Abstract
In this article, the improvement in electrical performance of poly(3,4-ethylenedioxythiophene)–poly(styrenesulfonate) (PEDOT:PSS) as the transparent electrode doped with different additives (ethylene glycol (EG), isopropyl alcohol) or treatment of sulfuric acid was enhanced that organic solar cells (OSCs) were produced by using poly(3-hexylthiophene-2,5-diyl):[6,6]-phenyl C61 butyric acid methyl ester. OSCs were fabricated by the doped or treated PEDOT:PSS films as transparent electrodes. The photoelectrical measurements were carried out and the effects of doping or treatment were compared. As a result, EG-added PEDOT:PSS electrode showed the best power conversion efficiency value of 1.87% among the PEDOT:PSS anodes.
Keywords
Introduction
The production of new materials, the developments in material engineering, and the developed device structures have resulted in an increase in the research and development efforts of organic solar cells (OSCs) in the last 40 years. 1 Despite the fact that mostly inorganic materials are used for photovoltaics (PVs), 2 many researchers have also spent tremendous efforts to improve OSCs within the last four decades. 3 -6
Despite their low power conversion efficiencies (PCEs), OSCs have many advantages, such as the processability with solution-based production techniques, low temperature processing resulting in low-cost producibility, and flexibility 7 that enables the OSCs to be applicable on flexible surfaces, roll-to-roll production, with the use of abundant materials. 8 Structure of OSC with the PV mechanism is shown in Figure 1.
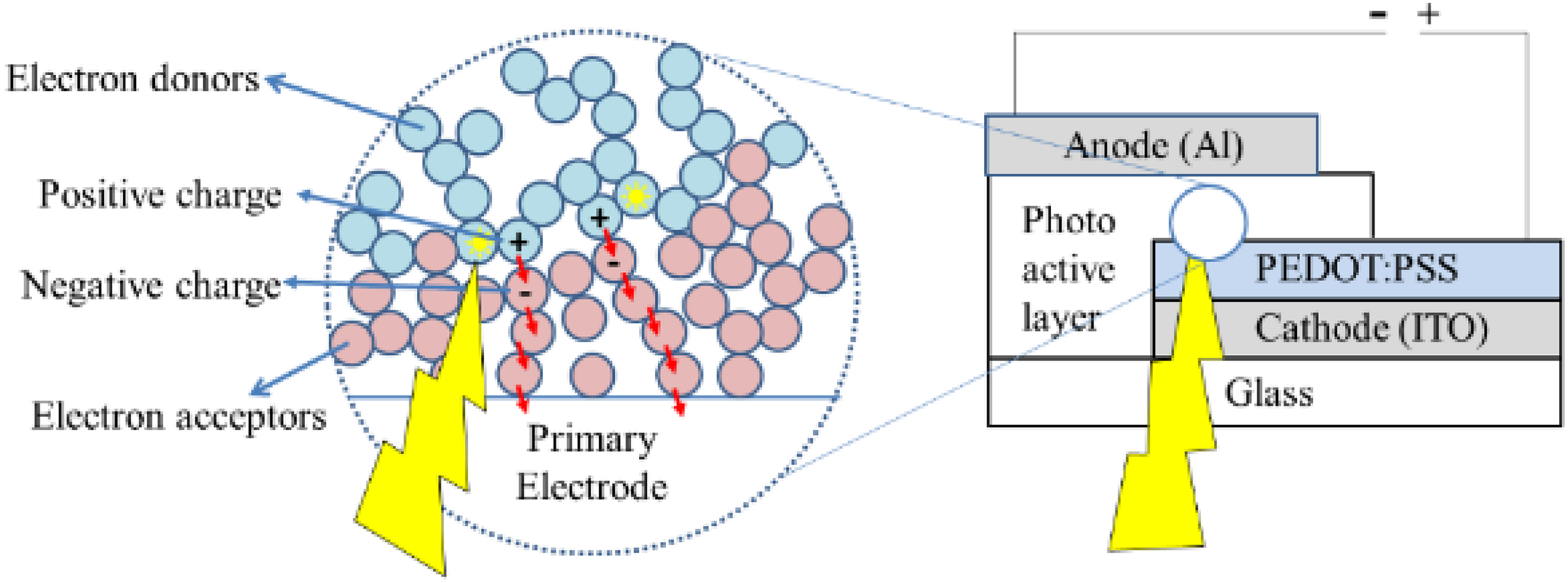
Depiction of bulk heterojunction solar cell structure and photovoltaic conversion.
