Abstract
Sulphur-vulcanised epoxidised natural rubber–polyaniline dodecylbenzenesulfonate (ENR-PAni.DBSA) blends with useful electrical conductivities (up to 10−1 S/cm) and adjustable electrostrictive behaviour were successfully produced via a thermomechanical mixing method. A reasonably high level of compatibility between ENR and PAni.DBSA was observed from both Fourier transform infrared spectroscopy and transmission electron microscopy studies. As found from both electrostrictive strain (Sz) tests, ENR-PAni.DBSA blends with 10.0–30.0 wt% of PAni.DBSA content possessed the best electrostrictive response (Sz up to 12.25%) with applied electric field strengths up to 50 V/µm. Apart from this, blends with very low and very high PAni.DBSA contents (i.e. ≤5.0 wt% and ≥40.0 wt%) had much poorer electrostrictive responses under the same level of applied electrical field, which is due to their lower dielectric properties and higher level of phase separation between ENR and PAni.DBSA.
Keywords
Introduction
In the early 1950s, Kuhn et al. 1 discovered that a film of polymer gel located between two metallic electrodes inside an electrolyte bent in opposite directions when submitted to opposed electric fields. From then on, electrostrictive polymers (ESPs) have gained much interest due to their exciting electromechanical properties. Some of these materials have exhibited very high electric field-induced strains and become potential candidates for electrical actuator, transducer and artificial muscle applications.
There are two major categories of ESP, that is, the ionic and the electronic ESPs. In ionic ESPs, the activation forces are created by the diffusion of mobile ions and require a driving voltage as low as 1–5 V. However, these materials have to be remained wet to operate. The electronic ESPs are driven by the electric field and have faster response than the ionic ones. 2 Some ESPs are good candidates for actuation, due to their large strains (>10%). 3 Polyurethane is a popular raw material used to make actuator and transducer, and this is due to its large deformation, high specific energy, and short response time. 4 Apart from this, it has recently been shown that the incorporation of nanosized fillers can greatly enhance the strains induced by an applied electric field. 4 -6 Several research works have reported that the electrostrictive abilities of a polymer depend on its dielectric permittivity and modulus. 7 -9 The dielectric permittivity is a crucial parameter because it directly influences the achievable electrical field-induced strains in actuator applications.
One of the most popular ways to raise the dielectric permittivity is by producing composites with various metals, carbon blacks or other highly conductive fillers to increase the interfacial polarisation of the composite materials. However, the geometric morphology of the phase dispersion between fillers and their polymer host matrix plays a very important role in determining the dielectric properties of the resulting composites. However, most of these conductive fillers tend to agglomerate easily when their loadings are over the optimised level. Therefore, polymer blends with conductive polymer fillers are of great interest for improving electrical properties and phase homogeneity.
Poly(vinylidene fluoride), 10 acrylic elastomers, 11 silicones 12 and polyurethanes 13 are the most common polymeric materials used to produce ESPs due to their high abilities to convert electrical energy into strain response. Pelrine et al. 14 -16 successfully developed some interesting ESPs with large electromechanical strain, fast response, high power to mass ratio, facile processability and low cost. However, all these ESPs demonstrate large actuation only at high electric fields, that is, ≥50 V/mm and consequently restricting their real applications.
Carbon powders and carbon nanotubes are reported to enhance the electrostrictive behaviour of their resulted composites. 17-18 The enhancement of electrostrictive behaviour was due to the increased dielectric property of the resulting composite in comparison with the polymer host matrix. However, a homogeneous dispersion of these fillers in the polymer host matrix is very difficult to obtain because of the strong attraction force between these filler particles, which lead to the formation of agglomerates. Agglomeration of these types of filler is a serious problem and can subsequently deteriorate the dielectric properties. According to Huang et al., 19,20 chemical grafting is another useful approach to enhance the dielectric properties of a composite. The chemical grafting of copper phthalocyanine oligomers and conductive polyaniline (PAni) oligomers to polyurethane showed increments of electrical actuation response due to the chemical bond between polymer backbone and graft that prevented agglomeration. However, some polymers (e.g. fluoropolymers) are difficult to be chemically modified due to their high chemical stability.
