Abstract
The objective of this study was to make a comparative analysis of the effect of formic and acetic acids as oxygen carriers on the epoxidation of soybean oil used with hydrogen peroxide as the oxygen donor. Comparative analysis between the use of formic acid (FA) and acetic acid (AA) was studied to obtain the most effective oxygen carrier that yielded high oxirane oxygen contents (OOC). The epoxidation reaction was carried out using a stoichiometric ratio of 1:0.5:1, 1:0.5:0.5, and 1:0.5:2 of soybean oil: formic/acetic acid: hydrogen peroxide. The synthesis was performed at three reaction times (2, 4 and 6 h) at a constant temperature of 50°C. Samples prepared using FA and AA were characterized using ASTM D1652-11 and confirmed by Fourier transform infrared (FTIR) spectroscopy. The result of this study proved FA to be an effective oxygen carrier compared to that of AA based on the high OOC and percent yield achieved. The optimum epoxidized soybean oil (ESO) sample using FA was obtained at a reaction time of 6 h using 2 moles of H2O2, yielding an OOC of 7.45 at a relative conversion to oxirane of 98%. Samples of FA were further characterized to prove the optimum parameters that gave the highest OOC using rheology and gel permeation chromatography (GPC). Rheology data revealed an increase in the viscosity that implied an increase in the degree of epoxidation. GPC indicated an increase in the molecular weight at low reaction times, then a decrease resulting in a change in the structure of the triglyceride and consequently an increase in the extent of epoxidation.
Introduction
A number of industries focus their attention on epoxy resins as an acceptable matrix (resin) system. Epoxy resins are known for their excellent mechanical and thermal properties 1 , good chemical resistance and the ability to bond well with reinforcing fibers 2 in fiber-reinforced composites. However, most resins offering excellent mechanical properties are derived from petroleum by-products; hence, they are neither ecofriendly nor bio-degradable. This provides an impetus to synthesize a bio-based epoxy resin system from vegetable oils.
The use of vegetable oils is one of the most inexpensive ways to create a resin system. Vegetable oils are sustainable, renewable, biodegradable with low toxicity, and for many of the vegetable oils, they are the most abundant biological feedstock available in large quantities 3 . The methods to obtain polymers from vegetable oils include direct polymerization through the double bonds or other reactive functional groups, chemical modification of the double bonds that introduces reactive functional groups, or chemical transformation to reduce fatty acid chains to monoglycerides 4 . Among these, chemical modification of the double bonds by way of the epoxidation mechanism is the most widely used method. This epoxidation method has demonstrated itself to be the most conventional and effective process used to produce polymers from vegetable oils, primarily because it is inexpensive and well-controlled5,6. The epoxidation method converts the double bonds in the vegetable oil to epoxide groups, thus going from a less reactive to a more reactive system because of the highly strained oxirane ring (epoxide). The presence of an epoxide group in vegetable oils makes for a good monomer for the replacement of the petrochemical diglycidyl ether of bisphenol A (DGEBA).
Epoxidation can be carried out in a number of ways that include the conventional chemical treatment; use of acid ion exchange resins (AIER), enzymes, or a metal catalyst 3 . Among all methods listed, the conventional chemical treatment method and the method using acid ion exchange resins are the most preferred ones. Both methods use carboxylic acid (formic acid or acetic acid) that acts as the oxygen carrier and hydrogen peroxide as the oxygen donor. It is important to know that hydrogen peroxide is considered to be the most promising oxidizing agent because it is cheap and pollution-free 7 . However in regard to the carboxylic acids, due to acetic acid being a weaker acid compared to formic acid, acetic acid is normally used with a catalyst which results in the acid ion-exchange resin method. This acid ion-exchange method uses an insoluble gel type catalyst such as Amberlite IR-120H 3 .
Reaction of the carboxylic acids (formic acid or acetic acid) and hydrogen peroxide together forms peracids (performic acid (PFA) or peracetic acid (PAA), respectively), that constitutes the aqueous phase of the system. The peracid further reacts with the double bonds in the oil to produce epoxide groups 8 in the oil phase. Thus, it is important to achieve a high oxirane oxygen content (OOC), which simply indicates the number of epoxide groups that will result in increased crosslinking and consequently increased properties. The ability to achieve desirable OOC requires precise optimization of the system resulting in reduction of production time and cost. However, there are a number of factors that can affect the epoxidation reaction, such as reaction time, reaction temperature, amounts of formic or acetic acid, amount of hydrogen peroxide and stirring speed9–12.
Monono et al. 13 carried out a synthesis reaction of epoxidized sucrose soyate while optimizing the reaction temperature (55, 60, and 65°C), moles of acetic acid (0.25, 0.375 and 0.5), moles of H2O2 (1, 1.5 and 2), catalyst amounts (1.5, 3.75 and 6 g), and reaction times (3.5, 4.5 and 5.5 h). A conversion rate of 95.9% from double bonds to epoxide groups was obtained when using 0.375 mol of acetic acid, 2 mol of hydrogen peroxide, and 6 g of catalyst for 4.5 h at 60°C through the American Oil Chemists Society (AOCS) method Cd 9-57. Similarly, other researchers have performed various tests to optimize the above mentioned parameters10,12,14–16. Optimized parameters varied with each study because researchers used different oils and hence had variation in fatty acid percentage. The major difference in optimizing parameters in the epoxidation of vegetable oils stems from the ability to determine the most effective oxygen carrier. Formic acid, being highly reactive, was chosen by Gupta et al. 17 and He et al. 18 , while others found acetic acid to be useful along with Amberlite IR-120H3,8,19.
Hence, this research focuses on determining the effect of varying the reaction time and molar ratios of hydrogen peroxide using both formic acid and acetic acid to synthesize an epoxidized soybean oil (ESO) system. The overall goal is to understand how the OOC changes because of these factors using two different peracids.
Experimental
Materials
Soybean oil used in this study was obtained from Sigma Aldrich in its raw form.
The fatty acids and the subsequent percent composition of each fatty acid of
soybean oil are shown in
Fatty acid percentage composition of soybean oil (Sigma Aldrich, USA)