Indium tin oxide (ITO) is commonly used in optoelectronic devices as hole transport layer with its prevailing properties of high conductivity (10–30 S/m), high optical transparency (over 90% transparency at 550 nm), and suitable work function (<4.8 eV). 9 However, such disadvantages as high-temperature processes, vacuum conditions, decreasing of indium resources, the requirement of acid processes, surface roughness, and brittleness when it is bent 10 -12 have forced the researchers to find alternatives to ITO. Carbon nanotubes, 8,13,14 carbon-based materials like graphenes, 15 -17 conductive polymers, 18,19,3 -6,20 -38 metal grids, 39 -41 nanowires, 42 -44 semitransparent metal layers, 45,46 metal oxides, 47,48 and combination of these materials can be given as examples for alternatives to ITO. These materials are indispensable materials for optoelectronic devices as transparent electrodes.
Poly(3,4-ethylenedioxythiophene)–poly(styrenesulfonate) (PEDOT:PSS) has been mostly used as hole transparent layer in OSCs. 49 The molecular structure of PEDOT:PSS is shown in Figure 2. By the advantages of the ability of usage on flexible surfaces, deposition with solution-based techniques, researchers attempted to use PEDOT:PSS as a transparent conductive layer substitute to ITO. 50 High conductivity is required to be used as an electrode for OSCs. 49 There are numbers of reports in the literature to enhance the electrical conductivity of PEDOT:PSS by treating the PEDOT:PSS in the solution form or after the film production. 2 -6,20 -38,51,52 PEDOT:PSS polymer consists of PSS as a counter ion, which is nonconductive that enables to be easily dispersed the conductive and cationic PEDOT.
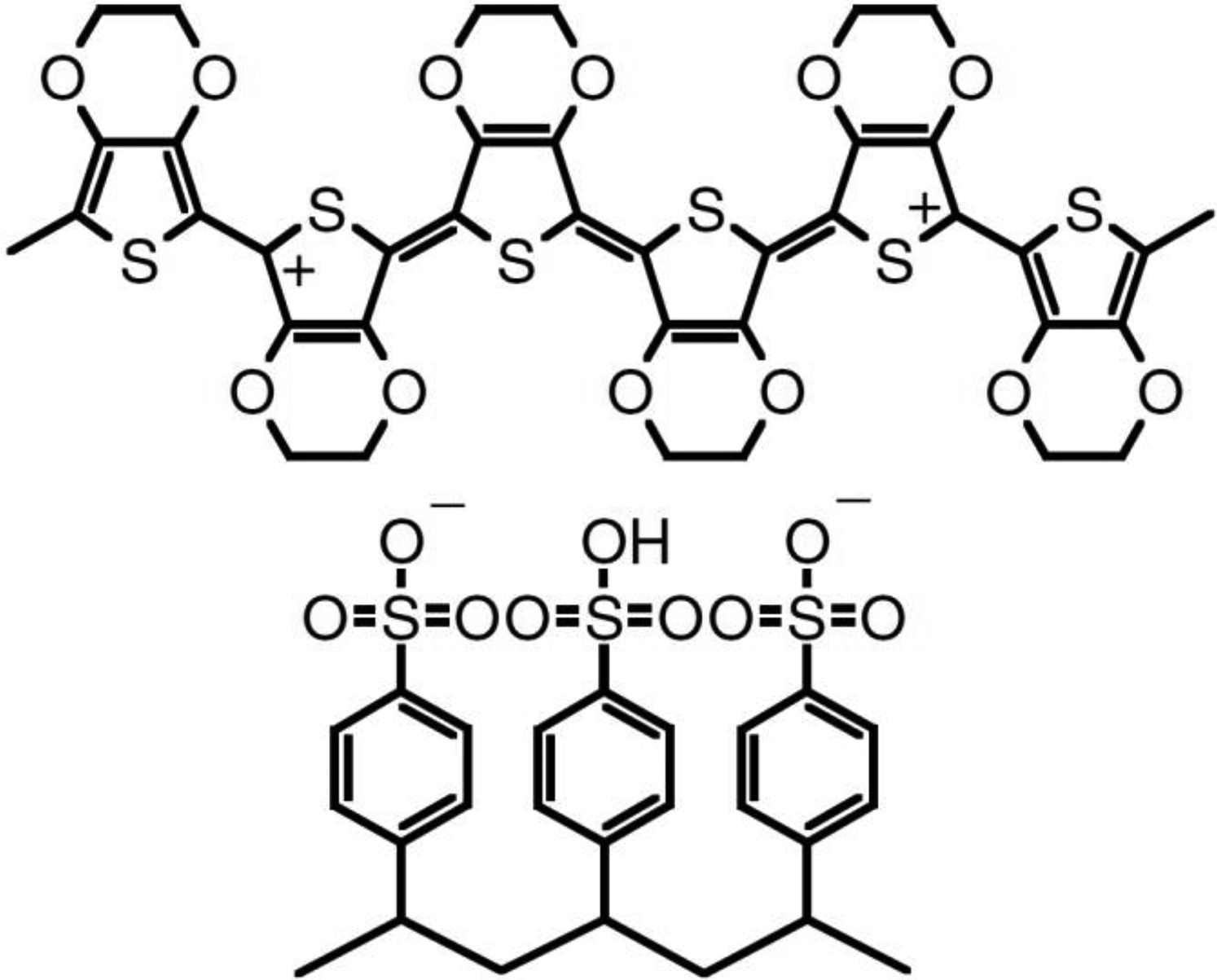
The molecular structure of PEDOT:PSS. 49 PEDOT:PSS: poly(3,4-ethylenedioxythiophene)–poly(styrene sulfonate).
In this study, additives with a high and low boiling point were added into highly conductive grade PEDOT:PSS, which were subsequently spin-coated on glass slides. However, the spin-coated pristine PEDOT:PSS films were treated by a mild acid solution in order to increase electrical conductivity to use as a transparent electrode for OSCs.
Materials and methods
