Abstract
Objective:
To examine for the in vitro existence of contractile nodules on the taut band of muscle fibers where myofascial trigger points (MTrPs) are located (using cell culture).
Methods:
Sixteen male Sprague–Dawley rats (7 weeks old) were randomly divided into experimental and control groups. A blunt striking injury and eccentric exercise were applied to the gastrocnemius muscle of rats in the experimental group once a week for 8 weeks to establish an MTrP model. Subsequently, the rats were reared normally and rested for 4 weeks. After modeling, the skeletal muscles at the MTrPs (and non-MTrPs at the same anatomical position) were extracted from the two groups of rats for in vitro cell culture experiments of single muscle fibers. Potential contractile nodules in the MTrP group were exposed to different concentrations of acetylcholinesterase, whereas non-MTrP cells were exposed to acetylcholine. The morphological changes of muscle cells in each group were observed.
Results:
By culturing MTrP cells in vitro, large contractile nodules remained in single MTrP muscle fibers, whereas some contractile nodules were twisted and deformed. After the addition of different acetylcholinesterase concentrations, no obvious morphological changes were observed in the contractile nodules in the MTrP group. After the non-MTrP cells were exposed to different acetylcholine concentrations, no significant morphological changes were observed in the single muscle fibers.
Conclusion:
MTrP cells can continue to maintain contractile morphology in vitro, but whether the recovery of such contractile nodules is related to acetylcholine remains uncertain.
Introduction
Myofascial trigger points (MTrPs) are highly sensitive sites in abnormal contractile nodules of skeletal muscle. 1 They are characterized pathophysiologically by typical morphological changes and spontaneous discharges that can be monitored by electromyography (EMG) at rest.1,2 Recent epidemiological data have indicated that the prevalence of chronic pain caused by MTrPs is 30%–85% in the United States and 18.7%–85.1% in Germany.3,4 Numerous studies have shown that MTrPs can directly affect the human motor system to cause soft tissue pain, as well as interfere with the adjacent fascia, blood vessels and nerves; they have been associated with vascular diseases, neurological diseases, pelvic floor dysfunction, menstrual disorders, alimentary system dysfunction, skin diseases and cardiovascular diseases.3 –10
The integrated trigger point hypothesis is currently the most credited theory to explain the pathogenesis of MTrP formation.11 –13 This hypothesis postulates that excessive acetylcholine (ACh) release from the resting motor nerve terminals causes a reduction in the postsynaptic membrane potential of the muscle fiber, leading to sustained sarcomere shortening and skeletal muscle contraction.11 –14 The final formation of a muscle taut band with contractile nodules is thought to be a typical clinical feature of MTrPs.12,13 Whether these differentiated MTrP myocytes can remain in a highly contractile state without motor innervation has not yet been studied. In other words, whether the maintenance of a MTrP myocyte survival state is related to factors other than the hyperexcitability of motor nerve endings and excessive ACh release is currently unclear. Anesthesiologists often use nerve blocks or peripheral nerve damage to suppress pain, but those treated often complain of residual pain after these nerve blocks.15,16 Based on the presumed mechanism of formation and the clinical characteristics of referred pain of MTrPs, we now highly suspect that this residual pain may be related to the inactivation of some associated MTrPs in the patient. Therefore, the primary purpose of the present study was to observe the survival state of differentiated MTrP myocytes in contractile nodules in vitro by muscle cell culture.
If these differentiated MTrP myocytes remain in a highly contracted state in vitro, based on the integrated trigger point hypothesis proposed by Simons, 11 hypothetically we could reverse the hypercontractile state to the normal skeletal muscle cell state by adding acetylcholinesterase to consume excess ACh on the surface of MTrP myocytes. Alternatively, if the hypercontractile state of differentiated MTrP myocytes disappears in vitro, hypothetically we could restore the abnormal state by adding ACh. Accordingly, the second purpose of this study was to observe the possible survival state of differentiated MTrP myocytes in vitro after adding acetylcholinesterase or ACh.
Methods
Experimental animals
Sixteen specific pathogen-free (SPF) male Sprague–Dawley rats (7 weeks old, weighing 220–240 g) were obtained commercially from Shanghai Laboratory Animal Center (Shanghai, China; animal license no. SCXK 2007-0003). They were housed in a pathogen-free animal facility maintained at 20–22 °C and 45%–55% relative humidity under a 12/12-h light/dark cycle with ad libitum access to food and water and with minimal environmental noise. After 1 week of environmental adaptation, the animals were randomly assigned to one of two groups (n = 8 each), namely an MTrP model group and a non-MTrP normal group. The National Institutes of Health (NIH) “Guide for the Care and Use of Laboratory Animals” (Bethesda, MD, USA) was followed for all experiments. All possible efforts were exerted to minimize the number of experimental animals used and reduce their suffering. The Committee on the Ethics of Animal Experiments of Shanghai University of Sport granted ethical approval for animal use (approval no. 2014012).
