Abstract
Objective:
To determine the effect of electroacupuncture (EA) on β1-adrenergic receptor (β1-AR) and post-receptor protein kinase A (PKA) signaling pathway after acute myocardial ischemia (MI).
Methods:
An MI model was established by ligating the left anterior descending coronary artery of wild-type (WT) C57/BL and β1-AR+/– mice (heterozygous for β1-AR gene deletion). EA treatment was administered at HT5-HT7 or LU9-LU8. We evaluated cardiac function by measuring ST segment displacement, ischemic area and serum levels of creatine kinase (CK)-MB and lactate dehydrogenase (LDH). Pathological morphology/apoptosis of myocardial tissue were examined using hematoxylin–eosin and terminal deoxynucleotidyl transferase dUTP nick end labeling staining. Norepinephrine (NE) levels in myocardial tissue were detected by ELISA. Levels of β1 and post-receptor PKA signaling components were evaluated by quantitative reverse transcription polymerase chain reaction (qRT-PCR) and Western blotting.
Results:
EA stimulation at HT7-HT5 could better regulate the level of β1-AR in myocardial tissue than that at LU9-LU8. Following EA, the ST segment, serum CK-MB/ LDH and area of myocardial infarction were decreased in WT mice, and the degree of myocardial pathology/apoptosis and expression of cleaved caspase-3 were decreased. Myocardial levels of Gs protein (Gs), adenylate cyclase (AC), cyclic adenosine monophosphate (cAMP), phosphorylated protein kinase A (p-PKA), L-type voltage-gated calcium channel α1C (Cav1.2), serine phosphate 16-phospholamban (p-PLBs16) and sarcoplasmic reticulum Ca2+-adenosine triphosphate (ATP)ase 2a (SERCA2a) increased after EA. However, these effects of EA were not replicated in β1-AR+/– mice. Interestingly, myocardial NE content decreased after EA in WT and β1-AR+/– mice.
Conclusion:
EA may enhance cardiac function and reduced MI area/apoptosis by restoring the activity of β1-AR and post-receptor PKA signaling.
Keywords
Introduction
Acute myocardial ischemia (AMI) is a high-incidence disease with high mortality rates in most developed countries, and is also the leading cause of heart failure (HF). 1 Studies have shown that myocardial ischemia (MI), arrhythmia and HF are closely associated with cardiac autonomic nerve dysfunction.2,3 Generally, several G-protein-coupled receptors (GPCRs) on the cytoplasmic membrane of cardiomyocytes respond to adrenergic and cholinergic stimulation, reflecting the two branches of the autonomic nervous system (ANS). Although they play a synergistic role in the regulation of cardiac function, abnormalities of the sympathetic adrenergic system appear to be the main cause of MI. 4
Changes in β-adrenergic signaling play a prominent role in cardiac remodeling in HF after MI, 5 particularly the β1-adrenergic receptor (AR). 6 It is also known that the β1-AR is the main adrenergic receptor of the heart and plays a significant role in maintaining its normal physiological function. Moreover, β1-AR signaling involves Gs proteins, which stimulate the effector adenylate cyclase (AC) to convert adenosine triphosphate (ATP) to cyclic adenosine monophosphate (cAMP), which then binds to cAMP-dependent protein kinase A (PKA) and phosphorylates different target substrates to increase intracellular free calcium and induce myocardial contraction. 7 During AMI, a large number of endogenous catecholamines are released by the sympathetic nerves, promoting β1-AR activation. However, sustained activation of the β1-AR ultimately results in the loss of sympathetic signals and HF. The loss of positive changes in time, force and conduction of the heart is mediated by desensitization and downregulation of β1-AR pathway intermediates. 6 Furthermore, the expression of β1-AR mRNA and cAMP gradually decreases with the exacerbation of HF, which further results in a reduction of PKA expression and abnormal Ca2+ transport, thereby leading to myocardial diastolic and systolic dysfunction. 8 To date, studies have demonstrated that the actuation of β-adrenergic signaling and the recovery of PKA activity contribute to the enhancement of cardiac function in rats with HF after MI.5,9 Therefore, restoring the activity of the β1-AR and post-receptor PKA signaling pathway may represent a therapeutic option to decrease the incidence of HF and promote heart function after MI.
Acupuncture, as an external treatment method of traditional Chinese medicine (TCM), has shown promise in the clinical management of stable angina pectoris.10–13 Electroacupuncture (EA) involves electrical stimulation of filiform needles using different current frequencies, intensities and amplitudes. EA allows standardization of stimulation frequency and has become a common intervention in scientific research and clinical practice. 14 Increasing evidence from animal studies has revealed that EA can protect cardiac function post-MI.15,16 In earlier studies, we found that EA at HT7 (Shenmen) and HT5 (Tongli) can reduce myocardial ischemic injury in rats by modulating autonomic nerve activity and norepinephrine (NE) release.17,18 However, its exact mechanism has not been identified.
Although EA could alleviate MI injury by regulating the autonomic nerves, the effect of EA on the cardiac β1-AR needs to be explored. Here, we hypothesized that β1-AR may be involved in the regulatory effect of EA on cardiac function after AMI. We used wild-type (WT) and β1-AR+/– mice (heterozygous for β1-AR gene deletion) to establish a model of MI, and the effect of EA on MI was assessed by electrocardiography (ECG), serum myocardial enzymes and determination of the myocardial ischemic area. Meanwhile, to explore whether EA improves cardiac function by regulating the β1-AR and post-receptor PKA signaling pathway, the levels of Gs, AC, cAMP and PKA in ischemic myocardial tissue were measured.
Methods
Animals
We obtained male WT C57 BL/6 mice (specific pathogen-free (SPF) grade, 8 weeks, weighing 20–25 g) from Hangzhou Ziyuan Experimental Animal Technology Co., Ltd. (Zhejiang, China). β1-AR+/– mice and C57 BL/6 background mice were bought from Jiangsu Jicui Yaokang Biotechnology Co., Ltd. (Nanjing, China). The animals were kept at ambient temperature (23 ± 1°C) under a 12 h light/dark cycle and fed with sterilized food and water.
In experiment 1, a total of 35 WT C57 BL/6 mice were used and three animals died during MI modeling. Thirty-two surviving mice were divided into one of the following four groups (n = 8 mice each): sham operation group (Sham), untreated MI group (MI), MI group receiving EA at HT7 and HT5 (EA1) or MI group receiving control EA at LU9 (Taiyuan) and LU8 (Jingqu) (EA2). In each group, five mice were designated for 2,3,5-triphenyltetrazolium chloride (TTC) staining and three for reverse transcription polymerase chain reaction (RT-qPCR) or Western blotting, respectively, (see Figure 1(a)–(c)).
