Abstract
The design and development of proton conducting polymer electrolyte membranes from a linear constituent, sulfonated poly (ether ether ketone) (SPEEK), and inorganic additive, niobium oxide (NBO), have been achieved. The degree of sulfonation of SPEEK was measured by back titration method and found to be 57%. The physicochemical properties such as water uptake ability, ion-exchange capacity, swelling ratio, proton conductivity, and thermal stability of the prepared polymer nanocomposite membranes were studied in detail. The distribution of NBO throughout the polymer matrix has been examined by scanning electron microscopic and X-ray diffraction analyses and found to be uniform. The SP-NBO-10 composite membrane shows 38.4% of water uptake, whereas the pristine membrane limits to 27.1%. The prepared electrolyte membranes exhibit good proton conductivity at temperature varying from 30°C to 90°C and possess less activation energy for the transportation of proton by the incorporation of NBO filler. The thermal studies demonstrated that the stability of the composite membranes was significantly enhanced by the impregnation of NBO. The filler NBO shows excellent improvements on the polymer nanocomposite, making it a very promising additive for other polymers and offers new roads for energy applications.
Introduction
Fuel cells appear to be environment-friendly energy conversion device which meets the technological needs for current issues related to power generation. Perfluorosulfonic acid membrane such as Nafion® produced by DuPont is a versatile polymer in proton-exchange membrane (PEM) because of its magnificent thermal and mechanical stability along with the proton conductivity as high as 0.1 S cm−1. 1 However, Nafion shows diminished mechanical and chemical stability with considerable loss in proton conductivity at high temperature. These restrictions associated with high cost confine the practical usage of Nafion in fuel cell (FC) applications. Cathode flooding is also one of the major problems that exists in low-temperature FCs which is not observed at high temperatures due to the existence of water vapour. 2 The key challenge for the researchers is to develop the best alternative commercial electrolyte membranes pertinent at high temperatures.
Non-fluorinated hydrocarbon-based polymer electrolyte membranes such as sulfonated poly (ether ether ketone) (SPEEK), sulfonated poly (ether sulfone), sulfonated polyimide, sulfonated polysulfone, and sulfonated poly styrene are promising substitute for Nafion because of their large hydrophilic/hydrophobic interface and narrow water channels. 3 To enhance the hydrophilicity for proton transportation, sulfonation of aromatic hydrocarbon is an essential process to develop polymer electrolyte membranes. Poly (ether ether ketone) (PEEK) has been chosen as the best base polymer because of its excellent mechanical and thermal stability and considerable cost-effectiveness. The SPEEK itself or making composite with various additives are reported and utilized as an effective membrane in FC applications. 4 -8 Incorporation of finely dispersed nanoparticles modifies the single-phase pure polymer structure. In general, the proton conductivity can be alleviated by the introduction of inorganic oxides into the pristine membrane. The presence of nanoparticles such as titanium dioxide, silicon dioxide, and zirconium dioxide in the virgin polymer formed a new series of nanocomposites and showed appropriate performance in hydrogen oxygen FC. 9 -11
The organic segment provides the benefit of flexibility and multifunctional reactivity, whereas the inorganic part provides high mechanical and thermal stability. 12 -17 The effective dispersion of inorganic nanoparticles in the polymer matrix modifies the physical and chemical properties. This approach alleviates the development of proton conducting composite membrane that works at temperatures above 100°C. Nanomaterials are known to play several role as polymer fillers, which enhances the thermal and mechanical properties. NBO has bounty applications in modern technology. It also acts as good corrosion resistance and an oxygen sensor material. It exhibits good water tolerance and provide thermal stability to the base polymer. Among the variety of transition metal oxides, oxides of tungsten, oxides of vanadium, niobium oxide (NBO) films exhibit interesting electrochromic behavior similar to metal oxides. 18 -20
Concisely, the modern developments of SPEEK/NBO nanocomposite membranes have been designed and synthesized. The effects of physicochemical properties upon introduction of the inorganic matrix into the pristine polymers were studied in this article. The water uptake capacity and proton conductivity of the membranes were optimized to achieve superior electrochemical performance.
