Abstract
Self-healing polyurethane (SHPU) containing disulfide was synthesized and used as a binder to investigate its effect on the performance of reduced graphene oxide–tin oxide electrodes compared to those of polyurethane (PU) and poly(vinylidene difluoride) (PVDF) binders in Li-ion battery (LIB). Structural and morphological characterization of the SHPU and electrode was performed using a tensile tester, Fourier transform infrared spectroscopy, X-ray diffractometer, and scanning electron microscopy. Electrochemical performance was investigated using Galvanostatic charge–discharge and electrochemical impedance measurements. The tensile properties and scanning electron microscopy photographs confirmed the self-healing characteristics of the synthesized SHPU. Electrochemical studies were conducted using an RGO-SnO2 electrode. The electrochemical measurements revealed that the SnO2-pillared carbon-based anode materials with SHPU binder showed improved cycling performances with an excellent reversible capacity retention compared to PU or PVDF. After 1000 cycles at 1C, the surface morphology of the electrode with SHPU showed no cracks or dendrites, while the PVDF-based electrode possessed some cracks and dendrites on its surface. The electrochemical results confirmed that SHPU binder improves the electrochemical performance of LIBs.
Introduction
Electrodes are crucial components for the overall performance of lithium-ion batteries (LIBs), where graphite is used as a standard anode material. 1 -3 The demand for higher energy density LIBs is increasing in many applications, such as automobiles, and group IV elements have received much interest for this purpose. However, the use of these elements is limited due to the large volumetric change (ca. 300%) that occurs during long-term charge–discharge cycling and results in undesirable rapid capacity fading, low initial coulombic efficiency, and poor rate performance. 4,5 A continuous large volume change in the lithiation–delithiation process results in disintegration and pulverization of the electrode. 6 -8
Among the various approaches employed to avoid this limitation, chemically modified graphene and SnO2 have been hybridized to accommodate the volume change and improve the capacity and cycling stability of the electrode material. 9 -11 A pomegranate structure, where single nanoscale silicones are encapsulated by a carbon layer, has been proposed and resulted in excellent cyclability and electrode–electrolyte contact area. 12 -16 SnO2 is believed to be a promising material due to its high theoretical lithium storage capacity, low cost, and safe working potential. However, large volume changes during charge–discharge cycling hinder the practical application of SnO2. Hybridization of SnO2 with carbonaceous materials has been used to circumvent this limitation. 17 -19 In particular, graphene has been used in hybridization because of its excellent electronic conductivity and high surface area. In situ synthesis approaches have been applied to obtain a homogeneous distribution of Sn nanoparticles in graphene sheets. 20 Jeevan Kumar Reddy et al. studied hybridization of SnO2 with long-chain alkylamine-grafted graphene oxide, and the specific capacities decreased with increasing alkylamine chain length. 21
Self-healing polymers can spontaneously repair mechanical damage, and this characteristic enhances the lifetime of energy storage materials. 22 -26 Wang et al. used a self-healing polymer as a binder and attained a 10 times longer life cycle. 3 Multiple network binder, poly(acrylic acid)-poly(2-hydroxyethyl acrylate-co-dopamine methacrylate), with rigid-soft bonds showed self-healing behavior and improved the rate of performance of LIB. 24 Feng et al. reported the self-healing behavior of polyurethane (PU)-based on the Diels–Alder reaction and thermal movement of molecular chains. 25
In the present study, we attempted to synthesize SnO2-nanopillared carbon structures using dodecylamine-grafted graphene oxide as a template. A PU-based self-healing polymer was synthesized and applied to LIBs as a binder for fabrication of electrodes. The effect of the self-healing characteristic on the performance of LIBs was evaluated and compared to that of PU or commercial poly(vinylidene difluoride) (PVDF) binders.
