Abstract
Background:
Neurogenic bladder (NB) is a form of neurological bladder dysfunction characterized by excessive contraction of the bladder detrusor. Protein kinase A (PKA) signaling is involved in the contraction of the detrusor muscle.
Aims:
To investigate whether PKA signaling mediates the effect of electroacupuncture (EA) on the excessive contraction of the bladder detrusor in NB.
Methods:
Sixty rats were randomly divided into control, sham, NB, NB + EA, and NB + EA + H89 (a PKA receptor antagonist) groups. The modified Hassan Shaker spinal cord transection method was used to generate a NB model. After EA intervention for one week, urodynamic tests were used to evaluate bladder function, hematoxylin and eosin staining was conducted to assess morphological changes, enzyme-linked immunosorbent assay (ELISA) was performed to measure the concentration of PKA, and Western blotting was conducted to measure the protein levels of phosphorylated myosin light chain kinase (p-MLCK)/p-MLC.
Results:
The results showed that NB resulted in morphological disruption, impairment of urodynamics, and decreases in the concentration of PKA and the protein levels of p-MLCK/p-MLC. EA reversed the changes induced by NB dysfunction. However, the improvement in urodynamics and the increases in the concentration of PKA and the protein levels of p-MLCK/p-MLC were inhibited by H89.
Conclusion:
Our findings indicate that the PKA signaling pathway mediates the beneficial effect of EA on excessive contraction of the bladder detrusor in a rat model of NB.
Introduction
Neurogenic bladder (NB) is acknowledged as the most severe complication following spinal cord injury (SCI).1,2 NB is a form of neurological bladder dysfunction characterized by urinary incontinence and urinary retention. 3 Dysfunction of the central nervous system, leading to hyperactivity of the detrusor reflex, noninhibitory contraction and high intravesical pressure, has been proposed as the major cause of NB in SCI patients. 3 Therefore, inhibiting excessive contraction of the detrusor may be an effective intervention for bladder dysfunction. In fact, in clinical practice, drugs targeting excessive contraction of the detrusor have also been shown to be effective at alleviating SCI-related symptoms and improving bladder function.4 –6
The contraction of the detrusor muscle is a calcium- and adenosine triphosphate (ATP)-dependent process. 7 Increasing the concentration of free Ca2+ in the cytoplasm results in the formation of Ca2+ and calmodulin (CaM) complexes, which bind to and activate myosin light chain kinase (MLCK), leading to the phosphorylation of the downstream myosin light chain (MLC).8,9 The phosphorylation of MLC results in the decomposition of enough ATP to induce the contraction of the detrusor muscle. MLCK can be phosphorylated by a variety of kinases, altering its affinity for the Ca2+–CaM complex and making it insensitive to calcium ions. Protein kinase A (PKA) is the most common kinase that phosphorylates MLCK/MLC.9,10
Increasing clinical and preliminary evidence has identified electroacupuncture (EA) as a potentially effective intervention for NB.11 –15 In our previous study, EA increased the maximum capacity of the bladder, reduced bladder pressure, and improved bladder compliance (BC). 16 However, it is still unclear whether EA exerts its effect by preventing excessive contraction of the bladder detrusor or by activating PKA-p-MLCK/p-MLC signaling. Thus, this study was designed to assess the effect of EA on detrusor contraction of the bladder in a rat model of NB and to explore the role of the PKA-p-MLCK/p-MLC signaling pathway.
Methods
Animals
Female Sprague-Dawley rats (8 weeks old) weighing 190–230 g were obtained from the Experimental Animal Center of Hunan University of Traditional Chinese Medicine. The rats were housed at room temperature (22°C–24°C) under a 12 h light/dark cycle and were provided with ad libitum access to food and water. The study was approved by the Animal Ethics Committee of Hunan University of Chinese Medicine (LL2019092303). All experimental procedures were conducted in accordance with the National Institutes of Health Guide for Care and Use of Laboratory Animals. Sixty rats were randomly divided into a control group, sham group, NB group, NB + EA group, and NB + EA + H89 group (n = 12 each).
