Abstract
The poly(
Introduction
Block copolymers have been combined with two or more chemical structures and have excellent properties. Recently, block copolymers have been widely applied as pressure sensitive adhesives, additives, drug delivery carriers, and smart materials. 1 -5 Polylactide and polyethylene glycol blocks copolymers (PLA-b-PEG) have an important position due to temperature sensitivity, good biocompatibility, and degradability. 6 -11 The strength and toughness of PLA-b-PEG copolymers mainly depend on the semi-crystalline structure. The interplay between PLA and PEG in the crystallization process is very important for the formation of final structure. 12 The crystallization process and crystal morphology of PLA-b-PEG copolymers were investigated. 13 -15 The crystallization behaviors and morphology of copolymers are different by comparing with the mixture of homopolymers. 16 Huang et al. 17 studied the phase separation process of diblock copolymers containing 24% PLA and 14% PEG. The differential scanning calorimetry (DSC) and small-angle X-ray scattering results revealed that the disorder-to-order process is driven by the crystallization of PLA. Sun et al. 18 exhibited the effect of PEG content from 7% to 52% on the crystallization of PLA chain, showing the crystallization rate of PLA is improved. Yang et al. 19 found that the crystallization rate of PEG chain increased when the components of PEG chain are between 50% and 67%. In addition, some scholars 13,16,20 also studied the crystal morphology of PLA-b-PEG by polarizing optical microscopy (POM), and found that the crystal morphology of PLA chain is affected by PEG chain. However, after PLA chain are crystallized, the crystallization process does not affect the crystal structure of the original PLA chain. Due to the presence of two different chains within the PLA and PEG diblock copolymer (PLA-b-PEG), the crystallization between PLA and PEG interacts with each other.
The most of above work focus on the influence of block components on the crystallization and morphology of PLA-b-PEG diblock copolymers. However, only a few studies report the effect of the chain length on the crystallization of copolymers. Yang et al.
21
studied the effect of PLA chain length on the crystallization behaviors of PEG chain. It is found that the restriction effect of PLA chain on PEG chain is gradually increased when the length of PEG chain remains unchanged. Hu et al.
22
found that PLA crystallization is limited when the length of PEG chain does not change and the length of PLA chain increases, which is attributed to the viscosity of the PLA system. Although some studies were reported on the synthesis and properties, the crystallization behaviors and spherocrystal morphologies of poly(
In this work, PLLA-PEG-PLLA triblock copolymers with different chain segment length were synthesized and their structures were analyzed by Fourier transform infrared spectroscopy (FTIR), 1H-nuclear magnetic resonance (NMR) spectroscopy, and gel permeation chromatography (GPC). The influences of PLLA and PEG chain segment length on the crystallization behaviors and the spherulite morphologies of PLLA-PEG-PLLA triblock copolymers were characterized and discussed in detail using POM, X-ray diffraction (XRD), and DSC. In addition, the crystallization mechanism of PLLA-PEG-PLLA triblock copolymers was also discussed in detail.
Experimental
Materials
Synthesis of PLLA-PEG-PLLA triblock copolymer
The typical procedure for synthesis of PLLA-PEG-PLLA triblock copolymers was previously reported.
24
In polymerization, the fixed ratio of PEG and
Characterization and measurements
FTIR spectra were collected using a Thermo Nicolet iS50 FTIR spectrometer (Waltham, MA, USA) in the wavenumber range of 400–4000 cm−1 with KBr pellet.
The chemical structures of the triblock copolymers in CDCl3 were analyzed by 1H-NMR (400 MHz Bruker, Germany). The molecular weights and compositions of the triblock copolymers were determined by NMR. The number-average molecular weight (Mn ) and PLLA-PEG-PLLA block ratio were calculated from the integration of 1H-NMR from the 1H-NMR spectra obtained in CDCl3. 7 Polydispersity was measured against polystyrene standards using GPC (Shimadzu HPLC LC-20A, Japan) with the flow rate of 1 mL min−1 at the room temperature. The column temperature was 35°C, and the THF was a mobile phase.
XRD patterns of the samples were performed using an X-ray diffractometer (X’pertPowder, PANalytical, Netherlands) with a Cu Kα radiation (λ = 0.154 nm) generated at 50 kV and 250 mA. Samples were scanned from 10° to 30° at a scanning rate of 2°C min−1.
DSC measurements were recorded with a DSC 910 of DuPont Instrument (Wilmington, USA) under N2 atmosphere. The samples were heated from room temperature to 180°C with a heating rate of 10°C min−1, and kept for 5 min to erase any thermal history. Then samples were cooled to −20°C with a cooling rate of 10°C min−1 and then heated to 180°C with a heating rate of 10°C min−1. The melt crystallization temperature (T mc), crystallization enthalpy (ΔH mc), melting temperature (T m), and melting enthalpy (ΔH m) were obtained. Degrees of crystallinity (X ci) were calculated from X ci = ΔH m,i/ωiΔH m,I, where ΔH m,i is the corresponding melting enthalpy, and ωi is the weight fraction for the block considered. 16 ΔH m,i is enthalpy of fusion of the corresponding 100% crystal structure. ΔH m,i values of 197 and 94 J g−1 were used for PEG and PLLA, respectively. 14
POM (AxioScope.A1, Carl Zeiss, Germany) was used to observe the crystal morphology of spherulites. The samples were melted at 180°C under N2 gas protection and observed crystal morphology under different heat treatment conditions. Overall crystallization behaviors of samples were also monitored by the digital images.
