Abstract
Background:
Postoperative gastrointestinal dysfunction (PGD) is one of the most common complications among patients who have undergone thoracic surgery. Acupuncture has long been used in traditional Chinese medicine to treat gastrointestinal diseases and has shown benefit as an alternative therapy for the management of digestive ailments. This study aimed to explore the therapeutic effectiveness of acupuncture as a means to aid postoperative recovery of gastrointestinal function in patients undergoing thoracoscopic surgery.
Methods:
In total, 112 patients aged 18–70 years undergoing thoracoscopic surgery between 15 June 2022 and 30 August 2022 were randomized into two groups. Patients in the acupuncture group (AG) first received acupuncture treatment 4 h after surgery, and treatment was repeated at 24 and 48 h. Patients in the control group (CG) did not receive any acupuncture treatment. Both groups received the same anesthetic protocol. Ultrasound-guided thoracic paravertebral block (TPVB) was performed in the paravertebral spaces between T4 and T5 with administration of 20 mL of 0.33% ropivacaine. All patients received patient-controlled intravenous analgesia (PCIA) after surgery.
Results:
Median time to first flatus [interquartile range] in the AG was significantly less than in the CG (23.25 [18.13, 29.75] vs 30.75 [24.13, 45.38] h, p < 0.001). Time to first fluid intake after surgery was significantly less in the AG, as compared with the CG (4 [3, 7] vs 6.5 [4.13, 10.75] h, p = 0.003). Static pain, measured by visual analog scale (VAS) score, was significantly different on the third day after surgery (p = 0.018). Dynamic pain VAS scores were lower in the AG versus CG on the first three postoperative days (p = 0.014, 0.003 and 0.041, respectively).
Conclusion:
Addition of acupuncture appeared to improve recovery of postoperative gastrointestinal function and alleviate posteoperative pain in patients undergoing thoracoscopic surgery. Acupuncture may represent a feasible strategy for the prevention of PGD occurrence.
Trial registration number:
ChiCTR2200060888 (Chinese Clinical Trial Registry)
Introduction
Lung cancer is the leading cause of cancer-related death and one of the most diagnosed cancers in the world. 1 Surgical intervention remains the main treatment for early lung cancer, and video-assisted thoracoscopic surgery (VATS) has emerged as an alternative to thoracotomy for the diagnosis and treatment of pulmonary nodules over the past few years. 2 Enhanced recovery after surgery (ERAS) protocols have the advantages of faster recovery, fewer complications, shorter hospitalizations and lower medical costs, and have been widely implemented in various surgical fields. 3 Early postoperative food intake is one of the specific measures within ERAS implementation. 4 Therefore, maintaining normal gastrointestinal function is considered to be an essential part of ERAS after thoracic surgery.
Postoperative gastrointestinal dysfunction (PGD) is one of the most common complications among patients who have undergone thoracic surgery, and it is often associated with administration of anesthesia, surgical retraction, postoperative fasting, use of opioid analgesics, and prolonged bed rest.5 –7 In the past few decades, some measures related to the promotion of postoperative gastrointestinal functional recovery have been used, including conventional treatments, such as prokinetic drugs;8,9 however, their therapeutic efficacy is less than ideal and their use is accompanied by various side effects. An appropriate measure that is effective and safe, and easily accepted by patients, would be most desirable for PGD treatment.
Acupuncture has long been used in traditional Chinese medicine (TCM) to treat gastrointestinal diseases and has gained popularity in Western countries. It is beneficial as an alternative therapy for the management of digestive ailments, such as postoperative nausea and vomiting (PONV),10,11 chemotherapy-induced nausea12,13 and postoperative ileus, 14 and other functional disorders including constipation, 15 diarrhea 16 and gastroesophageal reflux, 17 with minimal side effects. Clinical evidence suggests that acupuncture can promote the recovery of gastrointestinal function after major surgery, and both manual acupuncture and electroacupuncture have been reported to have preventive and therapeutic value.18 –20 However, the mechanism by which acupuncture promotes gastrointestinal peristalsis is still not clear. Acupuncture has been found to enhance gastric motility via the efferent parasympathetic pathway 21 and the N-methyl-D-aspartate (NMDA) receptor. 22 Previous studies have mostly been implemented in patients undergoing gastric or colorectal surgery.18,23,24 It is unclear whether acupuncture is equally effective in thoracic surgery; therefore, a prospective, randomized trial is needed to evaluate its potential role in this setting. The aim of this study was to investigate the therapeutic effectiveness of acupuncture for postoperative recovery of gastrointestinal function in patients undergoing thoracoscopic surgery. We anticipated that the results could help inform future decision-making as to whether to include acupuncture in ERAS protocols for lung surgery.
