Abstract
Background:
This study aimed to investigate the effects of electroacupuncture (EA) on cognitive recovery and synaptic remodeling in a rat model of middle cerebral artery occlusion (MCAO) followed by reperfusion and explore the possible mechanism.
Method:
Focal cerebral ischemia was modeled in healthy adult Sprague-Dawley rats by MCAO. The MCAO rats were classified into four groups: sham, MCAO, MCAO + GB20 (receiving EA at GB20) and MCAO + NA (receiving EA at a “non-acupoint” location not corresponding to any traditional acupuncture point location about 10 mm above the iliac crest). Neurological deficit scores and behavior were assessed before and during the treatment. After intervention for 7 days, the hippocampus was dissected to analyze growth-associated protein (GAP)-43, synaptophysin (SYN) and postsynaptic density protein (PSD)-95 expression levels by Western blotting. Bioinformatic analysis and primary hippocampal neurons with calcium-voltage gated channel subunit alpha 1B (CACNA1B) gene overexpression were used to screen the target genes for EA against MCAO.
Results:
Significant amelioration of neurological deficits and learning/memory were found in MCAO + GB20 rats compared with MCAO or MCAO + NA rats. Protein levels of GAP-43, SYN and PSD-95 were significantly improved in MCAO + GB20–treated rats together with an increase in the number of synapses in the hippocampal CA1 region. CACNA1B appeared to be a target gene of EA in MCAO. There were increased mRNA levels of CACNA1B, calmodulin (CaM), Ca2+/calmodulin-dependent protein kinase type II (CaMKII) and cyclic adenosine monophosphate response element binding (CREB) and increased phosphorylation of CaM, CaMKII and CREB in the hippocampal region in MCAO + GB20 versus MCAO and MCAO + NA groups. CACNA1B overexpression modulated expression of the CaM–CaMKII–CREB axis.
Conclusion:
EA treatment at GB20 may ameliorate the negative effects of MCAO on cognitive function in rats by enhancing synaptic plasticity. EA treatment at GB20 may exert this neuroprotective effect by regulating the CACNA1B–CaM–CaMKII–CREB axis.
Introduction
Ischemic stroke is one of the most serious manifestations of cerebrovascular disease. 1 It is the fifth leading cause of death and one of the leading causes of disability in the United States. In contrast to the downward trend of incidence in high-income countries, more than 2 million new cases occur each year in China, with a negative effect on disability-adjusted life years. 2 Due to the aging population and the continued high prevalence of hypertension, the burden of hospitalization caused by ischemic stroke is expected to increase further. Ischemic stroke can initiate several detrimental pathological cascades, such as apoptosis and calcium overload, usually causing cognitive impairment related to memory and attention.3,4 Many lines of evidence indicate that synaptic plasticity is necessary for information storage in the brain, providing insight into the association between brain ischemia and memory deficits.5,6 The pathology and mechanisms of stroke are complex, and the cellular signaling pathways involved in the recovery of cognitive function recovery are diverse.7,8 Ca2+/calmodulin-dependent protein kinase type II (CaMKII) necessitates the binding of calmodulin (CaM) complex to release autoinhibition and regulate cyclic adenosine monophosphate (cAMP) response element binding (CREB) gene expression.9,10 Accordingly, CaM–CaMKII–CREB with CaM and CaMKII are vital signaling molecules in hippocampal neurons that participate in learning and memory. 11
Cognitive rehabilitation is an important task after a stroke. Clinical trials have revealed that electroacupuncture (EA), when used as one component of a comprehensive rehabilitation strategy, achieves positive effects in poststroke rehabilitation. 12 Synaptic plasticity may be the potential bridge between EA and poststroke rehabilitation treatment. EA is a form of acupuncture therapy. 13 EA may mediate synaptic plasticity in rodents exposed to cerebral ischemia. 14 EA is characterized by parameter objectification with good reproducibility and the ability to standardize frequency, intensity and duration. 15 Therefore, EA has attracted considerable attention and is commonly recommended for clinical use in China and other countries.16,17 Clinical research studies support the role of EA as a valid therapy for various diseases, such as anxiety disorders, chronic low back pain and major depressive disorder, without significant adverse effects.18,19 Recently, EA combined with Western medicine has been used for the treatment of hypertension. 20 Although a few studies have explored the mechanisms of action underlying the effects of EA on cognitive impairment, 21 much still remains to be elucidated. We speculated that the use of current neurobiological theories to study the potential impact of acupuncture (and its mechanisms of action) on disease processes, and its interpretation in the context of mainstream medical concepts and terms, could further aid the evidence-based integration of EA into mainstream medicine. The aim of this study was to investigate the effect of EA on poststroke cognitive impairment using the middle cerebral artery occlusion (MCAO) rat model.