The modified Hassan Shaker spinal cord transection method was used to generate a NB model. 17 The rats were fasted for 24 h before modeling and received an intraperitoneal (i.p.) injection of penicillin sodium (200,000 U) 2 h before the operation to prevent infection. All NB model rats underwent spinal cord transection at the T10 level. After the operation, the rats received antimicrobial and rehydration therapy. Their cages were kept clean, manual urination was performed, and dressings were changed regularly. In the sham group, the spinal canal was opened but the spinal cord was not transected. Another 14 days were allowed to pass to confirm the stability of the NB model, ensuring that the experimental animals had passed the period of spinal cord shock.
EA administration
Rats in both the EA and EA + H89 groups were treated with EA (one 20-min session per day) for one week. 0.30 mm × 25 mm stainless steel acupuncture needles (Huatuo brand, JiangSu, China) were inserted to a depth of 10 mm at BL32 (Ciliao) and 5 mm at CV3 (Zhongji), SP6 (Sanyinjiao) and GV14 (Dazhui). These traditional acupuncture point locations were identified based on the 9th edition of Experimental Acupuncturology and Government Channel and Points Standard GB12346-90 of China and previous studies of acupuncture for NB.18 –20 All rats were anesthetized with inhalational 2% isoflurane and then fixed on a self-made rat board for the receipt of EA using dense waves at 10/50 Hz frequency (alternating at 3 s intervals) and 1.5 mA intensity (which caused limb tremor) for 30 min per day for a total of 7 days. Rats in the EA + H89 group were additionally pretreated with i.p. PKA receptor antagonist H89 (6 mg/kg) 2 h before EA treatment. 21 The EA intervention began on day 15 and stopped on day 22. During this period, the remaining groups of animals were managed identically except for the EA intervention.
Urodynamic testing
After the EA intervention for 1 week, all rats were anesthetized by i.p. injection of 1% pentobarbital (50 mg/kg) and fixed in a supine position. A catheter was slowly inserted to empty the bladder. Then, 0.9% saline (25°C~35°C) was infused at a rate of 100 μl/min. The change in the bladder pressure curve was recorded with an MP150 multichannel physiological recorder. The maximum cystometric capacity (MCC) was recorded as the perfusion volume at which fluid was first discharged from the urethral orifice. The bladder leak point pressure (BLPP) was recorded as the corresponding pressure at the point of MCC. BC was recorded as the ratio of the change in bladder volume to the change in pressure. The rats were euthanized by cervical dislocation immediately after urodynamic testing, while still under pentobarbital anesthesia.
Sample collection and processing
After euthanasia under terminal anesthesia, the rat bladder detrusor was isolated and divided into two parts. One part was fixed with 4% paraformaldehyde solution, and the other was stored at −80°C.
Hematoxylin and eosin staining
After the samples were incubated in 4% paraformaldehyde for 48 h, they were heated at 60°C for 1 to 2 h, then placed in xylene for 10 min followed by 100%, 100%, 95%, 85%, and 75% ethanol successively for 5 min each and finally immersed in distilled water for 5 min. The samples were stained with hematoxylin for 5 min, washed with distilled water, and incubated in phosphate-buffered saline. The samples were stained with E globulin for 3–5 min, rinsed with distilled water, and dehydrated using an ethanol gradient (95%–100%) for 5 min each. The samples were then removed, placed in xylene for 10 min, and sealed with neutral gum. Morphological changes in the bladder tissue were observed under a microscope (×400; BA210 T, Motic).
Measurement of the PKA concentration in bladder tissue
Bladder detrusor samples were collected in EDTA and centrifuged at 3000g for 15 min at 4°C, then the supernatant was stored at −80°C. The PKA concentration in the bladder tissues was measured with an enzyme-linked immunosorbent assay (ELISA) kit (CB04701792, Sigma-Aldrich, St Louis, MO, USA) according to the manufacturer’s instructions.