Methods
Recruitment and ethics
This study was a single-center, randomized, open-label trial. Patients scheduled for VATS lung surgeries between 15 June 2022 and 30 August 2022 at the Sun Yat-sen University Cancer Center (SYSUCC) were enrolled. The trial was approved by the Ethics Committee of SYSUCC. The study was prospectively registered at www.chictr.org.cn on 13 June 2022 (ChiCTR2200060888). All participants received information about the study and were screened in accordance with the eligibility criteria. All participants signed informed consent before enrollment.
Inclusion and exclusion criteria
Patients undergoing VATS lung surgeries (wedge resection, segmentectomy and lobectomy) were enrolled in the study. To meet the inclusion criteria, patients needed to: (1) have been diagnosed with a benign nodule or tumor without metastasis; (2) be aged between 18 and 70 years; and (3) be American Society of Anesthesiologists (ASA) class I–III. Exclusion criteria included: conversion to open surgery; poor preoperative pulmonary function; pre-existing severe cardiovascular, liver or kidney diseases; cognitive dysfunction or psychological disorders with inability to communicate or cooperate; gastrointestinal disorders; history of PONV; treatment with analgesic drugs; infection around the planned sites of acupuncture needling; coagulation disorders; and unwillingness to sign informed consent. If patients were lost to follow-up, cases were withdrawn from the study (per protocol analysis).
Randomization and blinding
The allocation sequence of the study in a 1:1 ratio was generated by Internet-based randomization software (http://www.randomization.com). Eligible patients were randomly allocated to either the acupuncture group (AG) or the control group (CG). Intraoperative and postoperative data were collected by investigators. Acupuncture was implemented by a qualified acupuncturist with more than 20 years of experience who holds a China Acupuncturist Certification.
By design, neither the participants nor the acupuncture provider could be blinded to treatment group allocation. However, the investigators, anesthetists and statisticians were unaware of study group assignments throughout the entire trial.
Intervention
The following traditional acupuncture point locations were selected for needling based on TCM theory and previous studies: ST36 (Zusanli), ST37 (Shangjuxu), PC6 (Neiguan) and LI4 (Hegu). Exact locations and depth of insertions were according to the 2021 National Standards of Nomenclature and Location of Acupuncture Points (GB/T12346-2021). 25
Patients in the AG first received acupuncture treatment at 4 h after surgery, and treatment was repeated on the next 2 days. Acupuncture was performed on patients in a supine position. The treatment sites were disinfected with 75% alcohol prior to needle insertion. Sterile, single-use, stainless steel acupuncture needles (0.30 × 60 mm, Suzhou Medical Supplies Factory Co., Ltd., China) were used. After insertion to a depth of 30–60 mm at ST36/ST37 or 10–30 mm at PC6/LI4, needles were stimulated through lifting-thrusting and twirling-twisting manipulations, resulting in the acupuncturist’s sensation of de qi. Needles were kept inserted for 30 min without any further stimulation. Patients in the CG did not receive any acupuncture treatment. Both groups followed standard ERAS protocols based on the guidelines for enhanced recovery for lung surgery. 26 ERAS protocols in this study included the following: (1) smoking cessation at least 4 weeks before surgery; (2) avoidance of sedatives to reduce anxiety preoperatively; (3) administration of intravenous antibiotics within the 60 min prior to skin incision; (4) maintenance of normothermia with convective active warming devices perioperatively; (5) use of lung-protective strategies during single-lung ventilation; (6) combination of paravertebral blockade and general anesthetic techniques; and (7) use of a single chest tube instead of two after anatomical lung resection.