Methods
Bioinformatics prediction
Genes related to EA and MCAO were collected from GeneCards (https://www.genecards.org/). A Venn diagram of overlapping genes was generated using the online Venny 2.1 interactive tool (https://bioinfogp.cnb.csic.es/tools/venny/index.html). The gene and protein names were unified by UniProt (https://www.uniprot.org/). These proteins were then interpreted in a biological context using STRING 11.0 (www.string-db.org) and OmicShare Tools (https://www.omicshare.com/tools/).
Establishment of the MCAO rat model
All animal procedures were performed according to the guidelines of the Institutional Animal Care and Use Committee of Jinshan Hospital of Fudan University regarding the protection of animals used for experimental and other scientific purposes. Healthy male Sprague-Dawley rats (250–280 g, Laboratory Animal Center of Southern Medical University) were freely fed food and water in a 12 h light/dark-cycle environment. Rats were assigned to one of four groups: (1) the MCAO group (n = 10), which underwent carotid artery embolization; (2) the sham group (n = 10), which underwent a sham procedure without embolization; (3) the MCAO + NA group (n = 10), which underwent carotid artery embolization and EA at a site not corresponding to any traditional acupuncture point location (a.k.a. “non-acupoint”); and (4) the MCAO + GB20 group (n = 10), which underwent carotid artery embolization followed by EA at GB20 (Fengchi). The rats were fasted overnight then anesthetized using isoflurane (induction 4%, maintenance 1.5%) during modeling. To establish the MCAO model, a silicon-coated monofilament (403756PK5Re; Doccol, Sharon, MA, USA) was inserted for external carotid artery embolization, and then removed to allow reperfusion.22,23 During the surgery, a flexible fiber probe linked to a MoorLDL-HR laser Doppler flowmeter (Moor Instruments, UK) was used to monitor and confirm successful MCAO and reperfusion. The neck incisions were then sutured, and rats were returned to their cages and given free access to food and water. Sham-operated rats underwent the same procedure except there was no occlusion of the MCA.
EA intervention
Two hours after the surgery, rats in the EA group received EA treatment for 30 min once daily for 7 consecutive days by experienced manipulators who were unaware of the group allocation. GB20 was located 3 mm lateral to the center of an imaginary line joining the two ears at the back of the head, and the “non-acupoint” was located about 10 mm above the iliac crest. 24 Stainless steel needles (0.25 mm in diameter and 13 mm in length; Suzhou Medical Instruments, China) connected to a XYD-III EA device (Xiangyu Medical, China) were inserted to a depth of 2 mm at GB20 and directed toward the opposite eye. The same depth of needling (2 mm) was applied at the “non-acupoint.” The parameters of EA were set as follows: dispersed waves, frequency 1 and 20 Hz (alternating every 10 s), pulse width 0.5 ms and intensity 0.5–1.0 mA, depending on the reaction of each rat. 25 At the end of treatment, euthanasia was performed via CO2 asphyxia, followed by rapid cervical dislocation.
