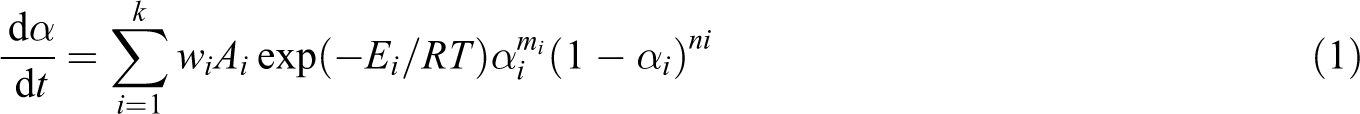Abstract
In this research, a method is proposed for optimising cure cycle of neat unsaturated polyester (UP) resins, and UP resins containing Cloisite 20A (UP/20A), in thin parts. Using the proposed optimisation method, optimised cure cycles for both neat UP resin system and UP/20A were calculated. Results of dynamic mechanical analysis tests show that samples cured by the optimised cure cycles are slightly different from those samples cured using a long-term cure cycle in terms of structural relaxation. This confirms that the proposed optimisation method not only reduces the cure cycle duration efficiently but also preserves the quality of the samples.
Introduction
Polymeric nanocomposites have attracted considerable interest recently. Nanoparticles cause special characteristics in various polymeric systems including unsaturated polyester (UP) resins. During this decade, numerous studies have been carried out on various characteristics of nanocomposites including their physical and chemical features, 1,2 gas barrier properties, 3,4 flame retardancy 5,6 and shrinkage control. 7,8
One of the research areas of interest is the effect of nanoparticles on the cure behaviour of UP resins. It has been demonstrated in various studies that nanoparticles, depending on their functional groups, have substantial effects on cure behaviour of UP resins. 9–12 These effects cause considerable changes in the curing rate of these resins.
In UP resins containing alkyd chains and styrene monomers, the styrene monomers sit between the alkyd chains and cause the formation of a network. 10 In this process, UP resins containing promoters exhibit a double peak in dynamic DSC tests, while resins without promoters have a single peak in this test. Xu and Lee studied the effects of nanoclay on the kinetics of reaction of UP resins under isothermal conditions at low temperatures. 13 They showed that nanoclay greatly affects the initiation stage of the curing reaction of UP resins. In another study, it was stated that nanoclays reduce the activation energy of the reaction through forming linkages with cobalt naphthenate (CoN) and, eventually, increase the reaction rate. 10 It is worth mentioning that the behaviour of resins containing nanoparticles changes during a curing process. This change in the behaviour results from increases in the temperature during the cure cycle that is caused by higher reaction rates. Therefore, new methods must be developed for curing parts in autoclaves.
Autoclave curing is a vital process in manufacturing of composites. In this process, the autoclave performs the thermal cure cycle based on a scheduled programme. Conventionally, thermal cure cycles are determined based on trial and error and through making an initial guess. In the current decade, researchers have made considerable efforts to optimise cure cycles using various methods. These optimisation techniques aim at minimising the duration of the curing process while preserving the quality of the manufactured products.
Li and colleagues used cure cycle optimisation methods to reduce duration of curing cycles up to 50% compared to the common curing processes. 14,15 Oh and Lee proposed an optimised cure cycle with heating and cooling stages to reduce thermal overshoot in epoxy/glass composites. 16 Kennedy and Hansen introduced a new semi-analytic gradient evaluation method called hybrid adjoint method to estimate temperature changes in autoclaves. 17 Their results indicated major improvements in the failure loads. Dmitriev et al. developed a mathematical model and studied methods for calculating optimised cure cycles for large size products of various thicknesses. 18 They showed that duration of the cure cycles increases considerably with increases in the thicknesses of the products.
Although many researchers have studied optimisation of cure cycles of thin and thick parts in autoclaves, yet effects of nanoparticles on optimised cure cycles for UP resins have not been investigated. The present research studies effects of nanoparticles on the optimised cure cycles for thin-part UP resins. The model used in this study considers two independent reactions in curing process of UP resins. This curing system employs CoN as the promoter and peroxide as the initiator. In this study, optimised cure cycles were firstly predicted for samples with small thicknesses. Then these samples were cured by applying the optimised cure cycle and subjected to dynamic mechanical analysis (DMA) test. Finally, networks formed in these samples were studied using segmental relaxation.