Fatty acid percentage composition of soybean oil (Sigma Aldrich, USA)

Other materials used in this study consist of sodium bicarbonate, sodium sulfate and distilled water used for neutralization and drying of the epoxidised oil to obtain the final product. Additionally, tetraethylammonium bromide (anhydrous), perchloric acid (60 %), acetic anhydride, methylene chloride, crystal violet indicator and potassium acid phthalate were used in the titration method to determine the OOC based on ASTM D1652-11.
Epoxidation of soybean oil was carried out by placing 1 mol of soybean oil and 0.5 mol of formic acid in a three-neck flask equipped with a thermometer and magnetic stirrer. The flask containing the solution was submerged in an oil bath on top of a magnetic stirring hot plate. The mixture was stirred at 900 rpm at 50°C. Once the desired temperature was achieved, the temperature was reduced by 5°C before the required amount of hydrogen peroxide was added. This reduction in the temperature was to avoid a drastic temperature rise as a result of the exothermic nature of reaction during the addition of hydrogen peroxide. With the temperature reduced by 5°C, the required amount of hydrogen peroxide was slowly added to the mixture using a titration tube to start the epoxidation reaction. After the completion of the addition of hydrogen peroxide, the reaction was allowed to continue until the end of reaction time. This exact procedure was performed for the varied reaction times (2, 4 and 6 h) and different ratios of soybean oil to hydrogen peroxide (1:1, 1:1.5 and 1:2 mol ratio).
The reaction using acetic acid was carried out in the same manner with the
addition of Amberlite IR-120H added during the addition of oil and carboxylic
acid. Amberlite IR-120H was used in the amount of 20 wt. % of oil. A schematic
representing the process used to perform the synthesis of epoxidized soybean oil
is shown in