Patients in both groups received the same anesthetic protocol. Anesthesia was induced with intravenous 2% lidocaine (1 mg/kg), propofol (2 mg/kg), rocuronium (0.6 mg/kg) and dexamethasone (5 mg). After insertion of a double-lumen tube, anesthesia was maintained with target-controlled infusion of propofol (3 μg/mL) using the Marsh model (Alaris PK Syringe Pump, Carefusion, Somerset, UK) and remifentanil (0.1 µg/kg/min). Ultrasound-guided thoracic paravertebral block (TPVB) was performed in the paravertebral spaces between T4 and T5 with 20 mL of 0.33% ropivacaine, which was administered when patients were laterally positioned. All patients received patient-controlled intravenous analgesia (PCIA) containing sufentanil (2 µg/kg) and palonosetron (0.5 mg diluted in normal saline to achieve a volume of 100 mL). The PCIA device was set to deliver a 2 mL/h background infusion and 1 mL on-demand bolus, with a 10-min lockout time. If patients still complained of a visual analog scale (VAS) pain score > 5 after boluses, an additional intravenous injection of parecoxib (40 mg) was administered as a rescue medication.
Blood pressure, heart rate and oxygen saturation were routinely recorded every 5 min after the patients entered the operating room. Target mean blood pressure was within 30% of baseline. Lung-protective strategies were used during single-lung ventilation and very restrictive or liberal fluid regimes were avoided in favor of euvolemia.
Measurements
The primary outcome of this study was time to first postoperative flatus. 18 This time period was measured as the interval between recovery from anesthesia and the first passage of flatus. The secondary outcomes included the time to first postoperative defecation, time to first fluid intake, time to first food intake, and time to first ambulation. 18 Other secondary outcomes included postoperative pain scores, nausea, vomiting, 19 and other relevant complications (e.g. anorexia, insomnia, dizziness). All outcome information was obtained through the patients or their family members and recorded by investigators. Postoperative pain was evaluated using the VAS (0–10, where 0 = no pain and 10 = worst pain).
Sample size calculation
Power Analysis and Sample Size (PASS) software version 15 (2017) (NCSS LLC, Kaysville, UT, USA), accessible at ncss.com/software/pass, was used to estimate the required sample size based on the primary outcome of time to first postoperative flatus. Based on our previous clinical practice, the mean time to first postoperative flatus after thoracoscopic surgery was 31.5 h, with a standard deviation of 14.6 h. It was estimated that a sample size of 52 patients per group would be needed to detect a 30% improvement in the time to first postoperative flatus, assuming α = 0.05, β = 0.1 and power = 0.9. Estimating a 10% dropout rate, we aimed to recruit a minimum of 116 subjects.
Statistical analysis
Data were analyzed using the Statistical Package for the Social Sciences (SPSS) version 26.0 (SPSS Inc., Chicago, IL, USA). Normal distribution of continuous variables was first evaluated using the Shapiro–Wilk test. Data were expressed as frequencies for categorical variables and mean ± standard deviation or median (interquartile range) for continuous variables, as appropriate. All data were compared between the groups using the chi-square test or Fisher’s exact test for categorical variables and Student’s t-test or Mann–Whitney U test for continuous variables, as appropriate. A p-value less than 0.05 was considered statistically significant.
Results
A total of 116 patients were assessed for eligibility between 15 June 2022 and 30 July 2022. Two dropped out because of changes in the surgery protocol, one refused acupuncture, and one patient was lost to follow-up. As a result, 112 patients were included after randomization into either the AG (n = 56) or the CG (n = 56). The study flowchart is shown in Figure 1. There were no differences in baseline characteristics or surgical parameters between the groups (Table 1).
