Abstract
In this study, the influence of length of flexible groups on the properties of poly(amide–imide)s (PAIs), three-model polymers (poly(amide–imide)-4-aminobutyric acid, poly(amide–imide)-6-aminocaproic acid, and poly(amide–imide)-11-aminoundecanoic acid) possessing different flexible methylene units ((CH2)3, (CH2)5, and (CH2)10) in the main chain were designed. With increasing the number of methylene units, it is found that the tensile strength of PAIs decreased from 75 MPa to 55 MPa; meanwhile, the elongation at break increased from 6% to 15%. On the other hand, the glass transition temperature decreased from 207°C to 112°C; fortunately, the starting decomposition temperature kept almost same with a high point around 400°C. Furthermore, the PAI with (CH2)10 unit in the main chain is a semicrystalline polymer, while the one with (CH2)5 or (CH2)3 unit is an amorphous material. In other words, the length of the flexible chain in the polymer backbone not only plays an important role in mechanical and thermal performances but also affects their phase transition. These findings highlight the important role of structural modification in high-performance polymers and may help in the further development of novel PAIs for their potential applications in advanced technology.
Introduction
Aromatic poly(amide–imide)s (PAIs) have been considered as one class of high-performance polymer possessing amide and imide groups in the main chain. 1 –10 It is generally recognized that amide groups could increase the solubility, and the aromatic imide groups are beneficial to provide good mechanical and thermal properties. Ideally, PAIs could possess good mechanical and thermal properties of polyimides as well as ease of processability of polyamides. In fact, it is still a challenge to get a good balance between processability and mechanical prosperity. On the other hand, the properties of PAIs depend on polymer structure significantly. Therefore, it is necessary to discuss the relationship between the property and the structural modification. Until now, there have been many reports that discussed the influence of structural modification on the properties of PAIs, 11 –20 but most of the literature focused on the aromatic imide groups. In other words, it is necessary to discuss the influence of the length of flexible groups on the properties for further development of novel PAIs.
For the investigation of the relationship between the property and the structural modification, the model PAI polymers should satisfy the following requirements. First, the reactants should be commercial and the target polymers should be prepared easily. Second, the model polymers should exhibit favorable solubility and molecular weight to compare their properties, such as thermal stability and mechanical strength. Third, the structural modification should be carried out without changing the amide and imide structures of the parent polymer. Finally, the structural modification should be controlled regularly.
Based on these considerations, a series of diimide-diacid (DIDA) monomers containing different methylene units ((CH2)3, (CH2)5, and (CH2)10) were designed through the dehydration cyclization of 4,4′-oxydiphthalic anhydride (ODPA) with aliphatic amino acid, and then PAIs were prepared from p-phenylenediamine (PPDA) and the synthesized DIDA monomers by phosphorylation polycondensation. The influence of this structural modification on the properties of PAIs was researched comprehensively.
Experiment
Materials
ODPA, 11-aminoundecanoic acid (OAUA), 6-aminocaproic acid (EACA), 4-aminobutyric acid (GABA), PPDA, triphenyl phosphite (TPP), pyridine (Py), and solvents were used without further purification. N-methyl-2-pyrrolidone (NMP) was distilled over calcium hydride. Commercially available anhydrous calcium chloride (CaCl2) and lithium chloride (LiCl) were dried under vacuum at 150°C overnight.