Western blotting
Bladder samples were homogenized in lysis buffer. The homogenates were centrifuged at 12,000g for 15 min at 4°C, and the supernatants were collected. The protein concentrations were determined by a bicinchoninic acid (BCA) assay. Equal amounts of protein (50–100 μg) from each sample were loaded onto 8% polyacrylamide gels and subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS PAGE), separated, and then electroblotted onto polyvinylidene difluoride membranes. The membranes were blocked with 5% nonfat dry milk in Tris-buffered saline (TBS) + 0.1% Tween-20 (TBST) for 1 h and incubated with the following primary antibodies at 4°C overnight: rabbit anti-phosphorylated (p)-MLCK (1:200; ab200809, Abcam), rabbit anti-p-MLC (1:200; ab15747, Abcam), and mouse anti-β-actin (1:4000; 6008-1-Ig, Proteintech). After being washed with TBST three times, the membranes were incubated with secondary goat anti-rabbit HRP-conjugated Affinipure (1:3000; SA00001-2, Proteintech). The blots were washed again three times in TBST, and the immunoreactive bands were detected using enhanced chemiluminescence (ECL) reagents.
Statistical analysis
All values are presented as mean ± standard error of the mean (SEM). Statistical differences were evaluated by one-way analysis of variance (ANOVA) followed by Fisher’s post hoc test of least significant difference (LSD). P < 0.05 was considered statistically significant.
Results
Effects of EA on urodynamics and morphological changes in NB model rats
As shown in Figure 1, the NB group exhibited significantly worse urodynamics (reflected by a lower MCC and BC, and higher BLPP) than the control group and the sham group, while no significant difference was found between the control group and the sham group. In addition, the three urodynamics parameters were better in the NB + EA group than in the NB group, indicating that EA improved bladder function in NB model rats.
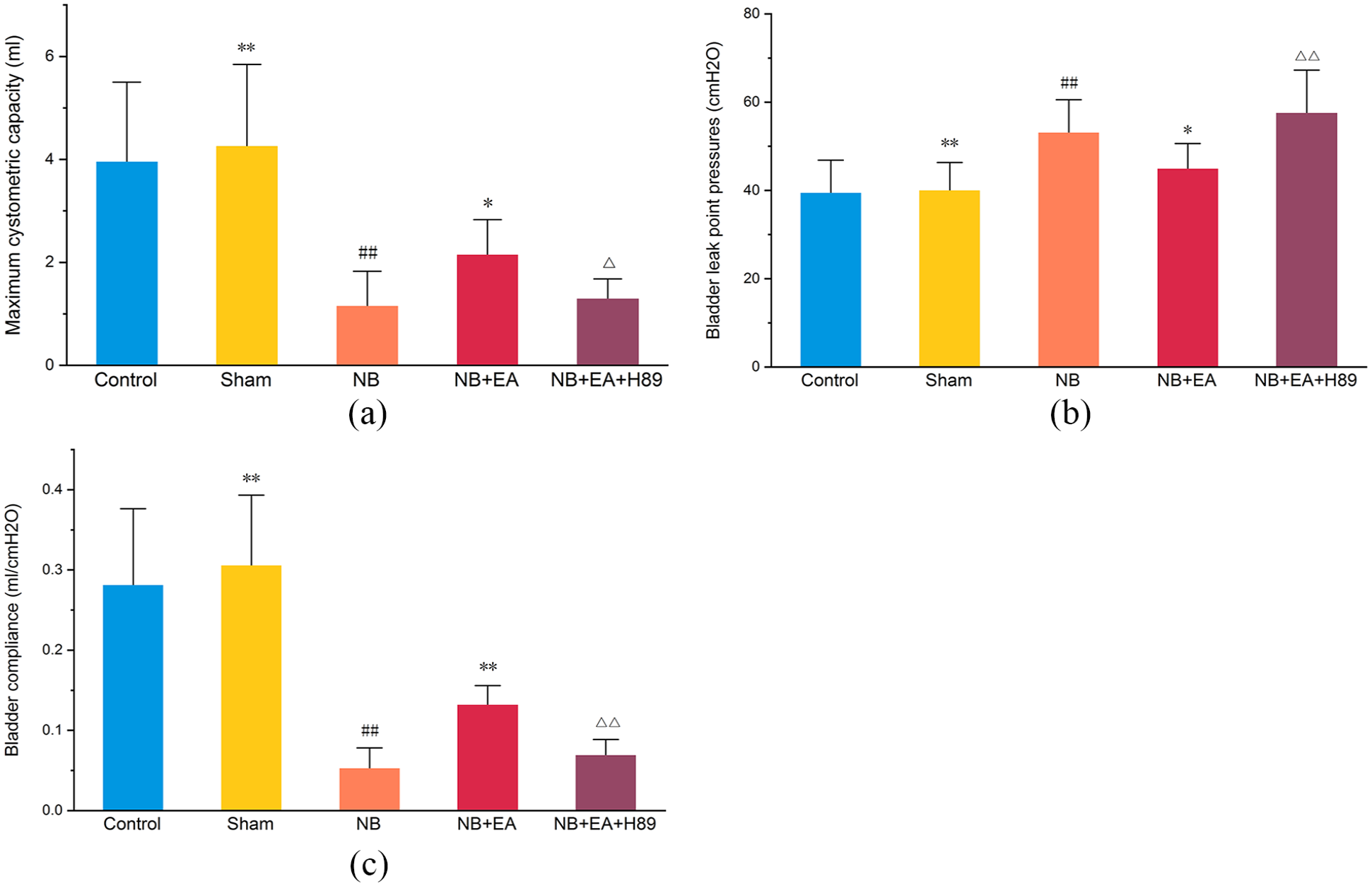
Bladder function in control, sham, NB (neurogenic bladder), NB + EA (electroacupuncture), and NB + EA + H89 groups: (a) maximum cystometric capacity (MCC), (b) bladder leak point pressures (BLPP), and (c) bladder compliance (BC). Values are expressed as mean ± SEM. #p < 0.05, ##p < 0.01 versus control group; *p < 0.05, **p < 0.01 versus NB group; △p < 0.05, △△p < 0.01 versus NB + EA group.