Thermal cure cycle modelling and optimisation
In this research, we intend to introduce an optimised cure cycle that could produce nanocomposite parts with a desirable quality in the shortest possible time. On the one hand, the quality of a part depends on the degree of conversion and the maximum temperature of the part during the curing process. On the other hand, to achieve a desirable degree of conversion, while the maximum temperature of a part is limited, a longer cure cycle is needed. Therefore, we are faced with an optimisation problem. The purpose of this optimisation problem is to reduce the duration of the cure cycle and simultaneously to increase the degree of conversion of the cure process and to reduce the maximum temperature of the part during the curing process.
The first step in optimising the curing process of composite parts is to model their curing process. The kinetic parameters of the curing reaction for UP resin and UP/20A are presented in reference. 11 Equation (1) shows the curing rate in the mentioned system 19
In equation (1), α is the degree of conversion for the curing process, wi is the ratio of the heat produced by the ith reaction to the total generated heat, Ai is the pre-exponential factor, Ei is the activation energy, R is the gas constant, T is the temperature, mi and ni are orders of the reactions and k is an integer determined based on the number of peaks observed in the DSC curve of the cured part. Assuming the composite part being cured in the autoclave is sufficiently thin, temperature distribution at various points of the part is expected to be uniform. Therefore, we can obtain the degree of conversion for the curing process at any point of time by using the equation of curing rate (equation (1)), provided that we have the time–temperature profile of the part being cured in the autoclave. Moreover, the maximum temperature of a part during the cure cycle can be obtained from the time–temperature profile. Therefore, having the time–temperature profiles of several cure cycles, the degrees of conversion and the maximum temperatures of the cured parts can be calculated and compared together to determine the best cure cycle.
Similar to any other optimisation problem, a vector of design variables (V), constraints (C) and an objective function (OF) must be determined to formulate the optimisation of the cure cycle. Design variables are parameters that the designer can adjust to obtain an optimum solution. Constraints are conditions that a sample solution must satisfy to be a practical and feasible solution, and the OF provides a suitable quantitative criterion for comparing merit of various sample solutions.
Figure 1 shows a typical five-stage cure cycle in which parameters such as Tultimate, t1, t2, t3, t4 and t5 are design variables that the designer can adjust to obtain a better cure cycle. Tultimate is the maximum temperature a sample reaches in the cure cycle, and t1 to t5 indicate the end times of phase 1 to phase 5 of the cure cycle, respectively.
A typical five-stage cure cycle.
Optimisation of the cure cycle is subject to constraints including Tmax, Tinit and Troom (see Figure 1). Tmax is the maximum temperature that can be applied to a composite part being cured, according to the quality considerations. Tinit and Troom are the temperature of the composite part at the start and at the end of the cure cycle, respectively. Other constraints are the maximum heating rate (HRmax) and the minimum cooling rate (CRmin) of the autoclave. These constraints limit the slope of various sections of the cure cycle diagram (m1, m2, m3) in Figure 1. Another constraint is Tlimit that is the maximum temperature that with regard to safety considerations can be applied to a part being cured, before the curing process reaches a conversion degree equal to alphalimit. This point is discussed in more detail in the ‘Results and discussion’ section.
As mentioned above, the purpose of the cure cycle optimisation problem is to produce a composite part with a desirable quality in the shortest possible time. Therefore, an OF must be defined in a way that it considers both the quality of the cured part and the cure cycle duration, to be able to evaluate cure cycles properly. Equation (2) shows an OF that considers these two factors
In this equation, Tultimate is the maximum temperature of a sample during the curing cycle, and Tmax is the maximum temperature a sample can be subjected to without any reduction in its quality. The final degree of conversion of a product at the end of the cure cycle is represented by (alphaultimate), and alphamin is the minimum acceptable degree of conversion that is necessary to preserve quality of a product. In this equation, tultimate is the length of the cure cycle, and tmax is the time required to cure a part at temperature Tmax until a degree of conversion equal to alphamin is reached. Furthermore, wT, walpha and wt are weight factors that represent the degree of importance of the three contributing parameters determining the merit of a sample cure cycle. These three parameters are the maximum temperature of the cured product (Tultimate), its final conversion degree (alphaultimate), along with the cure cycle duration (tultimate).
After determining the design parameters, the constraints and a suitable OF, the optimisation of the cure cycle can be formulated. Pseudocode 1 shows this formulation.

Formulation of the cure cycle optimisation problem.