Results and discussion
Chemical structure and composition of copolymers
Figure 1 shows FTIR spectra of pure PEG,

FTIR spectra of (a) PLLA-PEG-PLLA triblock copolymers, (b) PEG, and (c)
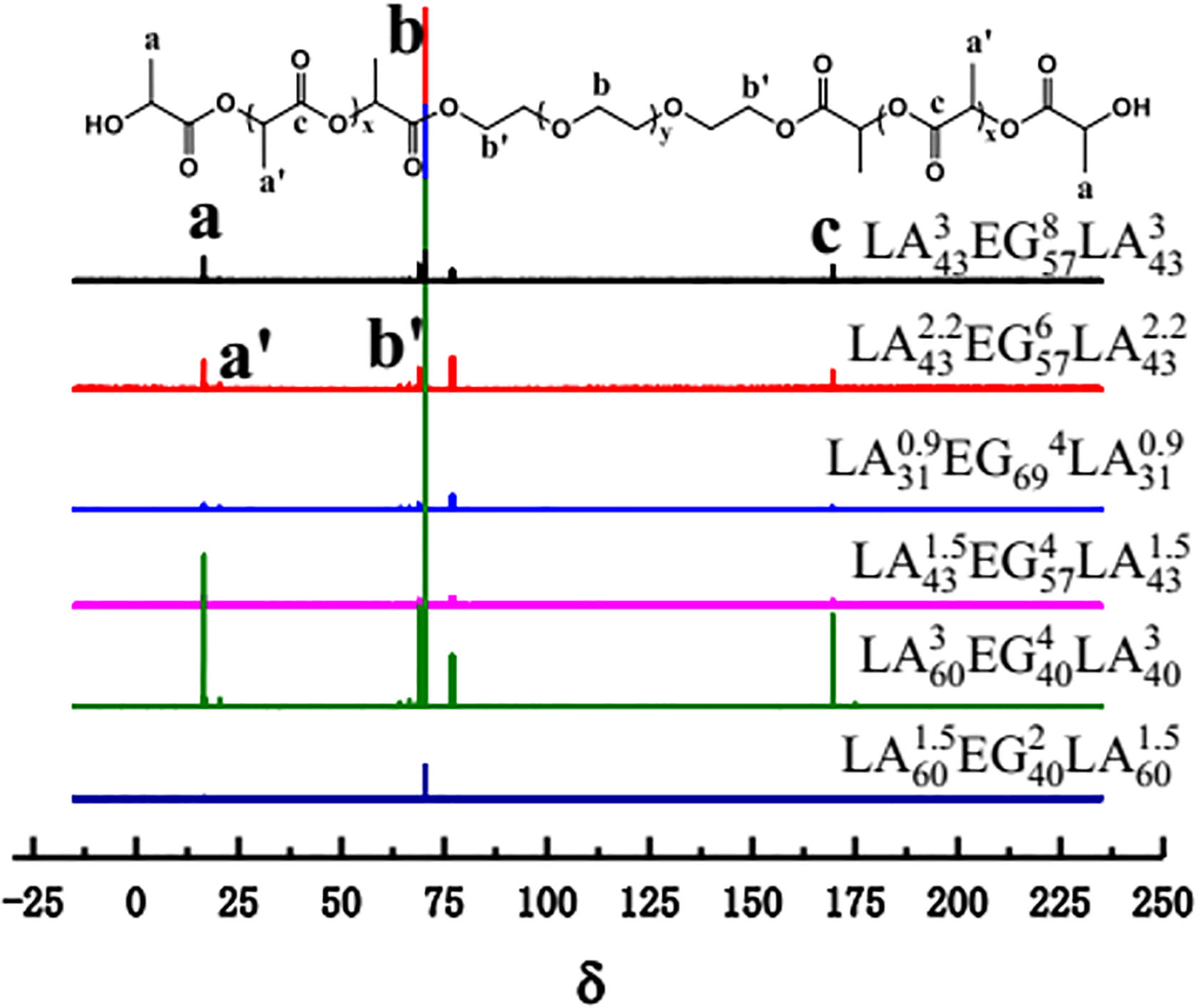
The 1H-NMR and 13C-NMR spectra of PLLA-PEG-PLLA triblock copolymers with different chain length are shown in Figures 2 and 3, respectively. As shown in Figure 2, the peaks at δ = 1.59 and 5.20 ppm are assigned to protons a and b of PLLA, respectively, and the area rate of the peak is about 3:1, which corresponds to the rate of H atoms in the PLLA chain. The peak at δ = 3.6 ppm is attributed to proton c of PEG. 23 Since the excessive PEG is removed by purification treatment, the proton b of PEG will not occur. The peak at δ = 7.36 ppm is assigned the proton peak of the residual solvent. It is observed from Figure 3 that the positions of the respective copolymer peaks are the same. The peaks at δ = 16.88 and 21.5 ppm are assigned to C atoms at proton a and a′ of PLLA chain. The peak at δ = 170.8 ppm is assigned C atoms at proton c of PLLA chain. The characteristic peaks at δ = 70.1and 66.8 ppm are assigned C atoms at proton b and b′ of PEG chain. At δ = 76.36 ppm, it should be the characteristic peak of the test solvent chloroform. All these FTIR and NMR results demonstrate the successful synthesis of PLLA-PEG-PLLA triblock copolymers with controlled PLA block length.