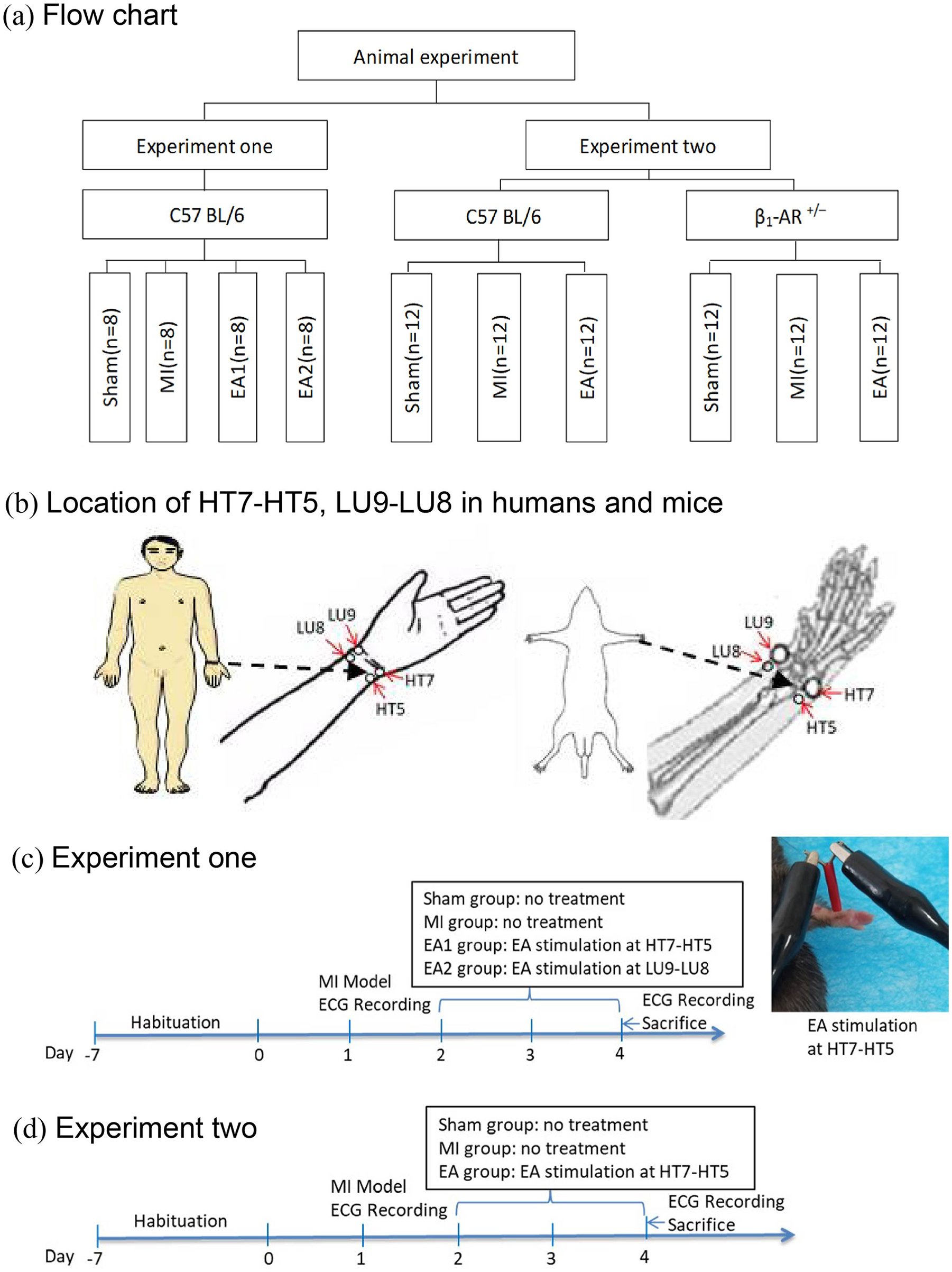
(a) Flow diagram of the study. (b) Location of traditional acupuncture points HT7 (Shenmen), HT5 (Tongli), LU9 (Taiyuan) and LU8 (Jingqu) in humans and mice. (c, d) Time chart and protocols of experiments 1 and 2.
In experiment 2, a total of 36 WT C57 BL/6 mice and 40 β1-AR+/– were used, and four β1-AR+/– mice died during the modeling process. Surviving β1-AR+/– mice (n = 36) were randomized into three subgroups (n = 12 mice each group): Sham group, MI group or MI group receiving EA at HT7-HT5 (EA). In each subgroup, five mice were designated for TTC staining, four for hematoxylin–eosin (H&E) and terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining and three for Western blotting, respectively (see Figure 1(a), (b) and (d)).
The study protocol was approved by the Animal Experiment Ethics Committee of Anhui University of Traditional Chinese Medicine and conformed to the “Guide for the Care and Use of Experimental Animals” of the National Institutes of Health (NIH; Bethesda, MD, USA).
Establishment of MI model
The MI model was developed as previously described. 19 The mice in the MI, EA, EA1 and EA2 groups were anesthetized with isoflurane (2% maintenance; Rayward Life Technology Co., LTD., Shenzhen, China) and fixed on an operating table. The left side of the chest was shaved and routinely disinfected. We made a 1 cm skin incision near the sternal margin and separated the pectoralis major and pectoralis minor, exposing the fourth intercostal space. Using hemostatic forceps, the right hand penetrated the intercostal muscle, pleura and pericardium at the intercostal space to create a small opening. The heart was squeezed with the left hand and the left anterior descending (LAD) arterial branch was ligated with 8-0 suture. In the Sham groups, only thoracotomy was performed (without LAD ligation).
EA intervention
In accordance with published traditional acupuncture point locations in experimental animals 20 and the anthropoid comparison method, EA was administered bilaterally at HT7 and HT5 or LU9 and LU8 (see Figure 1(b)) according to group allocation and under anesthesia. Stainless steel acupuncture needles (0.16 mm × 7 mm, Beijing Zhongyan Taihe Medical Instruments Co., Ltd., China) were inserted to a depth of 1 mm and stimulated for 30 min using an EA therapeutic instrument (SDZ-V type, Hua Tuo brand, Suzhou, China) at 1 mA intensity and 2/15 Hz alternating frequency (alternating 2 Hz sparse waves for 1.5 s and 15 Hz dense waves for 1.5 s, pulse width 0.2 ms). EA was administered once daily for a total of 3 days. In the Sham and MI groups, mice received isoflurane anesthesia at the same time and same dose as in the EA groups without any other intervention.