Synthesis
Synthesis of DIDA monomers
The DIDA monomers (DIDA-GABA, DIDA-EACA, and DIDA-OAUA) were synthesized by the condensation of ODPA with aliphatic amino acid through dehydration cyclization according to the literature. 21 The synthetic route of the DIDA monomers is depicted in Figure 1, and a representative experimental process to synthesize DIDA-EACA is detailed as follows. Into a 100-mL flask equipped with a mechanical stirrer and a nitrogen inlet and outlet, ODPA (9.3066 g, 0.03 mol), EACA (7.8702 g, 0.06 mol), and acetic acid (120 mL) were introduced. The reaction was refluxed for 8 h, cooled to room temperature, washed with acetic acid, filtered off, and washed again with deionized water. The monomers were dried at 80°C for 12 h under vacuum (yield: 93%). Proton nuclear magnetic resonance (1H-NMR) (trifluoroacetic acid-d (TFA-d), δ, ppm): 11.50 (s, 2 H COOH), 7.90–7.92 (d, 2 H, aromatic), 7.49–7.50 (d, 2 H, aromatic), 7.40–7.42 (d, 2 H, aromatic), 3.72–3.75 (t, 4 H, NCH2), 2.31–2.51 (m, 4 H, CH2),1.69–1.74 (m, 8 H, CH2), and 1.38–1.41 (m, 4 H, CH2). Fourier transform infrared spectroscopy (FTIR; potassium bromide (KBr), cm−1): 3401 (O–H, stretching), 2917 and 2848 (CH2, aliphatic chain), 1764 and 1688 (C=O, imide carbonyl asymmetric and symmetric stretching vibration), 1471 (C=C, aromatic ring), 1431 (C–C, aromatic ring), 3034 (C–H, aromatic ring), 1396, 1368, 1067, 746 (C–N–C, stretching vibration, imide), 1268 (–O–, Ph–O–Ph), and 1606 (C=C, Ph–O–Ph). N, N′-(2-hexyl) oxydiphthalic diimide (DIDA-GABA, yield: 92%) and N, N′-(2-undecyl)oxydiphthalic diimide (DIDA-OAUA, yield: 92%) were also synthesized from GABA and (OAUA), respectively.

The synthetic route of diimide-diacid monomers.
Synthesis of PAIs
PAIs (PAI-GABA, PAI-EACA, and PAI-OAUA) were synthesized by phosphorylation polycondensation in accordance with the literature. 22 –27 The synthetic route of PAIs is illustrated in Figure 2. The experimental details are described as below using PAI-EACA as an example. Into a 100-mL flask equipped with a mechanical stirrer and nitrogen inlet and outlet, DIDA-EACA (2.681 g, 5 mmol), PPDA (0.546 g, 5.05 mmol), and NMP (12 mL) were introduced. CaCl2 (2.0 g) and LiCl (1.5–2 g) were used as water absorbents. TPP (6 mL) and Py (5 mL) were used as dehydrants. The reaction mixture was refluxed for 8–12 h, cooled to room temperature, washed thoroughly with methanol and hot water, and then dried under vacuum at 80°C overnight. A white lightweight fabric (PAI-EACA) was obtained (yield: 98%). FTIR (KBr, cm−1): 3294 (N–H stretching), 1652 (C=O stretching, amide), 1514 (C=C, aromatic, PPDA), 1547 (C–N, amide), 2918 and 2848 (CH2, aliphatic chain), 1765 and 1687 (C=O, imide carbonyl asymmetric and symmetric stretching vibration), 1474 (C=C, aromatic ring), and 1431 (C–C, aromatic ring), 3034 (C–H, aromatic ring), 1396, 1368, 1067, and 746 (C–N–C, stretching vibration, imide), 1268 (–O–, Ph–O–Ph), and 1606 (C=C, Ph–O–Ph). 1H-NMR (TFA-d, δ, ppm): 7.75 and 7.06 (s, H, NH) 7.90–8.12 (d, 2 H, aromatic), 7.50–7.74(d, 4 H, aromatic), 7.49–7.50 (d, 4 H, aromatic), 3.72–3.80(dd, 4 H, NCH2), 2.51–2.75 (t, 4 H, CH2), 1.69–2.01 (m, 8 H, CH2), and 1.41–1.62 (m, 4 H, CH2).