Clevios PH1000 (high conductive grade) and conductive-grade (P VP AI 4083) PEDOT:PSS aqueous solutions, poly(3-hexylthiophene-2,5-diyl) (P3HT) with highest occupied molecular orbitals (HOMO) and lowest unoccupied molecular orbitals (LUMO) of P3HT are 5 and 3 eV, respectively; [6,6]-phenyl C61 butyric acid methyl ester (PC61BM) with HOMO and LUMO of PC61BM are 6.1 and 3.7 eV, respectively; ITO-coated glasses with 8–12 Ω/sq, anhydrous ethylene glycol (EG), anhydrous isopropyl alcohol (IPA), and chlorobenzene (ACS reagent) were purchased from Sigma-Aldrich. H2SO4 (1 M) in water solution was purchased from the market and used as is. All the materials were used without further purification.
Fabrication of the devices
Optical microscope slides were used as a substrate cut into square pieces by 1 inch and washed in distilled water, acetone, and IPA in an ultrasonic bath for 30 min, respectively, and then, dried in a vacuum oven and treated by UV-ozone for 30 min prior to use. This cleaning procedure was applied for all the following studies, where optical microscope slides were used.
P3HT (10 mg) (regioregular) and Phenyl-C61-butyric acid methyl ester (PCBM) (8 mg) were dissolved in 1 mL chlorobenzene and stirred overnight at room temperature prior to use.
The following OSC constructions were produced on glass substrates: ITO/PEDOT:PSS (P VP AI 4083)/P3HT:PCBM/LiF:Al (reference sample) EG (6%)-PEDOT:PSS (PH 1000)/P3HT:PCBM/LiF:Al PEDOT:PSS (PH 1000) (acid treated)/P3HT:PCBM/LiF:Al IPA (1:1)-PEDOT:PSS (PH 1000)/P3HT:PCBM/LiF:Al PEDOT:PSS (PH 1000)/P3HT:PCBM/LiF:Al
PEDOT:PSS (PH 1000) solutions were spin coated at 1500 r/min for 60 s on cleaned microscope slides and annealed in a vacuum oven at 100°C for an hour. Doped-PEDOT:PSS solutions with the mentioned amount of materials (EG and IPA) in volume ratio were stirred overnight at room temperature prior to deposition. The amount of doping solvents was chosen according to the reports in the literature. 9,53 -56
For the acid-treated samples, spin-coated PEDOT:PSS (PH 1000) layer was immersed into the 1% sulfuric acid solution (aq) and rinsed in deionized water and then dried in vacuum oven at 100°C for an hour.
PEDOT:PSS (P VP AI 4083) was spin coated as a buffer layer at 4000 r/min for 60 s onto ITO control devices only and annealed in a vacuum oven at 100°C for an hour.