ECG recording
ECG data were collected before and after model replication and after the last EA treatment. A Powerlab 16 physiological recorder was connected to record and detect the standard lead II ECG of mice in each group under anesthetized conditions and the ST segment displacement value was examined using built-in software.
Sample collection and processing
At the conclusion of the study, mice were deeply anesthetized with 5% isoflurane and blood was collected from the retro-orbital venous plexus. After being allowed to stand for 1 h, the supernatant was centrifuged (4°C, 1400 g, 15 min) and stored at −80°C. After blood collection, mice were sacrificed by decapitation. The hearts were immediately removed and washed in phosphate-buffered saline (PBS). Five hearts per group in both experiments were used for TTC detection. In experiment 1, the remaining three hearts per group were stored at −80°C for Western blotting and RT-qPCR. In experiment 2, three hearts were stored at −80°C for Western blotting and enzyme-linked immunosorbent assay (ELISA) and the hearts of the remaining four mice were fixed in 4% paraformaldehyde solution for 24 h, then dehydrated, paraffin-embedded and cut into slices (approximately 4 µm in thickness) for H&E staining and TUNEL detection.
Enzyme-linked immunosorbent assay
In both experiments 1 and 2, we measured serum levels of creatine kinase (CK)-MB and lactate dehydrogenase (LDH) using commercial ELISA kits (JL13793 and JL46089, respectively, Jianglai Biotechnology Co., Ltd. Shanghai, China). In experiment 2 only, we took ischemic myocardial tissue from three mice in each WT subgroup and the β1-AR+/– EA group, and detected the levels of NE, AC and cAMP in myocardial tissue using additional ELISA kits (JL13969, JL20605 and JL13362, Jianglai Biotechnology Co., Ltd).
TTC staining
Five hearts per group from both experiments were frozen at −20°C for 20 min. At least five sections of the heart (about 1 mm thick) were taken continuously from the conical tip at the lower left aspect of the heart, moving toward the bottom of the heart. Slices were then placed in 1% TTC, and incubated at 37°C in the dark for 20 min. Using this approach, the white and red parts of the tissue were taken to depict the ischemic and non-ischemic areas, respectively. Image analysis was carried out using Image J software (NIH), and the ratio of the infarcted area (%) was calculated as ischemic area/total area of slices × 100%.
H&E staining
Using xylene gradients and absolute ethanol, heart sections from four mice per subgroup in experiment 2 were dewaxed to water, washed with running water, stained with H&E and sealed with neutral gum. A Nikon optical microscope (Nikon, Japan) was used to observe the morphological changes associated with MI in each group. Four heart slices were selected from each mouse and three non-overlapping fields were randomly observed in each slice.
TUNEL assay
Paraffin sections were routinely dewaxed, treated with protease K for antigen repair and then incubated with membrane-breaking working solution at room temperature. An appropriate amount of terminal deoxynucleotidyl transferase (TDT) enzyme was added and incubated at 37°C for 1 h. Three washes in PBS were performed. A working solution of ovalbumin labeled by oxidase was reacted with for 30 min, washed four times with PBS and colored for 4 min using 3,3′-diaminobenzidine (DAB). Hematoxylin counterstaining was performed after washing, then sections were dehydrated, made transparent and sealed. The positive nuclei appeared brown under an optical microscope. Four heart slices were taken from each mouse and three non-overlapping fields were randomly selected from each section to count the number of TUNEL-positive cells.
RT-qPCR analysis
The myocardial tissues below the ligature of the LAD were washed several times in 0.9% sodium chloride solution then cut into pieces and homogenized with 1 mL Trizol (1596–026, Invitrogen, Carlsbad, CA, USA) on ice. After extracting total RNA from tissues, 40 µL of diethyl pyrocarbonate (DEPC; R1600, JRDUN Biotechnology Co. Ltd, China) was dissolved in water and stored at –80°C pending use. Total RNA was reverse transcribed into cDNA. Conditions for RT were 37°C for 60 min, 85°C for 5 min and 4°C for 5 min. The conditions for qPCR amplification were pre-denaturation at 95°C for 10 min, 95°C for 15 s and 60°C for 45 s for 40 cycles. Sequences of the forward and reverse primers for β1-AR were TATGGCTTACTGGCTTGTCTT and TTTCCATCGGGTCTTTT, respectively. The levels of mRNA expression were normalized to that of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and 2–∆∆Ct was used to calculate the relative expression of β1-AR mRNA.
Western blotting
Ischemic myocardial tissue from three mice in each WT subgroup and the β1-AR+/- EA group was used for Western blot analyses. The myocardial tissue was ground in liquid nitrogen, and radioimmunoprecipitation assay (RIPA) buffer (78510; Thermo Fisher Scientific, Waltham, MA, USA) was used to extract total protein. The concentration of samples was measured using a bicinchoninic acid (BCA) protein quantitative kit (PICPI23223, Thermo Fisher Scientific). After boiling in a water bath for 10 min, the supernatant was taken. After the gel was prepared, the samples were sealed for 1 h and then subjected to electrophoresis. After that, the film was transferred with 5% skim milk powder and then incubated with the following primary antibodies in the refrigerator overnight at 4°C: anti-β1 (1:1000; Ab8226, Abcam, Cambridge, UK); anti-Gs (1:500; Ab235956, Abcam); anti-phosphorylated (p)-PKA (1:2500; Ab75991, Abcam); anti-Cav1.2 (1:500; ACC-003, Alomone Labs, Israel); anti-p-phospholamban (PLB; 1:10,000; Ab92697, Abcam); anti-sarcoplasmic reticulum (anti-SR) Ca2+-ATPase 2a (SERCA2a; 1:10,000; A010–23S, Badrilla, China); anti-cleaved caspase 3 (1:500; Ab32042, Abcam); and anti-β-actin (1:1000; Ab8226, Abcam). Following three cycles of washing, the membranes were incubated with goat anti-rabbit horseradish peroxidase (HRP)-labeled secondary antibodies (1:10,000; ZB-2301, ZSGB-BIO, China) for 1 h at 37°C. Proteins were measured using an enhanced chemiluminescence (ECL) kit (WBKLS0100, Millipore, Germany).
Statistical analysis
Data are represented as mean ± standard deviation (SD). All statistical analyses were performed using GraphPad Prism 8.2.1 (GraphPad Software, San Diego, CA, USA). One-way analysis of variance (ANOVA) and post hoc least significant difference (LSD) multiple comparison tests were used to assess statistical differences between the groups. Pearson correlation analysis was performed to analyze the relationship between β1-AR and ST segment displacement values, serum CK-MB and LDH. p < 0.05 was considered statistically significant.