Experimental methods
Materials
PEEK was gifted by Gharda Chemicals Limited (Mumbai, Maharashtra, India) under the trade name of GATONE 1100. NBO was received from Department of Physics, Anna University, Chennai, Tamil Nadu, India. Concentrated sulfuric acid was purchased from Sigma-Aldrich Co. (St Louis, Missouri, USA). N, N-dimethyl formamide (DMF) was purchased from Merck (Mumbai, India).
Synthesis of SPEEK
Li et al. 21 proposed that SPEEK with degree of sulfonation (DS) greater than 60% is not appropriate for FC applications. Therefore, the sulfonation of PEEK was performed by maintaining proper temperature and time by the reported procedure. 22 The dried crystalline PEEK material was sulfonated using concentrated sulfuric acid under nitrogen atmosphere. A volume of 250 mL of sulfuric acid was taken in a round-bottomed flask, slightly warmed, and magnetically stirred at 40°C. An amount of 5 g of dried PEEK was slowly added over a period of 60 min under constant stirring. The temperature was slowly raised up to 45°C and maintained for 6 h. The sulfonated polymer was obtained by precipitating the viscous polymer in to large excess of ice-cold deionized water. The resultant sulfonated polymer was washed repeatedly with plenty of deionized water until the pH reached 6–7. The produced pink colored SPEEK materials were dried at room temperature for nearly 16 h and retained in a hot air oven at 105°C for 12 h.
Preparation of SPEEK/NBO nanocomposite membranes
The dried SPEEK was dissolved in DMF at room temperature to form a 15-wt% viscous solution. After the ample dissolution of SPEEK, desired quantity of NBO (2.5–10 wt%) were added to the viscous solution under vigorous stirring. The temperature of the medium was raised up to 60°C and maintained for nearly 1 h. The resultant solution was sonicated for 15 min, followed by casting onto the glass plate and dried at room temperature for 24 h in which the excess solvent was gradually removed. The membrane was further dried in hot air oven at 60°C for 8 h and 80°C for 24 h. The subsequent membrane was soaked in deionized water to peel the membranes from the glass plate. The prepared SPEEK/NBO nanocomposite membranes were thoroughly washed with deionized water and dried at 100°C under vacuum for 12 h. The schematic representation of the polymer nanocomposite membrane is illustrated in Figure 1.
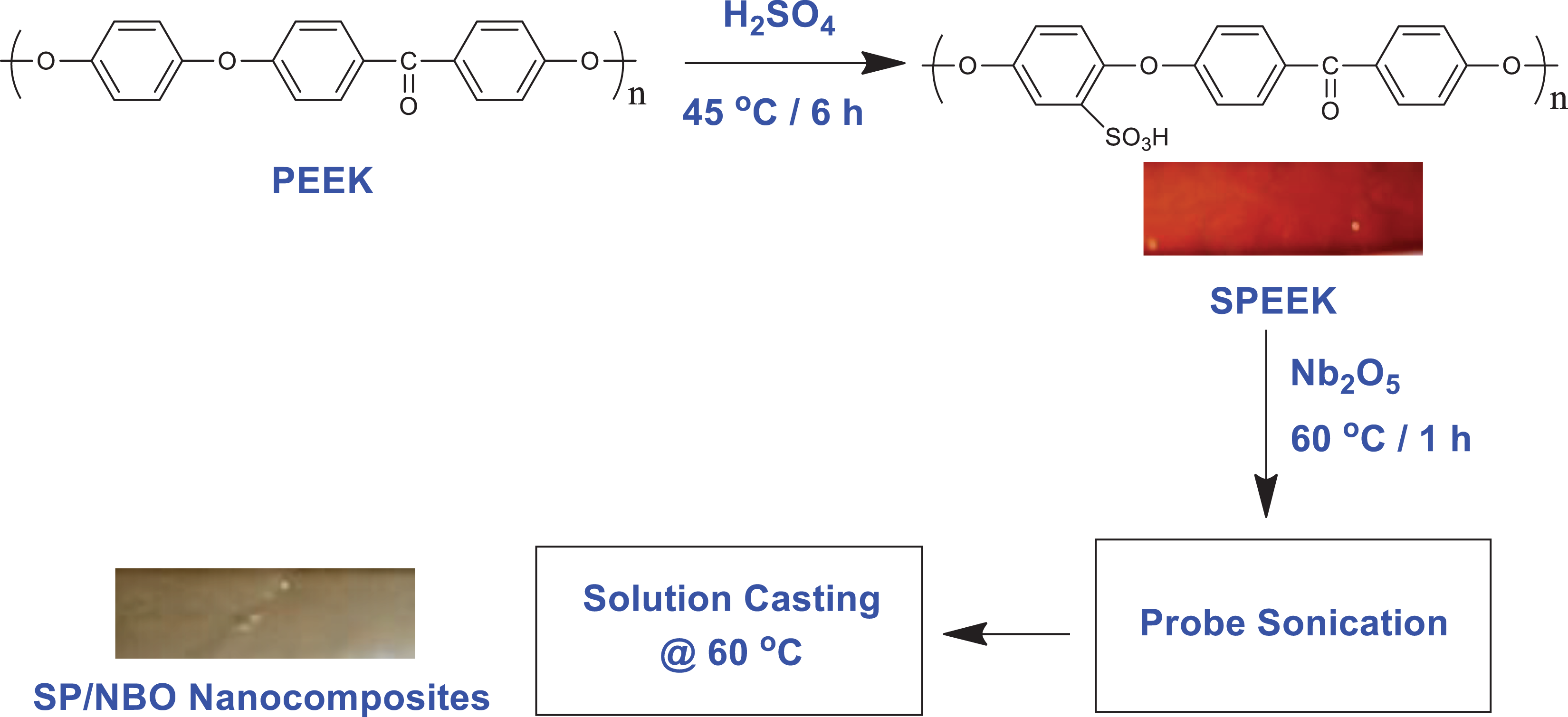
Pictorial representation of the preparation of SP-NBO polymer nanocomposites.
Measurements
FTIR analysis
Fourier transform infrared (FTIR)–attenuated total reflection spectra were recorded with a Shimadzu IRTracer-100 (Japan) in the range of 4000–400 cm−1 at a resolution of 4 cm−1.
XRD analysis