Experiment
SnO2-pillared dodecylamine-grafted graphene oxide (SnO2-GN) was prepared, as previously reported. 21,27,28
Self-healing polyurethane (SHPU) was synthesized using polycaprolactone diol (Capa™ Thermoplastics) with a molecular weight of ca. 900, 4,4′-methylenebis(phenyl isocyanate) (Sigma-Aldrich, St. Louis, MO, USA), and
To investigate the self-healing characteristics of SHPU, tensile stress tests (ASTM D638) were performed using a universal tensile tester (LF Plus, Lloyd Instruments) at room temperature with a crosshead speed of 5 mm min−1. The tensile specimen with a thickness of 2 mm was created from the sheet prepared at 150°C using a hot press. The center of the tensile specimens was cut to a depth of 1 mm, as shown in Figure 1, and the specimens were placed in an oven at 70°C for a predetermined period of time (1, 3, or 7 days). Field Emission (FE)-scanning electron microscopy (SEM) was used to observe the change of the cut area over time.

Schematic diagram of a tensile specimen.
The electrochemical performances of SnO2-GN were measured by fabricating 2032 type coin cells using Li metal as a reference electrode. Electrodes were prepared by casting a slurry with a composition of 80 wt% active material, 10 wt% conductive agent (super P, TIMCAL, Switzerland), and 10 wt% SHPU or PVDF (Kureha KF100) (for the reference sample) onto copper foil. The slurry was prepared by grinding the mixture in the presence of
Results and discussion
The Fourier transform infrared spectroscopy analysis results of the synthesized SHPU are shown in Figure 2. The peaks at 2865 and 2943 cm−1 represent –CH asymmetric stretching, and the peak at 1722 cm−1 belongs to C=O of the carboxylic group. The C=O in amide is overlapped with the carboxylic peak at ca. 1630–1680 cm−1. The peak at 511 cm−1 is attributed to the S–S bond, which induces the self-healing behavior of SHPU.
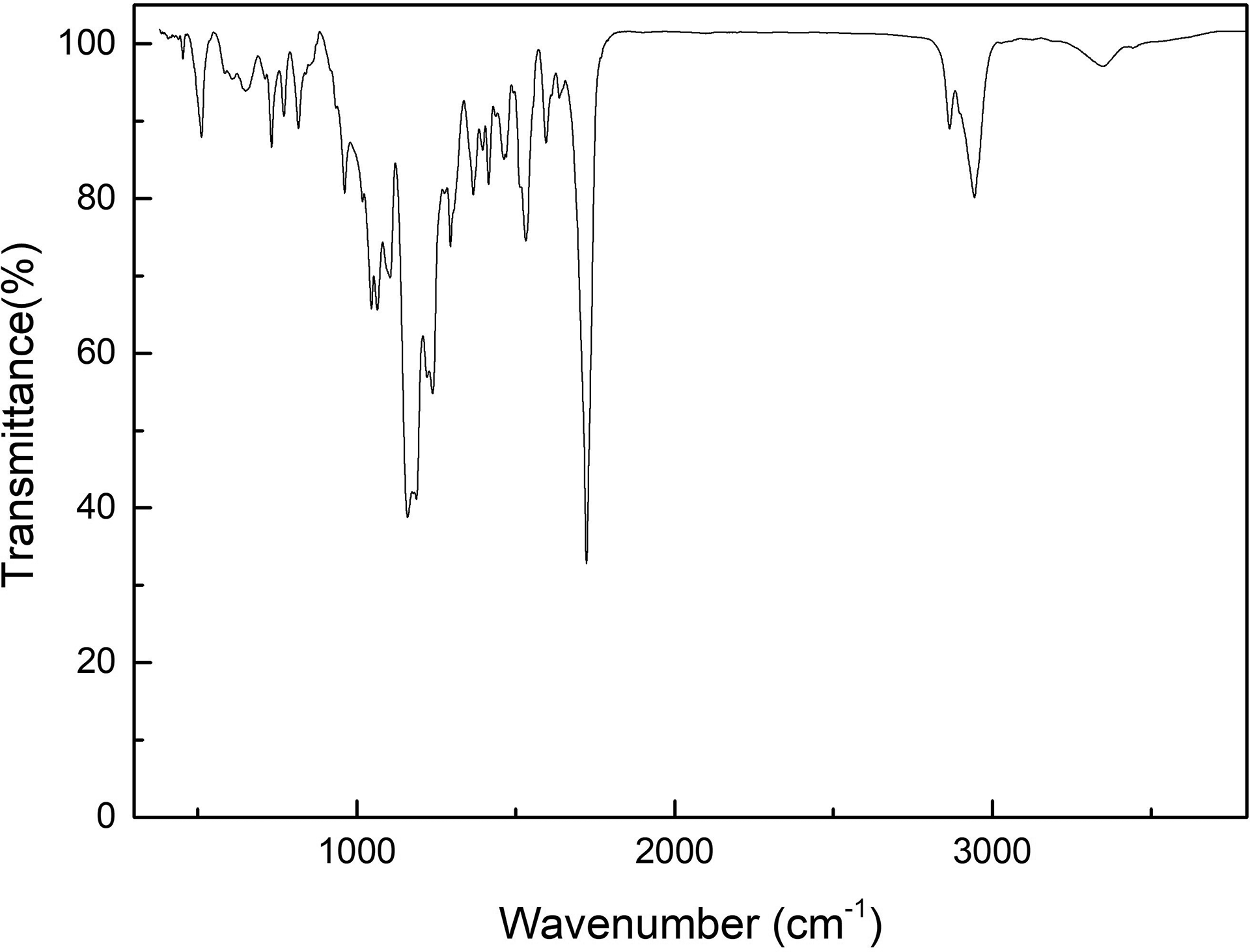
FTIR spectrum of the synthesized self-healing polyurethane.
The tensile properties of the half-cut SHPU specimens are shown in Figure 3. The tensile strength and elongation of the half-cut specimen were less than 30% of that of the original uncut specimen. The tensile properties of PU and SHPU without half-cut are similar. The tensile strength and elongation of half-cut SHPU held at 70°C for 1 day increase by 68% and 150%, respectively, and they further increase by 84% and 190% after treatment for 7 days compared to the half-cut specimen. These significant increases of tensile strength and elongation are attributed to the self-healing behavior of SHPU. It has been reported that the disulfide group is responsible for the self-healing property. 