Results
EA at HT7-HT5 (but not at LU9-LU8) improved myocardial injury and β1-AR expression in MI mice
We analyzed the ST segment, serum CK-MB and LDH content, MI area and the expression of β1-AR mRNA and protein in MI model rats that remained untreated or received EA at HT7-HT5 or LU9-LU8. Compared with the Sham group, ST segment displacement values (see Figure 2(a) and (b)) as well as serum CK-MB (see Figure 2(c)) and LDH (see Figure 2(d)) levels were significantly increased in MI mice. Conversely, these were decreased significantly after EA at HT7-HT5 (see Figure 2(a)–(d)) but not after EA at LU9-LU8. Moreover, the MI area increased significantly after MI and was decreased after EA at HT7-HT5 but not at EA at LU9-LU8 (see Figure 2(e) and (f)). We also examined the effect of EA stimulation on myocardial β1-AR in the MI model mice. Western blotting and RT-qPCR analysis revealed that MI was characterized by significantly reduced expression of β1-AR in myocardial tissue. Both protein and mRNA expression of β1-AR was significantly increased in the EA1 group but not the EA2 group (see Figure 2(g) and (i)). Finally, correlation analysis showed that β1 had a negative correlation with ST segment displacement values, serum CK-MB and serum LDH (see Figure 2(j)–(l)).
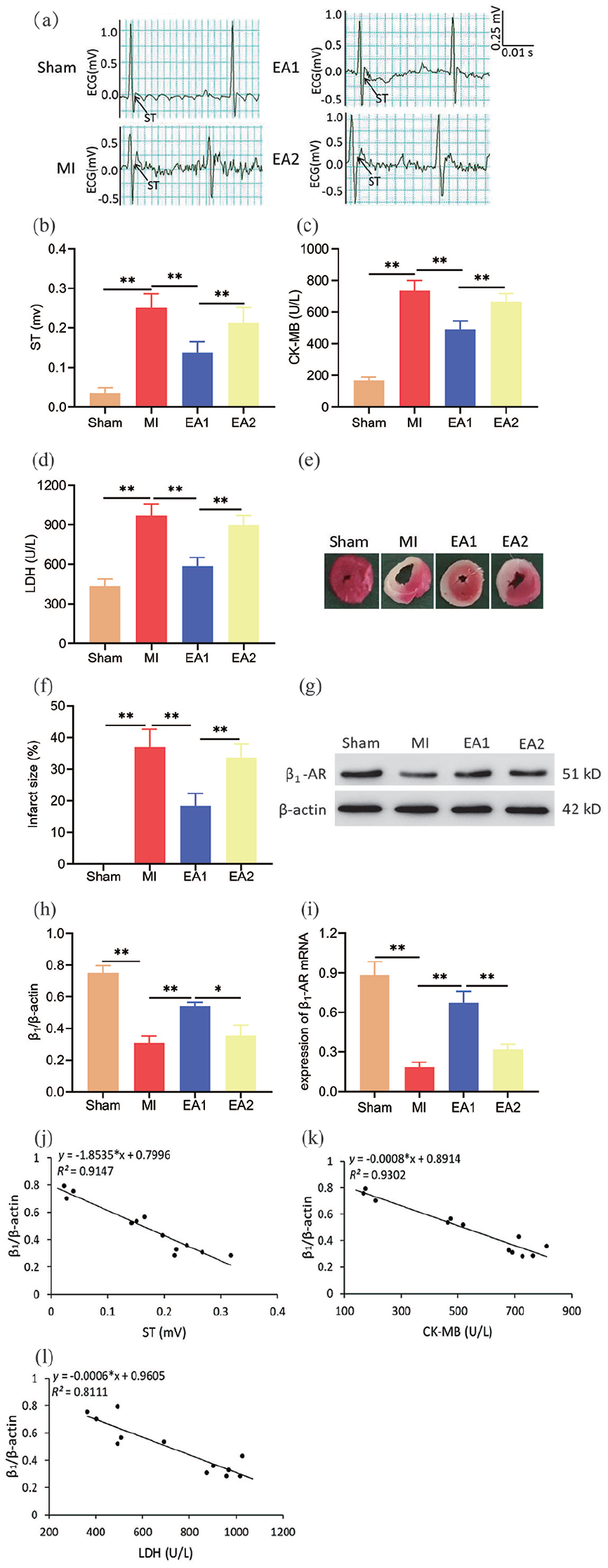
Cardiac function and β1 adrenergic receptor (β1-AR) expression levels in myocardial tissue after electroacupuncture (EA) at HT7-HT5 (EA1) and LU9-LU8 (EA2) in a mouse model of myocardial ischemia (MI). (a) Representative electrocardiogram (ECG) of mice in each group. (b) Quantitative analysis of ST segment displacement (n = 8 per group). (c, d) Serum levels of MB isoenzyme of creatine kinase (CK-MB) and lactate dehydrogenase (LDH; n = 8 per group). (e) Representative images of triphenyltetrazolium chloride (TTC) staining. (f) Infarction area (n = 5 per group). (g) Representative Western blot images of β1-AR. (h) Quantification of β1-AR expression levels (n = 3 per group). (i) mRNA level of β1-AR in myocardial tissue measured by RT-qPCR (n = 3 per group). Data are mean ± SD. (j–l) Correlation analysis of β1-AR with ST segment displacement, CK-MB and LDH.
EA at HT7-HT5 did not improve ST segment displacement, serum CK-MB/LDH content or infarction area in β1-AR+/– mice
ST segment displacement, serum CK-MB and LDH content and the MI area of mice were evaluated 3 days after MI injury to investigate the effects of EA on cardiac function. Overall, the results demonstrated that, compared with the Sham group, ST segment displacement of MI mice was significantly increased (p < 0.01), whereas it was significantly reduced after EA treatment in WT mice (p < 0.01). However, it did not change significantly when EA was given at HT7-HT5 after MI in β1-AR+/– mice (p > 0.05; see Figure 3(a) and (b)).