The phase separation measurement was carried out by XRD X’Pert HighScore Plus instrument (PANalytical, the Netherlands).
Morphology
The surface morphology of the nanocomposite membrane was explored using a scanning electron microscopy (FEI, Quanta FEG200, Hillsboro, Oregon, USA). The membrane samples were freeze-fractured under liquid nitrogen and vacuum sputtered with a thin layer of gold.
Proton conductivity
The proton conductivity measurements of the samples were carried out by our earlier reported article. 23
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was analyzed using a thermogravimetric analyzer (NETSZCH, Selb, Germany) at a heating rate of 15°C min−1 under nitrogen atmosphere.
Electrochemical performance
The electrochemical performance of the pure and composite membranes was studied using a FC test station (model-LCN4-25-24/LCN 50-24; Bitrode Instruments, St Louis, Missouri, USA). The complete technique for the measurements was described in our earlier paper. 23
Water uptake and swelling ratio
The water uptake of virgin polymer and polymer composite were determined by the difference in weight between the dry membrane and the swollen membrane. The acidified membranes were dried at 100°C under vacuum atmosphere for 24 h. Let W dry and L dry be the measured weight and length of the dried membranes. After this process, the dried membranes were soaked in deionized water. The liquid water on the surface of the wetted membrane was removed using a tissue paper before weighing, and it is recorded as W wet and L wet. The water uptake percentage and swelling ratio of the membrane were calculated using equations (1) and (2), respectively.
DS and IEC
DS is defined as the % of PEEK repeating units that has been sulfonated. Ion-exchange capacity (IEC) is the ability of a material to undergo displacement of ions previously attached and loosely incorporated into its assembly by oppositely charged ions present in the surrounding medium. The DS and IEC of the membrane can be evaluated with the classical titration method. 24 The pre-weighted sample is first immersed in to 0.10 M sodium chloride solution for 24 h to exchange all the protons with sodium ions. The H+ ions in the solution were back titrated with 0.01 N sodium hydroxide using phenolphthalein as an indicator. The IEC and DS were determined using equations (3) and (4), respectively.
where NSPEEK is the molar number of sulfonated PEEK units, NPEEK is the molar number of unsulfonated PEEK units,
According to the expressions of DS and IEC, the molar number NSPEEK unit in 1 g is given by
The molar number NPEEK unit in 1 g is given by
where MPEEK is the molecular weight of PEEK repeat unit (288 Da) and MSPEEK is the molecular weight of SPEEK repeat unit (368 Da).