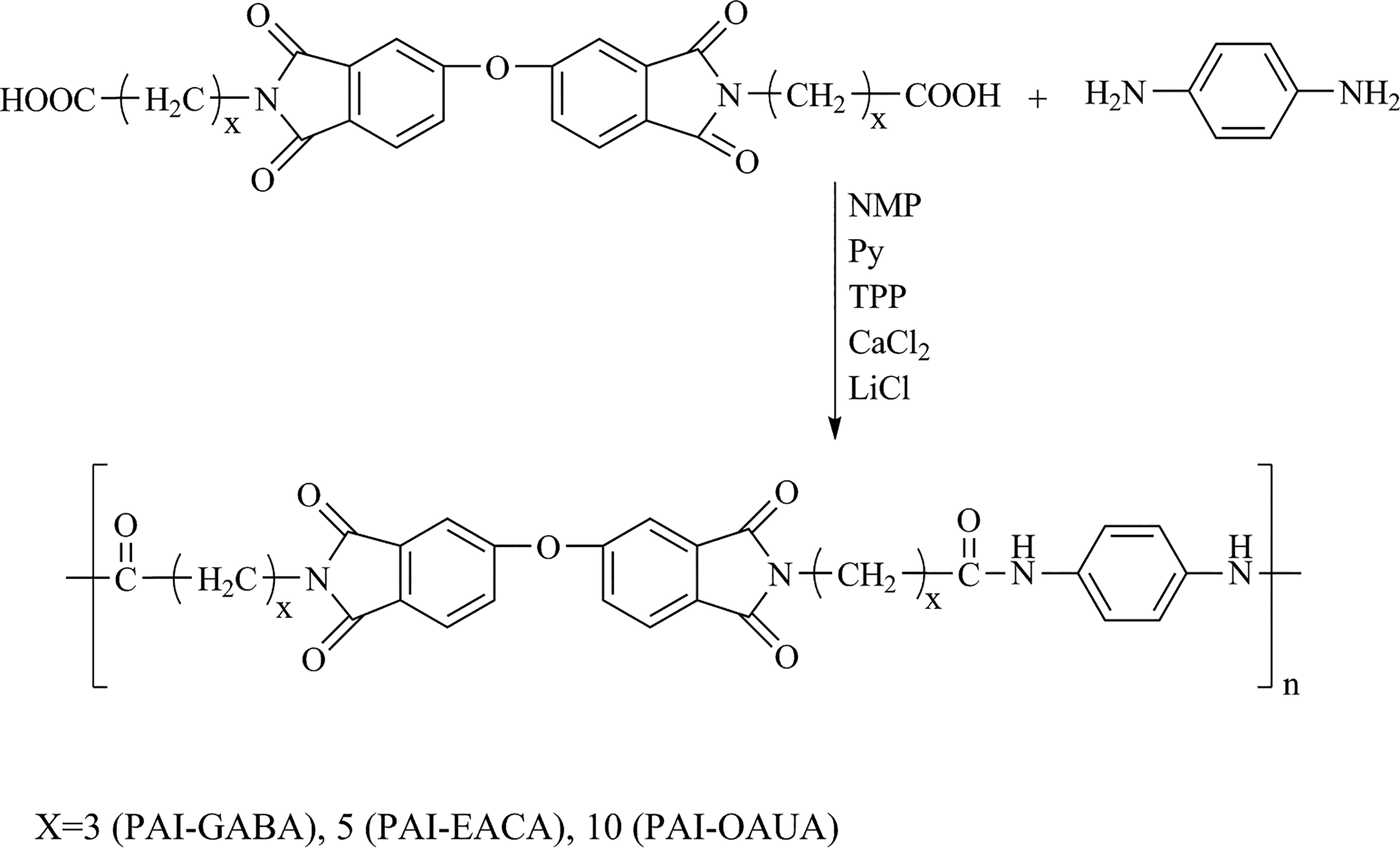
The synthetic route of poly(amide–imide)s.