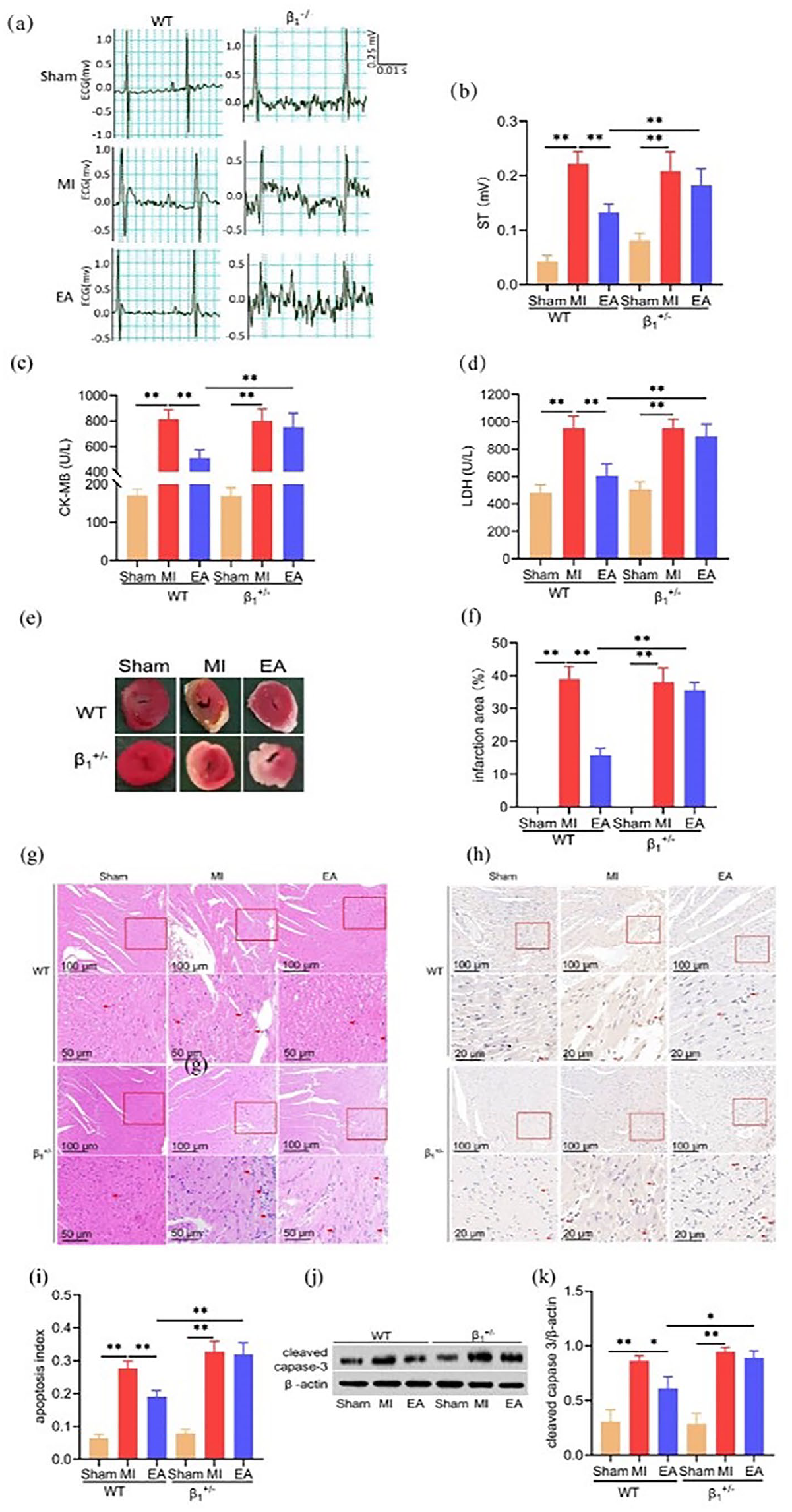
Effect of electroacupuncture (EA) on myocardial ischemia injury in mice. (a) Representative electrocardiogram (ECG) of mice in each group. (b) Quantitative analysis of ST segment displacement (n = 12 per group). (c, d) Serum MB isoenzyme of creatine kinase (CK-MB) and lactate dehydrogenase (LDH; n = 12 per group). (e) Representative images of infarction area by 2,3,5-triphenyltetrazolium (TTC) staining. (f) Quantitative analysis of infarction area (n = 5 per group). (g) Histological observation after hematoxylin and eosin (H&E) staining. (h) Cardiomyocyte apoptosis after terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining in each group. (i) Quantitative analysis of apoptosis (n = 4 per group). (j) Representative Western blot images of cleaved caspase-3. (k) Quantification of cleaved caspase-3 protein expression levels (n = 3 per group). Scale bar: 100 and 20 µm. Data are mean ± SD.
As compared to the Sham group, the serum levels of CK-MB and LDH significantly increased after MI and significantly decreased after EA in WT mice (both p < 0.01), while these did not change significantly when EA was given at HT7-HT5 after MI in β1-AR+/– mice (p > 0.05; see Figure 3(c) and (d)).
Compared with the Sham group, the infarction area in the MI group was significantly increased, and was significantly decreased after EA in WT mice (both p < 0.01); however, it did not differ significantly between the EA and MI groups of β1-AR+/– mice (p > 0.05; see Figure 3(e) and (f)).
EA at HT7-HT5 did not improve the pathological morphology or apoptosis of myocardial tissue in β1-AR+/– mice with MI
We examined the effects of EA on pathological morphology and apoptosis of myocardial tissue in mice using H&E and TUNEL staining. Histological observation of the Sham group exhibited that cardiac muscle cells were arranged in order and the myocardial fibers were compact (see Figure 3(g)). By contrast, mice in the MI group showed a disordered arrangement of myocardial fibers and interstitial edema with infiltration by a large number of inflammatory cells. Compared with the MI group, the degree of myocardial fiber injury and inflammatory cell infiltration in WT mice was significantly decreased after EA treatment, whereas in β1-AR+/– mice the EA and MI groups did not show any apparent differences upon histological evaluation (see Figure 3(g)).
Myocardial apoptosis and the expression of cleaved caspase-3 in the MI group were significantly increased compared to the Sham group (p < 0.01) and decreased after EA in WT mice (p < 0.01; see Figure 3(h)–(k)). On the contrary, the apoptosis and cleaved caspase-3 levels did not significantly differ between the EA and MI groups of β1-AR+/– mice (p > 0.05; see Figure 3(h)–(k)).
EA at HT7-HT5 reduced NE content in myocardial tissue in vivo
The levels of NE in mouse myocardial tissues were measured by ELISA. Our results showed that the level of NE in the myocardial tissue of MI mice was significantly higher than that of the Sham group (p < 0.01). Interestingly, after EA, it was significantly decreased in both WT and β1-AR+/– mice (both p < 0.01; see Figure 4(a)).