1H-NMR spectra of PLLA-PEG-PLLA triblock copolymers.
13C NMR spectra of PLLA-PEG-PLLA triblock copolymers.
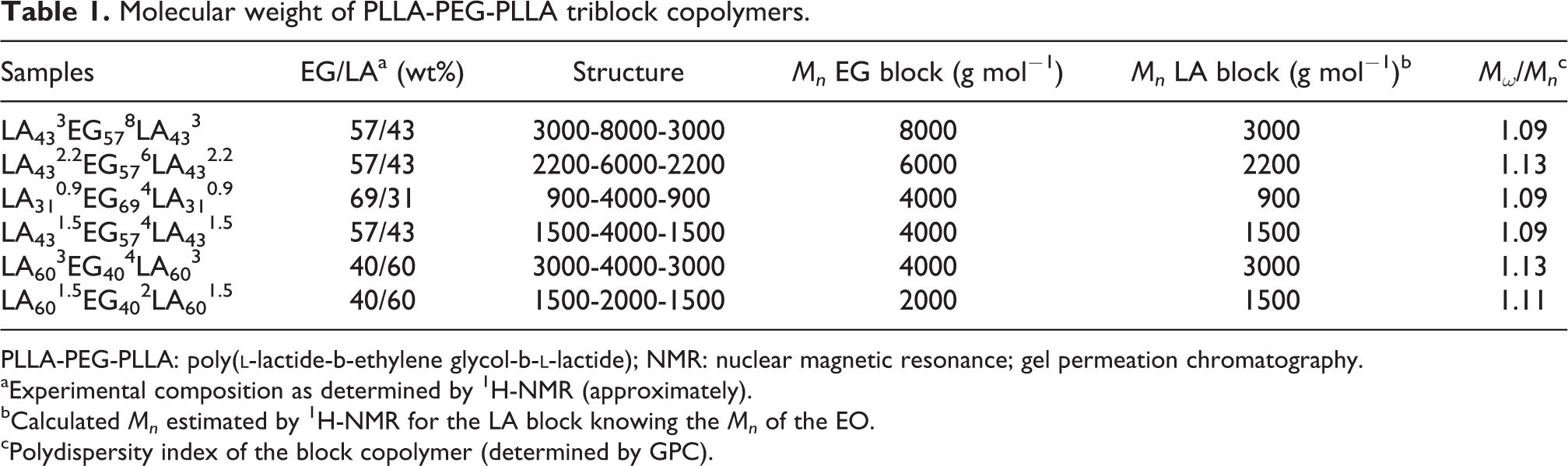
Molecular weights of PLLA-PEG-PLLA triblock copolymer are also measured by GPC and calculated from NMR spectrum by comparing NMR peak intensities. The GPC curves of samples are shown in Figure 4 and the corresponding data are listed in Table 1. As can be seen from Figure 4 and Table 1, the triblock copolymer has a very narrow molecular weight distribution, and the lowest molecular weight distribution index is 1.09, which is close to a single distribution. This further proves that the synthesized product is a copolymer rather than a blend of PLLA and PEG.
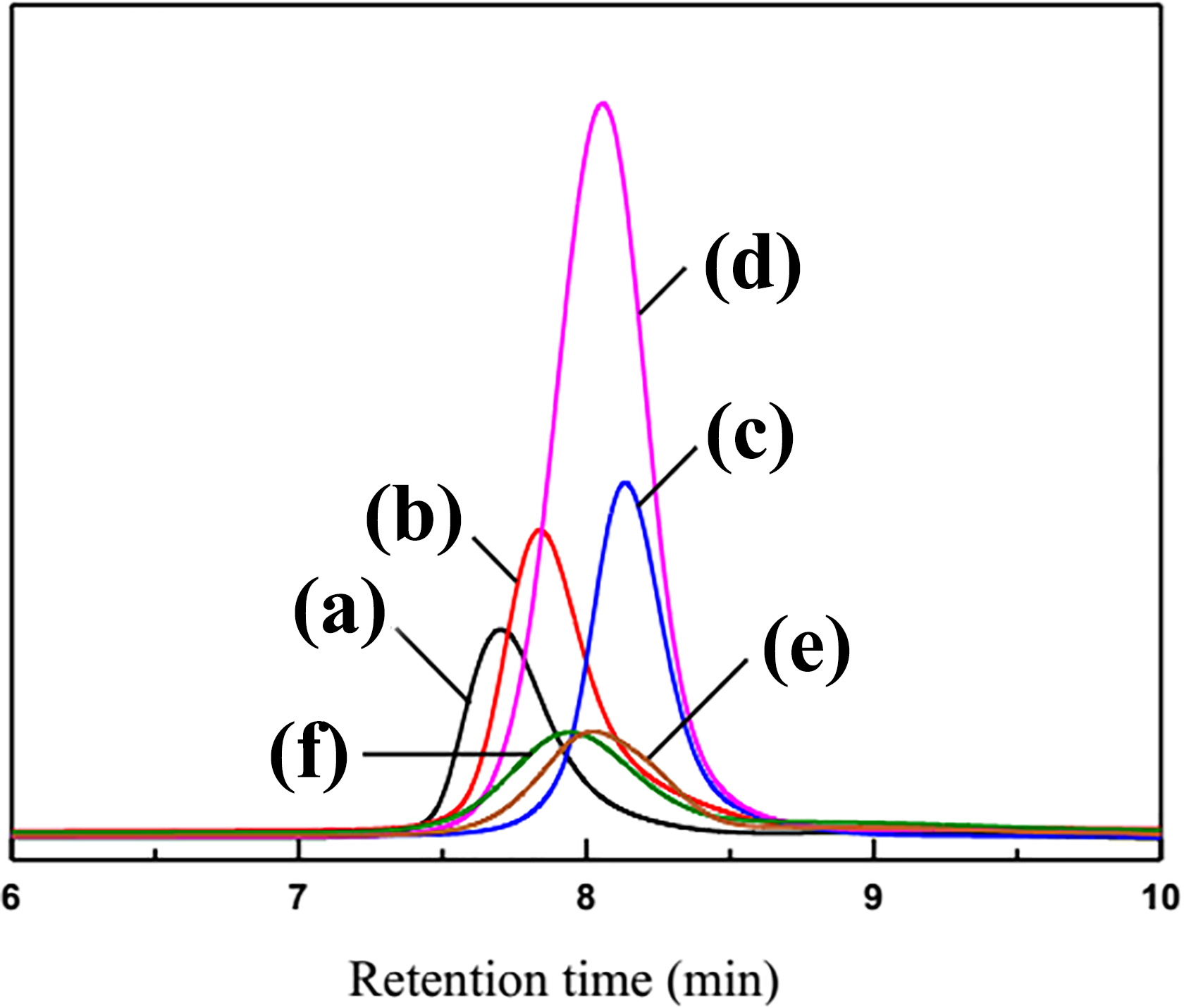
GPC curves of PLLA-PEG-PLLA triblock copolymers: (a) LA43 3EG57 8LA43 3, (b) LA43 2.2EG57 6LA43 2.2, (c) LA31 0.9EG69 4LA31 0.9, (d) LA43 1.5EG57 4LA43 1.5, (e) LA60 3EG40 8LA60 3, and (f) LA60 1.5EG40 2LA60 1.5.
Molecular weight of PLLA-PEG-PLLA triblock copolymers.
PLLA-PEG-PLLA: poly(
aExperimental composition as determined by 1H-NMR (approximately).
bCalculated Mn estimated by 1H-NMR for the LA block knowing the Mn of the EO.
cPolydispersity index of the block copolymer (determined by GPC).
XRD analysis
The XRD patterns of PLLA-PEG-PLLA copolymers are shown in Figure 5. The crystal of the PEG chain is a monoclinic crystal form, and the 2θ values are 15, 19.1, and 23.2°, corresponding to the plane (110), (120), and (032), respectively. 20 For PLLA, it has been proved that the ordinary crystals are pseudo-orthorhombic (α crystal, 17 β crystal, 25 and γ crystal). 