P3HT:PCBM was spin coated at 1500 r/min for 60 s onto PEDOT:PSS layer.
LiF (0.7 nm) and Al (100 nm) was evaporated at 10−6 Torr and annealed in a glove box at 150°C for 5 min. The schematical drawing of the solar cells with PEDOT:PSS cathode treated with different additives is shown in Figure 3.
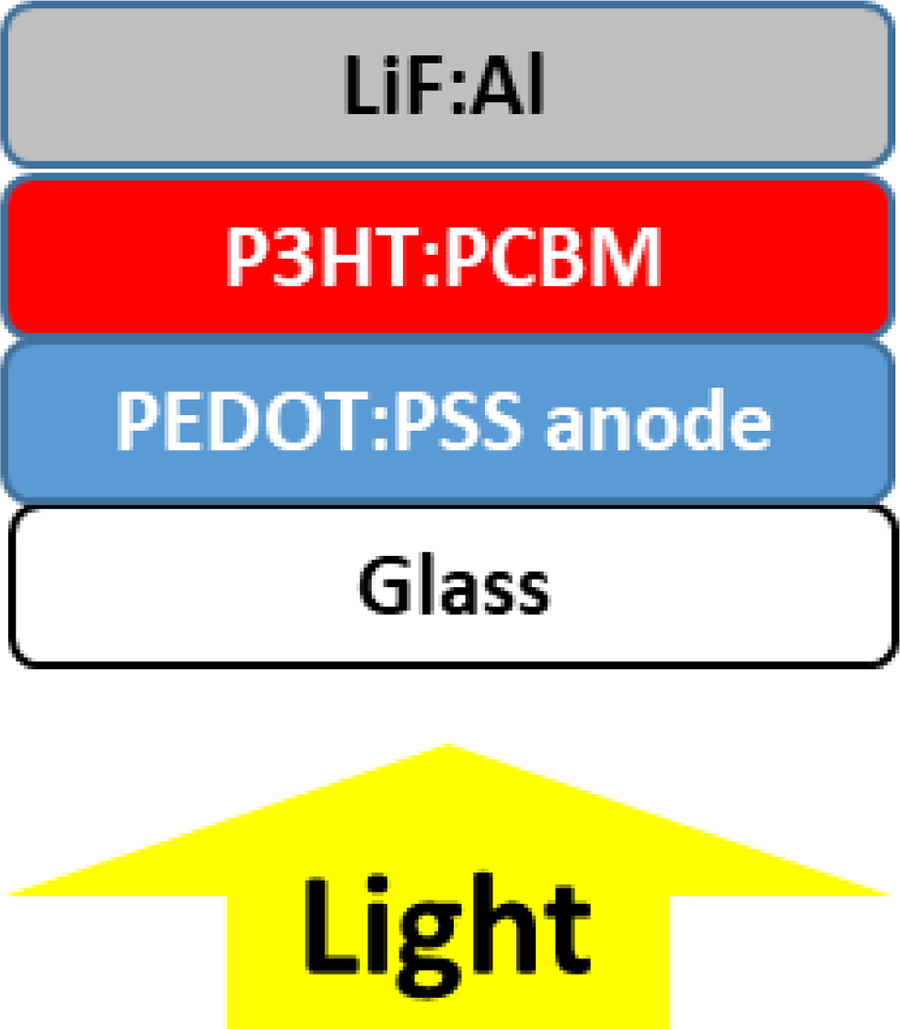
The schematical drawing of the solar cell with PEDOT:PSS anode.
Characterization
Two-probe resistivity measurements of the PEDOT:PSS electrodes were carried out with a multimeter prior to fabricating PV devices. The measurement was taken by the contacting two probes of multimeter with 1 cm distance in between and the value on the screen is noted.
The photoelectrical results of the solar cells are mostly rated in terms of their efficiency with respect to standard reporting conditions (SRC) defined by temperature, spectral irradiance, and total irradiance. The SRC for the performance of the PVs is as follows: 1000 W/m2 irradiance, AM 1.5 (AM: air mass) global reference spectrum, 57 and 25°C cell temperature.
The PCE (η) of a PV cell is given as follows
where Imax and Vmax are the maximum values at the peak power of the solar cell, Pin is the total incident irradiance, Isc is the current where the voltage is zero, and Voc is the voltage where the current is zero. A LOT-Quantum Design GmbH solar simulator is used as a light source and Keithley 2400 source-meter was used to measure the photoelectrical result of the solar cells. An ideal current–voltage curve is shown in Figure 4.