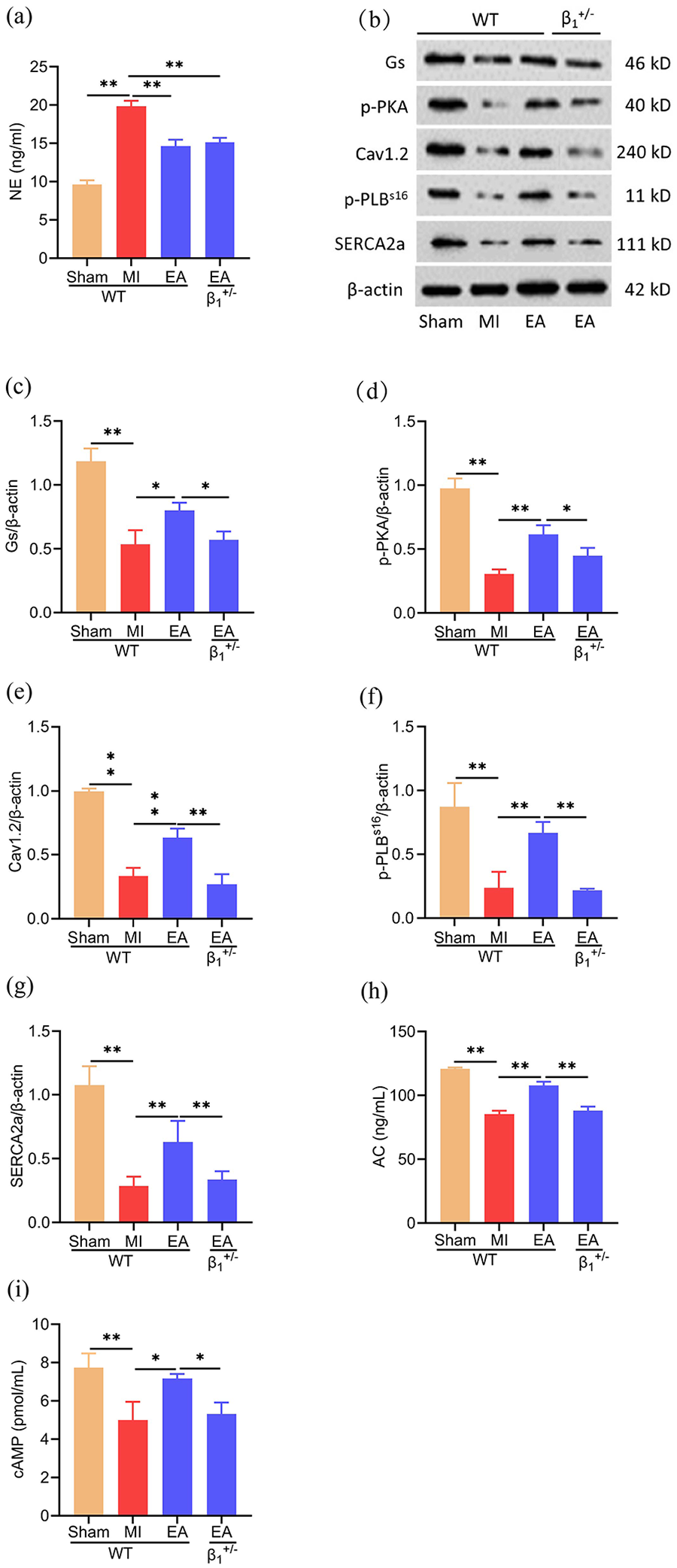
Effects of electroacupuncture (EA) on the level of norepinephrine (NE), post-receptor protein kinase A (PKA) signaling pathway and expression levels of L-type voltage-gated calcium channel α1 C (Cav1.2), serine phosphorylated 16-phospholamban (p-PLBs16) and sarcoplasmic reticulum (SR) Ca2+-adenosine triphosphate (ATP)ase 2a (SERCA2a) in myocardial tissues in vivo. (a) NE levels in myocardial tissue (n = 3 per group). (b) Representative Western blot images of stimulatory G protein (Gs), phosphorylated PKA (p-PKA), Cav1.2, p-PLBs16 and SERCA2a. (c–g) Quantification of Gs, p-PKA, Cav1.2 p-PLBs16 and SERCA2a protein expression levels (n = 3 per group). (h, i) Levels of adenylate cyclase (AC) and cyclic adenosine monophosphate (cAMP) in myocardial tissue (n = 3 per group). Data are mean ± SD.
EA reversed the inhibition of post-receptor PKA signaling pathway in vivo
The expression of Gs, AC, cAMP, p-PKA, Cav1.2, p-PLBs16 and SERCA2a in myocardial tissues were detected by Western blotting or ELISA to observe the effect of EA treatment on the post-receptor PKA signaling pathway of β1-AR mice. Our results showed that protein expression of Gs, p-PKA, Cav1.2, p-PLBs16 and SERCA2a, as well as levels of AC and cAMP in myocardial tissue, were decreased in MI versus Sham groups (p < 0.01) but markedly increased in WT mice after EA (p < 0.01 and p < 0.05, respectively; see Figure 4(b)–(i)). However, these indicators showed no significant differences after EA in β1-AR+/– mice with MI (p > 0.05; see Figure 4(b)–(i)).
Discussion
In this study, we examined the effects of EA on cardiac function and β1-AR in AMI mice and found that EA at HT7-HT5 can alleviate MI injury and improve the expression of β1-AR in myocardial tissue. Furthermore, our study showed that EA might potentially alleviate MI injury by enhancing the activity of the PKA signaling pathway facilitated by β1-AR, regulating calcium channel protein levels to maintain the balance of intracellular and extracellular calcium ions, and reducing apoptosis.
In order to examine the cardioprotective effect of EA, a MI model was quickly and effectively established in mice by ligating the LAD coronary artery. In addition to yielding acute ST segment elevation infarction, this method reduces the sample size required to detect differences between groups, 16 and is especially appropriate for modeling of MI in transgenic mice. 19 In prior studies, it has been shown that EA treatment of 1 mA intensity, 2/15 Hz frequency and 30 min duration for 3 days is effective.17,18 Since serum levels of myocardial enzymes reaches their peak within 24–72 h of AMI, these could potentially mask the effect of EA if only given for 1 day, hence our choice of a 3-day treatment course. Our previous studies have also confirmed that EA at HT7-HT5 alleviates MI injury in rats,17,18 and that the abnormal ST segment and serum myocardial enzymes are closely related to AMI. 18 In this study, EA at HT7-HT5 reduced ST segment elevation, serum CK-MB, LDH content and myocardial infarction area in AMI mice when compared with control EA at LU9-LU8; thus, it appears that EA at HT7-HT5 can help protect myocardial tissue, although the physiologic basis underlying these differential effects at treatment locations that are anatomically very close to one another (especially in the mouse) remains unclear.