This work aims to produce a smart elastomeric material via a simple, practical technique, which also exhibits excellent physical and electrical field-induced electrostrictive behaviour that can be targeted for electrical actuator applications. As a result, rubber blends based on epoxidised natural rubber (ENR) with different proportions of dodecylbenzenesulfonate (DBSA) acid-doped PAni (PAni.DBSA) in the presence of hydroquinone as compatibiliser 21 were prepared in the hope fulfilling these criteria. These blends were prepared by thermomechanical mixing, that is, using an internal mixer, and they were also vulcanised via an optimised sulphur-based curing system. ENR was chosen here due to its good elastomeric properties and chemical resistance. 22 Meanwhile, PAni.DBSA was selected due to its low toxicity, good thermal stability, high electrical conductivity, good compatibility with ENR and well-established synthesis method. 23 -31
In this work, the basic tensile properties and electrical conductivities of these ENR-PAni.DBSA blends were determined. Fourier transform infrared (FT-IR) spectroscopy and morphological study using transmission electron microscopy (TEM) were conducted to assess the compatibility level of the two constituent polymers. Two efficient techniques were used to study the electrostrictive behaviour of these blends, that is, measurement of electrostrcitive strain (Sz) using (a) laser displacement sensors and (b) deflection response.
Experiment
Chemicals and raw materials
Aniline monomer (99 wt%, Sigma-Aldrich, Malaysia), ammonium persulphate (APS; 98 wt%, Sigma-Aldrich), 36.5–38 wt% HCl solution (GPR), anhydrous iron (III) chloride (FeCl3; 98 wt%, Sigma-Aldrich), 33 wt% ammonia solution (GPR, Sigma-Aldrich), 70 wt% DBSA solution in 2-propanol (GPR, Sigma-Aldrich) and methanol (GPR, Sigma-Aldrich) were used. ENR (grade ENR 50 with 48 ± 3 mole % of epoxide level, manufactured by Felda Rubber Industries, Malaysia) was used. Hydroquinone (benzene 1,4-diol) was used as received (ReagentPlus®, ≥99 wt%, Sigma-Aldrich). The conventional sulphur vulcanisation system was chosen here. Sulphur and benzothiazole disulphide (Perkacit MBTS, sourced from Bayer Malaysia Sdn. Bhd., Malaysia) were used as the curing agent and accelerator, respectively. Zinc oxide and stearic acid (both sourced from Luxchem Sdn. Bhd., Malaysia) were also added as the vulcanisation system activators. Conductive carbon grease (grade 846) was sourced from MG Chemicals. Polydimethylsiloxane (PDMS; 100 cSt) was purchased from Sigma-Aldrich as the liquid medium for the study of deflection response.
Synthesis of PAni.DBSA
Conductive emeraldine salt PAni.HCl was synthesised by oxidative chemical polymerisation based on an oxidant (APS)/monomer (aniline) initial mole ratio of 1.0. 50 mL of aniline and 125.2 g of APS were dissolved in 375 mL and 276 mL of 2 M HCl, respectively, which had been pre-cooled to 1°C. The aniline solution was placed in an ice bath with an overhead stirrer. After the addition of about 1 mL of FeCl3 solution as a catalyst, the APS solution was added gradually over a period of 20 min, with constant stirring to ensure thorough mixing. The temperature of the polymerisation mixture was maintained to be ≤2°C, while the pH was adjusted to be ≤1.0 by the addition of concentrated HCl. The mixture was left for 6 h to complete the polymerisation, and the precipitate was allowed to settle before decanting and final washing with distilled water until the filtrate was colourless. PAni emeraldine base (EB) was prepared by deprotonating the wet PAni.HCl in 33% aqueous ammonia solution (about 5000 mL) with 24 h stirring, followed by washing and reprotonation in 1 M aqueous DBSA at a molar ratio EB:DBSA of 1:1. Filtration was performed with a sintered-glass funnel of grade 3 porosity (16–40 μm). All products were washed with large quantities of distilled water until the filtrates were colourless and odourless. Final drying of the powdered filter cake was done in a vacuum oven at 50°C for 24 h. A sample of PAni.DBSA was submitted for elemental analysis and was estimated thereby to have a 42% protonation level on the basis of the S:N atomic ratio.
Preparation of ENR-Pani.DBSA blends and addition of curing system
An internal mixer (HAAKE PolyLab OS, RheoDrive 16, Germany) was first used to prepare ENR-PAni.DBSA blend masterbatches with different compositions of ENR wt%: PAni.DBSA wt% (i.e. 97.5:2.5, 95.0:5.0, 92.5:7.5, 90.0:10.0, 80.0:20.0, 70.0:30.0, 60.0:40.0 and 50.0:50.0). 5.0 pphr of zinc oxide and 2.0 pphr of stearic acid were added to each blend masterbatch. A fill factor of 0.80 for the internal mixer was chosen to perform the mixing. The starting temperature of each mixing was 120°C, and the mixer rotor speed was set at 100 r/min. Table 1 summarises the stages of each mixing.
Stages of mixing for ENR-PAni.DBSA blend masterbatch using an Internal Mixer (HAAKE PolyLab OS, RheoDrive 16, Germany)