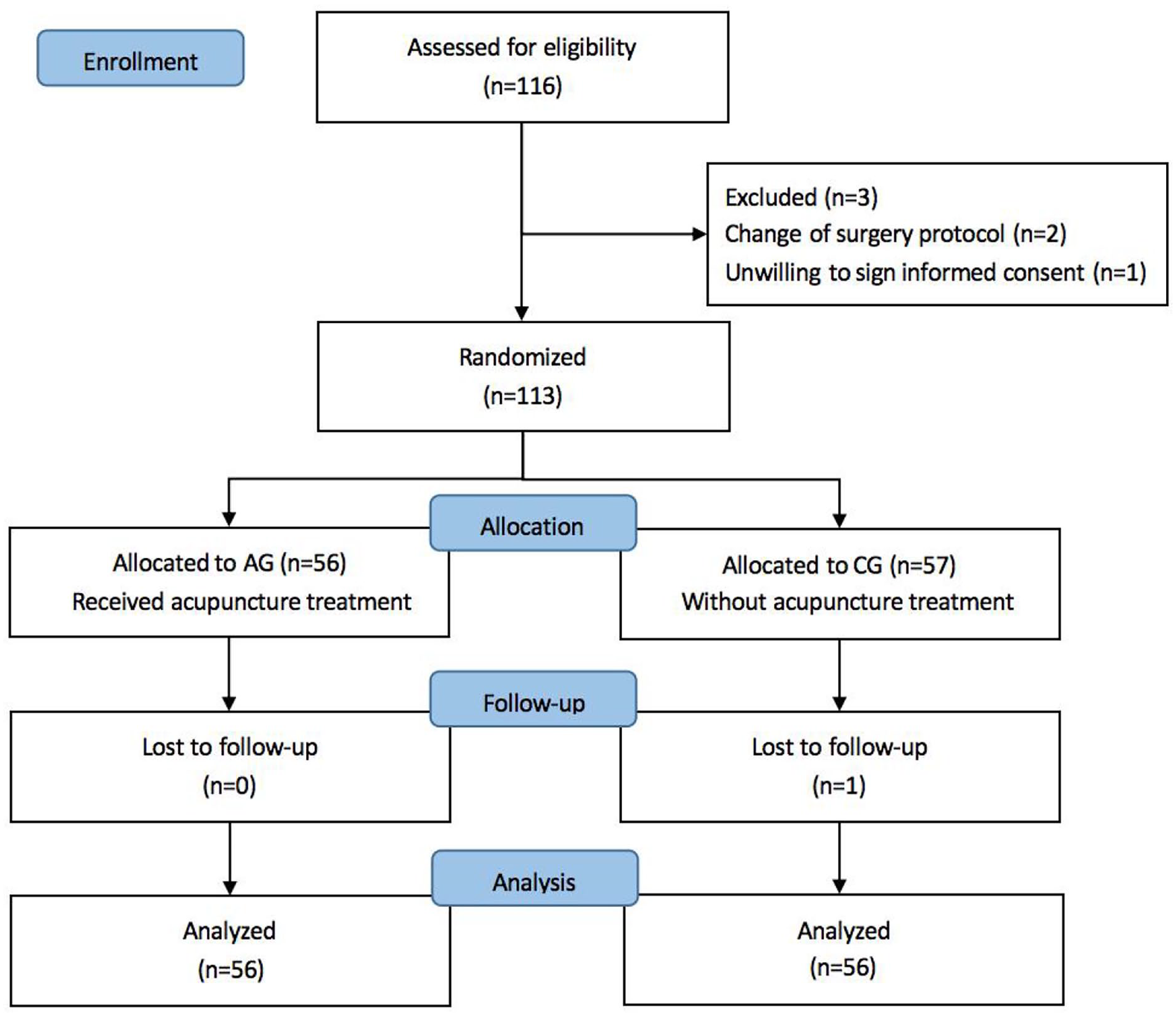
Flowchart of patient selection.
Baseline characteristics and surgical parameters of patients.