β1-ARs maintain normal heart rate, rhythm, myocardial contractility and cardiac output. However, under pathological conditions, sustained β1-AR activation can lead to sympathetic signal loss and result in HF. 21 Of note, the downregulation of β1-AR in myocardial cells may be the primary cause of declining myocardial systolic and diastolic function and HF post-MI. 22 β-AR blockers are often used to inhibit receptor and ligand binding, thereby enhancing the positive inotropic effect of the myocardium and reducing the risk of sudden death. 23 This suggests that increased expression of β1-AR may contribute to the maintainenance of cardiac function after ischemia. In this study, the findings of experiment 1 indicated that mRNA and protein levels of β1-AR in the MI group were decreased significantly compared to the Sham group, whereas their levels remarkedly increased after EA treatment. Thus, the β1-AR might be involved in the protective effect of EA on ischemic heart disease. We also found that β1-AR levels demonstrated a negative correlation with MI markers. Overall, these results indicate that myocardial β1-AR might be involved in the protective effect of EA on the ischemic heart.
Next, we investigated whether β1-AR affects the protective effect of EA on MI using gene-knockout mice. Because most β1-AR–/– mice (homozygous for β1-AR gene mutation) die prenatally and strain dependence occurs in lethality penetrance, 24 it is difficult to cultivate homozygous mice that can meet actual needs. Thus, experiment 2 was completed with β1-AR+/– mice. Our findings revealed that the β1-AR+/– genotype weakened the effect of EA on the ST segment, serum CK-MB, LDH and myocardial ischemic area after MI. Likewise, based on the findings of H&E staining and TUNEL staining, EA significantly restored myocardial fiber structure and reduced apoptosis in WT mice, which suggests that EA has a protective effect on MI injury; however, this effect was not seen in β1-AR+/– mice.
NE is an important neurotransmitter that connects the nervous system and the heart; in excess, it can lead to heart injury through Ca2+ overload, oxidative stress and mitochondrial dysfunction. 25 However, increased cardiac sympathetic nerve activity is the main source of NE in HF. 26 A previous study has shown that reducing sympathetic NE release during MI is helpful in protecting the myocardium from ischemia-reperfusion (I/R) injury. 27 We found that, in WT and β1-AR+/– mice, the level of NE in myocardial tissue was significantly reduced after EA treatment compared to no treatment, which is consistent with earlier studies.16,18 Although EA stimulation could reduce the level of NE in the myocardial tissues of β1-AR+/– mice, the protective effect of EA on MI was weakened by the defect in β1-AR, suggesting that the β1-AR might be the main target of EA for AMI.
It is well-known that β1-AR is a GPCR, which can be coupled with Gs protein on the cardiomyocyte membrane to regulate the phosphorylation of various target proteins through AC/cAMP/PKA signaling, thereby mediating positive chronotropic, inotropic and conduction effects on the heart. 28 According to previous research, Gs and AC activity of the endocardium underlying ischemic myocardium was significantly reduced and persisted compared with non-ischemic endocardium in dogs. 29 Ferrara et al. 6 reported that the reduction in β-AR reactivity associated with aging results in reduced AC activity and decreased cAMP production, causing a reduced ability to activate PKs, which is associated with the pathogenesis of HF. It is worth noting that cAMP is responsible for controlling PKA-dependent phosphorylation activity in myocardial cells. 30 In addition, diminished cardiac reserve and exercise capacity due to reduced PKA activation may result in an increased risk of HF. 31 Recent studies have shown that increasing PKA activity in myocardial tissue has a beneficial effect on the recovery of heart function in rats with HF.9,32 This means that the abnormal regulation of heart function by PKA is associated with the incidence of heart disease.
Overall, when MI promotes HF, then reduced expression of β1-AR and the decrease in downstream signaling results in cardiac dysfunction. Restoring the activity of β1-AR and AC–cAMP–PKA pathways might, therefore, represent an effective way of improving cardiac dysfunction. Given we observed no significant difference in myocardial ischemic injury between MI and EA groups of β1+/- mice in this in vivo study, we only examined the post-receptor pathway in subgroups of WT mice and the EA group of β1-AR+/- mice. We found that the protein levels of AC, cAMP, Gs, p-PKA, Cav1.2, p-PLBs16 and SERCA2a in myocardial tissues were decreased in the MI versus Sham groups and increased by EA treatment in WT but not β1-AR+/- mice. These findings suggest that EA enhances the activity of the post-β1 receptor PKA signaling pathway and may protect cardiac function following MI.
It has been revealed that PKA phosphorylates various membrane proteins (e.g. phosphoprotein PLB, L-type calcium channel and SR) 6 On one hand, the L-shaped voltage-gated calcium channel, Cav1.2 is located on the plasma membrane and can control heart rate and nerve excitability.33,34 Inactivation of the AC–cAMP–PKA pathway may weaken Cav1.2 phosphorylation after MI, which affects Ca2+ influx and has an adverse effect on myocardial systolic function. On the other hand, SERCA2a is responsible for clearing 70% of the Ca2+ in the cytoplasm. 35 During diastole, SERCA2a pumps cytosolic Ca2+ into the SR of cardiac myocytes, causing muscle relaxation. Moreover, several studies have shown that SERCA2a downregulation decreases the calcium transport capacity from the cytoplasm to the SR in cardiomyocytes, ultimately causing cardiomyocyte apoptosis or necrosis. 36 It has been reported that unphosphorylated PLB inhibits SERCA2a; however, phosphorylated PLB alleviates the inhibition of SERCA2a. 37 Another study has revealed that the degree of apoptosis of cardiomyocytes can be attenuated by increasing the expression of SERCA2a and the phosphorylation level of PLB. 38 Therefore, it is of great significance to reduce MI injury and improve cardiac function by increasing PLB phosphorylation and restoring the activity of SERCA2a to maintain intracellular Ca2+ homeostasis and reduce the apoptosis of myocardial cells. Besides, previous studies have confirmed that EA-induced neuroprotection in ischemic disease might involve inhibition of apoptosis via various pathways.39,40 In this study, we observed that EA increased the levels of Cav1.2, p-PLB16 and SERCA2a within myocardial tissue and decreased the level of cleaved caspase-3, indicating reduced apoptosis in WT mice. By contrast, these effects of EA were not replicated in β1-AR+/– mice. Consequently, EA may enhance the expression of Cav1.2, p-PLBs16 and SERCA2a through the PKA signaling pathway mediated by β1-AR, thereby maintaining intracellular calcium balance, inhibiting apoptosis and ultimately alleviating MI injury. In summary, this study has further clarified the protect mechanism of EA treatment of MI, indicating that β1-AR and the post-receptor PKA signaling pathway could be potential targets of EA for MI injury. This provides a theoretical framework and experimental basis for future research on EA treatment and rehabilitation of these diseases.