26 In addition, the PLLA chain can also crystallize at low temperature to form α′ crystal, which is converted to α crystal after heating. The 2θ values of the α crystal of PLLA chain are 15, 16.7, 19.1, 21, 22.3, and 27.4°, which corresponds (010), (110/200), (203), (204), (015), and (207), respectively. XRD spectra show the peaks at 2θ values of 16.4, 18.7, 24.5° for α′ crystal. However, as shown in Figure 5, the diffraction peaks mainly appear at 2θ = 16.6, 19.1, and 23.2°, suggesting that the PLLA chain mainly forms α crystal in the triblock copolymer.

XRD patterns of various PLLA-PEG-PLLA triblock copolymers.
It is found from LA43 3EG57 8LA43 3, LA43 2.2EG57 6LA43 2.2, and LA43 1.5EG57 4LA43 1.5 that the intensity of the diffraction peak is enhanced with growth of PEG and PLLA chains, indicating that the growth of the chains promotes crystallization of the chain itself. This is consistent with the results of Luo et al. 27 The three samples show a dispersion peak at 2θ = 16.6°, which is attributed to the amorphous region in the PLLA chain. Interestingly, the three samples have strong peaks at 2θ = 19.1°, which may be the peak of (203) crystal plane of PLLA chain or (120) crystal plane of PEG chain. It is observed from LA31 0.9EG69 4LA31 0.9, LA43 1.5EG57 4LA43 1.5, and LA60 3EG40 4LA60 that the diffraction peak intensity at 2θ = 23.2° becomes weaker with the PLLA chain increase, indicating the growth of PLLA chain confines crystallization of the PEG chain. While the enhancement of the PLLA crystallization peak at 2θ = 16.6° indicates that the growth of the PLLA chain promotes the crystallization itself. It is seen from LA43 3EG57 8LA43 3, LA60 3EG40 4LA60 3, LA43 1.5EG57 4LA43 1.5, and A60 1.5EG40 2LA60 1.5 that the growth of PEG chain promotes crystallization itself but confines the crystallization of PLLA chain.