29,30 SEM photographs of specimens are shown in Figure 4 and clearly show that the cut area becomes narrower with increasing time and eventually it is fully healed. These photographs support the notion that the improvement of tensile properties shown in Figure 3 is dependent on the self-healing capability of SHPU.

Tensile stress–strain data of SHPU under various treatment periods at 70°C.

FE-SEM photographs of half-cut specimens of SHPU after various treatment periods.
The electrochemical properties of the SnO2-GN-PU, SnO2-GN-SHPU, and SnO2-GN-PVDF anodes in lithium-ion half-cells are evaluated with respect to constant current charge–discharge cycling in the potential range of 0.1–2.0 V. The charge–discharge profiles of SnO2-GN-SHPU and SnO2-GN-PVDF at the 1st, 100th, and 1000th cycles measured at a 2C rate are shown in Figure 5. The first cycle discharge and charge capacities of SnO2-GN-PU and SnO2-GN-SHPU are observed to be 835/530 and 836/537 mA hg−1 with first columbic efficiencies of 63.5% and 64%, respectively. Similarly, the first discharge and charge capacities of SnO2-GN-PVDF are observed to be 843 and 539 mA hg−1, respectively, with a columbic efficiency of 64%. However, as cycling proceeded to the 100th cycle, the discharge and charge capacities became 346/342 and 417/416 mA hg−1, respectively, with columbic efficiencies of ca. 100% for SnO2-GN-PU and SnO2-GN-SHPU. For SnO2-GN-PVDF, the discharge and charge capacities are 419 and 417 mA hg−1, respectively, with a columbic efficiency of 100%. At the end of the 1000th cycle, the discharge and charge capacities are 233/232 and 389/387 mA hg−1 with columbic efficiencies of ca. 100% for SnO2-GN-PU and SnO2-GN-SHPU, respectively, while SnO2-GN-PVDF shows discharge and charge capacities of 339 and 337 mA hg−1. The capacity retention of SnO2-GN-SHPU (72%) is much higher than that of SnO2-GN-PU (44%) from the 1st to 1000th cycle and even higher than that of SnO2-GN-PVDF (62%), as shown in Figure 6. It has been reported that avoiding cracking and pulverization of the electrode can result in enhanced cycling stability. 23 -25 Thus, the constant cycling performance with excellent capacity retention of SnO2-GN-SHPU can be attributed to the self-healing nature of the SHPU binder.