Measurements
FTIR spectra were acquired with an Avatar 370 spectrometer (Thermo Nicolet, Ramsey, Illinois, USA). Spectra were collected in the region of 4000–400 cm−1 with a spectral resolution of 0.1 cm−1. 1H-NMR spectra were acquired at room temperature for DIDA monomers and PAIs on an Avance III 400 M NMR (Bruker, Rheinstetten, Germany) in TFA-d containing tetramethylsilane as the internal reference. The wide-angle X-ray diffraction (WAXD) measurement was obtained in a D/Max 2500 PC diffractometer (Rigaku Corporation, Japan) with curved graphite crystal filtered. The corresponding WAXD patterns were collected in transmission mode at room temperature under a tube current of 300 mA and an acceleration voltage of 60 kV. The thermal properties of the sample were measured with a PE Pyris 1 differential scanning calorimetry (DSC) (PerkinElmer, Waltham, MA, USA). About 7 mg of the sample was sealed in an aluminum pan; an empty sealed pan was used as the reference. The sample was heated to 300°C at a rate of 10°C min−1 and 20°C min−1, and the thermogram was recorded from the DSC first and second heating scan. Decomposition characteristics of the samples were determined by TG 209 F3 (Netzsch, Tarsus, Germany). About 5 mg of sample was placed in the pan and heated from 50°C to 750°C at a heating rate of 10°C min−1 under nitrogen atmosphere.
Results and discussion
Structure characterization of monomers and polymers
The chemical structure of DIDA monomers and PAIs was confirmed by FTIR and 1H-NMR. Firstly, representative FTIR spectra of monomer DIDA-EACA and polymer PAI-EACA are discussed in the following. As shown in Figure 3, the FTIR spectrum of DIDA-EACA exhibits characteristic bands of the imide ring at 1764 cm−1 (C=O asymmetric stretching), 1688 cm−1 (symmetric C=O stretching), 1396 cm−1, 1368 cm−1, 1067 cm−1, and 746 cm−1 (C–N–C, stretching vibration, imide), which demonstrates the successful formation of the imide group. On the other hand, in the FTIR spectrum of PAI-EACA, the broad peak around 3450 cm−1 ascribed to COOH group disappeared; instead, some new vibration peaks ascribed to amide group at 3294 cm−1 (N–H stretching), 1652 cm−1 (C=O stretching, amide), 1514 cm−1 (C=C, aromatic, PPDA), and 1547 cm−1 (C–N, amide) appeared. Meanwhile, those vibration bands associated with the imide ring of PAI-EACA are still observed at the almost same wave numbers of DIDA-EACA, which indicate that the imide ring is stable during Yamazaki phosphorylation reaction.

FTIR spectra of DIDA-EACA and PAI-EACA.
The chemical structure of DIDA monomers and PAIs was further characterized via 1H-NMR. In the 1H-NMR spectrum of DIDA-EACA as shown in Figure 4, the protons of carboxylic acid (H8) and aromatic protons of the dianhydride unit (H1–H3) appeared in the downfield at 11.5 ppm and 7.40–7.92 ppm, respectively. Peaks as a triplet at 3.72–3.75 ppm are assigned to the protons of the methylene-bonded imide group (H4). The other methylene protons (H5–H7) locate in the range of 1.38–2.51 ppm. In the 1H-NMR spectrum of PAI-EACA, peaks at 7.26 ppm and 7.50–8.12 ppm are assigned to NH (H1) of the amide linkage and aromatic proton (H2), respectively. The other methylene protons (H3–H8) appeared in the range 1.38–3.70 ppm. The results of FTIR and 1H-NMR support the successful preparation of DIDA monomers and PAI polymers.