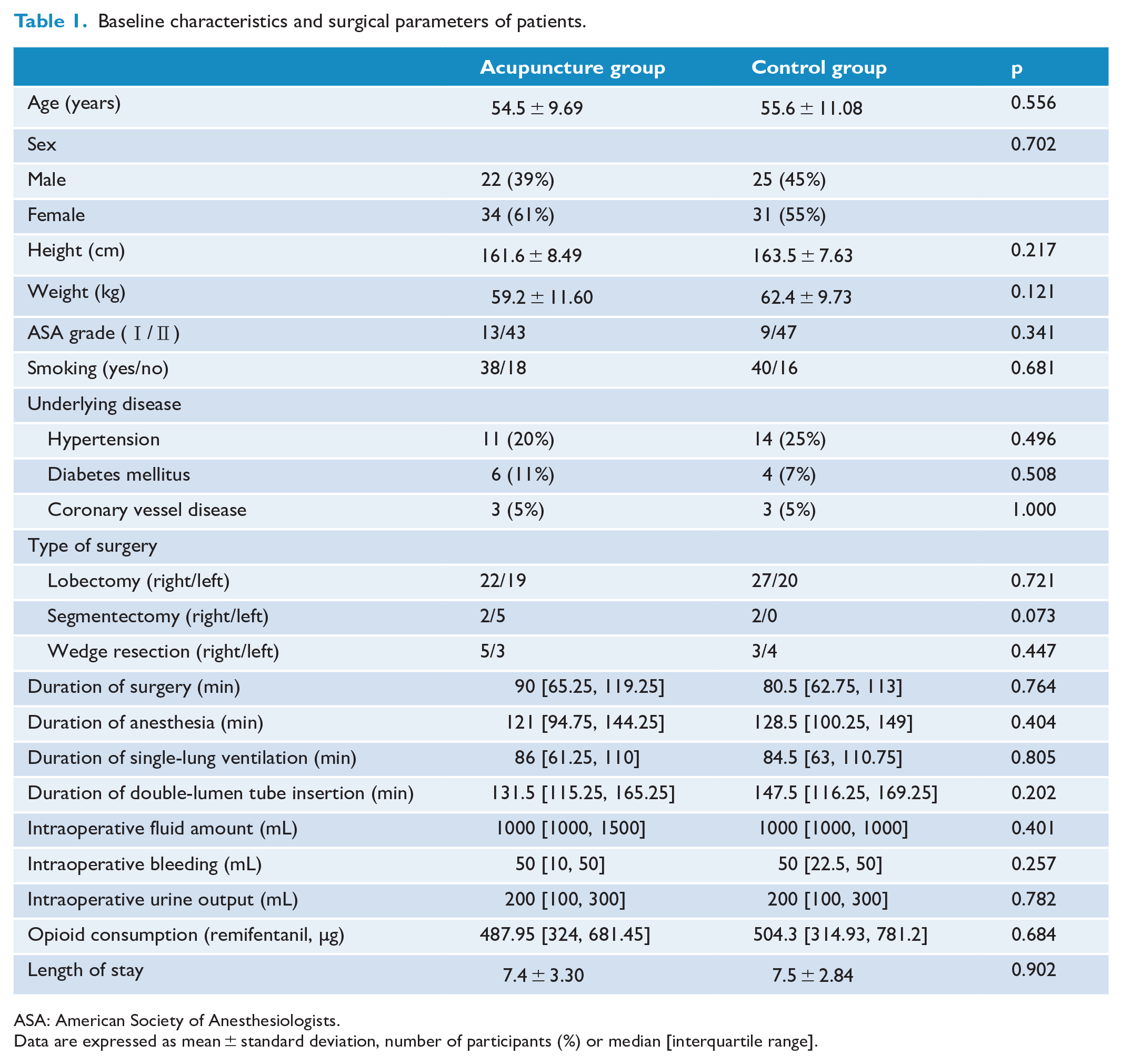
ASA: American Society of Anesthesiologists.
Data are expressed as mean ± standard deviation, number of participants (%) or median [interquartile range].
For the primary endpoint, shown in Table 2, median time to first flatus [interquartile range] in the AG was significantly less than in the CG (23.25 [18.13, 29.75] vs 30.75 [24.13, 45.38] h, p < 0.001). Time to first defecation did not differ between the two groups, and neither did time to first ambulation, which was 41 [25, 45] and 41.45 [35.25, 45] h in the AG and the CG, respectively (p = 0.383). Time to first fluid intake after surgery was significantly lower in the AG, as compared with the CG (4 [3, 7] vs 6.5 [4.13, 10.75] h, p = 0.003). However, there was no difference between two groups in the time to first food intake (p = 0.445).
Primary outcome and selected secondary outcomes of patients.