Schematic representing the synthesis of epoxidized soybean oil
The epoxidized oil was allowed to cool to room temperature after the completion of the reaction. The post treatment of the synthesized oil involved filtering and washing the oil several times with distilled water to remove any excess acid and separate the oil phase from the water phase. The reaction using acetic acid and Amberlite IR-120H was first filtered through a filter paper to separate the bead-like catalyst from the epoxidized oil before washing with distilled water. Sodium bicarbonate in the amount of 5 wt. % dissolved in 100 mL of distilled water was placed in a titration tube and used to neutralize the reaction to obtain a pH of 7. The epoxidized oil solution was placed in a beaker on a magnetic stirring plate to allow for constant mixing, while the solution of sodium bicarbonate was titrated into the beaker. The pH was checked regularly using pH strips. The neutralized epoxidized soybean oil was then dried over sodium sulfate in the amount of approximately 20 wt. % of the undried epoxidized oil and placed in an oven overnight at 70°C.
Oxirane oxygen content (OOC) and epoxy equivalent weight (EEW) were calculated according to ASTM D1652-11. Fourier transform infrared (FTIR) spectroscopy was used to determine the chemical bonds and change in functional groups. This test was carried out using Shimadzu FTIR 8400s setup equipped with MIRacle TM ATR. Each sample was scanned from 400 to 4000 cm−1 with a resolution of 4 cm−1. The key goal for FTIR was to verify the presence of epoxide groups as a result of varied molar concentrations of hydrogen peroxide and varied reaction times.
For FA samples, rheological studies were carried out using TA Instruments AR-2000 equipped with environmental testing chamber (ETC) and 25 mm parallel plates. Scans were performed at room temperature with varying shear rate from 0 to 300 s−1. Each test recorded the change in viscosity as a function of the shear rate in order to determine processing and performance conditions. The results for rheology were further analyzed using TA Instruments Data Analysis software. Gel permeation chromatography studies were carried out at North Dakota State University using Waters Corporation: Module system with Refractive Index detector. Tetrahydrofuran (THF) was used as the mobile phase at a flow rate of 1 mL/min, and columns were maintained at 40°C. The samples were first filtered then added to a 1.5 mL glass vial with THF at a concentration of 1 mg/mL. Astra V software was used to calculate the molecular weight.
Results
Determination of OOC using Titration method based on ASTM D1652-11
The oxirane oxygen content (OOC) is an important parameter in the characterization of epoxidized vegetable oils and is used to determine the number of oxirane groups formed in place of double bonds. Theoretically, the OOC value of the soybean oil used in this study was found to be 7.58. The theoretical OOC (OOCt) was calculated based on equations used by Cooney 20 . These equations consisted of calculating the theoretical iodine value (IVt) shown in equation (1) that is used in the theoretical OOC (OOCt) equation shown in equation (2).
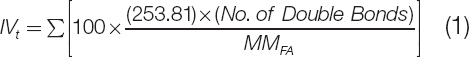