After formulating an optimisation problem, an appropriate method must be used to solve it. Random search method and grid search method are two common techniques employed in solving optimisation problems. 20 -23 In this research, we propose that instead of randomly or exhaustively sweeping parameters space, as is performed in random search and grid search methods, only those parts of the parameters space that satisfy the constraints of the problem be searched. In other words, in this method all sample solutions satisfy problem constraints. This way, redundant calculations on inappropriate sample solutions are removed and the simulation is accelerated. We call this method directed grid search (DGS) method.
Pseudocode 2 shows our proposed cure cycle optimisation method. In this pseudocode, initialisation of the variables is performed in lines 1–3. Lines 4–13 indicate an iteration loop in which the main optimisation task takes place. This loop is repeated until a cure cycle with an OF less than a desired OF (OFdesired) is produced or until the loop is iterated a desired number of times (n). In this loop, at first, using the proposed DGS method and considering the constraints C1, C2, and C3 (which are explained in Pseudocode 1), a sample cure cycle is generated (line 7). Then, OF for the sample cure cycle is calculated (line 9). If the calculated OF for the sample cure cycle (OFcur) is smaller than the minimum OF calculated so far (OFmin), the sample cure cycle is considered as the best cure cycle found to that moment (Voptimal; lines 10–13). Finally, after the loop is repeated the suitable number of times, Voptimal is reported as an optimal cure cycle (line 14).

Proposed cure cycle optimisation method.
Experimental
The UP resin (FARAPOL 101) used in this work was supplied by Farapayeh Chemical Industry (Iran). FARAPOL 101 is an ortho resin with the average viscosity of 380 cP at 25°C. Cloisite® 20A (20A) is a natural montmorillonite modified with dimethyl dehydrogenated tallow quaternary ammonium chloride. 20A is a modified clay from Southern Clay. The initiator was MEKP (50% in phthalate plasticiser) containing 9.9% active oxygen supplied by Pamokale Co. (Iran). The promoter used was CoN (10% in white spirit from Chekad Co., Iran). Hydroquinone (HQ; Merck, Germany) was used as an inhibitor. All samples contain 1.5 wt% MEKP, 0.4 wt% CoN and 300 ppm HQ. HQ was added to the samples to allow an efficient mixing of MEKP with the UP mixture.
A five-step procedure was used to prepare the mixture of the UP and 20A. A presuspension-containing 10 wt% 20A was prepared and diluted by adding neat UP resin up to 1 wt%. The diluted suspensions were subjected to high shear mixing at velocity of 5000 r min−1 using a homogeniser, equipped with a powerful dispersing aggregate, at 55°C for 12 h. A radial flow stirrer was installed next to the dispersing aggregate to make the suspension homogeneous and to prevent formation of vortexes near to the dispersing aggregate. More information on this mixing method can be found in Poorabdollah et al. 12
A sheet with a thickness of about 2.5 mm was prepared by casting resin in a Teflon mould. DMA samples were prepared by cutting this sheet into 30 mm × 7.5 mm × 1 mm parts. Small thickness of a sample guarantees that all regions of the sample would have a uniform temperature. In our experiments, we employ a long-term cure cycle. In the long-term cure cycle, a sample is first kept at 25°C for 24 h, and then it is placed in an oven at 120°C for 2 h.
Samples were cured by use of NUVE EV018 oven (NUVE, Turkey). Dynamic mechanical thermal properties of all UP and UP/20A composites were measured using a dynamic mechanical thermal analyzer (DMA-Triton) with a three-point bend fixture at the condition of 1 Hz, 0.05 mm displacement and −30 to 200°C range. The DMA specimens (30 mm long, 7.5 mm wide and 2–2.7 mm thick) were cut from the composite samples.
Results and discussion
In this research, the minimum acceptable degree of conversion (alphamin) for a cured part is supposed to be 0.95. The maximum heating rate (HRmax) is 5°C min−1, and the minimum cooling rate (CRmin) is −5°C min−1. The initial temperature of the autoclave (Tmin) and the room temperature are 25°C.
As mentioned, there are some constraints in the cure process optimisation problem. The first constraint is the flash point of styrene (which is 32°C). Heating free styrene to exceed its flash point is dangerous because it can produce a substantial quantity of vapour that is harmful for the health of the operators and, additionally, the heated styrene may cause fire. Moreover, the rate of styrene evaporation at this temperature is very high and, therefore, if the temperature exceeds the flash point before the sample turns into gel, the amount of styrene in the boundary layers decreases compared to the inner layers. Since styrene plays the role of a cross-linker in UP resins, the difference in the quantities of styrene in the various layers of the sample causes heterogeneity in the final product. Therefore, the temperature of the sample must be kept below Tlimit = 32°C before it reaches the gel point. The gel point for UP resin is when the degree of conversion reaches about alphalimit = 5%. This constraint is formulated as C3 in Pseudocode 1 and induces phases 1 and 2 in the cure cycle proposed in Figure 1.