Animal modeling
According to our previously reported methodology,1,2 an MTrP model was established by blunt striking of the gastrocnemius muscle and eccentric-based exercise for 8 weeks along with 4 weeks of recovery. In a typical procedure, the left proximal gastrocnemius muscle of rats in the model group was marked and struck by a homemade stick device dropped from a height of 20 cm with a kinetic energy of 2.352 J to induce muscle contusion on the first day of each week. Then, rats were run on a treadmill (DSPT-202; Duanshi Co., Hangzhou, China) at a −16°downward angle and speed of 16 m/min for 90 min on the second day.
When the rats exhibited the corresponding characteristics of MTrPs described in the literature,1,2 the model was considered to have been constructed successfully. Initially, a contractile nodule in a taut band was palpated at the hit site of the left gastrocnemius muscle, into which an electrode needle was then inserted. If a local twitch response was observed to be elicited by needling, the contractile nodule was considered to be a possible MTrP. For further confirmation, an EMG device with three fine-needle electrodes (Φ 0.3 mm; NeuroCare-E, NCC Medical Co., Ltd., Shanghai, China; sampling frequency = 50,000 Hz) was used to record myoelectrical signals at the contractile nodule. When spontaneous EMG activity with a long duration (>120 s) of high frequency and amplitude (>130 µV) was detected, it was considered to represent a genuine active MTrP.
Cell culture
The muscle cells of each group were extracted by the single muscle fiber separation method and subjected to primary culture.17,18
Preparation before the experiment
Before the experiment, different specifications of Petri dishes were placed in a CO2 cell incubator for >4 h, irradiated with an ultraviolet lamp for >30 min, and washed once with phosphate-buffered saline (PBS). The surgical instruments were then sterilized under high pressure at 120 ℃ before drying and irradiating with ultraviolet light for 30 min before use.
Separation of muscle tissue
Rats were first euthanized by decapitation under deep anesthesia with pentobarbital sodium (30–50 mg/kg; Sinopharm Chemical Reagent Co. Ltd, Shangai, China). Immediately afterward, these rats were soaked in 75% alcohol for 5 min, and then their gastrocnemius muscles were stripped using surgical instruments. The gastrocnemius muscle at the sites of the marked MTrPs was extracted for cell culture. Visible fat and fascia were removed as much as possible, and the size of the sample was limited to a small piece of skeletal muscle corresponding to the location of the tip of the electrode needle when needling at the MTrPs. This muscle tissue was placed in penicillin (400,000 boli units/l) and streptomycin (400 mg/l) in cold PBS and transferred onto a 35-mm Petri dish. The muscle tissue was cut longitudinally into thin muscle fiber bundles on an ultraclean workbench and washed twice with serum-free medium to remove the supernatant and floating tissue. For the digestion of muscle tissue, 4 ml of mixed digestive enzymes (containing 2 mg/mL collagenase type I (SCR103; Sigma Aldrich, St. Louis, MO, USA), 5 mg/mL disperse II (04942078001; Sigma Aldrich) and 0.28 mg/mL CaCl2 (53219961; Sinopharm)) was added to the Petri dish, pipetted for 5 min, and placed in a 5% CO2 cell incubator for 1.5–2 h at 37 ℃ for digestion. After incubation, muscle tissue was transferred to a new 35-mm culture dish and pre-warmed collagenase I–free separation medium (containing 2 mg/ml collagenase type I (SCR103; Sigma Aldrich), Dulbecco’s modified eagle medium (DMEM) (SH30285.02; Hyclone™, Cytiva, Marlborough, MA, USA), 2% fetal bovine serum (FBS) (SH30071.04; Hyclone™, Cytiva) and penicillin streptomycin solution (10378016; Gibco™, Thermo Fisher, Watham, MA, USA)) was added. A 1-ml syringe was used to grind the muscle tissue and separate it as much as possible, and tweezers were used to remove larger fragments to generate a single muscle fiber. If the number of single muscle fibers was insufficient, the muscle tissue was placed in isolation medium again for 1–1.5 h, and the aforementioned steps were repeated.
Extraction of single muscle fibers
We selected straight, thick, transparent and uncrimped single living fibers under a microscope, gently aspirated them with a drawn micro-Pasteur pipette (with a very small mouth) and placed them in a 24-well culture plate (two to three muscle fibers per well). Extracellular fluid was added to the culture plate to maintain the internal environment for the survival of muscle fibers. After adding four times the volume of muscle tissue of serum-containing DMEM (containing 90% DMEM and 10% FBS) to the culture dish to terminate the digestion, it was filtered through a 200-mesh sieve. The filtrate was centrifuged at 100 g for 10 min, the supernatant was removed, and serum-containing DMEM containing penicillin streptomycin solution (containing 90% DMEM, 10% FBS and 1% penicillin streptomycin solution (15140148; Gibco™, Thermo Fisher) - that is, growth medium) was added.