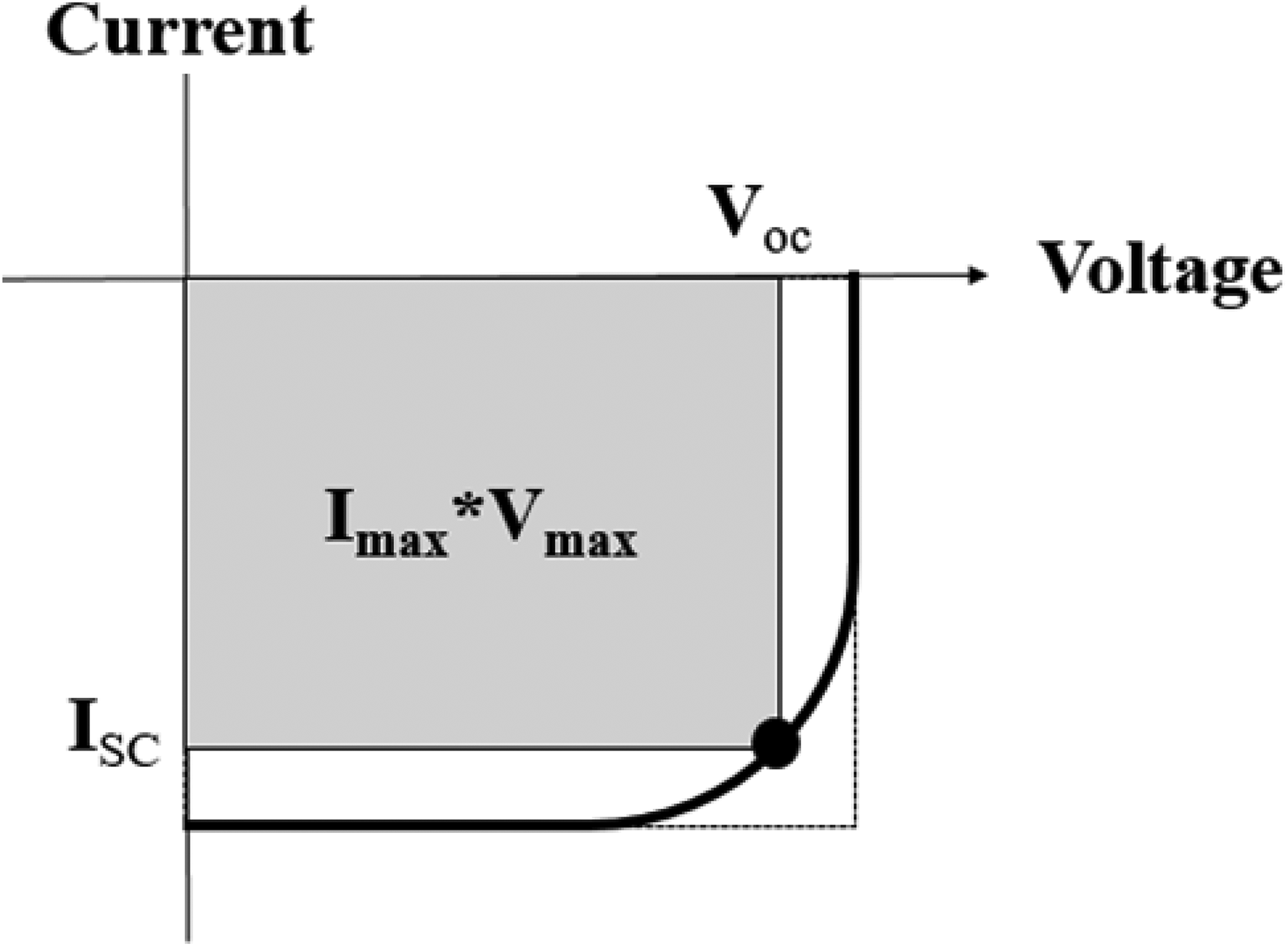
The illuminated IV curve. IV: current–voltage.
UV-Vis spectra of PEDOT:PSS and ITO were obtained with Perkin Elmer Luminescence Spectrometer.
Dektak profilometer was used to measure the film thickness of spin-coated PEDOT:PSS films.