Neurological assessment
The neurological assessment was performed in a blinded fashion.
Neurological deficit scores
The recovered rats were scored using the improved Bederson score (0–4) method after modeling, as well as 1, 3 and 7 days after EA treatment. Rats whose scores were a 0 or 4 were excluded from the study to ensure the success of MCAO modeling and the survival rate during the experiment. Ultimately, one rat was excluded from the sham group, while no rats were excluded from the other three groups.
Balance beam walking test
The beam walking test was introduced to assess the motor coordination of rats by traversing a narrow and horizontal beam (3 cm width, 90 cm length and 50 cm height) at the end of modeling and 1, 3 and 7 days after EA treatment. Before MCAO, rats were trained for 2 days with four trials per day, to ensure that all rats could pass smoothly. A six-point scoring system developed by Ohlsson was used to assess the rats. 26
Y-maze test
The effect of EA on MCAO rats was explored by Y-maze test in a Y-maze experiment video analysis system (Nanjing, China) at the end of modeling and 1, 3 and 7 days after surgery. Each rat was placed in the center of the maze and allowed to move freely for 8 min. Total arm entries and spontaneous alternation (consecutive entries in three different arms) were recorded. The percentage of alternation was analyzed as follows: (number of alternations/number of total arm entries − 2) × 100%.
Number of synapses in hippocampus
The number of synapses was quantified by histopathological synaptic ultrastructural analysis. The left ischemic hippocampus from each rat was cut into 1 mm3 cubes after being fixed for 24 h in 1% paraformaldehyde/lanthanum nitrate tracer, 24 h in 3% glutaraldehyde and 2 h in 1% osmium tetroxide. Then, samples were dehydrated by graded ethanol containing 1% lanthanum nitrate tracer. The hippocampal slices were stained with lead citrate and uranyl acetate. A JEM-1400FLASH electron microscope (JEOL, Japan) was used to count the neuronal synapses in randomly selected microscopic fields.
Reverse transcription quantitative polymerase chain reaction
The mRNA expression of calcium-voltage gated channel subunit alpha 1B (CACNA1B), CaM, CaMKII and CREB were determined by reverse transcription quantitative polymerase chain reaction (RT-qPCR). Total RNA was extracted from the brain tissue (0.05 g) of rats from the different groups using Invitrogen™ TRIzol™ Reagent (Thermo Fisher, Waltham, MA, USA). One μg of total RNA (as the template) was reverse transcribed to cDNA. RT-qPCR was performed using an ABI Step One Plus System with universal TaqMan 2×PCR SYBR green mastermix. β-actin was used as an internal reference to standardize each mRNA. The relative transcript levels were calculated by the 2−ΔΔCt method.
Western blot analysis
Hippocampal tissues from the four groups were homogenized, and then the protein was obtained by brain tissue protein extraction kits (Biolabs, Beijing, China). Protein levels were determined using total protein assay kits (Biolabs). Then, equal amounts of proteins were electrophoresed with sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) protein loading buffer (Biolabs) and transferred to polyvinylidene fluoride (PVDF) membranes using Western wet membrane fluid. The membranes were blocked with Western blotting (WB) blocking solution (Biolabs), followed by overnight incubation (at 4°C) with the following primary antibodies: mouse monoclonal anti-growth–associated protein (GAP)-43 (1:2000, ab277627, Abcam, Cambridge, UK), mouse monoclonal anti-synaptophysin (SYN, 1:500, ab8049, Abcam), mouse monoclonal anti-postsynaptic density protein (PSD)-95 (1:1000, ab13552, Abcam), phosphorylated (p)-CREB (1:1000, ZRB1118, Sigma-Aldrich, Burlington, MA, USA), p-CaMKII (1:1000, WH0000815M1, Sigma-Aldrich), CaM (1:500, C2969, Sigma-Aldrich) and β-actin (1:1000, E4D9Z, Cell Signaling). Then, a 2-h incubation with secondary horseradish peroxidase (HRP)–conjugated Affinipure goat anti-rabbit antibody (1:7000, SA00001-2, Proteintech, Rosemont, IL, USA) was performed at room temperature. The specific bands were visualized and analyzed using Image J software (National Institutes of Health, Bethesda, MD, USA). β-actin was used as a loading control.