Thermal properties
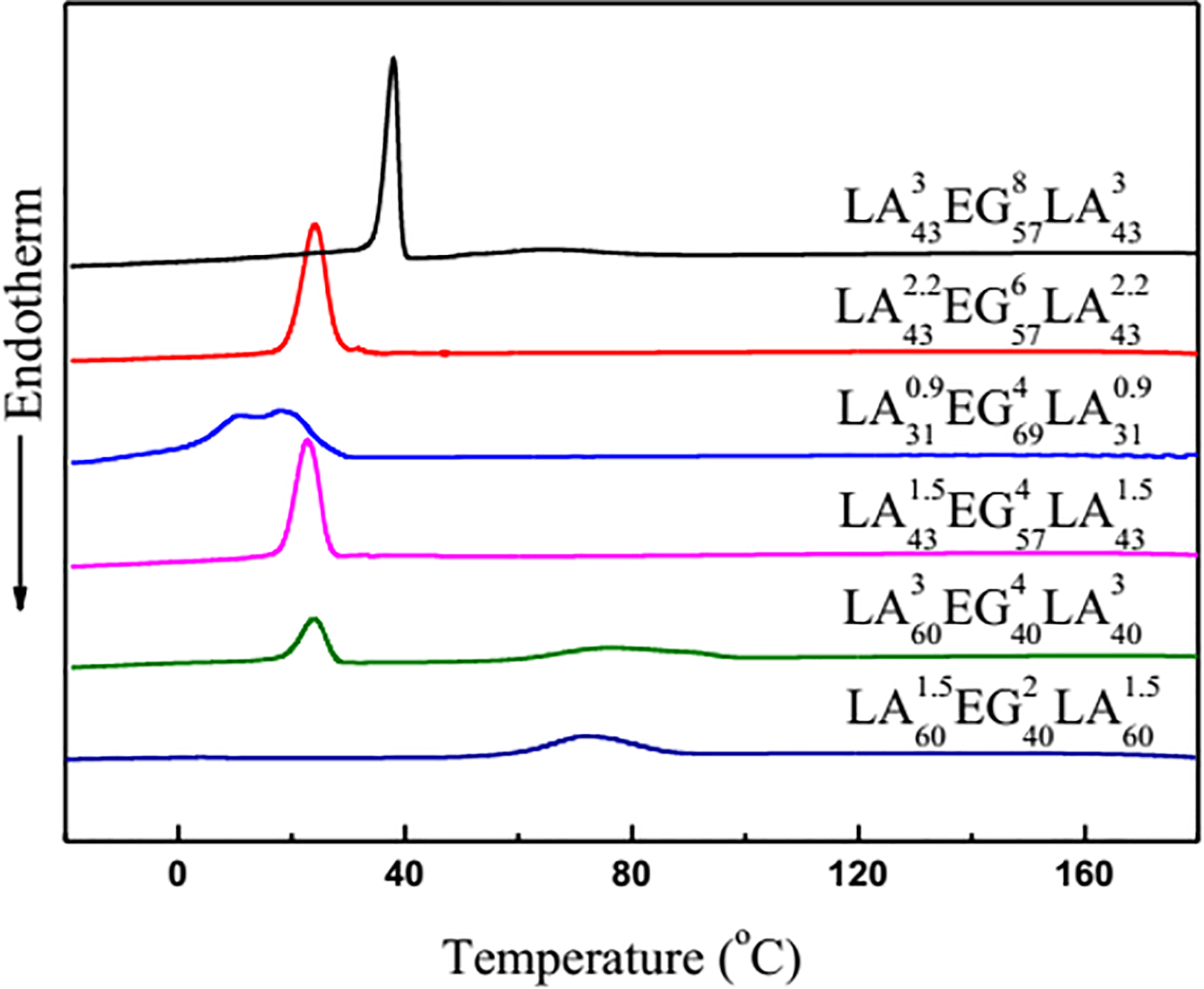
Thermal properties of PLLA-PEG-PLLA triblock copolymers are investigated by DSC, and the results are shown in Figures 6 and 7. The low temperature peak (T mc-PEG) and high temperature peak (T mc-PLLA) are assigned to the crystallization peak of PEG and PLLA, respectively. The crystallization is completed in two steps. Firstly, the chain with higher crystallization temperature crystallizes to form a crystal region with a miscible or partially miscible amorphous region system. Secondly, the chain with lower crystallization temperature crystallizes within the system when the temperature is continuously lowered, and crystallization in the zone or amorphous zone is limited by the microphase separation and other chains, which leads to be restricted at the crystallization of the chains at low temperatures. 28 -30 In the case of EO/LA > 1, both LA43 2.2EG57 6LA43 2.2 and LA43 1.5EG57 4LA43 1.5 exhibit only one T mc-PEG. However, LA43 3EG57 8LA43 3 shows two crystallization peaks. The double crystal phenomenon was also observed in other work. 28,31 On the one hand, the increase of PLLA chain length will promote the crystallization of PLLA. On the other hand, since relaxation time is proportional to molecular weight, PEG segmental motion induced the orientation of PLLA decays much more slowly for long chains than short chains, which is beneficial to the formation of PLLA crystal precursor and promotes the crystallization of PLLA. 22 Long-chain PEG may produce larger flow fields than short-chain PEG. Therefore, two crystal peaks appear for LA43 3EG57 8LA43 3. T mc value of PEG increases with increasing PEG chain length, indicating that the homogeneous nucleation of PEG chain is easier (as listed in Table 2). In the case of EO/LA < 1, T mc-PLLA value of LA60 3EG40 4LA60 3 is 10.5°C, which is higher than that of LA60 1.5EG40 2LA60 1.5. This indicates that the growth of PLLA chain promotes the heterogeneous crystallization itself. Compared to LA43 3EG57 8LA43 3 and LA60 3EG40 4LA60 3, LA43 1.5EG57 4LA43 1.5 and LA60 1.5EG40 2LA60 1.5, it is found that the growth of PEG chain is beneficial to the crystallization of PEG chain. However, the crystallization of PLLA chain is confined with increasing PEG chain length and vice versa. That is because that the movement of PEG chain is not conducive to the orientation of PLLA chain with increasing PEG chain length. In addition, the crystallization enthalpy of PEG chain is reduced and the peak shape becomes narrower with increasing PLLA chain length when PEG chain length is constant, indicating PLLA chain confines crystallization of PEG chain. For LA43 2.2EG57 6LA43 2.2 and LA43 1.5EG57 4LA43 1.5, the crystallization peak appears in XRD patterns, however, there is no crystallization peak in DSC curves. The possible reason is that the rapid cooling is too late to crystalline for PLLA chain.