Results and discussion
Spectral studies
The FTIR spectra of NBO, SPEEK, and nanocomposite membranes are shown in Figure 2. The typical symmetric stretching vibration of sulfone (O=S=O) group was noticed at 1021 cm−1. The stretching of carbonyl group in SPEEK was confirmed by a peak observed at around 1651 cm−1. The peaks at 1595 and 1465 cm−1 represent the vibrations of aromatic ring skeleton. The intense band observed at 1219 cm−1 indicates the presence of aromatic ether linkage. The characteristic peak viewed at 768 cm−1 confirms the incorporation of NBO into the polymer matrix.
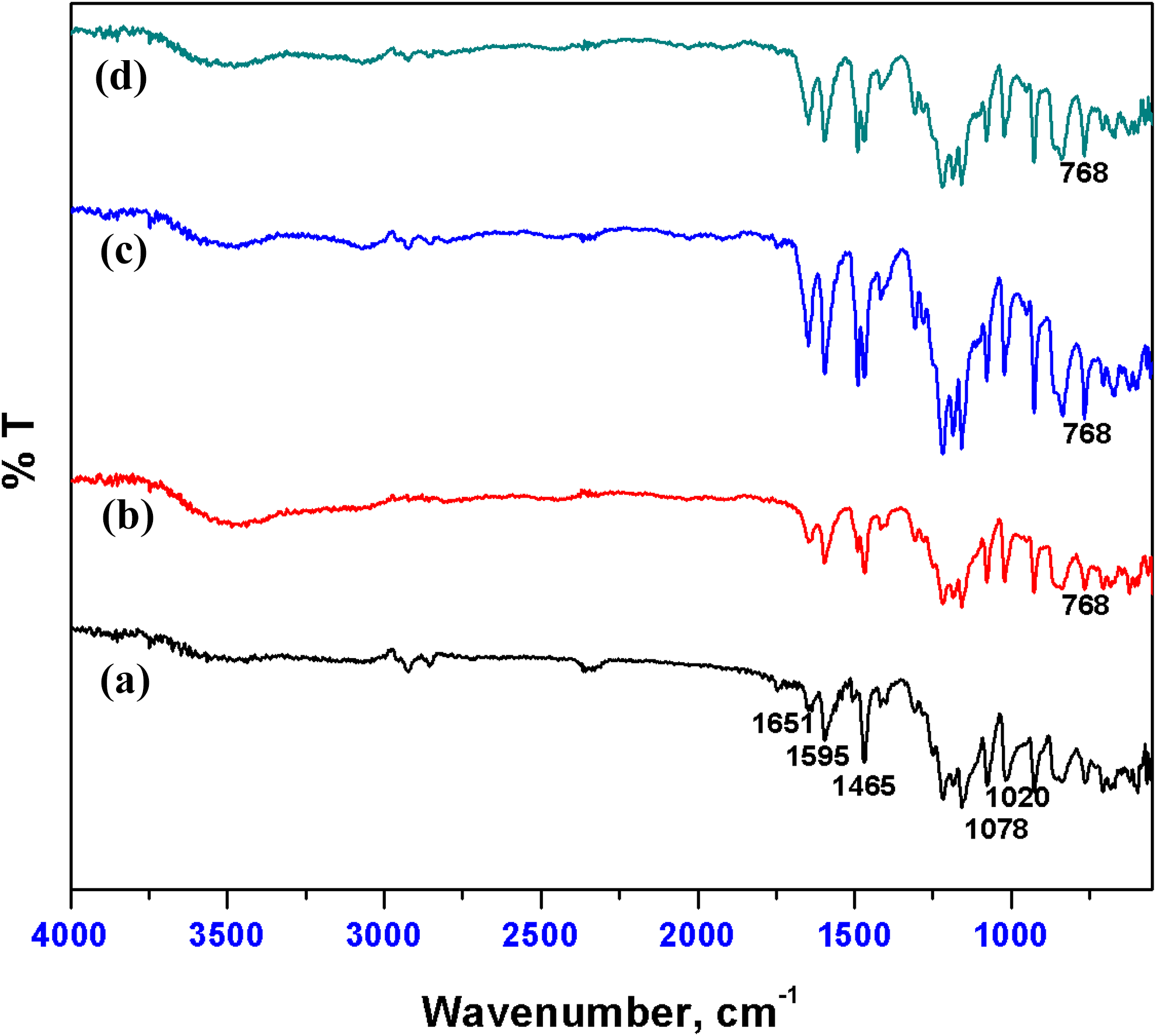
FTIR spectra of (a) SPEEK, (b) SP-NBO-5, (c) SP-NBO-7.5, and (d) SP-NBO-10.