Based on the previous research, the suitable post cure temperature for UP is 120°C. 8,12 A sample can be kept safely in this temperature for 2 h to form a homogeneous network through the sample and produce a high-quality product. Therefore, another constraint for the cure cycle optimisation problem is the post cure temperature of 120°C. Therefore, Tmax is adjusted to 120°C to preserve quality of the product. Considering the discussed constraints, the proposed optimised cure cycle consists of five stages and its general form is shown in Figure 1.
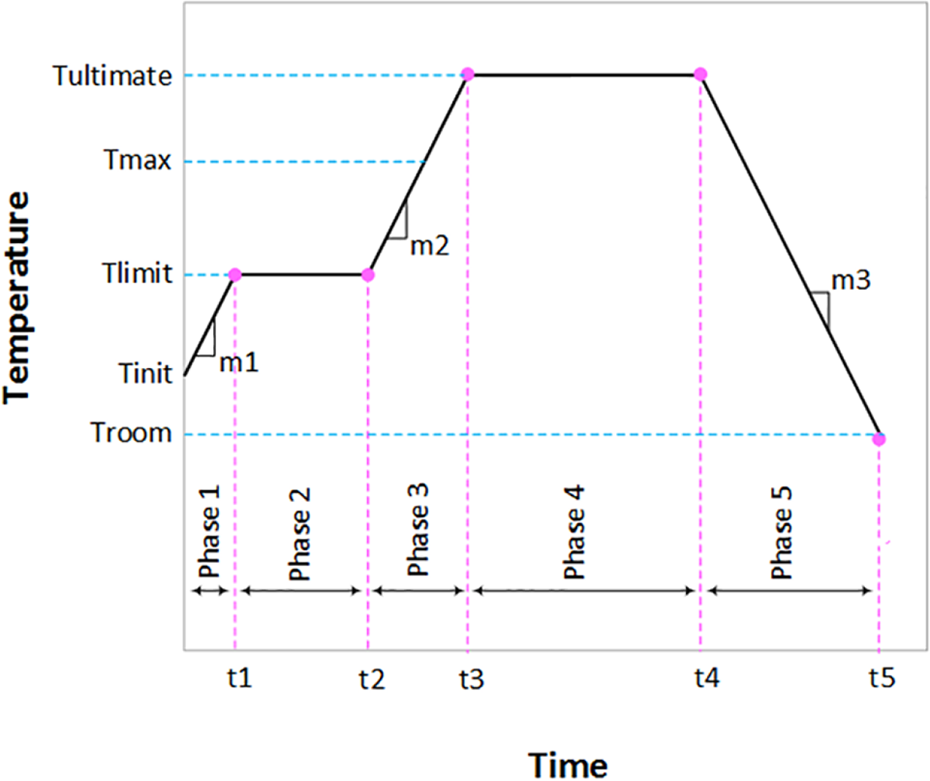
Figure 2 presents a cure cycle for UP resin which is optimised by use of our proposed cure cycle optimisation method. As shown, the sample stays for a considerable length of time at Tlimit = 32°C, but application of this constraint is unavoidable. The maximum temperature of the sample (Tultimate) in the cure cycle of Figure 2 is about 143°C.

Optimised cure cycle for neat UP resin. UP: unsaturated polyester.
It has been reported in previous research studies that nanoparticles increase the rate of reaction in UP resins through reducing the activation energy in the redox region. 10,11 Figure 3 shows an optimal cure cycle for UP/20A. Comparison of Figures 2 and 3 indicates that duration of the first isotherm region (phase 2 in Figures 2 and 3) declines from 943 s to 671 s due to the presence of nanoparticles. Temperature during the second isotherm region reduces from 143°C for neat UP resin to 133°C for UP/20A. Moreover, the residence time in the second isotherm region (phase 4) for UP/20A is shorter than that for the neat UP resin.