As shown in Figure 2, hematoxylin and eosin staining revealed tissue edema as well as a slight infiltration of inflammatory cells and fibroblasts in the lamina propria of the bladder tissues from the NB group, while EA appeared to abolish the destructive lesions in NB model rats.
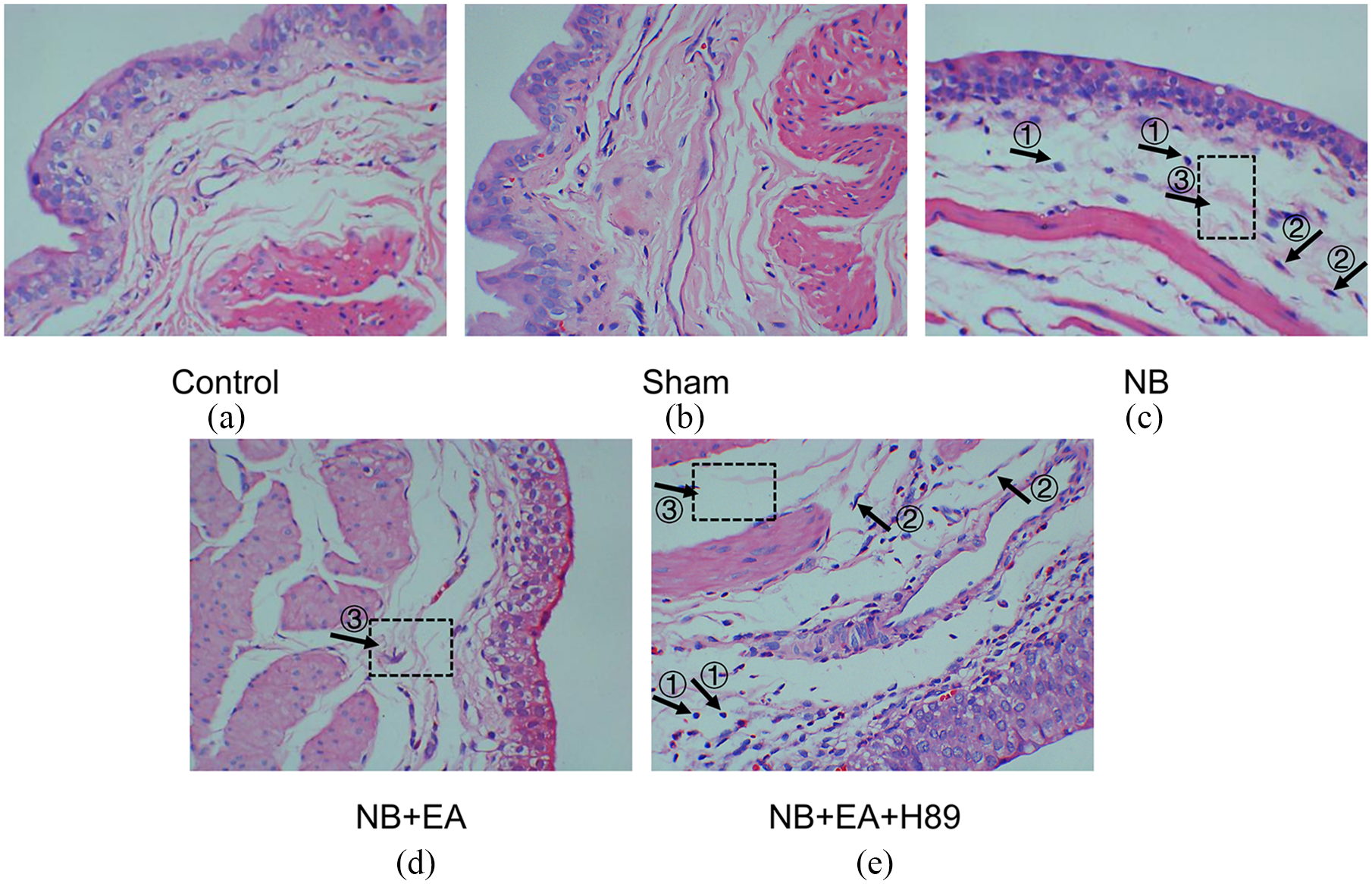
Morphological changes of bladder tissue in different groups under light microscopy after hematoxylin and eosin staining (×400): (a) control group, (b) sham group, (c) NB (neurogenic bladder) group, (d) NB + EA (electroacupuncture) group, and (e) NB + EA + H89 group. ① Inflammatory cells. ② Fibroblasts in the lamina propria. ③ Bladder tissue edema.
PKA-p-MLCK/p-MLC signaling was inhibited in NB model rats
To explore the possible key role of PKA-p-MLCK/p-MLC signaling in the impairment of bladder function in NB model rats, we examined the concentration of PKA (Figure 3), as well as the expression of p-MLCK and p-MLC (Figure 4) in bladder tissues. Lower levels of PKA, decreased expression of p-MLCK, and increased protein expression of p-MLC were found in the NB group compared with the control and sham groups.