Isolation, transfection and oxygen-glucose deprivation/reperfusion of primary hippocampal neurons
Primary hippocampal neurons were isolated from the hippocampi of Sprague-Dawley rat embryos at 17 days of gestation as previously described. 27 An overexpressing pcDNA CACNA1B construct (GenScript ProBio, China) was used to transfect hippocampal neurons to overexpress CACNA1B. The transfection efficiency was confirmed using RT-qPCR 3 days after transfection. For the oxygen-glucose deprivation/reperfusion (OGD/R) model, hippocampal cultures were incubated in deoxygenated Neurobasal™-A medium (Invitrogen, Carlsbad, CA, USA) with no glucose under an anoxic atmosphere (85% N2, 10% H2, 5% CO2) at 37°C in an anaerobic incubator for 3 h. Then, the hippocampal cultures were cultured in Neurobasal™ Plus medium in a normoxic incubator (5% CO2 and 95% air) for 24 h, to form the OGD/R+pcDNA-CACNA1B group. Primary hippocampal neurons transfected with pcDNA control (NC) subjected to the same OGD/R proceedures were designated as the OGD/R+pcDNA-NC group. Finally, the control group was established by culturing primary hippocampal neurons in normal medium under normal conditions (37°C, 95% air and 5% CO2). The mRNA expression of CaM, CaMKII and CREB was detected by RT-qPCR.
Statistical analysis
Statistical comparisons were performed by t-test, for comparison between two groups. For comparison among three or more groups, one-way analysis of variance with post hoc Tukey multiple comparisons test or two-way analysis of variance (fixed factors: time of treatment and treatment group for comparison of neurobehavioral testing results; treatment group and pathway factors for comparisons of the levels of tested molecules) using GraphPad Prism 8 software (GraphPad Software Inc., San Diego, CA, USA). p < 0.05 was considered significant.
Results
Effect of EA on neurological deficits and behavior
The neurological deficit scores, balance beam scores and attention percentages in MCAO versus sham groups verified the success of modeling (Figure 1). Neurological deficit scores were evaluated to assess the neuroprotective function of EA. The results showed that rats receiving EA at GB20 (MCAO + GB20 group) exhibited significant improvement after cerebral injury compared to MCAO rats remaining untreated or receiving EA at a “non-acupoint” (p < 0.05; Figure 1(a)). Neurological behavior was assessed by the balance beam walking and Y-maze tests. For the beam walking test, scores for the MCAO + GB20 group were higher than the MCAO and MCAO + NA groups on the third and seventh days (p < 0.05; Figure 1(b)). As for the Y-maze test, rats in the MCAO + GB20 group demonstrated a decreased alteration percentage compared to the MCAO and MCAO + NA groups on the third and seventh days (p < 0.05; Figure 1(c)). These results suggest that EA at GB20 can mitigate neurological impairment caused by cerebral ischemia–reperfusion injury.