ENR: epoxidised natural rubber; PAni: polyaniline; DBSA: dodecylbenzenesulfonate.
Then, 1.0 wt% of hydroquinone, 21 2.0 pphr of sulphur and 1.0 pphr of Perkacit MBTS were added later manually to each masterbatch on a pre-warmed 50°C two-roll mill with gap distance adjusted to approximately 3.0 mm. Each masterbatch was removed from the two-roll mill in sheet form after about 5 min of mixing.
Preparation of sulphur-vulcanised test pieces
Appropriate amounts of each ENR-PAni.DBSA blend were cut from the sheet and fed into different shapes of mould according to the testing requirement. The mould together with the blend was sent for hot pressing and curing (at temperature 150°C, pressure 60 psi and duration of curing level 90%, tc90 as determined by a Monsanto Moving Die Rheometer, MDR 2000E-see Table 2). Test pieces were also prepared from the non-PAni.DBSA added sulphur-vulcanised ENR blend for control purpose. It was not possible to prepare pure PAni.DBSA test pieces using this hot press-moulding technique due to its glassy behaviour.
Curing duration (tc90) of sulphur vulcanising agent-containing ENR and ENR-PAni.DBSA blends as determined by a rheometer.

ENR: epoxidised natural rubber; PAni: polyaniline; DBSA: dodecylbenzenesulfonate.
Measurement of tensile properties
Basic tensile properties (tensile strength, elongation at break percentage (EB %), modulus at 100% elongation (M100) and modulus at 300% elongation (M300)) tests were carried out according to standard BS ISO 37. 32 The thickness for each test piece was approximately 2 mm. Six test pieces of each sample were used to repeat each test to define the final result as the mean value of all measurements.
FTIR spectroscopy
Infrared spectra of PAni.DBSA, sulphur-vulcanised ENR and sulphur-vulcanised ENR-PAni.DBSA blends were recorded on a Nicolet Magna-IR 560 infrared spectrometer (UK). A small amount of each vulcanised sample was hot-pressed (at 150°C, 5 min) to obtain a thin (6.0 μm), transparent film before it was put onto a potassium bromide (KBr) window (as the substrate) for characterisation. Chloroform-based solution of pure PAni.DBSA (0.0167 g PAni.DBSA/mL solvent) was also prepared here. A small amount of the PAni.DBSA solution was later cast onto the KBr window to obtain a reasonably thin (approximately 6.0 μm), transparent film.
Determination of electrical conductivity
The direct current (DC) electrical conductivity values for pure PAni.DBSA, sulphur-vulcanised ENR and sulphur-vulcanised ENR-PAni.DBSA blends were calculated from electrical ‘resistance’ (voltage/current) values measured by two-probe technique. A Keithley 6517A electrometer (USA) (with built-in voltage source up to 1 kV) was used for this purpose. Hot press moulded samples (at 150°C, 0.42 MPa and 38 min) of all materials with dimensions 25 mm (length) × 25 mm (width) × 2 mm (thickness) were used in this work. All measurement setups were exactly the same as those used for cast film samples, 33 except that no microscope slides were used here since all prepared test pieces were thick enough to support themselves. Meanwhile, test piece for pure PAni.DBSA (1 cm (diameter) × 2 mm (thickness)) was prepared from powder form using a KBr press.
Electrical conductivities were calculated from equation (1) using a two-probe configuration for all samples.

where σ is the electrical conductivity, R is the mean value of apparent resistance (voltage/current ratio) for the pressed sample, L is the measured electrode spacing and A is the cross-sectional area of pressed sample between the current-carrying electrodes.
Morphological study (TEM)
All sulphur-vulcanised ENR-PAni.DBSA blends were studied with a TEM (Philips CM12 at an accelerating voltage of 80 kV, Netherlands). All observed images were captured digitally. Ultrathin samples (approximately 150 nm) were obtained through microtome technique. Samples for TEM were collected on standard copper grids.
Determination of Sz via laser displacement sensor technique
The electrostrictive strain (Sz) of a test piece was measured using two laser displacement sensors (Keyence LKG80) under the application of step DC voltage. Each blend film with carbon grease electrodes was mounted between the rigid frames with a 15 mm diameter. The blend film was mounted on a rigid circular frame, as shown in Figure 1. Incident laser beams were exposed vertically to the film surface. The details of the thickness strain measurement setup were described in the literature. 34,35 For the Sz measurement, the electrical field strength was generated by a DC power supply (Keithley 2280S). Five duplicated test pieces from each sulphur-vulcanised NBR-PAni.DBSA blend were prepared through hot press in a mould with dimensions 8 mm diameter × 1 mm thickness, respectively, to present the result as a mean value.

A schematic illustration of test piece under applied electrical field: (a) power is switched off and (b) power is switched on.
When an electric field is applied across the film thickness, a film of ESP is compressed in the longitudinal direction and spreads in the lateral planar direction, as shown in Figure 1. The Sz can be defined using equation (2). 14 -16

where t and t0 are the thickness and initial thickness of the test piece.
Determination of Sz via deflection response technique
The Sz was determined by measuring the deflection of test pieces which was carried out at room temperature (23°C) under three different levels of electrical field strength, that is, 10, 30 and 50 V/µm. (refer to Figure 2). One end of the test piece was fixed and suspended vertically in a glassware containing PDMS (viscosity 100 cSt) between two parallel copper electrodes. The DC power was supplied by a Keithley 2280S. The bending distance of a test piece was measured along the x (d) and y (l) axes at the edge of the test piece using a Scion Image Program (version 4.0.3.2). Five duplicated test pieces from each sulphur-vulcanised ENR-PAni.DBSA blend were prepared through hot press in a mould to present the result as a mean value. Each test piece has dimensions of 100 mm (length) × 5 mm (width) × 1 mm (thickness). Sz can be defined using equation (3). 36