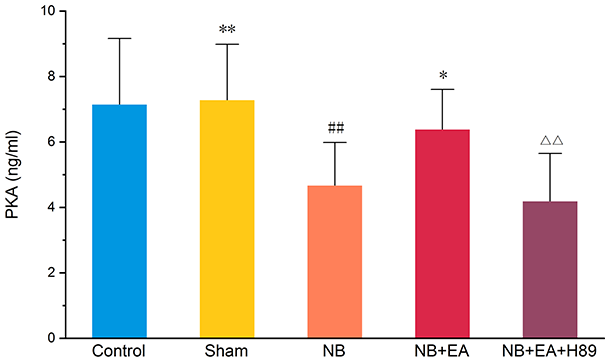
Concentration of protein kinase A (PKA), measured by enzyme-linked immunosorbent assay (ELISA), in control, sham, NB (neurogenic bladder), NB + EA (electroacupuncture), and NB + EA + H89 groups. Values are expressed as mean ± SEM. ##p < 0.01 versus control group. *p < 0.05 versus NB group. △△p < 0.01 versus NB + EA group.
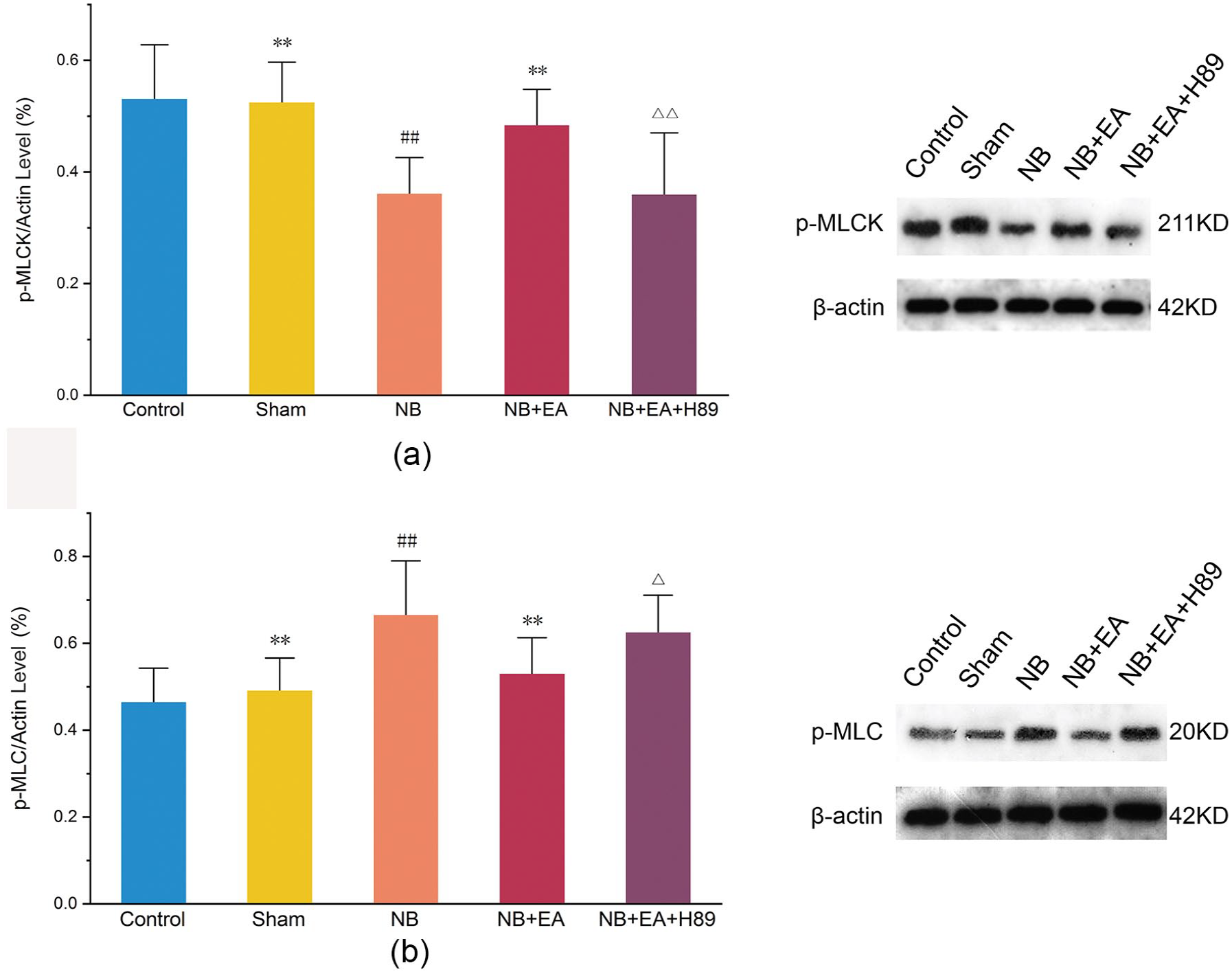
Protein expression of phosphorylated myosin light chain kinase (p-MLCK) and p-MLC, measured by Western blotting, in control, sham, NB (neurogenic bladder), NB + EA (electroacupuncture), and NB + EA + H89 groups: (a) Quantification of p-MLCK expression, and bands of p-MLCK expression, in detrusor tissue. (b) Quantification of p-MLC expression, and bands of p-MLC expression, in detrusor tissue. Values are expressed as mean ± SEM. ##p < 0.01 versus control group. **p < 0.01 versus NB group. △p < 0.05, △△p < 0.01 versus NB + EA group.
EA improved bladder function by activating PKA-p-MLCK/p-MLC signaling
EA reversed the inhibition of PKA-p-MLCK/p-MLC signaling (Figure 3). Increased levels of PKA, increased expression of p-MLCK, and decreased expression of p-MLC were observed in the NB + EA group compared with the NB group.
Inhibition of PKA impaired the beneficial effect of EA by blocking PKA-p-MLCK/p-MLC signaling
To further validate the key role of PKA-p-MLCK/p-MLC signaling in the beneficial effect of EA on bladder function, H89 (a PKA inhibitor) was administered to EA-treated rats. The results showed that the administration of H89 to EA-treated rats reversed EA-associated improvements in urodynamic parameters by inhibiting downstream p-MLCK/p-MLC signaling (Figure 4).
Discussion