DSC heating curves of block copolymers at heating rate of 10°C min−1.
DSC cooling curves of block copolymers at cooling rate of 10°C min−1.
Thermal parameters of PLLA-PEG-PLLA triblock copolymers.

PLLA-PEG-PLLA: poly(
In the case of EO/LA > 1, for LA43 3EG57 8LA43 3, LA43 2.2EG57 6LA43 2.2, and LA43 1.5EG57 4LA43 1.5, the value of ΔH m-PEG increases when the contents of PLLA and PEG chains are constant and the length of PEG and PLLA chains increases. LA43 3EG57 8LA43 3 also shows a melting peak at a high temperature, however, LA43 2.2EG57 6LA43 2.2, LA43 1.5EG57 4LA43 1.5, and LA31 0.9EG69 6LA31 0.9 do not show the melting transition because copolymers PLA chain is too short in the case of EO/LA > 1, indicating that the growth of the chains promotes the crystallization itself. However, in the case of EO/LA < 1, the values of ΔH m-PLLA of LA60 3EG40 4LA60 3 and LA60 1.5EG40 2LA60 1.5 become smaller and the crystallinity becomes lower when PLLA chain length increases, as listed in Table 3, which is attributed to a change in the proportion of block. For LA31 0.9EG69 4LA31 0.9, LA43 1.5EG57 4LA43 1.5, and LA60 3EG40 4LA60 3, the Tm and ΔHm values of PEG chain gradually become smaller when the length of PEG chain is constant and the length of PLLA chain increases. This indicates that the restriction of PEG chain is enhanced by the growth of PLLA chain. For LA43 3EG57 8LA43 3 and LA60 3EG40 4LA60 3, the Tm and ΔHm values increase, however, the Tm and ΔHm values of PLLA decrease for LA43 3EG57 8LA43 3 when length of PLLA chain is constant and the length of PEG chain increases, which reveals that restriction of PLLA is enhanced by growth of PEG chain.
Crystallinity of PLLA-PEG-PLLA triblock copolymers.

PLLA-PEG-PLLA: poly(
Crystallization morphologies
POM images of isothermal crystallization of LA43 1.5EG57 4LA43 1.5 at 70 and 25°C are shown in Figure 8. As can be seen from Figure 8(a), the number of spherulites in the copolymer increases with increasing time, but the growth rate of spherulites is still very slow. This is consistent with the above analysis of XRD and DSC. For LA43 1.5EG57 4LA43 1.5, although PLLA chain still crystallizes, the crystallization rate is very slow. No crystallization peak of PLLA chain appears on DSC cooling scans at 10°C min−1. PEG chain can form spherulites at 25°C, and the growth rate is faster, however, the spherulitic morphology is incomplete, as shown in Figure 8(b). It is very interesting to observe that the spherulites of PEG chain gradually cover the spherulites of PLLA chain during the process of growth. This indicates that the crystallization of PEG and PLLA chain is carried out separately in different temperature regions, which is consistent with the report of diblock copolymer by Arnal et al. 12

POM micrographs after isothermal crystallization of LA43 1.5EG57 4LA43 1.5 copolymers at different temperatures: (a) 70°C and (b) 25°C.
Figure 9 shows pictures of isothermal crystallization of LA43 3EG57 8LA43 3 and LA60 3EG40 4LA60 3 at 80°C. Generally, the melting point of PLLA is between 145°C and 190°C, and the melting point of PEG is around 66°C. 16 The PEG chain is still in a state of motion and does not form crystallization at 80°C. According to Hoffman theory, 32 the Regime III temperature region of PLLA is confirmed at the range of 70–95°C. In this triblock copolymer, the PEG segment hinders the PLLA crystallization. Therefore, the crystallization of triblock copolymer is very hard compared with a homopolymer. It is seen from Figure 9 that the growth rate of PLLA chain spherulites is reduced from 1.14 to 0.20 μm s−1 with increasing the length of the PEG chain. This is reduced by 5.7 times. The spherulite morphologies show a distinct dendritic bifurcation. This may be because the increase of PEG block length provides more hindrance to the folding of PLLA chains in crystal thereby reducing crystallinity of PLLA, resulting in the spherulite growth rate of PLLA chain decreases and destruction of the spherulite morphology.

POM and radial growth rate of spherulites for isothermal crystallization of PLLA-PEG-PLLA at 80°C: (a) LA43 3EG57 8LA43 3 and (b) LA60 3EG40 4LA60 3.