A schematic illustration of deflection response experiment setup: (a) power is switched off and (b) power is switched on and the free-end side of the test piece bends towards the anode site, where l0 is the initial length, l is the length after bending and d is the distance of bending.

where d is distance of bending, t is thickness of test piece and l0 is initial length of test piece.
Results and discussion
Measurement of tensile properties
Results from the tensile properties test are shown in Figures 3 to 6. PAni.DBSA acted as a reinforcing agent for the blends, where both tensile strengths and EB% of blends enhanced with the PAni.DBSA proportion (up to an optimum level, i.e. 30.0 wt%). Tensile strengths and EB% started to decrease for blends with ≥40.0 wt% of PAni.DBSA (due to the increase of phase separation between rubber host and PAni.DBSA 23 -25 ). It is proposed that the phase-separated PAni.DBSA regions had started to determine the blends’ properties, making their tensile values decrease and approach those of ‘thermoplastic like’ pure PAni.DBSA. 37 Results of M100 and M300 (refer to Figures 5 and 6) are also included here to show the good elastomeric features of these blends in general.

Tensile strength (error ±0.5 MPa) of sulphur-vulcanised ENR-PAni.DBSA blends. ENR-PAni.DBSA: epoxidised natural rubber–polyaniline dodecylbenzenesulfonate.
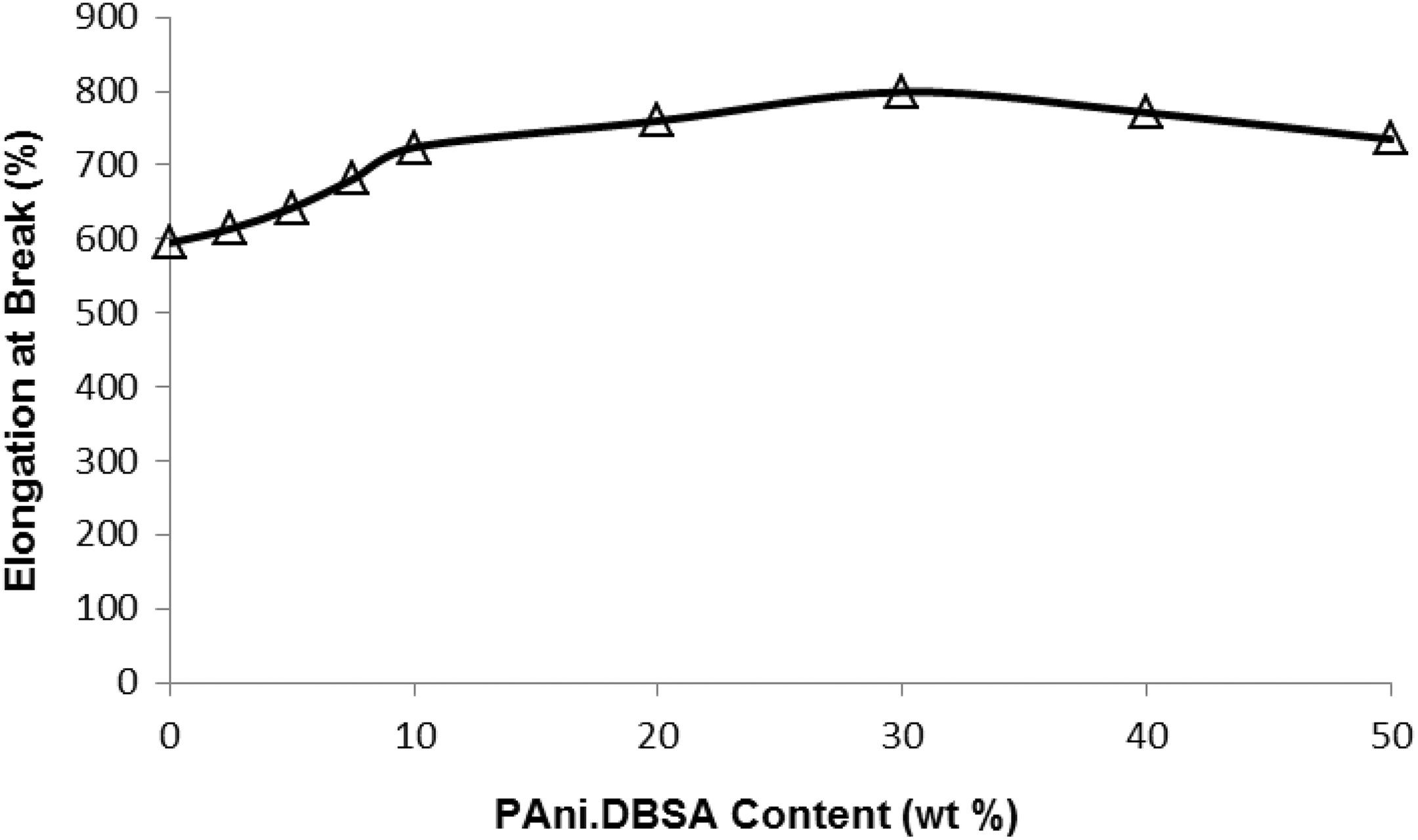
Elongation percentage at break (EB%, error ±20%) of sulphur-vulcanised ENR-PAni.DBSA blends. ENR-PAni.DBSA: epoxidised natural rubber–polyaniline dodecylbenzenesulfonate.