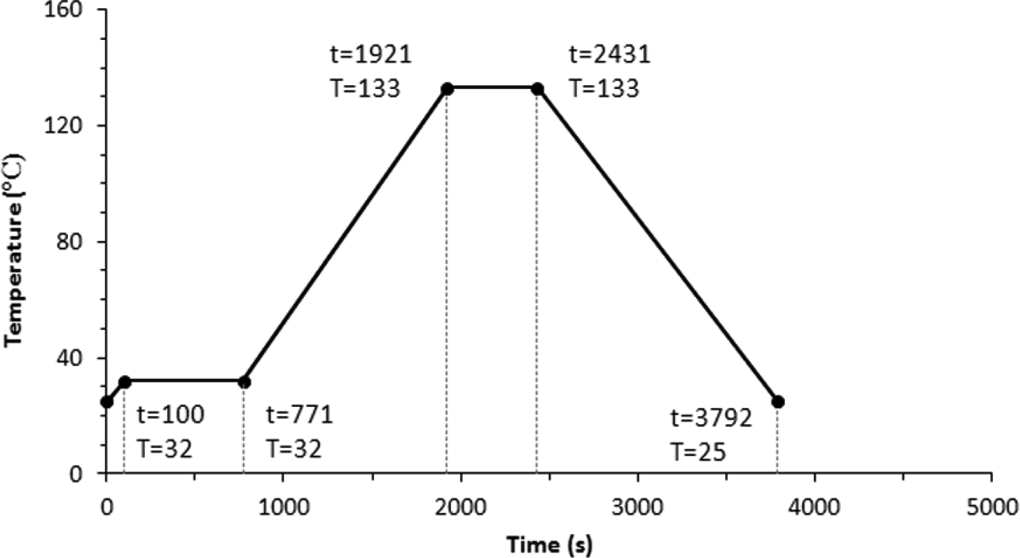
Optimised cure cycle for UP/20A resin. UP: unsaturated polyester.
To better justify the effectiveness of our proposed cure cycle optimisation method, we should study the characteristics of the networks formed in the composite parts after they are cured by the optimal cure cycles and compare them with the parts produced using the long-term cure cycles. In this direction, we conducted several DMA tests. DMA samples were produced by casting resin in Teflon moulds with a small thickness of about 2.5 mm. As mentioned, the large surface area of the samples compared to their thickness guarantees the uniformity of temperature throughout the samples.
Figure 4 presents storage modulus curves (G′) obtained from DMA tests for neat UP resin and UP/20A that are cured by applying a long-term or an optimal cure cycle. As shown in Figure 4, by use of the optimal cure cycle, the storage modulus of neat UP resin in the glassy region slightly decreases, but this reduction is negligible. This confirms the efficiency of the proposed cure cycle optimisation method. Two reasons can be offered for the slight reduction in storage modulus in the glassy region. The first reason is that the final degree of conversion for the optimal cure cycle is smaller than the final degree of conversion in the long-term cure cycle. This can cause a slight reduction in the storage modulus of the system with reducing the degree of networking. The second reason may be the greater network heterogeneity in the part cured by the optimal cure cycle. In the long-term cure cycle, the extent of migration of styrene to the surface is negligible due to the application of low temperatures in a wide range of time. Therefore, the network becomes more homogenised due to the slow curing. A more homogenised network causes a greater consistency along the cooperative rearrangement region (CRR), leading to a more uniform reaction in the segmental relaxation of these regions. 24 Moreover, the number of short length segments in the long-term cure cycle is less than that in an optimal cure cycle. The response of the regions with short CRR lengths to the DMA tests will result in a lower storage modulus in the glassy region. However, for further investigation of these regions, loss modulus diagrams (G″) must be studied.
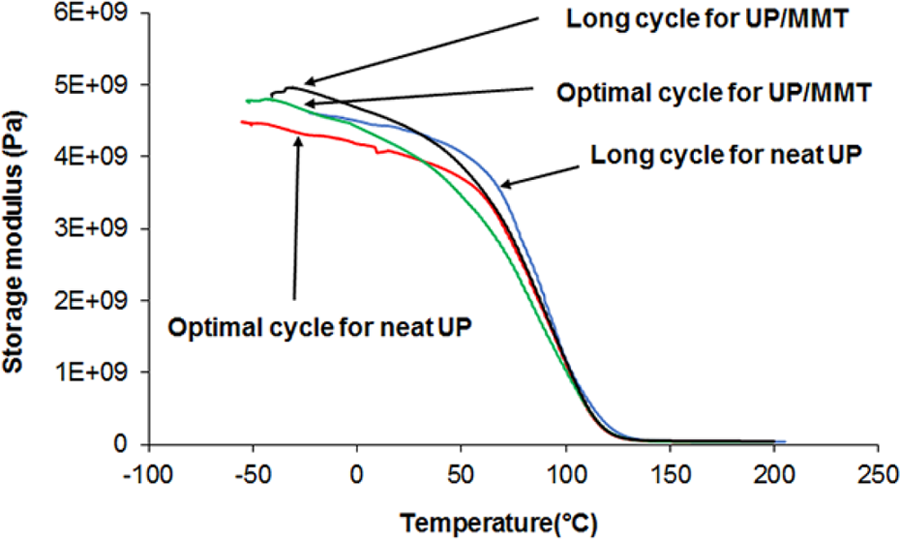
Storage modulus of neat UP and UP/20A in terms of temperature in optimal and long-term cure cycles. UP: unsaturated polyester.