Results
UV-Vis transmittance of pristine PEDOT:PSS on microscope glass was measured between 350 and 800 nm wavelengths and compared with ITO-coated glass. The thickness of the pristine PEDOT:PSS film which was spin-coated at 1500 r/min for 60 s was measured as 120 ± 5 nm. The transmittance graph is given in Figure 5. As can be seen from Figure 5, PEDOT:PSS layer is so thin so that light can go through the structure easily.

The transmittance graph of pristine PEDOT:PSS and ITO on glass.
Volume resistivity measurement of PEDOT:PSS electrode layers is given in Table 1.
The volume resistivity measurements of PEDOT:PSS layers.

PEDOT:PSS: poly(3,4-ethylenedioxythiophene)–poly(styrenesulfonate); EG: ethylene glycol; IPA: isopropyl alcohol.
As it is known, pristine PEDOT:PSS is due to its insufficient conductivity, problematic as an electrode of OSCs. However, ITO which is brittle and expensive is not suitable for flexible applications. Therefore, in order to improve the photoelectrical results of the SCs having PEDOT:PSS layer as transparent electrode, EG and IPA have been separately added into PEDOT:PSS solution before spin coating and so formed layer was used as an anode and the other OSC layers were deposited thereof while pristine PEDOT:PSS-coated film has been treated with sulfuric acid mentioned. Table 2 presents the photoelectrical results of the OSCs with ITO anode as well as the pure PEDOT:PSS anode as reference values.
Photoelectrical results of solar cells with modified PEDOT:PSS electrode.

PEDOT:PSS: poly(3,4-ethylenedioxythiophene)–poly(styrenesulfonate); ITO: indium tin oxide; EG: ethylene glycol; IPA: isopropyl alcohol; SD: standard deviation; FF: Fill factor.
aThe average values and standard deviations are calculated from 15 devices.
Despite the fact that EG and IPA improve the PV performances in comparison to pure PEDOT:PSS, both of these additives do not create the expected result to exceed the ITO performances.
The current–voltage curves of the investigated OSCs with modified PEDOT:PSS electrodes are given in Figure 6.
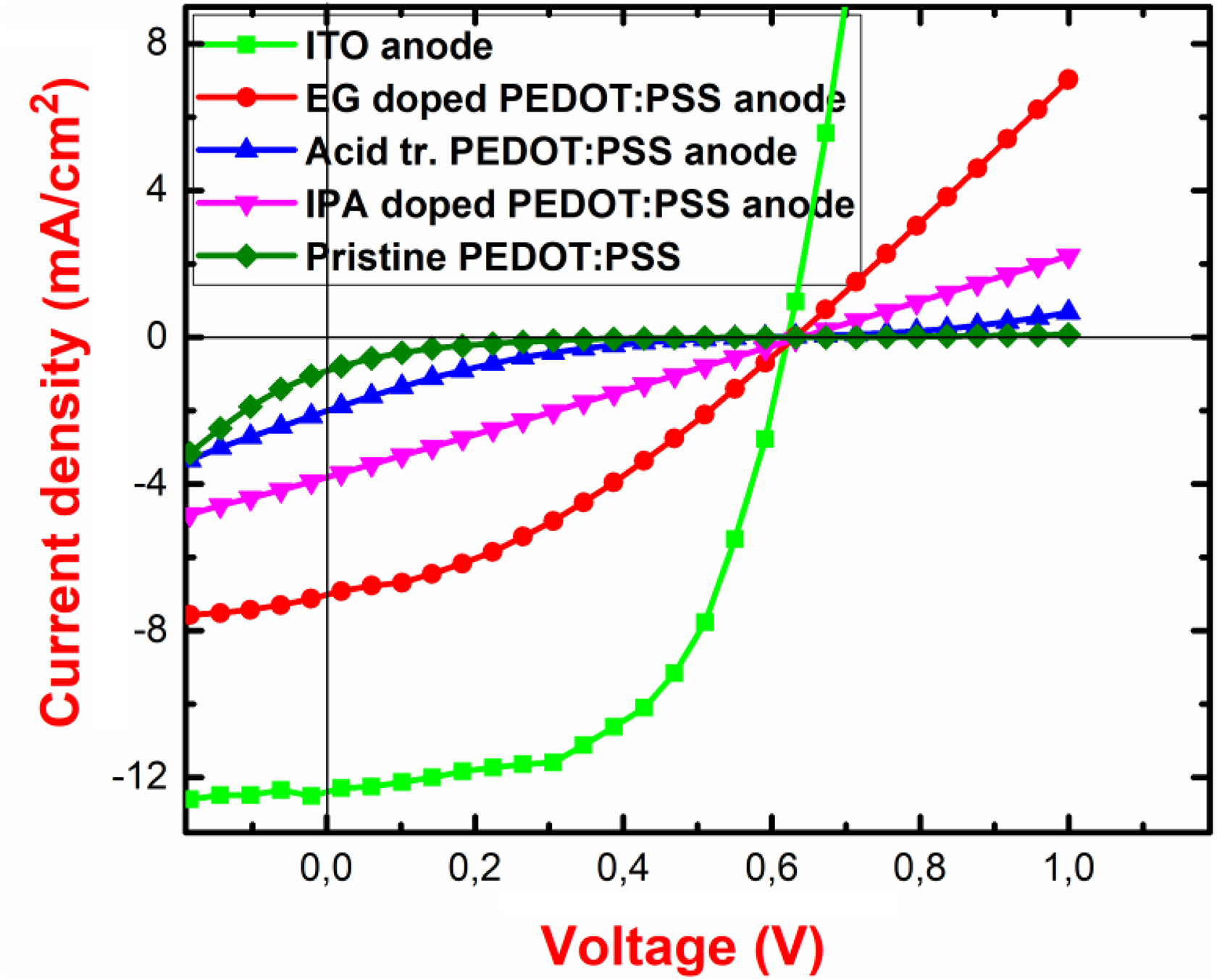
The IV curves of the investigated OSCs with PEDOT:PSS electrodes. PEDOT:PSS: poly(3,4-ethylenedioxythiophene)–poly(styrenesulfonate); OSC: organic solar cell; IV: current–voltage.
Addition of IPA improves the wetting and sticking of PEDOT:PSS on glass substrates, 58,59 but obviously, it did not improve the PCE of OSCs with IPA-added PEDOT:PSS electrodes here. Produced solar cells with the pristine PEDOT:PSS and IPA-doped PEDOT:PSS electrodes showed lower PCEs compared to EG-doped PEDOT:PSS and acid-treated PEDOT:PSS electrodes.