M100 (error ±0.5 MPa) of sulphur-vulcanised ENR-PAni.DBSA blends. ENR-PAni.DBSA: epoxidised natural rubber–polyaniline dodecylbenzenesulfonate.

M300 (error ±0.5 MPa) of sulphur-vulcanised ENR-PAni.DBSA blends. ENR-PAni.DBSA: epoxidised natural rubber–polyaniline dodecylbenzenesulfonate.
FTIR spectroscopy
Figure 7 shows some FTIR spectra examples of PAni.DBSA, sulphur-vulcanised ENR and sulphur-vulcanised ENR-PAni.DBSA blends. The FTIR spectra of sulphur-vulcanised ENR and all ENR-PAni.DBSA blends show characteristic absorptions at 1250 cm−1 (epoxy ring bond stretching), 875 cm−1 (C–C of epoxy ring stretching), 796 cm−1 and 837 cm−1 (ring deformation), 2859 cm−1 (C–H stretching vibration of epoxy ring) and 3390 cm−1 (–OH stretching vibration of ‘diol’ ring opening). The spectrum of PAni.DBSA cast film also exhibits characteristic absorptions at 3448 cm−1 (=N–H stretching), 1558 cm−1 (N=quinoid=N), 1477 cm−1 (N–benzoid–N), 1294 cm−1 (quinoid=N–benzoid), 1240 cm−1 (C aromatic-N stretching) and 1031 cm−1 (S=O stretching).
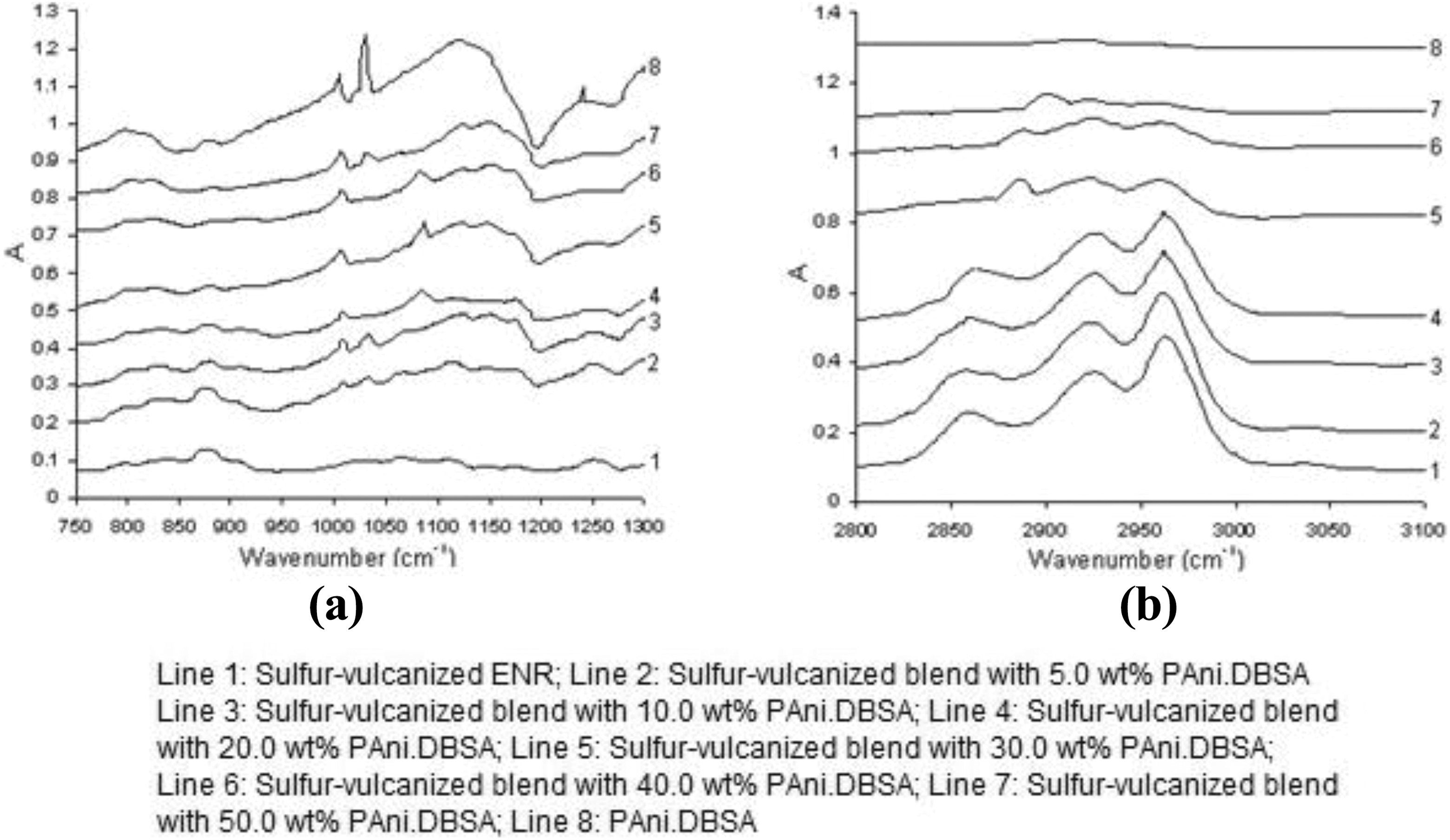
FTIR spectra of PAni.DBSA, sulphur-vulcanised ENR and sulphur-vulcanised ENR-PAni.DBSA blends: (a) 750–1300 cm−1 and (b) 2800–3100 cm−1. FTIR: Fourier transform infrared; ENR-PAni.DBSA: epoxidised natural rubber–polyaniline dodecylbenzenesulfonate.
Detailed examination of FTIR spectra of the blend films reveals significant shifts of certain key bands. The compatibility level of the ENR and PAni.DBSA can be assessed by studying some of these shifts (see Table 3). It is proposed that the larger the peak shift, the higher the intermolecular interaction of the two constituent polymers. The band at 1031 cm−1 (S=O stretching) is solely corresponded to PAni.DBSA, while that at 2859 cm−1 derives from ENR (solely corresponding to the epoxide ring). In the spectra of all blends, these bands have intensities comparable to those for the pure polymers. However, two large peak shifts can be seen, that is, an increase to 2894 cm−1 for the C–H stretching of epoxy ring and an increase to 1082 cm−1 for the S=O stretch. This phenomenon was attributable to changes in the intermolecular interactions. The peak positions only revert to those of the pure PAni.DBSA at a high level of PAni.DBSA loading, that is, ≥40.0 wt%.
Representative FTIR spectroscopy peaks for PAni.DBSA, sulphur-vulcanised ENR and sulphur-vulcanised ENR-PAni.DBSA blends.