Figure 10 exhibits POM photos and radial growth rate of spherulites of triblock copolymers at 25°C. The samples are rapidly cooled from the melting state and the presence of crystals is not observed in other temperature regions. The spherulitic morphologies of the triblock copolymers are not much different. The growth rate of spherulites of PEG chain for LA31 0.9EG69 4LA31 0.9 and LA43 1.5EG57 4LA43 1.5 are 4.39 and 1.41 μm s−1, respectively, which is attributed to PEG chain confined by the frozen PLLA chain. In addition, the growth rate of spherulites increases from 1.14 μm s−1 to 2.46 μm s−1 when the proportion of the chain is constant and the chains grow synchronously for LA43 2.2EG57 6LA43 2.2. This indicates that the growth of the chains can promote crystallization itself.
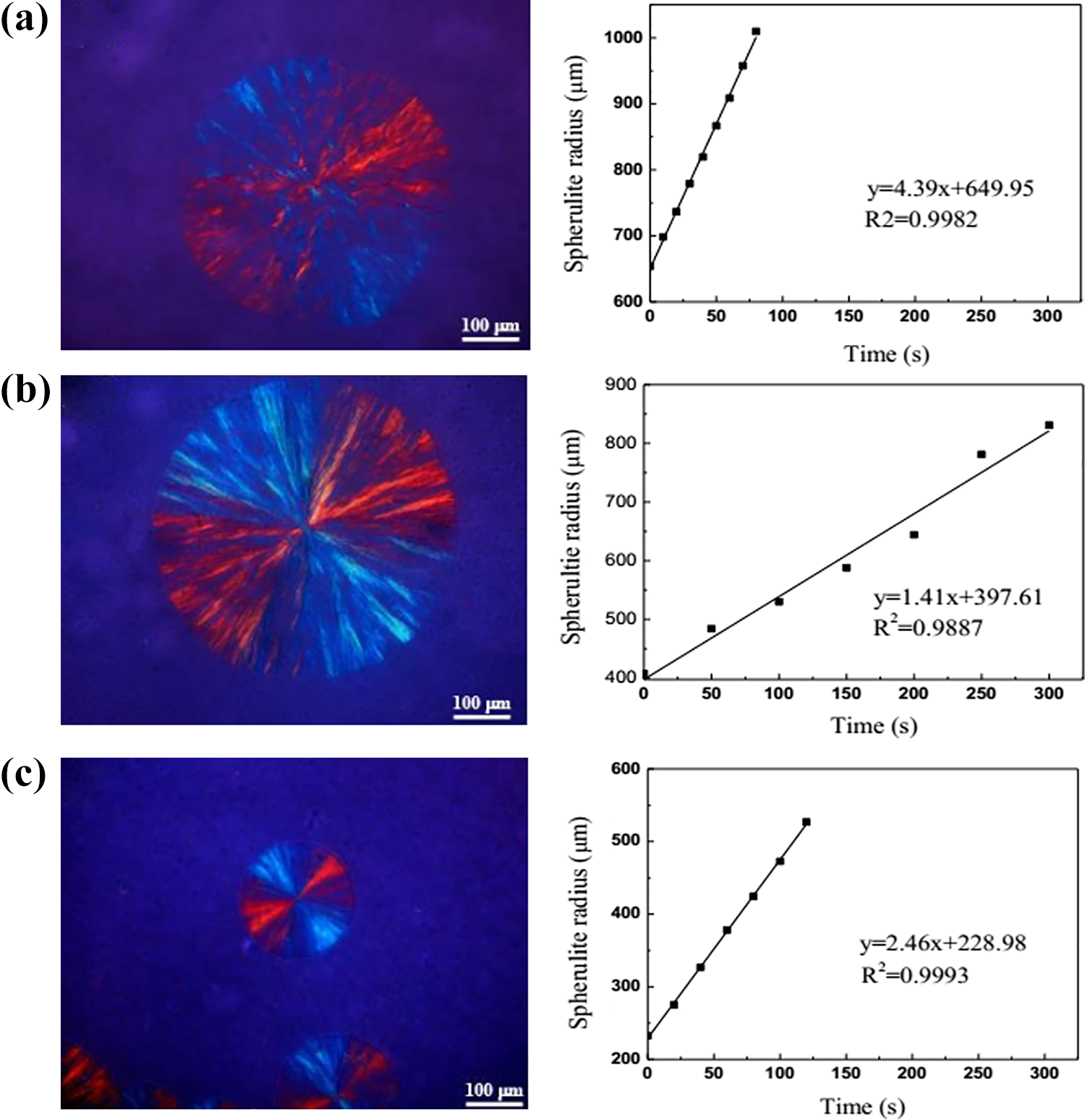
POM and radial growth rate of spherulites for isothermal crystallization of PLLA-PEG-PLLA at 25°C: (a) LA31 0.9EG69 4LA31 0.9, (b) LA43 1.5EG57 4LA43 1.5, and (c) LA43 2.2EG57 6LA43 2.2.