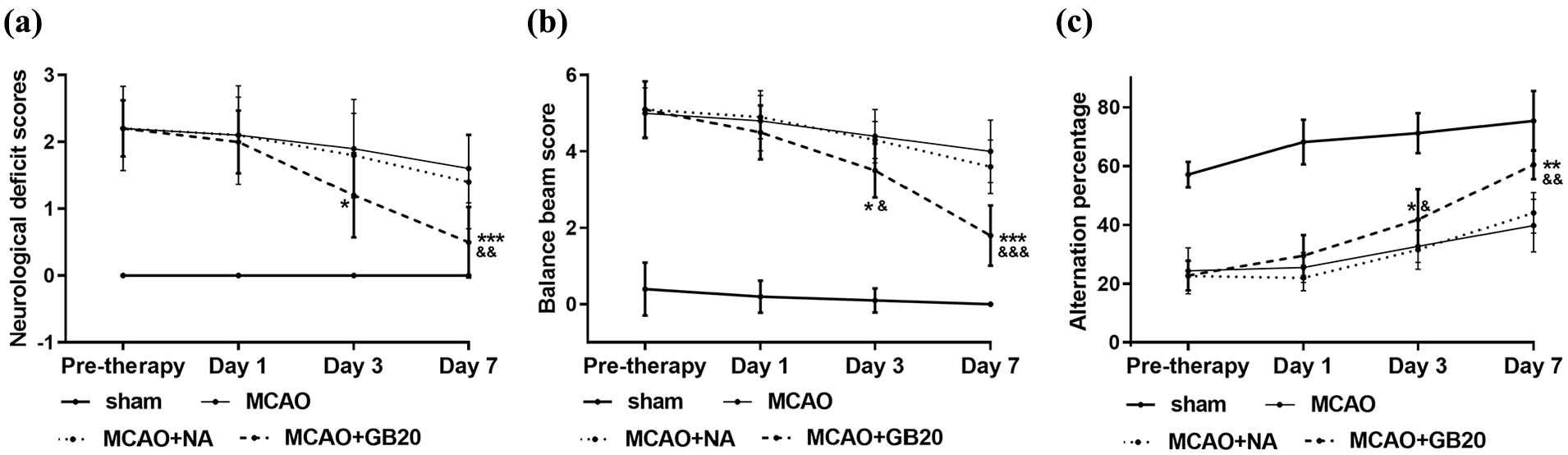
Electroacupuncture (EA) treatment attenuated cerebral ischemia–reperfusion injury in middle cerebral artery occlusion (MCAO) rats. (a) Neurological deficits, as detected by Bederson (0–4) scoring. (b) Results of balance beam walking test. (c) Results of Y-maze test. *p < 0.05; **p < 0.01; ***p < 0.001 (MCAO + GB20 vs MCAO groups). &p < 0.05; &&p < 0.01; &&&p < 0.001 (MCAO + GB20 vs MCAO + NA groups).
Effect of EA on synaptic plasticity
The effects of EA on synaptic plasticity in rats were assessed by the levels of expression of SYN, GAP-43 and PSD-95, along with the number of synapses in the ischemic hippocampus. As shown in Figure 2(a), WB demonstrated significantly increased PSD-95, SYN and GAP-43 protein levels in MCAO + EA versus MCAO and MCAO + NA groups (p < 0.05). With regard to the number of synapses, EA was found to significantly reverse the reduction in the number of synapses caused by cerebral ischemia–reperfusion injury (p < 0.05; Figure 2(b)).
