even though the activation energy obtained from the Kissinger method is slightly higher than that from the Ozawa method. However, Starink et al. 30 found that the Kissinger method is more accurate than the Ozawa method. So, the activation energy of the Kissinger method for both 618A/593 and 618A/593/FA was chosen in this article.
To calculate the reaction order of 618A/593 and 618A/593/FA, the relationship between lnβ and 1/TP is depicted in Figure 5. Based on equation (9), the reaction order of 618A/593 and 618A/593/FA is 0.8873 and 0.8975, respectively.

Plots of lnβ versus 1/TP (crane) for (a) 618A/593 and (b) 618A/593/FA.
To exhibit the effect of the FA more intuitively, the activation energy, pre-exponential factor, and reaction order of 618A/593/FA were summarized in Table 2. Obviously, the activation energy, pre-exponential factor, and reaction order of 618A/593/FA were increased due to the presence of FA.
Kinetic parameters of the Kissinger model for non-isothermal cure of 618A/593 and 618A/593/FA.
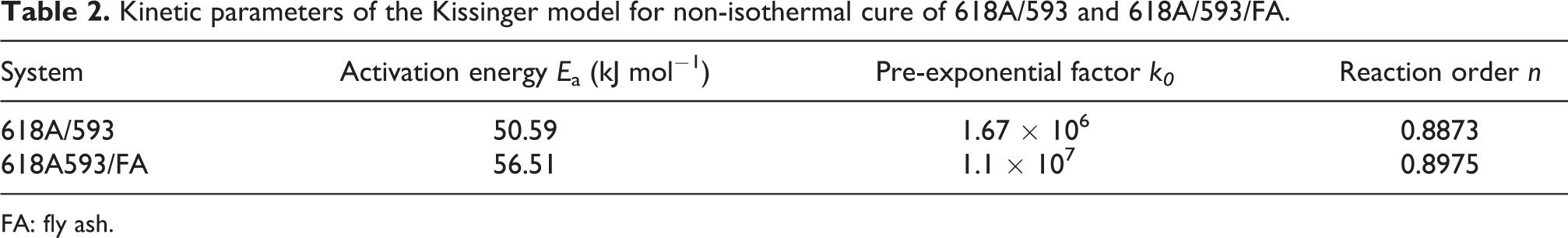
FA: fly ash.
Curing characteristic of 618A/593 and 618A/593/FA based on the DSC curves
Curves of curing degree versus time
Based on Figure 1 and equation (1), the curves of curing degree versus time for 618A/593 and 618A/593/FA at different heating rates were displayed in Figure 6(a). It can be clearly seen that both 618A/593 and 618A/593/FA curves at different heating rates exhibit a sigmoidal contour. The curing degree of 618A/593 and 618A/593/FA first increased gradually and then increased remarkably and finally increased slightly to the maximum curing degree.

(a) Curing degree versus time, (b) curing degree versus temperature, (c) curing rate versus time, and (d) curing degree versus time of 618A/593 and 618A/593/FA.
As shown in Figure 6(a), the reaction times to reach the same conversion for 618A/593 are dramatically decreased with ascending heating rates, which indicated that the curing rates increased with the increasing heating rates. Similar results were obtained in the curing process of 618A/593/FA. These results were consistent with the previous finding shown in the “Dynamic DSC curves” section.
It can also be seen from Figure 6(a) that the total curing time of 618A/593/FA is longer than 618A/593 with the same heating rates, which suggested that the curing rate reduced when the FA is incorporated in the 618A/593.
Curves of curing degree versus temperature
To exhibit the relationship between curing degree and temperature more intuitively, the conversional curves of the non-isothermal cure of 618A/593 and 618A/593/FA at different heating rates were illustrated in Figure 6(b) according to Figure 1 and equation (3). Both 618A/593 conversional curves and 618A/593/FA conversional curves exhibit a sigmoidal contour and shift to a higher and broader temperature range with the increased heating rate, demonstrating that the temperature for the reaction to reach the same conversion is systematically increased. 23 As shown in Figure 6(b), the temperature for reaching the same conversion of 618A/593/FA is greater than 618A/593.
Curves of curing rate versus time
To demonstrate the effect of the FA and heating rates on the curing rates of 618A/593, the relationship between curing rate and time is shown in Figure 6(c). It can be seen from Figure 6(c) that the curing rate of 618A/593 and 618A/593/FA at different heating rates first increased and then decreased with the increasing time. With the increase of heating rates, the maximum curing rates of 618A/593 and 618A/593/FA increased remarkably, whereas the total curing time decreased gradually.
It can also be observed in Figure 6(c) that the maximum curing rates of 618A/593 displayed a slight decline with the addition of FA, which was consistent with the previous finding shown in the “Dynamic DSC curves” section. Therefore, we can conclude that the incorporation of FA decreased the curing rate of 618A/593. In addition, the maximum curing rates difference of 618A/593 and 618A/593/FA decreased with the increasing heating rates. In other words, the increases of heating rates reduced the influence of FA on the curing rate of 618A/593.
Curves of curing rate versus temperature
For further study the FA and heating rates on the curing rates of 618A/593, the relationship between curing rate and temperature is depicted in Figure 6(d). Obviously, both 618A/593 and 618A/593/FA curves at different heating rates exhibit a parabolic contour. With the increase of heating rate, that is to say, the curing rate of 618A/593 and 618A/593/FA at different heating rates initially increased and then decreased, and the temperature of maximum curing rate increased remarkably.
It can also be observed clearly in Figure 6(d) that the maximum curing rate reduced when the FA was incorporated in the 618A/593. In the meantime, the curves of curing rates versus time shift to a broader temperature range after the FA were added into 618A/593.
As a summary, the FA has an important influence on the curing characteristic of PC. It should be noted that this study has not discussed the effect of the mass ratio of resin and curing agent and the mass content of FA on the curing characteristics of epoxy, because the optimal mechanical strength was obtained when the mass content of FA in 618A/593/FA is 33.3%, and the mass ration of resin and curing agent is 4:1. In addition, future work should be focused on the curing process of PC and the isothermal curing characteristics.
Conclusions
Curing characteristics of PC are highly related to the FA, and FA has a greater effect on the curing characteristics of epoxy resin and its additives according to this article. In addition, this study may provide great importance to optimize the curing process of PC. The following conclusions can be drawn based on the above experimental results. Both the peak temperatures of 618A/593 and 618A/593/FA increase with the increasing heating rates, and the peak temperature of 618A/593 is greater than that of the 618A/593/FA with the same heating rates. This conclusion can provide an important reference for controlling the curing temperature and preventing the thermal deformation of PC. The curing rates of 618A/593 and 618A/593/FA increased with the increasing heating rates, the curing rate of 618A/593, and 618A/593/FA at different heating rates first increased and then decreased with the increasing time. With the increase of heating rates, the maximum curing rates of 618A/593 and 618A/593/FA increased remarkably and the maximum curing rates difference of 618A/593 and 618A/593/FA decreased with the increasing heating rates. Therefore, we can improve the heating rates to accelerate the curing rates of PC in the later period. The activation energy, pre-exponential factor, and reaction order of 618A/593/FA were increased due to the presence of FA. The curing rate of 618A/593 reduced when the FA is incorporated.
Footnotes
Acknowledgements
The author thanks editors and anonymous reviewers for helpful comments and suggestions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work of this article is supported by the Scientific Research Foundation of Taishan University (grant no. Y012016011) and the Natural Science Foundation of Shandong Province (grant no. ZR2018LE012).