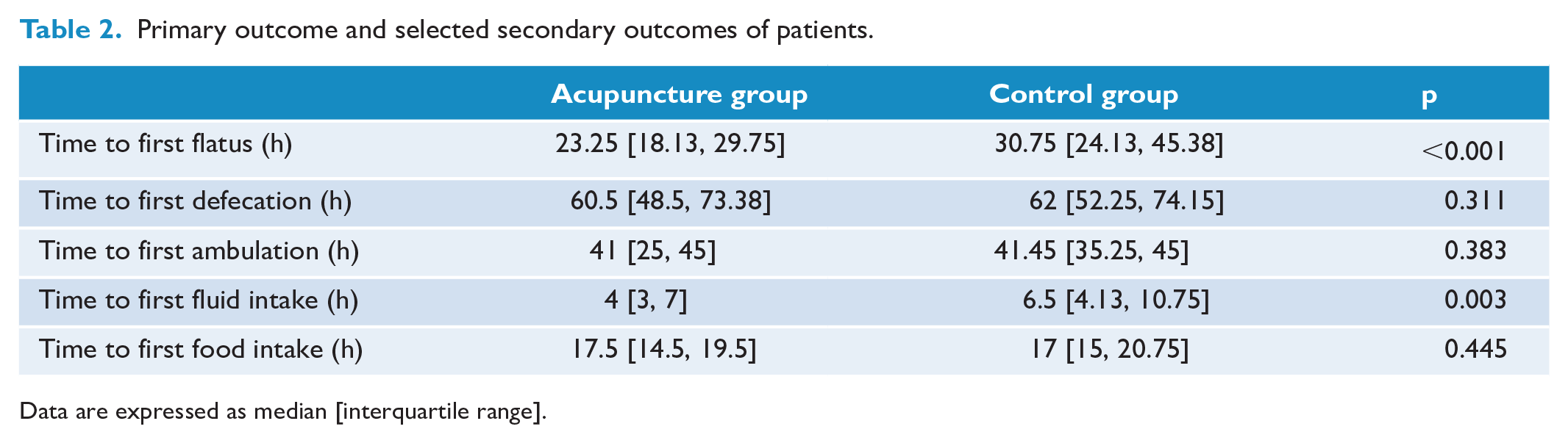
Data are expressed as median [interquartile range].
Table 3 shows the other clinical outcomes of the patients. There were no differences in PONV between groups at 0–24, 24–48 and 48–72 h after surgery. Moreover, anorexia, insomnia and dizziness did not differ between the two groups within the first 3 days after surgery. Regarding postoperative static pain (measured by VAS), no differences were observed between the groups on the first and second day, but there was a significant difference on the third day (p = 0.018). Dynamic pain VAS scores were lower in the AG compared with the CG on each of the first three postoperative days (p = 0.014, 0.003 and 0.041, respectively). Severe pain (VAS score ⩾ 7) with 24 h of surgery occurred in 14 of 56 patients (25%) in the CG, as compared with 3 of 56 (5%) in the AG. On the second and third postoperative days, a VAS score ⩾ 7 was less common in the AG than in the CG (21% vs 46% and 11% vs 21%, respectively). The actual PCIA boluses and rescue analgesia use in the first 24 h after surgery were comparable in both groups, but there were significant differences at 24–48 and 48–72 h between groups (p < 0.05).
Other secondary outcomes of patients.
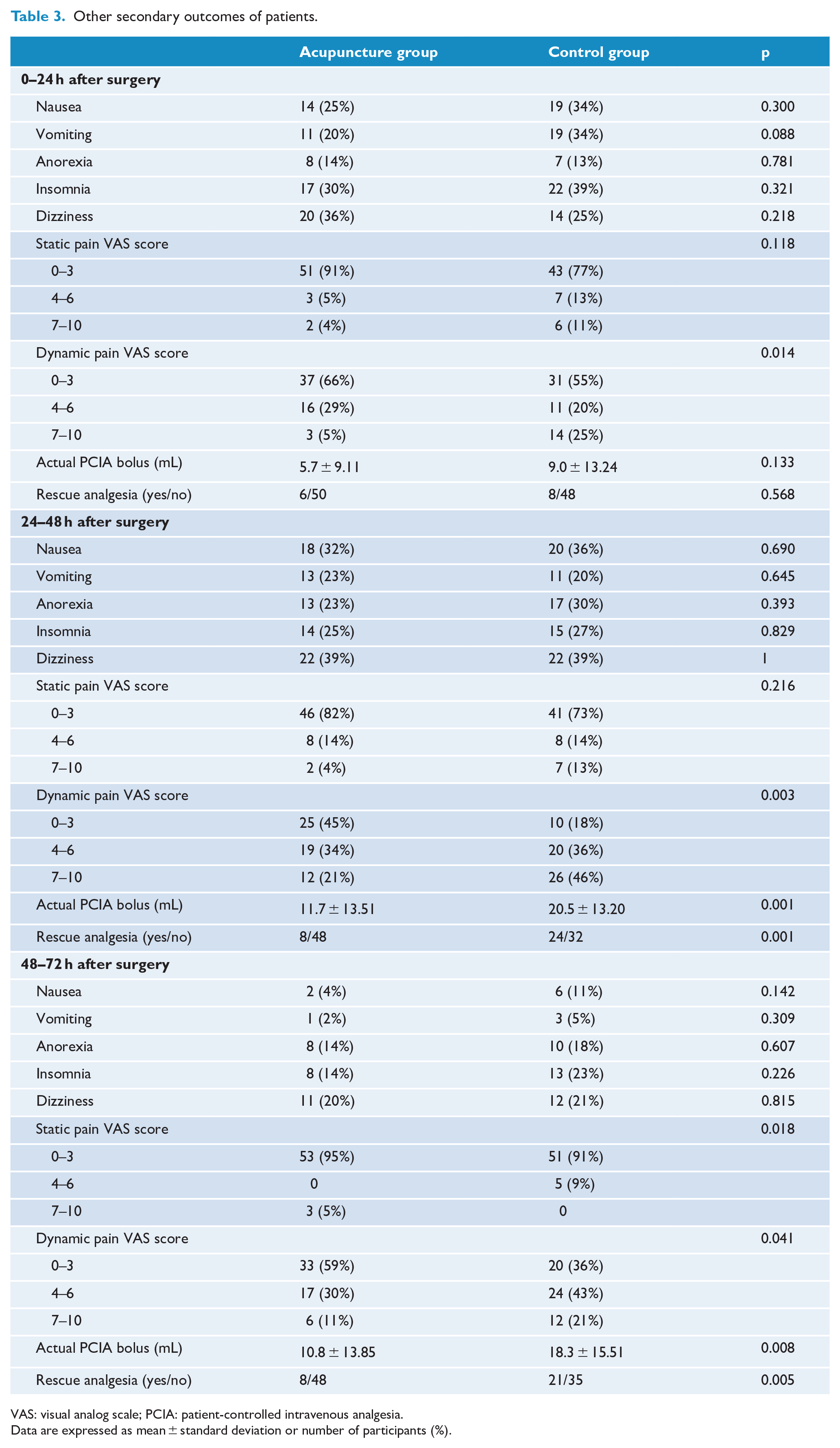
VAS: visual analog scale; PCIA: patient-controlled intravenous analgesia.
Data are expressed as mean ± standard deviation or number of participants (%).
No serious acupuncture-related adverse events were reported throughout the study. Some patients reported normal bodily sensations of acupuncture, such as slight pain and numbness, which disappeared after acupuncture and were considered transient and acceptable.
Discussion
In this prospective randomized controlled trial, we found that patients receiving acupuncture at ST36/ST37/PC6/LI4 had a shorter time to flatus after VATS lung surgeries, compared to those not receiving acupuncture. This is consistent with a prior report by Yang et al. 27 Meanwhile, time to first fluid intake was also shorter in patients receiving acupuncture. No difference was found between groups in time to first defecation, although significant differences in this parameter have been reported by others. 