3.2 Morphology
Figure 3 depicts the morphologies and structural properties of NBO and polymer nanocomposites. The SEM images of NBO show the structure of the particles consisting of regular cubic clusters of well-defined shapes and sizes of approximately 500 nm. The morphology studies evidently reveal that all the nanocubes retain their intact structure together during membrane preparation endowing the probability to create continuous pathway for proton transfer by embedding these nanocubes in the polymer matrix. The polymer nanocomposite generates void formation due to adhesion between the organic and the inorganic interface which attributes to good proton conduction.

SEM images of (a) SPEEK, (b and c) NBO, (d and e) SP-NBO-5, and (f) SP-NBO-10.
XRD analysis
The XRD pattern of metal oxide and polymer nanocomposite is depicted in Figure 4. In general, sulfonation of pristine polymer strongly reduces its crystallinity and increases the amorphous nature. 25,26 The incorporation of crystalline metal oxide into the polymer via blending, composite, interpenetrating polymer networks (IPNs), leads to enhancement of the amorphous behavior, thereby improving the proton conductivity. The XRD spectrum of NBO displays the polycrystalline nature and SP-NBO-10 polymer nanocomposite exhibits amorphous phase, confirming the fine dispersion of NBO and thereby enhancing the water uptake ability and proton conductivity.

XRD pattern of (a) NBO and (b) SP-NBO-10.
Thermogravimetric analysis
The thermal stability of the electrolyte membrane is an important parameter of its operation in FC assembly. The thermograms of NBO, SPEEK, and nanocomposite membranes recorded over 600°C are depicted in Figure 5. The pure and composite membrane exhibits three-step degradation pattern, whereas the crystalline NBO holds good thermal stability. The initial mass loss in SPEEK and composite membranes is due to physically and chemically bound water observed at nearly 100°C. The second stage of the membranes that was found between 350°C and 450°C can be attributed to the degradation of sulfonic acid moiety. Finally, the membranes were decomposed due to the breakdown of polymer scission entities. The thermal stability of the composite membrane was enhanced due to the annexation of NBO as inorganic filler.

TGA plot of (a) NBO, (b) SP-NBO-10, and (c) SPEEK.
Water uptake and swelling ratio
In general, the water absorption characteristics of PEM can be increased by the incorporation of inorganic oxides. 27 Table 1 exhibits the physicochemical properties of the prepared pure SPEEK and nanocomposite membranes. The major part of water molecules present in ionomer membrane is responsible for the dissociation of protons from sulfonic acid group and also serves as a better platform for vehicular transport. The higher concentration of acid content leads to excess swelling which affects the mechanical durability of the membrane. Since the membrane morphology plays a vital role in enhancing the performance of the FC, moderate increase in water uptake is preferable as it has a direct proportion with swelling ratio. Since the hygroscopic affinity increases with increasing concentration of inorganic fillers, the water uptake behavior of SP-NBO-10 shows the highest value of 38.4%.
Physicochemical properties of SP-NBO nanocomposite membranes.

SPEEK: sulfonated poly (ether ether ketone); IEC: ion-exchange capacity; NBO: niobium oxide.