ENR: epoxidised natural rubber; PAni: polyaniline; DBSA: dodecylbenzenesulfonate.
a wt% ENR:wt% PAni.DBSA.
b Indication of peak intensity: W = weak; M = medium.
According to literature, 23,27 the positively charged N–H hydrogens of PAni.DBSA could have a strong propensity for hydrogen bonding with the negative sulphonate oxygens, and hence the FTIR frequency changes suggest that this bonding was disrupted by polar interactions with the ENR matrix in the dilute, well-dispersed blends. The more aggregated PAni.DBSA particles in concentrated blends feature smaller specific surface areas. As a result, strong H-bonding characteristic of bulk PAni.DBSA (peak positioned at 1031 cm−1) could be detected again by the FTIR spectrometer for these blends.
Determination of electrical conductivity
Figure 8 shows the calculated mean electrical conductivities for sulphur-vulcanised ENR-PAni.DBSA blends. The sulphur-vulcanised ENR without addition of PAni.DBSA is a very good electrical insulator, with conductivity in the region of ×10−16 S/cm. The in-house synthesised PAni.DBSA showed a high electrical conductivity of 2.6 ± 0.5 S/cm.

Electrical conductivities for sulphur-vulcanised ENR-PAni.DBSA blends as a function of PAni.DBSA content (wt%). ENR-PAni.DBSA: epoxidised natural rubber–polyaniline dodecylbenzenesulfonate.
The electrical conductivities were generally enhanced with the increasing content of PAni.DBSA. The conductivity percolation threshold for sulphur-vulcanised ENR-PAni.DBSA blends was estimated by fitting data from Figure 8 to a simple percolation model as defined by equation (4) 38

where c is a constant, t is the critical exponent, f is the volume fraction of the conductive medium and fp is the volume fraction at the percolation threshold. All the weight fractions referred above were converted into volume fractions for this analysis. By fitting the data to a plot of log electrical conductivity (σ) versus log (f − fp), it was possible to estimate the values of t and correlation coefficient (R) for each set of data. The estimated electrical conductivity percolation threshold for the ENR-PAni.DBSA blends is 3.75 wt%/3.3 volume% of PAni.DBSA (with t = 3.8 and R = 0.99). A reasonably low electrical conductivity percolation threshold can be achieved for the sulphur-vulcanised ENR-PAni.DBSA blends; which was attributed to the addition of hydroquinone as a compatibiliser.
Morphological study
All sulphur-vulcanised ENR-PAni.DBSA blends were studied using a TEM, but only examples of micrographs of blends with 10 and 40 wt% PAni.DBSA are shown in Figure 9. The darker coloured regions (rich in PAni.DBSA) are large conductive particles and their agglomerates result from some degree of phase separation. The lighter coloured regions (rich in ENR) are related to well-blended PAni.DBSA and NBR. In Figure 9, the PAni.DBSA particles start to cluster or form conductive pathways, which is consistent with the enhanced conductivities of both blends.