Figure 5 shows curves of tan δ for neat UP system obtained by DMA tests in the long-term and also in the optimal cure cycles. Figure 6 shows these curves for UP/20A. Results indicate that in both systems, application of an optimal cure cycle instead of the long-term cure cycle will not cause tangible changes in the width and the location of the peaks of the curves.
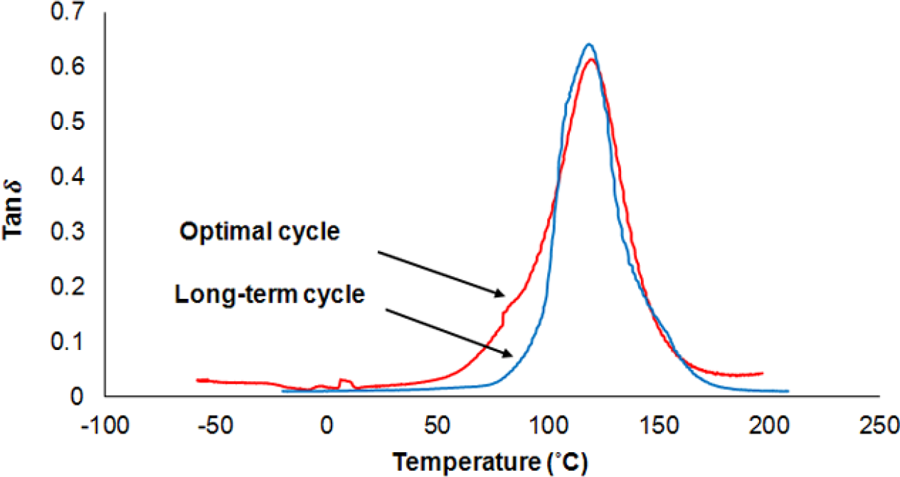
Tan δ versus temperature for neat UP in optimal and long-term cure cycles. UP: unsaturated polyester.
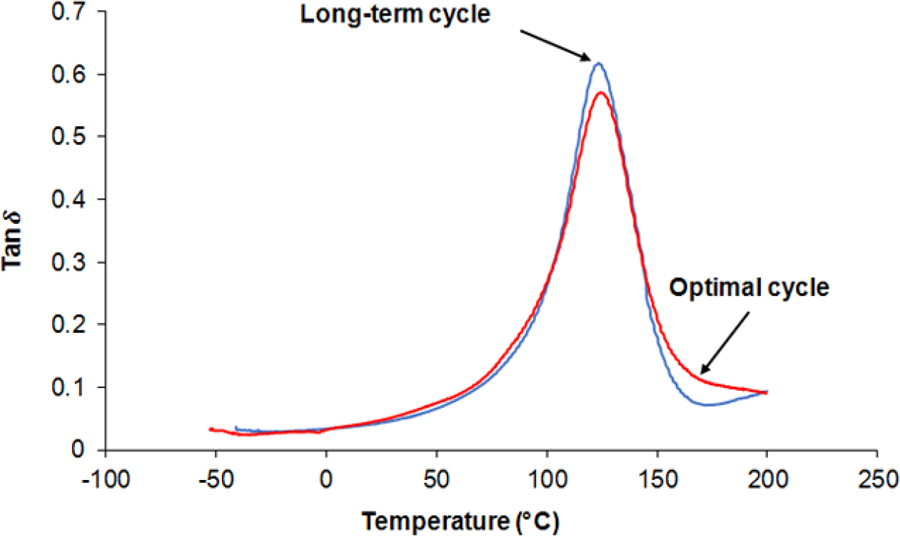
Tan δ versus temperature for UP/20A in optimal and long-term cure cycles. UP: unsaturated polyester.