Figure 11 shows POM diagrams of PLLA-PEG-PLLA with different chain segment length. PLLA chain first forms crystals when the sample is cooled from the melting state. PLLA forms spherulites that are a template of morphology of the block copolymer because the second block that crystallizes at lower temperature (i.e. PEG) is forced to crystallize in the interlamellar and/or interfibrillar regions. 12 It has been concluded from DSC curves that LA43 3EG57 8LA43 3 and LA60 3EG40 4LA60 3 show double crystallization, however, LA43 2.2EG57 6LA43 2, LA31 0.9EG69 4LA31 0.9, LA43 1.5EG57 4LA43 1.5, and LA60 1.5EG40 2LA60 1.5 show single-chain crystallization. As shown in Figure 11(a) and (c), the spherulites of LA43 3EG57 8LA43 3 and LA60 3EG40 4LA60 3 have no obvious cross-extinction phenomenon due to the formation of spherulites of PEG chains in the crystal region of PLLA. Therefore, the spherulite morphology is destroyed. POM images of LA43 2.2EG57 6LA43 2, LA31 0.9EG69 4LA31 0.9, and LA43 1.5EG57 4LA43 1.5 present obvious spheroid morphologies of cross-extinction, indicating that the influence of PLLA on the spherulite morphology is very small below a certain threshold molecular weight. The spherulite morphology of LA60 1.5EG40 2LA60 1.5 is destructed to some extent, which shows that the moving PEG chain destroys the spherulite structure of PLLA. In addition, it can be found from Figure 11(c) and (d) that the number of spherulites gradually increases in the field of view. That is because that the rigid PLLA chain promotes nucleation of PEG spherulites as PLLA chain grows.

POM micrographs of cooling to 25°C at 5°C min−1 for block copolymers after melting for 5 min different triblock copolymers at 25°C: (a) LA43 3EG57 8LA43 3, (b) LA43 2.2EG57 6LA43 2, (c) LA31 0.9EG69 4LA31 0.9, (d) LA43 1.5EG57 4LA43 1.5, (e) LA60 3EG40 4LA60 3, and (f) LA60 1.5EG40 2LA60 1.5.
Crystallization mechanism of PLLA-PEG-PLLA triblock copolymer
The crystallization mechanism diagram of PLLA-PEG-PLLA triblock copolymer is exhibited in Figure 12. The PLLA and PEG chains are in a free motion state when the triblock copolymer is heated to 180°C. The PLLA chain is folded and crystallized when the temperature is in the crystallization zone of PLLA chain, while the free-moving PEG chain may have two effects on PLLA chain: (1) plasticization and (2) limitation. In the diblock copolymers, plasticization makes PLLA chain easier to fold into spherulites and increases the crystallization rate of PLLA chain. 18,19 However, the results show that the increase of block length of PEG provides more hindrance to the folding of PLLA chains in crystal thereby reducing crystallinity of PLLA in this work. As shown in Figure 12(a), the increase of block length of PEG makes PLLA block more crowded with PEG block, which limits the crystallization of PLLA and destroys the spherulite morphology of PLLA. For example, the spherulite growth rate and crystallinity of LA60 3EG40 4LA60 3 are 1.14 and 0.28 μm s−1, respectively, which is higher than that of LA43 3EG57 8LA43 3. The spherulite morphology of LA60 3EG40 4LA60 3 is also more complete than that of LA43 3EG57 8LA43 3. PEG chain is in metastable state and can be folded to form crystals when the temperature reduces to the crystallization region of PEG, however, PLLA chain is in stable state, which can build a stable space around PEG chain. As shown in Figure 12(b), PEG crystallizes inside of the PLLA crystallization. The orientation and crystallization of PEG chain is confined by PLLA chain when the metastable PEG chain begins to fold and form crystallization. For example, for LA31 0.9EG69 4LA31 0.9 and LA43 1.5EG57 4LA43 1.5, the spherulite growth rate of PEG is 4.39 and 1.41 μm s−1, and the crystallinity is 0.54 and 0.49, respectively, as PLLA chain grows.

Diagram of crystallization mechanism of PLLA-PEG-PLLA triblock copolymers.
Conclusions
In this article, the effect of composition and length of chain segment on the crystallization behaviors and morphologies of PLLA-PEG-PLLA is evaluated by XRD, DSC, and POM, and the crystallization mechanism of the triblock copolymer is proposed from the perspective of the interaction between the chains. The molecular weight of PEG ranges from 2 kg mol−1 to 8 kg mol−1 and its content in the synthesized copolymers varies are 40, 57, 69%. The molecular weight of PLLA block ranges from 0.9 kg mol−1 to 3 kg mol−1, and its content in the synthesized copolymers varies are 31, 43, and 60%. The XRD and DSC results show that the growth of PLLA (PEG) chain promotes its own nucleation crystallization and the restriction on the crystallization of PEG (PLLA) chain. The triblock copolymer exhibits a double crystallization phenomenon when the length of PLLA (PEG) chain is kept constant, and the length of PEG (PLLA) chain increases. Isothermal crystallization of LA43 1.5EG57 4LA43 1.5 indicates that the PLLA and PEG blocks crystallize in different temperature regions. Keeping PLLA (PEG) chain unchanged, the growth of PEG (PLLA) chain reduces, however, the spherical growth rate of PLLA (PEG) chain increases. The non-isothermal crystallization of POM exhibits that the spherulite morphology of the varies copolymers is severely damaged when copolymers exhibit double crystallization, however, the destruction is not significant when copolymers exhibit single crystallization. PLLA chain has a nucleating effect on the formation of spherulites of PEG chain when PEG chain grows.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Natural Science Foundation of China (51663006), International Science and Technology Cooperation Project of Sichuan (2019YFH0047), Guizhou Province Graduate Research Foundation (KYJJ2017012), Opening Project of State Key Laboratory of Polymer Materials Engineering (Sichuan University) (2017-4-02), Guizhou Province Science and Technology Program Project ([2017]5788, [2017]5630, [2017]1091), and Guizhou Province Key Topic of Education Teaching Reform for Postgraduates (JGKT2017011).