TEM micrographs of sulphur-vulcanised ENR-PAni.DBSA blend. ENR-PAni.DBSA: epoxidised natural rubber–polyaniline dodecylbenzenesulfonate: (a) blend with 10 wt% PAni.DBSA and (b) blend with 40 wt% PAni.DBSA.
Determination of Sz via laser displacement sensor
Figure 10 shows Sz of sulphur-vulcanised ENR and sulphur-vulcanised ENR-PAni.DBSA blends as a function of applied DC electric field. The Sz of all sulphur-vulcanised ENR-PAni.DBSA blends increased with the strength of the applied electric field, which indicates that they are all actuated like the ESP actuators as shown by equation (2). 14 -16 Apart from this, the Sz values also increased with the PAni.DBSA loading up to 30.0 wt%. In Figure 10, sulphur-vulcanised ENR blends with 10.0–30.0 wt% PAni.DBSA loadings show the highest actuation (Sz) values, that is, ≥8.1% at a pretty low electrical field of 10 V/µm. This is due to their highest compatibility level between the two major constituent polymers (i.e. ENR and PAni.DBSA). Other synthetic polymer-based composites, for example, chemically-grafted poly(methyl methacrylate) and hydrogenated rubber styrene–ethylene/butylene–styrene only showed Sz of 6% and 1%, respectively at 16 V/µm. 39
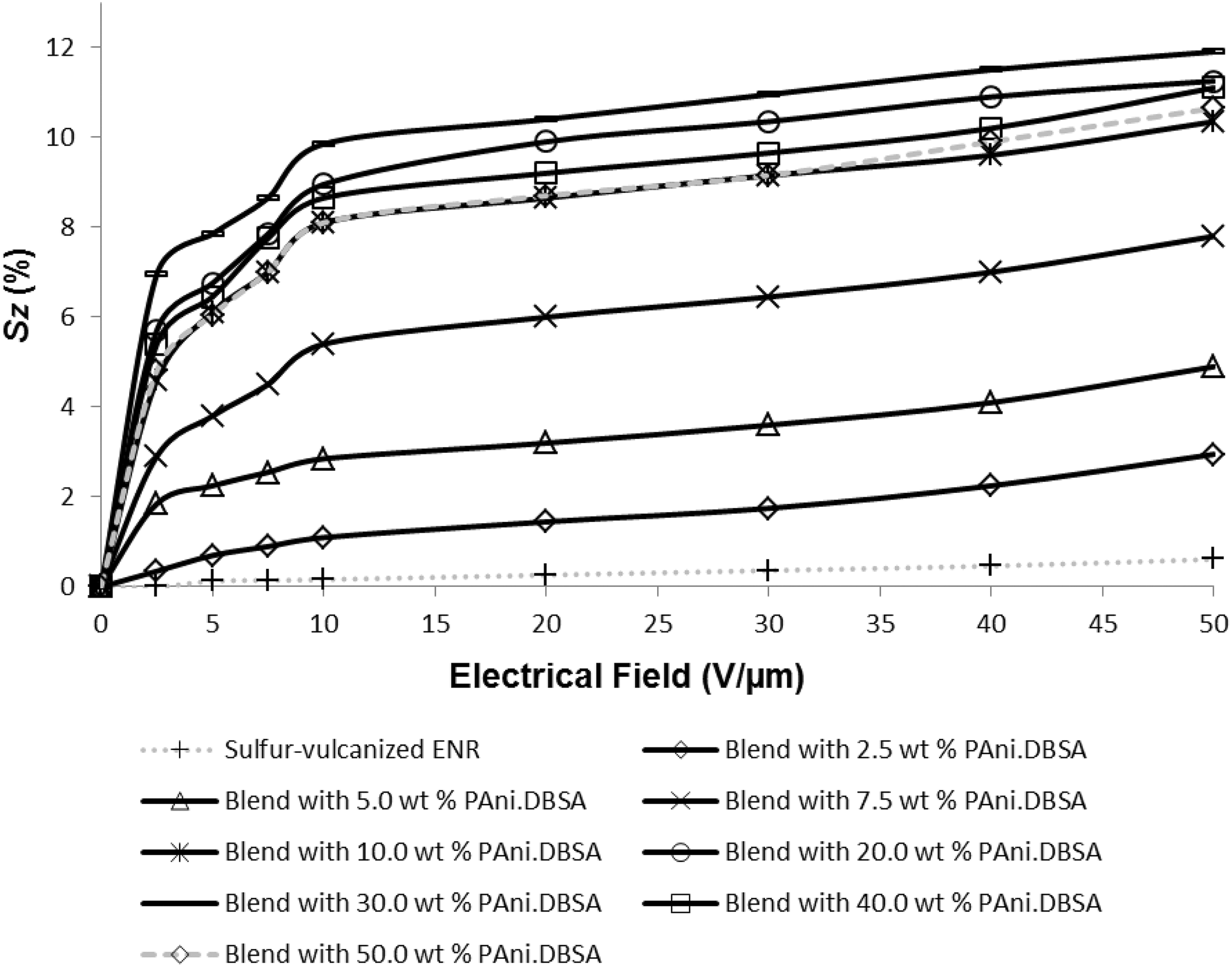
Sz (error ±0.2%) of sulphur-vulcanised ENR and sulphur-vulcanised ENR-PAni.DBSA blends as a function of the applied electrical field. ENR-PAni.DBSA: epoxidised natural rubber–polyaniline dodecylbenzenesulfonate. Sz: electrostrictive strain.
As discussed before, the geometric morphology of the phase dispersion between fillers and their polymer host matrix plays a very important role in determining the composite’s dielectric properties. It is proposed that the agglomeration of these phase-separated PAni.DBSA fillers (for blends with ≥40.0 wt % PAni.DBSA contents) can subsequently deteriorate the dielectric property of these blends. Meanwhile, sulphur-vulcanised ENR without any addition of PAni.DBSA shows the lowest Sz values also because of its having the poorest dielectric properties among all analysed samples.
Determination of Sz via deflection response technique
The deflection response of sulphur-vulcanised ENR and all sulphur-vulcanised ENR-PAni.DBSA blends were investigated at 23°C at three different levels of electric field strength, that is, 10, 30 and 50 V/µm. Figure 11 shows the Sz values of sulphur-vulcanised ENR and sulphur-vulcanised ENR-PAni.DBSA blends that were determined via the deflection response technique.