Some researchers have suggested that it is better to study G″ diagrams to investigate segmental relaxation (that is rooted in the formed network). 25 Figure 7 compares G″ diagrams of the neat UP resin for the long-term and the optimal cure cycles. It is worth mentioning that the location and the intensity of the G″ peaks indicate segmental relaxation. 25 As shown in Figure 7, application of the optimal cure cycle instead of the long-term cure cycle does not considerably change the location and the intensity of G″ peaks, confirming the effectiveness of the proposed cure cycle optimisation method.
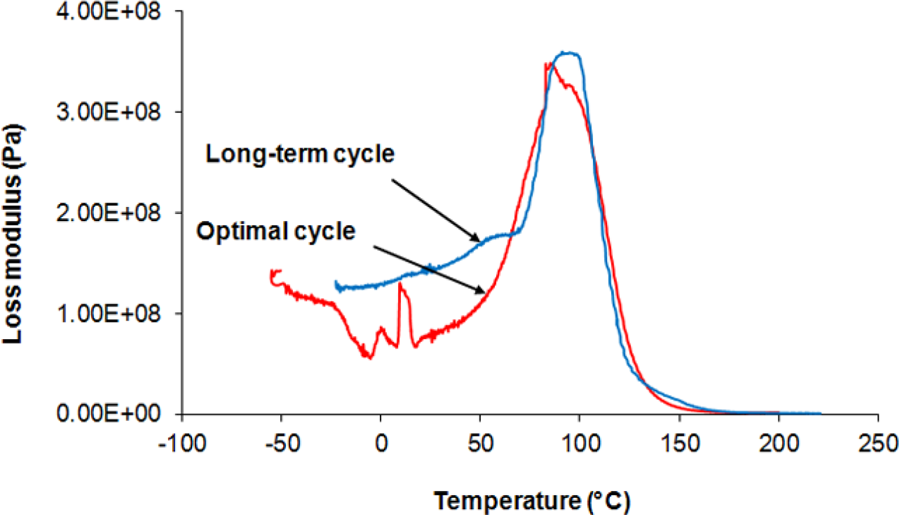
Loss modulus versus temperature for neat UP resin in optimal and long-term cure cycles. UP: unsaturated polyester.
To investigate the effect of nanoparticles on the UP resin system, we compare G′ curves of the neat UP resin with the G′ curves of UP/20A. Comparison of the G′ diagrams reveals that the storage modulus of UP/20A is somewhat larger than that of the neat resin (see Figure 4). This could be related to interactions between the formed network and the nanoparticles. However, a closer look at the mentioned diagrams indicates that the glass transition region in UP/20A is wider. To investigate the reasons of this widening and its extent, G″ diagrams must be studied.
Figures 5 and 6 show tan δ for the neat UP system and tan δ for UP/20A, respectively. As shown, addition of nanoparticles causes changes in these curves due to its effect on G″ and G′. However, as previously mentioned, researchers have suggested that it is better to use G″ curves to study segmental relaxation. Comparison of the G″ curves of the neat UP system with the G″ curves of UP/20A reveals considerable changes in the shapes of these curves (see Figures 7 and 8). The reason for this can be found in the formed network. The UP resin contains alkyd chains and styrene monomers that are of different polarities. The alkyd chains contain COOH and OH groups and, hence, enjoy greater polarity compared to styrene monomers. 10,24 Therefore, the tendency to absorb styrene and alkyd chains between nanoclay platelets varies depending on the degree of polarity of the surfactant that is present between these platelets.
As mentioned before, in this research, 20A nanoparticles were used. Because of the low polarity of the surfactant between the platelets of these nanoparticles, they exhibit a greater tendency to absorb styrene between the platelets. Moreover, the length of alkyd chains and styrene molecules also influences the extent of this absorption. 10,24 Alkyd chains are much larger than styrene molecules. Hence, according to the small distance between the nanoclay platelets, a small number of the alkyd chains compared to the number of styrene monomers can penetrate between the platelets. Therefore, the polarity factor, in alignment with the size of the alkyd chains, increases the concentration of styrene between the nanoclay platelets compared to its concentration outside of the platelets, and this cause heterogeneity in the formed network. A large concentration of styrene between the platelets would even result in homopolymerisation of the styrene. 10,24 In other words, concentration of styrene outside of the nanoclay platelets in UP system containing nanoclay is less than its concentration in the neat UP system. Therefore, the length of some CRR in the UP/20A is less than that in the neat UP system due to the lower amounts of styrene. 24 This makes the G″ curves shift to lower temperatures. Furthermore, the peak region of G″ curves of UP/20A is wider than that of the neat UP system, which clearly indicates the formation of a more heterogeneous network in UP/20A.