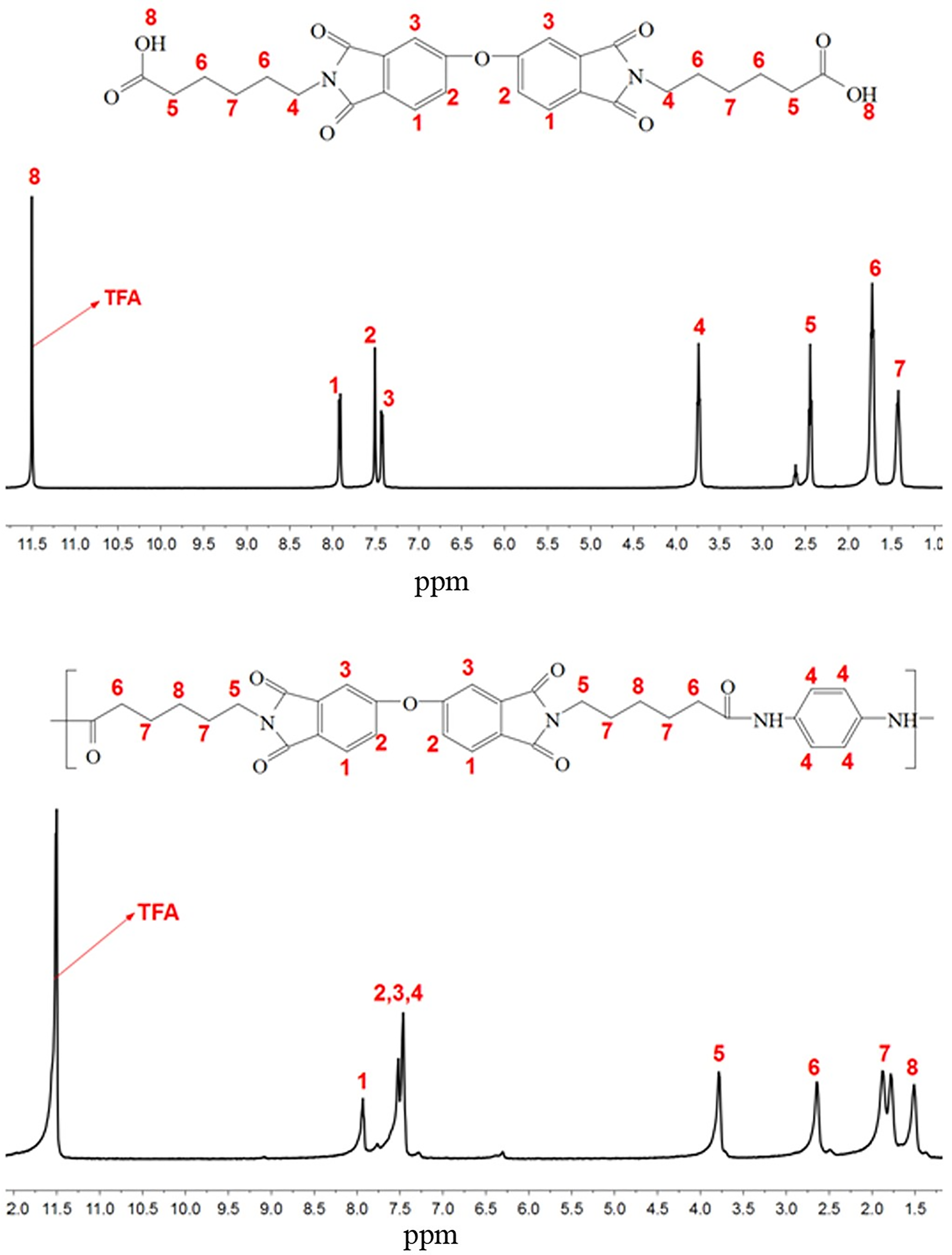
1H-NMR spectra of (a) DIDA-EACA and (b) PAI-EACA.
Solubility and molecular weight
Generally, the full aromatic PAIs have poor solubility in common organic solvents due to their rigid backbone. It is expected that the solubility of the synthesized PAIs could be improved through incorporation of biphenyl ether and methylene units into the polymer main chain, which can decrease the hydrogen bonding between the amide groups. The qualitative solubility of PAIs in various solvents is listed in Table 1, and a 10% (w/v) concentration has been taken as criteria. All the synthesized PAIs have excellent solubility in some polar aprotic solvents, such as m-cresol, TFA, and sulfuric acid at room temperature and are also soluble in hot NMP. However, no clear relationship is found between the solubility and the number of methylene units. This might be probably because the solvents which can dissolve the PAIs are polar, and the methylene unit is a nonpolar group. On the other hand, the inherent viscosities of PAIs were evaluated to be 0.88 dl g−1 for PAI-GABA, 1.18 dl g−1 for PAI-EACA, and 1.92 dl g−1 for PAI-OAUA; the relatively high inherent viscosities indicated that the molecular weights of PAIs are high enough for potential technical applications.