3.6 Ion-exchange capacity
IEC of the polymeric membrane usually represents the amount of exchangeable groups in its structure. In general, the water adsorption ability of the electrolyte membrane facilitates the transportation of ions, thereby enhancing the proton conductivity. The IEC of SPEEK is found to be increased with respect to DS because of the existence of sulfonic acid group in the polymer chain. 28,29 In addition, further impregnation of the inorganic metal oxide into SPEEK alleviates the IEC of the nanocomposites. 30 The introduction of 2.5–10.0 wt% of NBO gradually increases the water uptake capability of the composite membranes. The results clearly indicate that the IEC value follows the same trend as water uptake capacity. The reason for the enhancement of IEC is owing to the hydration regions near the ions overlapping and the reduced fraction of the unfunctionalized region. The maximum IEC value was found as 1.80 meq. g−1 for SP-NBO-10, whereas the pure polymer has 1.62 meq. g−1. Thus developing a successful PEM with an increase in IEC value up to a certain level shows appreciable performance. Figure 6 illustrates the variation of IEC and water uptake of the pristine and composite membranes. This result indicates that the modest change in IEC of the polymeric membrane leads to the production of number of ionic sites.
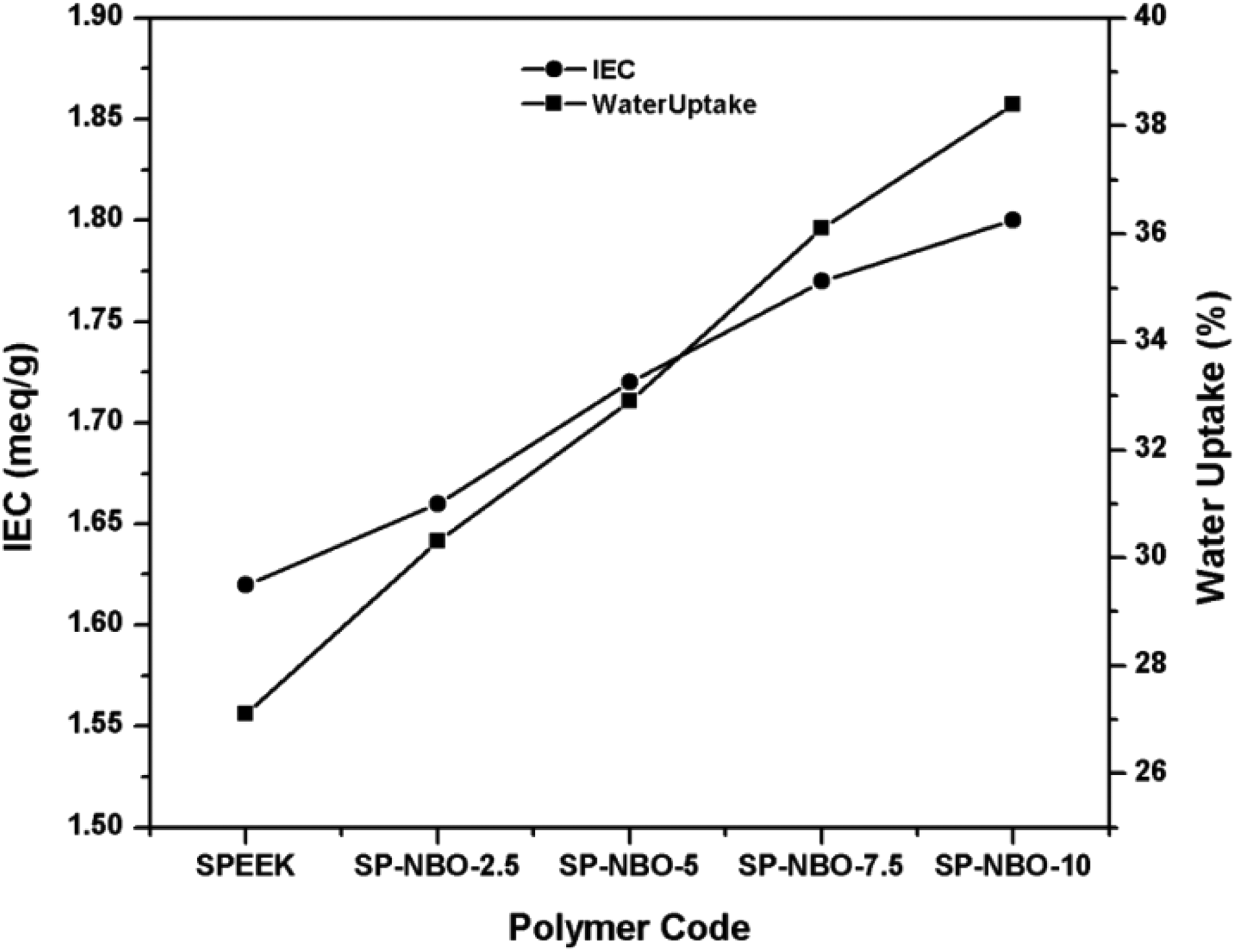
Comparison between IEC and % water uptake capacity of the electrolyte membranes.