Microscopic observation and intervention
The morphological characteristics of isolated skeletal muscle fibers from the two groups were observed under a microscope (Leica, Germany, 50×). If the muscle fibers from the experimental group still had large contractile nodules, concentrations of acetylcholinesterase (9000-81-1; BioDuly, Nanjing, China) were added. When adding acetylcholinesterase, we set 10 different concentrations (0.1, 0.2, 0.4, 0.5, 0.8, 1, 2, 3, 4 and 5 mmol/l, respectively, added dropwise from lowest to highest)19,20 to observe for serial changes in contractile nodules. If there were no significant contractile nodules in the muscle fibers of the control group, increasing concentrations of ACh (LS825B-5 g; EKEAR Bio, Shanghai, China) were added dropwise (0.1, 0.2, 0.4, 0.5, 0.8, 1, 2, 3, 4 and 5 mmol/l, respectively) from lowest to highest.21,22 Morphological changes in muscle fibers after ACh intervention were observed.
Statistical analysis
All measurement data were subjected to the Shapiro–Wilk test for normal distribution. After confirming normality of distribution, groups were compared by Student’s t-test. All statistical analyses were completed using PRISM version 5.01 (GraphPad Software, Inc., La Jolla, CA, USA). The level of significance of all tests was set at p < .05. The results are presented as the mean and standard deviation.
Results
By culturing the trigger point muscle fibers in vitro, we observed that large contractile nodules remained at the MTrP muscle fibers (as indicated by the black arrows in Figure 1(b) and (d)). Some contractile nodule distortion also occurred. After the skeletal muscle cells in Figure 1(b) and (d) were exposed to different concentrations of acetylcholinesterase, no obvious morphological changes occurred in contractile nodules in normal muscle fibers or MTrP muscle fibers. Similarly, after adding different concentrations of ACh to the skeletal muscle cells in Figure 1(a) and (c), no significant morphological changes were observed in the contractile nodules of normal muscle fibers or MTrP muscle fibers.
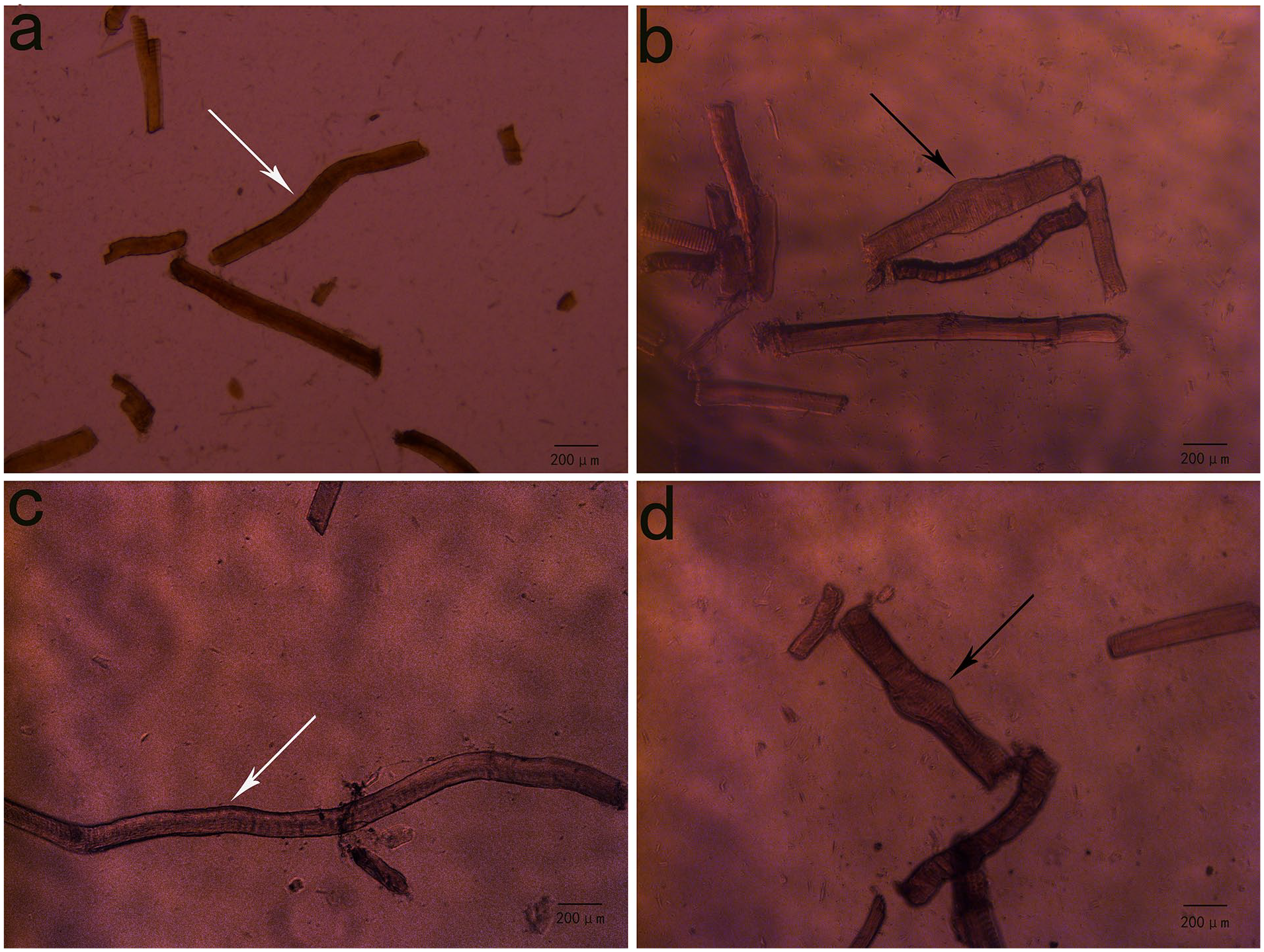
Primary culture results of rat skeletal muscle cells. (a) and (c): Skeletal muscle cells in the normal group. (b) and (d): Skeletal muscle cells at the myofascial trigger point (MTrP) in the MTrP model group. Cultures (a) and (b) were used for the dropwise addition of acetylcholinesterase, and (c) and (d) were used for the dropwise addition of acetylcholine.
Discussion
During the formation of MTrPs, contractile nodules often form in skeletal muscle. Most studies have speculated that these abnormal skeletal muscle contractile nodules may form through the excessive release of ACh caused by the dysfunction of the motor end plate at MTrPs. However, to our knowledge, no experimental verification in cell culture has been conducted to date. Moreover, to our knowledge, no research institution has performed scientific experiments on an in vitro primary culture of MTrP cells. Whether MTrP cells can continue to survive in vitro and maintain a contractile shape is still undetermined. Our