The solubility of poly(amide–imide)s in some common solvents.
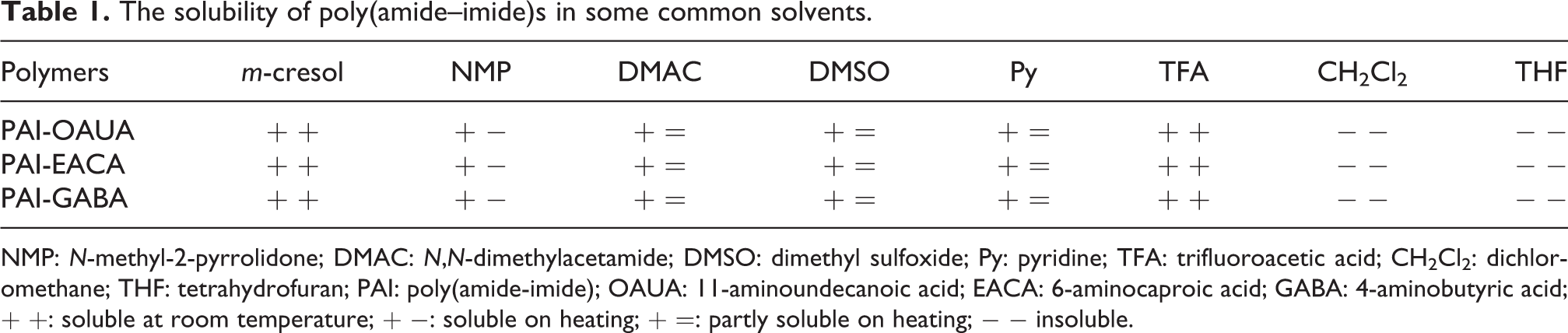
NMP: N-methyl-2-pyrrolidone; DMAC: N,N-dimethylacetamide; DMSO: dimethyl sulfoxide; Py: pyridine; TFA: trifluoroacetic acid; CH2Cl2: dichloromethane; THF: tetrahydrofuran; PAI: poly(amide-imide); OAUA: 11-aminoundecanoic acid; EACA: 6-aminocaproic acid; GABA: 4-aminobutyric acid; + +: soluble at room temperature; + −: soluble on heating; + =: partly soluble on heating; − − insoluble.
Mechanical properties
The mechanical properties of these thin films are summarized in Table 2. As can be seen, the data suggested that the PAIs could afford good quality films due to rigidity of the chain structure derived from the phenyl rings, hydrogen bonds, and symmetry of the molecular structure. Normally, with the increasing number of the methylene units in the main chain, the tensile strength of the PAIs decreased from 76.6 MPa to 55.1 MPa and Young’s modulus also decreased from 2.96 GPa to 1.85 GPa; meanwhile, the elongation at break was improved obviously, increased to 14.6% for PAI-OAUA, which was three times of PAI-GABA. In brief, the structural modification had great effects on the mechanical properties of PAIs.
The mechanical properties of poly(amide–imide) thin films.

PAI: poly(amide–imide); OAUA: 11-aminoundecanoic acid; EACA: 6-aminocaproic acid; GABA: 4-aminobutyric acid.