The best efficiency results were obtained from the EG-doped PEDOT:PSS anode. This could be explained as follows: The polar solvents with high boiling point such as EG, dimethyl sulfoxide (DMSO) remarkably increase the electrical performance of PEDOT:PSS, thus contributing the PCE in OSCs beside the high screening effect and important morphological changes. 60 -62 EG forms a hydrogen bond with PSS in PEDOT:PSS, thus the interaction between PEDOT and PSS is lowered 63 with the addition of EG, due to the removal of PSS, the conformation of PEDOT forms from a coiled structure to linear or extended-coil structure. 64 By the posttreatment of sulfuric acid, PSS could be removed away and improve the crystallization and optimize the conformation of PEDOT. 63
Conclusions
PEDOT:PSS films were deposited by spin coating on glasses. The effects of addition of IPA and EG into PEDOT:PSS solution and sulfuric acid treatment of spin-cast films were carried out to improve electrical properties in regard to use as a transparent electrode in OSCs. The surface resistivity of PEDOT:PSS films was measured. OSCs were fabricated and PV performances were compared. EG-doped PEDOT:PSS film has shown the best efficiency among the test samples.
Considering complicated production methods of ITO-based solar cells, PEDOT:PSS can be a good alternative transparent electrode for OSCs and flexible PVs since they can be applied via solution-based coating techniques.
It is believed that one of the reasons for the low efficiency obtained from the devices with PEDOT:PSS anode is coming from the very thin of PEDOT:PSS layer (about 120 nm whereas ITO thickness is about 120–160 nm 65 ) as an electrode together with the nonuniform of that layer on a glass substrate. Therefore, the thickness and the uniformity of PEDOT:PSS anode layer should be further improved in the future work. However, the low performance is not due to the low thickness but also about the relatively low conformal changes in the polymer.
Footnotes
Acknowledgments
Authors would like to thank Professor Dr. David Carroll and Dr. Jiwen Li of the Center for Nanotechnology and Molecular Materials at Wake Forest University for their valuable contributions. Professor Dr Ali Demir and Associate Professor Dr Ayşe Celik Bedeloğlu are also acknowledged for their scientific suggestions and discussions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Turkish Scientific and Technical Research Council, TUBITAK, Program No: 2214/A.