Charge–discharge profiles of SnO2-GN-PU, SnO2-GN-SHPU, and SnO2-GN-PVDF at (a) 1st cycle, (b) 100th cycle, and (c) 1000th cycle.

Comparison of the capacity retention of SnO2-GN-PU, SnO2-GN-SHPU, and SnO2-GN-PVDF.
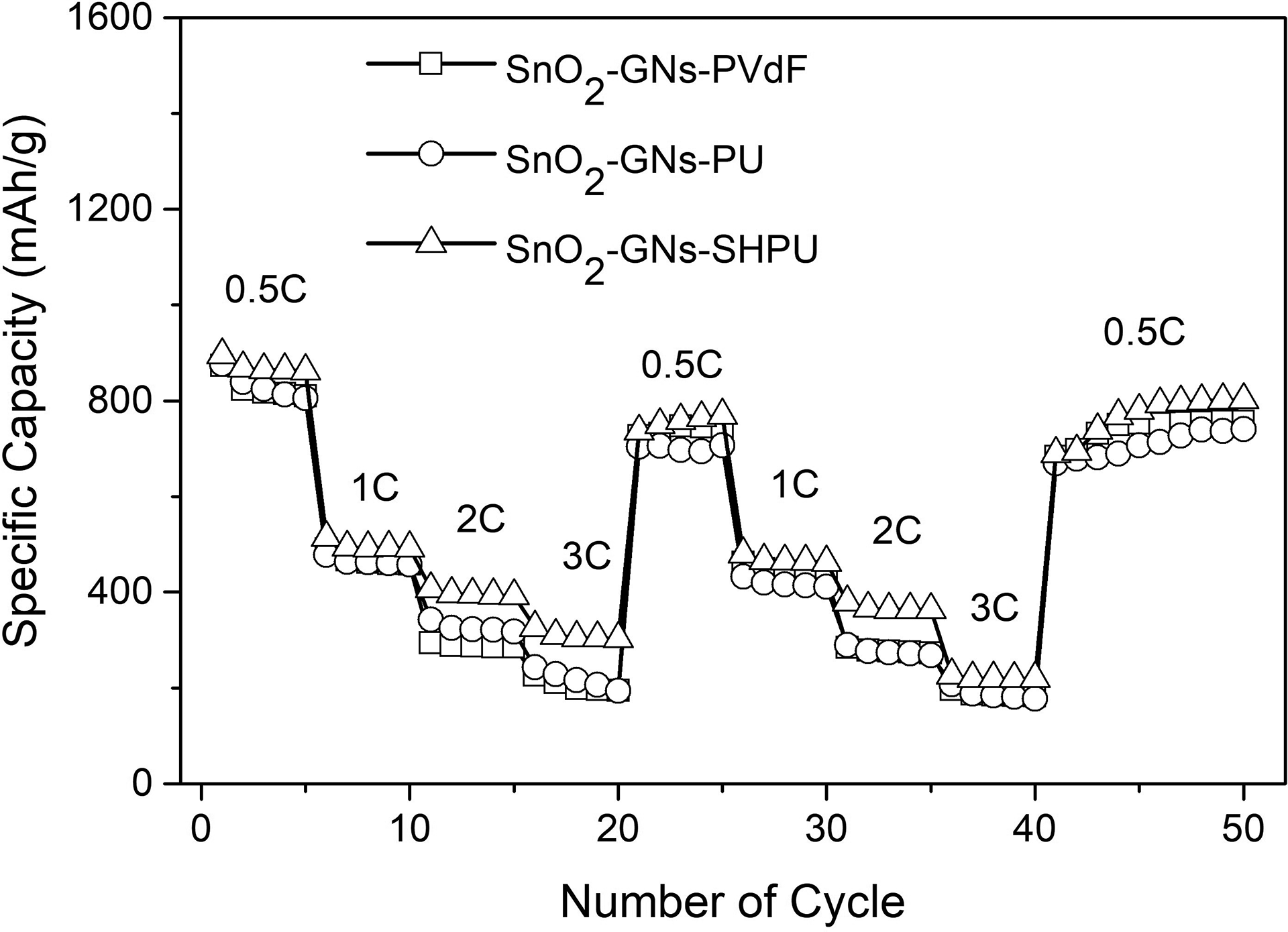
The specific capacities of SnO2-GN-SHPU and SnO2-GN-PVDF are evaluated at various current rates, and the results are shown in Figure 7. The specific capacity decreases with increasing current rate for both SHPU and PVDF. However, the relative decay of specific capacity for SHPU is relatively less than that of PVDF. When the C-rate returns to the initial 0.5C rate at the 50th cycle, SnO2-GN-SHPU and SnO2-GN-PVDF recover up to 90% and 86% of their initial specific capacity values, respectively. This supports better reversibility and cyclability of SnO2-GN-SHPU compared to SnO2-GN-PVDF.
Electrochemical impedance spectroscopy (EIS) results of SnO2-GN-PU, SnO2-GN-SHPU, and SnO2-GN-PVDF.
Electrochemical impedance spectroscopy (EIS) of SnO2-GN-SHPU and SnO2-GN-PVDF is performed to understand the interfacial electrochemistry and reaction mechanism in the frequency range of 100 kHz–0.01 Hz. Typical Nyquist plots of the AC impedance before and after cycling are shown in Figure 8(a) and (b). In general, the impedance spectrum consists of a depressed arc followed by a straight line inclined at 45° angle. The equivalent circuit adopted for calculations is shown in Figure 8(c).

Specific capacity of SnO2-GN-SHPU and SnO2-GN-PVDF at various current rates.
Values of

SnO2-GN: SnO2-pillared dodecylamine-grafted graphene oxide; PVDF: poly(vinylidene difluoride).