23 Patients in the AG exhibited lower dynamic VAS scores within 3 days of surgery, while static VAS scores differed between groups only on the third postoperative day. Regarding the VAS pain scores, our results are consistent with the study by Li et al., 19 but not that of Yang et al. 27
Acupuncture at different sites may have variable therapeutic effects. For gastrointestinal (GI) disorders, LI4, PC6, ST36 and ST37 are commonly chosen traditional acupuncture point locations. Acupuncture at LI4 is often used in the treatment of large intestine-related conditions and as an alternative to non-pharmacologic analgesic methods. 28 Acupuncture at PC6 has been shown to regulate GI function and increase gastric motility. 29 Stimulation at PC6 is also associated with the relief of PONV due to the use of opioids and other medications. 30 Acupuncture at ST36 improves blood flow distribution in the GI tract, regulates motilin and somatostatin production, and promotes peristalsis through regulation of nitric oxide (NO) and angiotensin.31,32 Needling at ST37 is often used in conjunction with ST36 stimulation to treat large intestine-related conditions. 33 Some studies have demonstrated that acupuncture may have regulatory effects on GI motility, especially that of the stomach and colon by stimulation at ST36 in normal rats.34,35 Acupuncture at ST36 has been found to enhance gastric motility via the efferent parasympathetic pathway 21 and NMDA receptors, which are associated with vagal afferent nerve fibers that play a critical role in mediating gastric motility. 22
In this study, there was no difference in time to first defecation between groups, possibly because food intake was minimal on the first and second days after surgery. There was also no difference in PONV between the groups in this study. To achieve sufficient pain control, opioids may be used during or after surgery, which may lead to continuous PONV for a few days.