Thermal properties
The thermal properties of the PAIs were determined byDSC and thermogravimetric analysis. The thermal behavior data of PAIs are summarized in Table 3. DSC experiments were conducted at a heating rate of 20°C min−1 under nitrogen atmosphere as shown in Figure 5. Rapid cooling from 300°C to room temperature gave rise to amorphous samples, so the glass transition temperatures (Tgs) of all the PAIs could be easily recorded in the subsequent heating DSC traces. It is found that the Tg of PAIs decreased obviously with increasing the length of the flexible chains, which could increase the flexibility of the polymer backbone and chain mobility. As expected, PAI-OAUA revealed the lowest Tg value of 112°C due to the longest flexible chain in the polymer backbone, and the Tg values for PAI-GABA and PAI-EACA were 207°C and 151°C, respectively. Encouragingly, as shown in Figure 6, all the PAIs displayed single-stage degradation process, and high starting decomposition temperatures (T5%) around 400°C, which indicated that these PAIs had fairly good thermal stability regardless of the introduction of flexible groups in the polymer backbone. Interestingly, the thermal stability of PAIs was strengthened slightly rather than weakened with increasing the number of methylene units; this is probably because the PAIs with longer flexible chain possess higher molecular weight.
The thermal properties of poly(amide–imide)s.

T5: temperature at 5% weight loss; T10: temperature at 10% weight loss; Tmax: temperature at the maximum degradation; PAI: poly(amide–imide); OAUA: 11-aminoundecanoic acid; EACA: 6-aminocaproic acid; GABA: 4-aminobutyric acid; LOI: limiting oxygen index.

DSC spectra of poly(amide–imide)s with a heating rate of 20oC min−1 under nitrogen atmosphere.

(a) TGA and (b) DTA curves of poly(amide–imide)s under nitrogen atmosphere.
The char yield can be considered as a key factor for estimating the limiting oxygen index (LOI) of polymers according to the equation of van Krevelen and Hoftyzer: 28

where CR is the char yield. The LOI values of PAIs decreased due to the lower relative contents of benzene moiety when the number of flexible methylene units increased, and the LOI values of PAIs were calculated to be 41.9 for PAI-GABA, 35.9 for PAI-EACA, and 26.9 for PAI-OAUA. In other words, all the synthesized PAIs can be classified as self-extinguishing polymers.
Morphology
To investigate the morphology of PAIs, the DSC experiment was conducted at a slower heating rate of 10°C min−1 under nitrogen atmosphere. As shown in Figure 7, an apparent endothermic peak at 225°C is observed for PAI-OAUA, which indicates that PAI-OAUA is a semicrystalline material. 29 On the other hand, the other two PAIs (PAI-GABA and PAI-EACA) display no significant endothermic peaks, suggesting that PAI-GABA and PAI-EACA are amorphous. These results are further proved by the WAXD experiments. As illustrated in Figure 8, there is a broad halo pattern in the WAXD spectra of PAI-GABA and PAI-EACA, which demonstrates that the two polymers are amorphous. 30 , 31 Compared with PAI-GABA and PAI-EACA, PAI-OAUA exhibits an additional sharp peak at 5.7°, which is characteristic of semicrystalline material. These demonstrate that the molecular chain of PAI-GABA and PAI-EACA arranged irregularly, and the shorter flexible chain ((CH2)3 or (CH2)5) produced a steric hindrance and prevented close chain packing. With increasing the number of methylene units, the flexible chain of PAI-OAUA could offer enough room for PAI to rearrange regularly.

DSC spectra of poly(amide–imide)s with a heating rate of 10oC min−1 under nitrogen atmosphere.