To better understand the performance of SHPU as an efficient binder at a higher current rate of 1C, the surface morphologies of the electrodes after 1000 cycles are examined, and the FE-SEM photographs are shown in Figure 9. Figure 9(a) and (b) corresponds to electrodes prepared using PVDF. These photographs show cracks on the surface and dendrites due to the side reaction on the electrode materials during the charge–discharge process. Dendrites are formed due to the surface and interfacial reaction between the electrode surface and components in the electrolyte. 31 However, in the case of Figure 9(c) and (d), which are prepared using SHPU as a binder, the photographs show a normal surface without any cracks or dendrites. This indicates that most of the surface cracks formed by the charge–discharge process were healed, which can be attributed to the self-healing characteristic of the SHPU binder. Although the exact self-healing mechanism during charge–discharge process is under investigation, this surface morphology study also supports the improved electrochemical performances of SnO2-GNs-SHPU compared to SnO2-GNs-PVdF.

Surface morphologies of (a, b) SnO2-GN-PVDF and (c, d) SnO2-GN-SHPU electrodes after 1000 cycles.
Conclusion
Polyurethane-based self-healing polymer (SHPU) was prepared successfully using polycaprolactone diol and 4,4′-methylenebis(phenyl isocyanate) as starting materials followed by the self-healing additive,
Footnotes
Authors contribution
The authors YJK and JKRM have equally contributed to this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