Figure 8 compares the G″ curve of UP/20A in a long-term cure cycle with the G″ curve in an optimal cure cycle. As shown, these two curves have slight differences. Therefore, it can be concluded that the networks formed by these two cure cycles are very similar, and this confirms the effectiveness of our cure cycle optimisation method. However, a small shoulder is observed in the G″ curve of the optimal cure cycle (see Figure 8), which implies few changes in the segmental relaxation of the system in the optimal cure cycle. In other words, CRR with shorter lengths is existing in the sample cured by the optimal cure cycle. This indicates the existence of regions containing less styrene in this sample because of the formation of a heterogeneous network after application of an optimal cure cycle. However, the extent of this heterogeneity is not considerable, confirming the effectiveness of our proposed cure cycle optimisation method.

Loss modulus versus temperature for UP/20A in optimal and long-term cure cycles. UP: unsaturated polyester.
In summary, addition of nanoparticles in UP systems increases the reaction rate and decreases the optimal cure cycle duration. It may be possible to make other uses of this increase in the reaction rate. In samples containing fibre, with larger thicknesses, the temperature varies between different layers of the sample. The temperature of boundary layers is lower than that of the internal ones. This difference in the temperature causes a difference in the degree of conversion in the various layers of the sample. Moreover, shrinkage is dependent on the degree of conversion. Therefore, shrinkage will vary in the various layers of the part. Because of this difference in the shrinkage of the various layers, large residual stresses are produced between these layers, causing very small cracks in the sample that eventually decrease the mechanical properties of the part. Addition of nanoparticles to the resin impregnating the fibres of the boundary layers will bring the reaction rate and consequently the degree of conversion in these layers closer to that of the internal ones, although these layers are at lower temperatures. Consequently, another application of nanoparticles is to make the degrees of conversion in the various regions of a part more homogeneous and, hence, to improve the mechanical properties of the part.
The results of this research show that the network formed in a part after application of an optimal cure cycle is very similar to the network formed in a part after application of the long-term cure cycle, and their segmental relaxations have slight differences. This shows the effectiveness of the optimised cure cycles generated by our optimisation method.
Conclusions
In this research, we proposed an optimisation method called DGS method to optimise cure cycle of UP resin and also UP resin containing 1 wt% nanoparticles (UP/20A). Results show that the addition of nanoparticles to UP resin reduces activation energy and, hence, decreases cure cycle duration.
Comparison of the G′ diagrams for neat UP resin and for UP/20A indicates that the storage modulus of UP/20A is somewhat higher than that of neat UP resins. This can be related to the interaction between the formed network and the nanoparticles. Comparison of the storage modulus of samples cured by the optimal cure cycles with the storage modulus of the samples produced by the long-term cure cycle indicates that the storage modulus in the glassy region of the samples produced by the optimal cure cycle has decreased slightly (in both the neat UP resin system and UP/20A). This can result from the slight reduction in the final degree of conversion in the optimal cure cycle or from the heterogeneity of the network produced in the optimal cure cycle. In the long-term cure cycle, the degree of migration of styrene to the surface is negligible due to the application of low temperatures in a wide range of time, and the network becomes more homogeneous because of the slow curing process. The production of a more homogeneous network causes a greater consistency in the lengths of the CRR, which in turn causes a uniform response in the segmental relaxation of these regions.
Comparison of G″ curves of the neat UP resin system with the G″ curves of UP/20A reveals considerable changes in the shapes of these curves. The surfactant between the nanoparticle platelets has a greater tendency to absorb styrene monomers instead of alkyd chains because of its low polarity. On the other hand, alkyd chains are much larger than styrene molecules and, therefore, only a small amount of the alkyd chains compared to the styrene monomers can penetrate between the nanoparticle platelets. The low polarity of the surfactant, consistent with the large size of alkyd chains, increases the concentration of styrene between nanoparticle platelets. In the other words, the concentration of styrene between the platelets is greater than its concentration outside the platelets, and this difference causes heterogeneity in the network.
Comparison of DMA curves of the samples cured in the optimal cure cycle with those cured in the long-term cure cycle (in both the neat UP resin system and UP/20A) shows that the segmental relaxation in these parts differ slightly, confirming the similarity of the network formed in these samples. Therefore, the proposed cure cycle optimisation method not only shortens the cure cycle efficiently, but it also preserves the quality of the parts.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.