Pain following thoracic surgery is often severe and can be due to retraction, injury to the intercostal nerves, or irritation of the pleura or intercostal bundles by chest tubes. 26 A standardized multimodal analgesic strategy was implemented in our study including TPVB analgesia and PCIA. Static pain VAS scores in the first 2 days after surgery were low, most likely owing to this multimodal analgesic strategy. After the PCIA pumps were withdrawn, the static VAS scores showed differences between groups on the third day. Patients in the AG demonstrated lower VAS scores, probably due to the known analgesic properties of acupuncture at LI4, PC6 and ST36. Acupuncture at LI4 and PC6 was effective in relieving postoperative pain and upregulated expression of γ-aminobutyric acid (GABA) released by neurons and astrocytes. 36 Acupuncture at ST36 can induce analgesia and a concomitant increase in the degranulation ratios of mast cells and collagen fibers may contribute to the analgesic effect in rat. 37 Previous studies have also shown that peripheral afferent nerve fibers including both A and C fibers contribute to acupuncture analgesia. 38 Although adequate analgesia may decrease static pain, it was evident that dynamic pain was unavoidable, likely due to chest tubes irritating the pleura when patients coughed or ambulated. In our study, dynamic pain VAS scores in the AG were lower than the CG on the first, second and third days after surgery. Acupuncture effectively alleviated dynamic pain in postoperative patients undergoing thoracic surgery, especially those with high-intensity pain.
There are several limitations to our study. First, all participants receiving acupuncture were Asian. Studies recruiting participants of other ethnicities/races should be proposed to gain more clinical evidence. Second, the study lacked a sham acupuncture control group and, as a result, blinding of the study subjects was not feasible. Third, the follow-up duration was 3 days after surgery. Given postoperative GI discomfort may last for a prolonged time, studies with a longer follow-up period are needed. In addition, we did not perform an intention-to-treat analysis. One patient lost to follow-up in the CG was not included in our per-protocol analysis, which may have a slight impact on the results. Despite these limitations, the study adds to our knowledge regarding the general effects of acupuncture on postoperative recovery and provides novel evidence in support of the inclusion of acupuncture into ERAS protocols.
In conclusion, acupuncture at ST36/ST37/PC6/LI4 appeared to promote postoperative recovery of GI function. Application of acupuncture, which can also reduce postoperative pain, may be recommended for ERAS protocols in lung surgery patients.
Footnotes
Acknowledgements
The authors thank the Department of Thoracic Surgery at Sun yat-sen University Cancer Center for providing the research facilities for screening volunteers and implementing acupuncture. They also thank Dr Shiyang Kang for his kind help in grammatical editing and writing advice.
Contributors
YZ, CO, HO and SW devised and wrote the study protocol. All authors took part in conducting the study and collecting data. YZ, CO and HO contributed to the data analysis. All authors read and approved the final version of the manuscript accepted for publication.
Availability of data and materials
The dataset of the current study has been uploaded to the Research Data Deposit (RDD) system of Sun Yat-sen University Cancer Center. All datasets and materials are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the H.O.’s Clinical Medical Scientists Project of Sun Yat-sen University Cancer Center (PT09090101).
Ethical approval and consent to participate
The study protocol and informed consent were approved by the Ethics Committee of Sun Yat-sen University Cancer Center. All participants gave their informed consent for inclusion before participating in the study.
Consent for publication
Not applicable.