In the current study, we demonstrated a positive effect of EA on excessive contraction of the bladder detrusor in NB model rats and revealed an important role for the PKA-p-MLCK/p-MLC signaling pathway in mediating this effect.
Accumulating clinical evidence has confirmed that EA can improve the bladder function of patients with NB after SCI above the sacral level.11 –13 Preclinical studies have revealed that acupuncture can alleviate the deleterious effects of SCI in NB model rats.3,12,15 Consistent with previous findings, our study demonstrated that EA was effective at treating the excessive contraction of the bladder detrusor in NB model rats.
Excessive contraction of the bladder detrusor is regarded as the core pathological basis of NB secondary to SCI. 3 Interventions addressing the excessive contraction of the bladder detrusor have been reported in prior studies. 22 Giannantoni et al. 6 and Elkelini et al. 5 demonstrated that the injection of onabotulinum toxin-A (an acetylcholine receptor antagonist) into patients exhibiting excessive contraction of the bladder detrusor can significantly increase the maximum bladder volume. Chancellor et al. 4 reported that the administration of a muscarine receptor antagonist into the bladder can inhibit the excessive contraction of the bladder detrusor induced by the excessive release of acetylcholine. The aforementioned evidence suggests that interventions addressing the excessive contraction of the bladder detrusor are effective in the treatment of NB. Our findings revealed that EA also improved bladder function through modulation of the excessive contraction of the bladder detrusor. Notably, EA could be considered a more cost-effective and safer approach than pharmacological interventions.