Strengths and weaknesses
In this study, the effect of EA on the expression of β1-AR was observed, but no other subtypes of β-AR were detected. It might be considered a limitation that there was no control group receiving conventional medication and that we did not examine the post-receptor pathway in the Sham or MI groups of β1-AR+/- mice. Considering that EA was only used as in vivo stimulation, Ca2+ levels in cardiomyocytes were not measured.
Conclusion
In conclusion, our results demonstrate that alleviation of AMI injury by EA might involve inhibition of NE release from autonomic nerves and altered β1-AR and post-receptor PKA signaling in the myocardium, which may regulate the expression of calcium channel protein to maintain intracellular calcium homeostasis and reduce apoptosis, thereby potentially enhancing cardiac function (see Figure 5). Future studies should focus on the relationship between β1-AR and inflammation in the ischemic heart to further elucidate the possible mechanisms underlying the potentially beneficial effects of EA on MI.
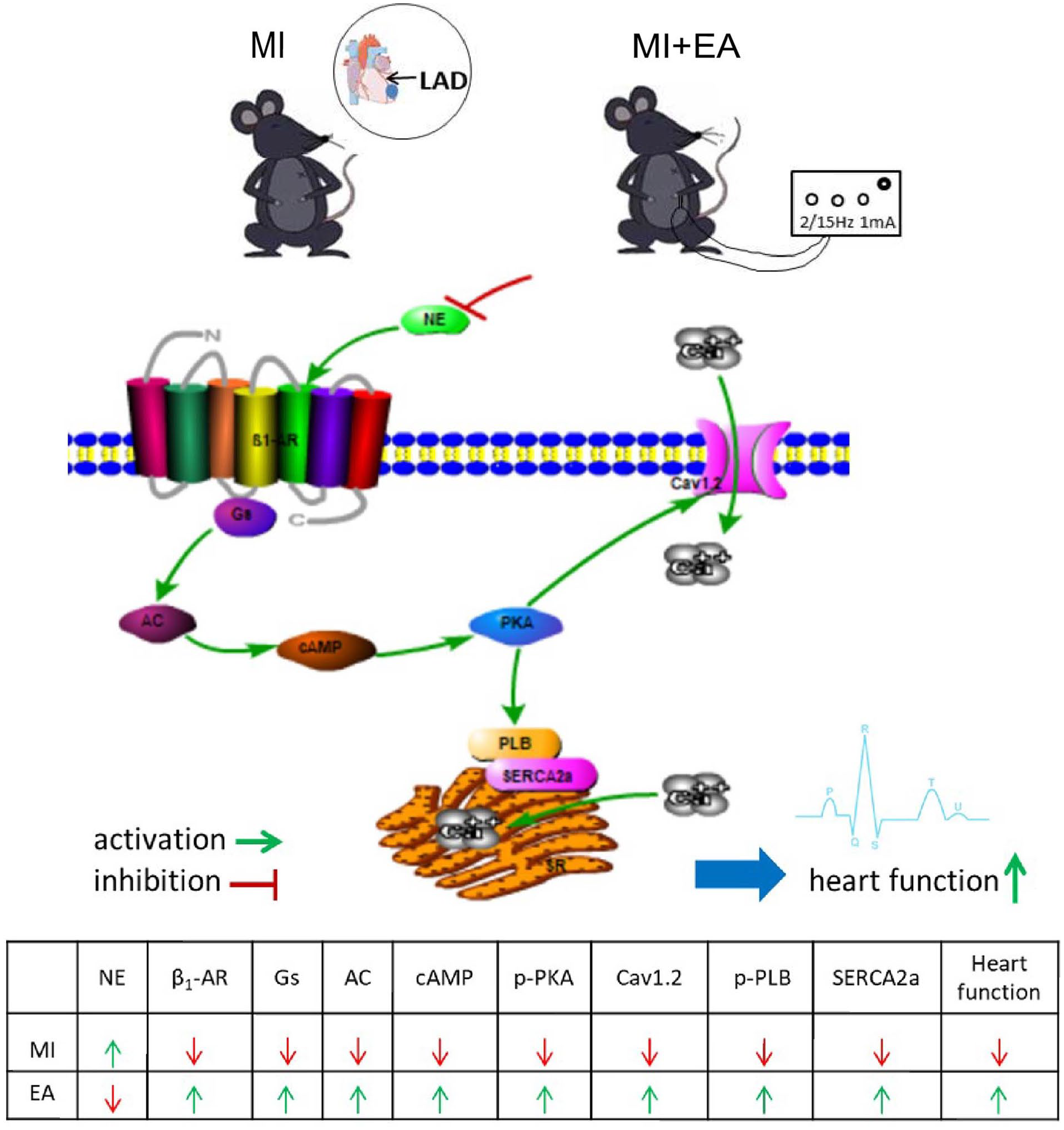
Schematic representation of the cardiac β1 adrenergic receptor (β1-AR) and post-receptor protein kinase A (PKA) signaling pathway regulated by electroacupuncture (EA). MI: myocardial ischemia; LAD: left anterior descending coronary artery; NE: norepinephrine; Gs: stimulatory G protein; AC: adenylate cyclase; cAMP: cyclic adenosine monophosphate; Cav1.2: L-type voltage-gated calcium channel α1 C; p-PLBs16: serine phosphate 16-phospholamban; SERCA2a: sarcoplasmic reticulum Ca2+-adenosine triphosphate (ATP)ase 2a.
Footnotes
Acknowledgements
The authors would like to thank Jiangsu Jicui Yaokang Biotechnology Co., Ltd for providing the β1-AR+/- mice, and all the participants for their valuable contributions.
Contributors
MZ conceived and supervised the study. MZ and HZ designed the experiments. HZ, QQ, YT, XW and SW performed the experiments. QQ, YT and LW analyzed the data. HZ, QQ and YT wrote the article. All authors reviewed the results and approved the final version of the article accepted for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Key Research and Development Plan (2018YFC1704600), the National Natural Science Foundation of China (81674058, 81273858), the Anhui Natural Science Foundation (1908085MH289) and Support Plan of Anhui University of Traditional Chinese Medicine (2022rcyb023).
Disclosure
Qiaoyu Qu and Yan Tong are the co-first authors.
Ethical approval
The experimental protocols for animal use were reviewed and approved by the Animal Ethics Committee of Anhui University of Traditional Chinese Medicine (NO.AHUCM-2020040).