Proton conductivity
The ionic cluster domains in the polymer matrix always have a strong interconnection with acidic functional groups. 31 -34 The proton transfer through the membrane can be determined by channel continuity, channel size, and IEC. In general, the proton conductivity of the electrolyte membranes enhances with increasing concentration of inorganic additives. 27 The proton conductivity results of pure and nanocomposite membranes with increasing temperature are plotted in Figure 7. The proton conductivity of SPEEK was found to be 0.016 S cm−1, whereas the conductivity values of membranes having NBO content from 2.5 wt% to 10wt% were found to be in the range between 0.019 S cm−1 and 0.028 S cm−1. These results designate that the addition of NBO improves the trapping ability of inorganic particles from the membranes which in turn improves the ionic conductivity. The vehicular proton transport mechanism involved in sulfonic acid-based ionomeric membrane leads to adsorbion and retention of water molecules effectively.

Proton conductivity of pure SPEEK and polymer nanocomposite membranes.
Electrochemical performance
The single-cell performance of SPEEK and nanocomposite membranes with 10% NBO content for PEM FC using platinum as both the cathode and the anode catalysts are displayed in Figure 8. The measurements were made using H2 and O2 as fuel at 60°C. The maximum power density of 601 mW cm−2 was produced by the nanocomposite membrane, whereas the pristine membrane limits to 497 mW cm−2. The reason for increasing the trend of current density and power density of the composite membrane may be explained by few points: (i) The vehicular proton transport mechanism involved in sulfonic acid-based ionomeric membrane leads to adsorption and retention of more water molecules; (ii) the nanosized inorganic fillers associated with hydrophilic segments through self-assembly influences the proton transport pathway, thereby enhancing the power density; and (iii) the proton conductivity of the composite membrane of thickness 33 µm is relatively high (0.029 S cm−1) when compared to pristine SPEEK (0.018 S cm−1), and the power density of the membrane is increased for the composite membrane. The electrochemical studies demonstrated that the polymer electrolyte membrane derived from SPEEK and NBO proved to be a feasible material for PEM FC applications.

Polarization and power density curves of SPEEK and SP-NBO-10 at 60°C.
Conclusions
In this article, the preparation, physicochemical properties, and applications of polymer nanocomposite membranes based on SPEEK and NBO have been reported. The high engineered polymer GATONE™ 1100 PEEK was sulfonated by post-sulfonation process. The structural and morphological studies of the membranes were performed by FTIR, XRD, and SEM analyses. The IEC of the prepared membranes was found in the range between 1.62 meq g−1 and 1.80 meq g−1. The amorphous nature of the polymer nanocomposite which facilitates the ionic conductivity was examined by XRD. It was found that the ordered acid–base pairs formed along the SPEEK/NBO interface serves as a facile proton transfer pathway and affords the hybrid membrane-elevated proton conductivity and electrochemical performances. The TGA studies reveal that the nanocomposite have superior thermal stability to pure SPEEK membrane. The electrochemical performance of the SP-NBO-10 membrane has been explored and renowned the potentiality of the prepared nanocomposite membranes as a promising candidate for FC applications.
Footnotes
Acknowledgements
The authors wish to gratitude Nanotechnology Research Centre (NRC), SRM Institute of Science and Technology (IST) for providing SEM and XRD analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: One of the authors (DP) wishes to acknowledge SRM-IST for providing financial assistance under SRM Selective Excellence Scheme and also wishes to thank DST-FIST (fund for improvement of S&T infrastructure) for the financial assistance at Department of Chemistry, SRM IST, No.SR/FST/CST-266/2015(c).