IVt was calculated based on the percent fatty acid composition of
soybean indicated in
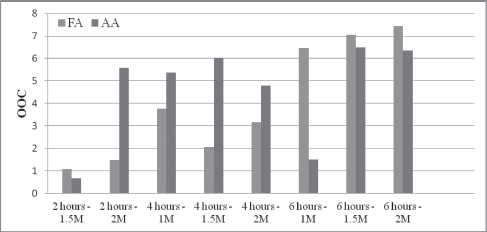
Comparison of OOC values of epoxidized soybean oil using formic and acetic acids at different reaction times and H2O2 concentration
A more stable reaction occurs by increasing the reaction time to 6 h with increasing molar concentrations of hydrogen peroxide resulting in an increase in the OOC. It is believed that more time is needed to break the double bonds in the oil and generate PFA to create higher epoxide content compared to values at 2 and 4 h. Therefore, the optimum epoxidized soybean oil using FA at 50°C was achieved when using a reaction time of 6 h at a 2 mol ratio of hydrogen peroxide. These optimum parameters yield an OOC of 7.45.
On the other hand, results from the epoxidation reaction of soybean oil using AA and Amberlite IR-120H revealed a variation in the OOC with increasing reaction time and increasing molar concentration of hydrogen peroxide. It is believed that the variation in the OOC values is affected by the concentration and use of Amberlite IR-120H catalyst. Most of the degradation occurs due specifically to the external surface of the Amberlite IR-120H catalyst that results in oxirane ring opening26,27. The beadlike structure of the catalyst acts as an adsorbent in the presence of AA and other aqueous solutions including water that is generated from the production of PAA; hence, resulting in swelling of the catalyst. Therefore, AA was found to be a less effective and stable oxygen carrier compared to FA.
On the other hand, the epoxide equivalent weight (EEW) using both FA and AA was seen to have an inverse relationship with the OOC. The EEW is defined as the weight of resin in grams, which contains 1 g of the equivalent 28 . This parameter is important in determining the amount of hardener to use when curing the resin. A high epoxide equivalent weight indicates low epoxide groups formed. Consequently, a high epoxide equivalent weight indicates less curing agent needed due to less reactivity of the epoxidized oil which indicates less functional epoxide groups. Therefore, the amount of curing agent needed for the optimum epoxidized soybean oil system (50°C for 6 h using 2 mol of hydrogen peroxide) would be close in weight to the grams of resin being used.
EEW and RCO values of epoxidized soybean oil with varied molar ratios of hydrogen peroxide and reaction time

FTIR was used to verify the formation of epoxide groups. The data obtained showed
a number of functional groups associated with the epoxidation reaction as listed
in
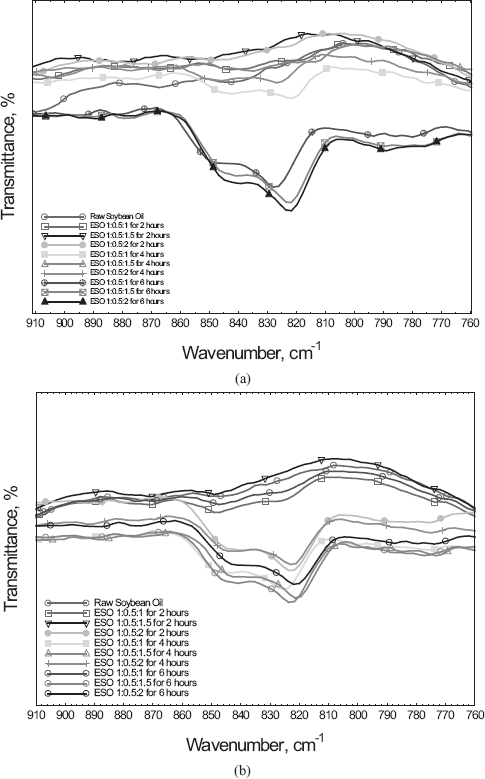
FTIR spectrum highlighting the presence of epoxide groups at 822 cm−1 using (a) formic Acid and (b) acetic Acid
FTIR functional groups and corresponding frequency peaks present in epoxidized soybean oil samples