Accumulating evidence has confirmed that there is a close connection between PKA and smooth muscle relaxation. Prior studies have found that PKA can phosphorylate MLCK and that phosphorylation of MLCK can affect the affinity of MLCK for the Ca2+–CaM complex, making it insensitive to calcium ions, thereby reducing the phosphorylation of downstream MLC and causing smooth muscle relaxation.8,13,23 In addition, the effect of PKA in promoting smooth muscle relaxation may be related to myosin light chain phosphatase (MLCP). 24 PKA can also phosphorylate the myosin phosphate targeting subunit 1 (MYPT1) of MLCP and activate MLCP. 25 Activated MLCP can dephosphorylate MLC, and dephosphorylation of MLC reduces ATP decomposition, leading to smooth muscle relaxation. 26 The aforementioned studies show that the PKA-p-MLCK/p-MLC signaling pathway plays a very important role in regulating smooth muscle relaxation.
We observed that, in addition to urodynamic indicators, PKA content in the detrusor muscle was significantly lower in the model group than in the normal group, indicating that PKA is involved in the regulation of detrusor contraction. The PKA content in the NB + EA group was significantly higher than that in the model group, indicating that the PKA pathway may be a key signaling pathway through which EA inhibits detrusor contraction. To confirm this possibility, we directly administered a PKA inhibitor to NB model rats prior to EA, and found that the EA-induced detrusor relaxation was blocked by the PKA inhibitor.
In addition, our results showed that the phosphorylation levels of MLCK and MLC, which are downstream of PKA, were also changed (increased expression of p-MLCK and decreased expression of p-MLC) in the NB model rats, which indicates that the PKA-p-MLCK/p-MLC signaling pathway plays an important role in the excessive contraction of the bladder detrusor. After 7 days of acupuncture treatment, the changes in PKA-p-MLCK/p-MLC signaling and the excessive contraction of the bladder detrusor were reversed; however, these treatment effects were abolished by PKA inhibition. Taken together, our results reveal that EA can effectively improve detrusor relaxation by activating the PKA-p-MLCK/p-MLC signaling pathway.
In the present study, we chose to administer EA at CV3, SP6, BL32, and GV14 because these are the most commonly used traditional acupuncture point locations in the treatment of NB. 18 The tissues at CV3 are innervated by the T12/L1 segments of the spinal cord, while those at SP6 are innervated by the S2 segment. BL32 is located in the second posterior sacral foramen, below the second sacral nerve branch. Acupuncture at SP6 and BL32 can stimulate the second sacral nerve and regulate the S2-S4 parasympathetic center. Collectively, acupuncture at these three sites in conjunction with GV14 has been shown to improve excessive contraction of the bladder detrusor muscle in rats, which may be related to the modulation of the sympathetic and/or parasympathetic nerves innervating the bladder detrusor.19,20
Conclusion
In conclusion, this study found for the first time (to our knowledge) that EA can alleviate NB after suprasacral SCI through the PKA-MLCK-MLC signaling pathway. EA prevented noninhibitory contraction of the detrusor muscle and improved the function of the bladder, providing preclinical evidence that acupuncture might be used to treat detrusor hyperreflexia. Considering that Ca2+ also plays an important role in smooth muscle contraction, it will be of importance to reveal the roles of Ca2+ in the process of EA regulating the concentration of bladder detrusor muscle in future studies.
Footnotes
Acknowledgements
We would like to thank all the participants in this study.
Contributors
QL and Q-LQ contributed equally to this article.
Author contributions
Study design: K.A., L.Z. Project administration: K.A. Experimental implementation: Q.R.Q., Q.L., M.X. and J.S.L. Data analysis: F.Q., X.Q.Y. and Q.R.Q. Paper writing: Q.R.Q., Q.L., and K.A. Paper review and editing: H.Z. Experimental support: K.A. and L.Z. All authors read and approved the final version of the manuscript accepted for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific Research Project of Hunan Provincial Department of Education (grant no. 21B0369), National Natural Science Foundation of China (grant no. 81874510), Natural Science Foundation of Hunan Province (grant no. 2022JJ40301) and Key Scientific Research Projects of Hunan Administration of Traditional Chinese Medicine (grant no. 201704).