Sz values (error ±0.2%) of sulphur-vulcanised ENR-PAni.DBSA blends as a function of the PAni.DBSA content. ENR-PAni.DBSA: epoxidised natural rubber–polyaniline dodecylbenzenesulfonate. Sz: electrostrictive strain.
The Sz values of all sulphur-vulcanised blends increased with the increment of applied electrical field. From the experiment, it was found that the free end of each test piece bended toward the anode (positive charge) site under an applied electric field due to the electrostriction effect (refer to Figure 2(b)). It is believed that the chemical structure of hard PAni.DBSA tends to form some polar crystalline domains. These regions are polarised with the applied electrical field and lead to some deformations within the overall polymer matrix. An attractive force between unlike charges is created and induces the test piece to deflect towards the anode side when an electric field is applied. 34 In general, the Sz values also increased with the content of PAni.DBSA up to 30.0 wt%. The increased phase separation between ENR and PAni.DBSA for blends ≥40.0 wt% of PAni.DBSA content may deteriorate their dielectric properties. All results from here are in agreement with the ones as determined using the laser displacement sensor.
The induction time and the recovery time of each deflection response are summarised in Table 4. The induction time is the time required for a test piece to bend toward the maximum displacement under an applied electric field. The recovery time is the time for a test piece to deflect back to its original position after the electric field is switched off. All sulphur-vulcanised blends show fast deflection response and they are also comparable to other types of potential electrostrictive elastomer. 36,39 The deflection response time is also decreased with the PAni.DBSA content due to the enhancement of dielectric properties of the blend.
Comparison of response times for sulphur-vulcanised ENR-PAni.DBSA blends under the deflection response test.

ENR: epoxidised natural rubber; PAni: polyaniline; DBSA: dodecylbenzenesulfonate.
Conclusion
Sulphur-vulcanised ENR-PAni.DBSA blends with adjustable electrostrictive properties were successfully produced via the simple, practical thermomechanical mixing method. This type of blend possesses good electrical conductivities and elastomeric properties. As observed from both FT-IR spectroscopy and TEM morphological studies, a reasonably high level of compatibility between the ENR and PAni.DBSA was achieved via the introduction of 1.0 wt% of hydroquinone. Based on the results of both electrostrictive behaviour studies, ENR-PAni.DBSA blends with 10.0–30.0 wt% of PAni.DBSA loadings have the best potential for fast-response electrical actuator applications. This is attributed to their best compatibility level and lowest phase separation problem between the two major constituent polymers.
Footnotes
Acknowledgements
The author would like to thank the Malaysian Rubber Board for providing permission to publish this work. Assistance given by Mr Hassanal Baharom throughout this study is also highly appreciated.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project is fully funded by the Malaysian Rubber Board.