research group has attempted to culture MTrP cells in vitro many times. We have found that, after the isolation and culture of a single MTrP muscle fiber, contractile nodules with a large diameter can still be observed in skeletal muscles at the site of an MTrP. Some contractile nodules were also distorted, indicating that MTrP cells can continue to survive in vitro. After acetylcholinesterase was applied to skeletal muscle at the MTrP, morphological changes of contractile nodules did not occur in response to the change in acetylcholinesterase concentration. This finding suggests that MTrP cells in vitro may not be affected by acetylcholinesterase. However, we cannot rule out the possibility that the contractile nodule may have been deformed by other factors, such as the change in ion permeability of the motor endplate membrane in vitro.
The irreversible phenomenon of MTrP contractile nodules in our research results may suggest that there are some specific proteins in MTrP cells that cannot be reversed by the external acetylcholinesterase administration. Accordingly, we plan to conduct a systematic study of proteomic expression between MTrP skeletal muscle and normal skeletal muscle in the future to determine whether differential protein profiles exist between MTrP cells and normal skeletal muscle cells. We will also aim to provide a theoretical basis for the precise immunobiological treatment of MTrP diseases.
In addition, the observation that contractile nodules are still highly present at MTrP sites in the completely denervated state also provides insights into the many ways in which a nerve block or neurectomy is used clinically to stop neuropathic pain.15,16 Several studies have reported that some patients with neuralgia can still feel residual pain a short time after radiofrequency ablation or nerve block treatment.15,23 Based on the results of our current study, we suspect that such residual pain is closely related to the residual abundance of MTrP contractile nodules and the fact that a nerve block may compress sensory nerve endings in surrounding fascial tissues to produce pain. Notably, studies have shown that long-term single MTrP injection techniques can treat refractory neuropathic pain.10,24 Therefore, the treatment of MTrPs may be necessary to eliminate residual pain after peripheral nerve block or neurodestruction.
In conclusion, MTrP cells can continue to maintain their contractile morphology in vitro; however, whether or not the recovery of contractile nodules is related to ACh remains uncertain.
Footnotes
Contributors
L.L. and Q-.M.H. conceived and planned the study. L.L. and Q-.G.L. wrote the paper and established the animal model and cell culture. L.L., Q-.M.H. and Q-.G.L. analyzed the results, wrote the discussion and revised the manuscript. Q-.M.H. and Q-.G.L. further revised the manuscript. All authors read and approved the final version of the manuscript accepted for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (grant no. 32000829 and 81470105) and “Qinglan Project” of the Jiangsu Higher Education Institutions of China.