Other peaks observed from the FTIR spectrum are 3530 cm−1 (OH
stretching), 3012 cm−1 (C-H and C=C stretching) 2920 cm−1
(C-H stretching of CH3), 2850 cm−1 (C-H stretching of
CH2), and 1738 cm−1 (C=O stretching vibration of
carbonyl ester) are listed in
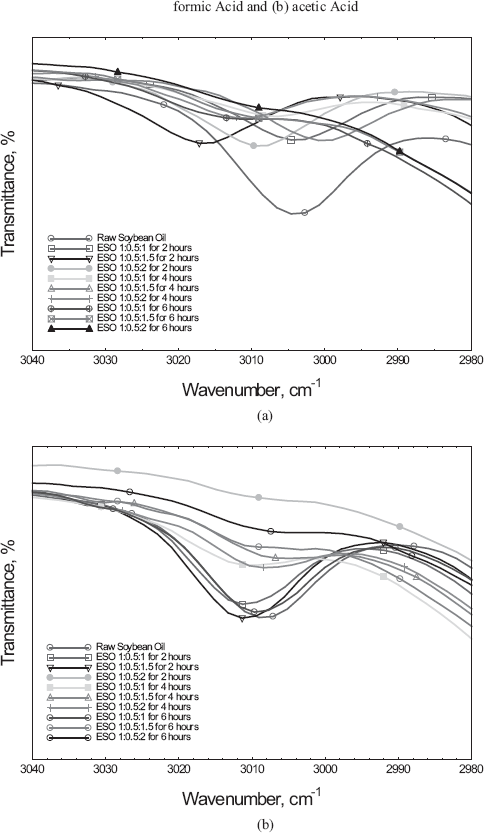
FTIR spectrum highlighting the disappearance of C=C at 3012 cm−1 using (a) formic acid and (b) acetic acid
With FA proven to be the most effective oxygen carrier compared to acetic acid,
this section focuses specifically on using this acid to verify the trend
obtained from ASTM D1652-11 and FTIR. Rheology tests were performed on the
samples at room temperature at shear rates from 0 to 300 s−1. The
results of the viscosity versus shear rate of samples synthesized using FA at
50°C with varying reaction time and molar concentration of hydrogen peroxide are
shown in

Rheology plot of epoxidation reaction using formic acid at 50°C

GPC plot of major molecular peak using formic acid at 50°C
GPC Results showing average molecular weight and PDI as a result of varied molar ratio of hydrogen peroxide and reaction time of epoxidized soybean oil using formic acid

This study focused on the effects of reaction time and molar concentration of hydrogen peroxide on the synthesis of epoxidized soybean oil using FA and AA. The system was optimized by varying the reaction times at 2, 4 and 6 h with varied molar concentrations of hydrogen peroxide at 1, 1.5 and 2 mol to yield high conversions of epoxide groups. Compositional properties to determine the amount of double bonds converted to epoxide groups (oxirane oxygen content (OOC)) was characterized using the titration method based on ASTM D1652-11. Further characterization focused on the verification of the presence of epoxide formation using Fourier-transform infrared (FTIR) spectroscopy. Based on results from ASTM D1652-11 and FTIR, further characterization was performed using rheology and gel permeation chromatography for the carboxylic acid that yielded the most effective results.
It was revealed that FA was proven to be the most effective oxygen carrier compared to AA based on the high percentage yield and OOC obtained. Samples using FA revealed complete decomposition of the reactants compared to the regeneration of Amberlite IR-120H used with AA. As a result, samples using AA resulted in varied OOC with increasing reaction times and molar concentrations of hydrogen peroxide. Low OOC resulted in the presence of side reactions such as hydrolysis, generation of glycerol and oxirane cleavage. Overall, optimum conversion to oxirane was achieved using FA at 6 h using a 2 mol ratio of hydrogen peroxide at 50°C. Based on results from ASTM D1652-11, the optimum system yielded an OOC of 7.45 and a RCO of 98%. This result was verified using Fourier-transform infrared (FTIR) spectroscopy shown at frequencies of 822 cm−1. Rheology and GPC verified the results from ASTM D1652-11 and FTIR for samples synthesized using FA. High viscosities were revealed for samples that resulted in high degree of epoxidation. Similarly, a high average molecular weight and low PDI at short reaction times of 2 and 4 h reveal little to no change in the structure of the triglyceride. Lower average molecular weight and high PDI signifies the presence of oligomers as a result of the formation of epoxide groups. With FA being the most effective oxygen carrier, this work is in its initial stages to characterize and synthesize a bio-based epoxy resin system for use in natural fiber-reinforced composites.
Footnotes
5.
The authors are grateful to NSF-EPSCoR (Grant No. 1158862), NSF-CREST (Grant No. 1137681), and Alabama Commission on Higher Education (ACHE) for the financial support to carry out this research. Special thanks to Dr. Ulven, Dr. Webster, Andrew Mahoney and Ali Amiri for performing GPC testing at North Dakota State University.