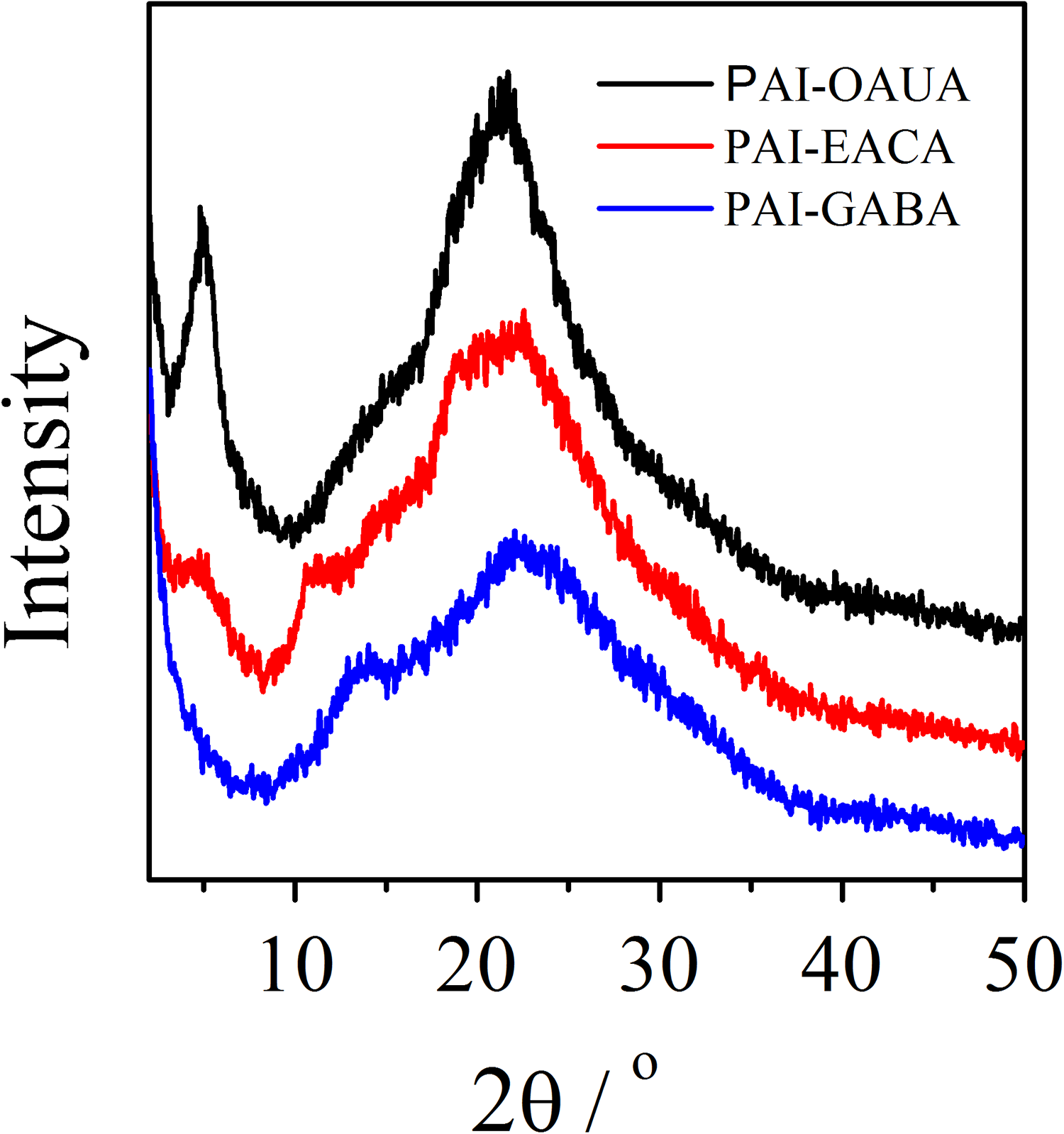
WAXD curves of of poly(amide–imide)s.
Conclusion
We have demonstrated that the structural modification plays a significant role in the properties ofPAIs. With increasing the length of the flexible chain in the backbone, a pronounced decrease in the Tg from 207°C to 112°C is accompanied by a slight reduction of tensile strength; meanwhile, elongation at break was improved obviously. The well-established correlation between the structural modification and the rigidity of the polymer chain is responsible for the changed properties ofPAIs. We believe that this finding is generally applicable and could be used to guide further development of novel PAIs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by the Natural Science Foundation of Jiangsu Province (BK20160280) and Double Plan of Jiangsu Province, Science and Technology Project of Changzhou University (ZMF15020116).