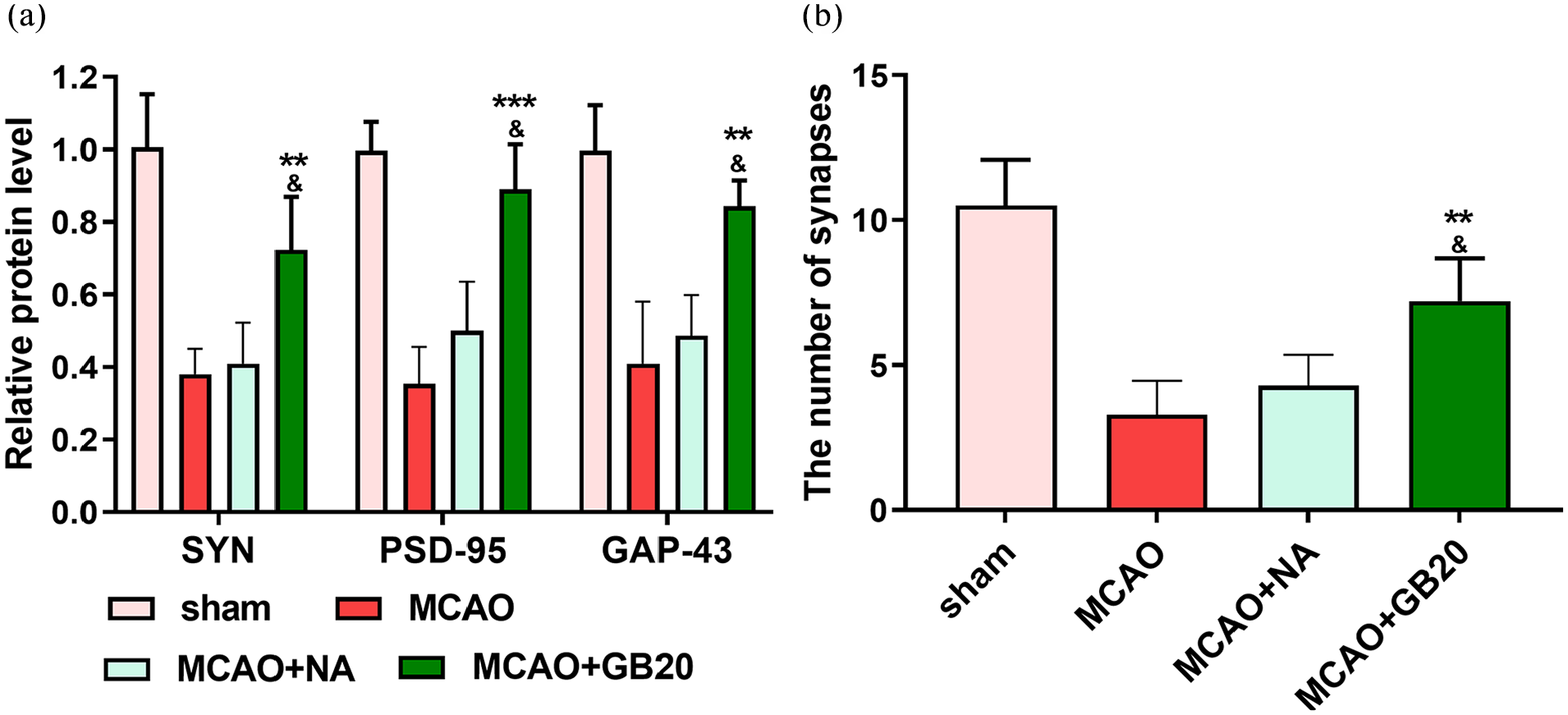
The relative levels of synapse-associated proteins and synapse number in middle cerebral artery occlusion (MCAO) rats. (a) Protein expression levels of synaptophysin (SYN), postsynaptic density protein (PSD)-95 and growth-associated protein (GAP)-43 in each group. (b) The number of synapses in each group. **p < 0.01; ***p < 0.001 (MCAO + GB20 vs MCAO groups). &p < 0.05 (MCAO + GB20 vs MCAO + NA groups).
CACNA1B appeared to be a target gene of EA in MCAO rats
By searching GeneCard and integrating with Venny, we screened 95 genes targeted by both EA and MCAO (Figure 3(a)). The 95 relevant genes were explored using STRING and OmicShare Tools to archive functional protein association networks, Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis and Gene Ontology (GO) analysis. GO analysis revealed these 95 genes were enriched in signaling receptor binding (Figure 3(c)), while KEGG pathway functional enrichment showed that these genes were first categorized into neuroactive ligand–receptor interaction (Figure 3(d)). In the result of the functional protein association networks by STRING, the calcium signaling pathway (related genes shown in red in Figure 3(b)), especially involving CACNA1B, piqued our interest and thus was selected for further study.
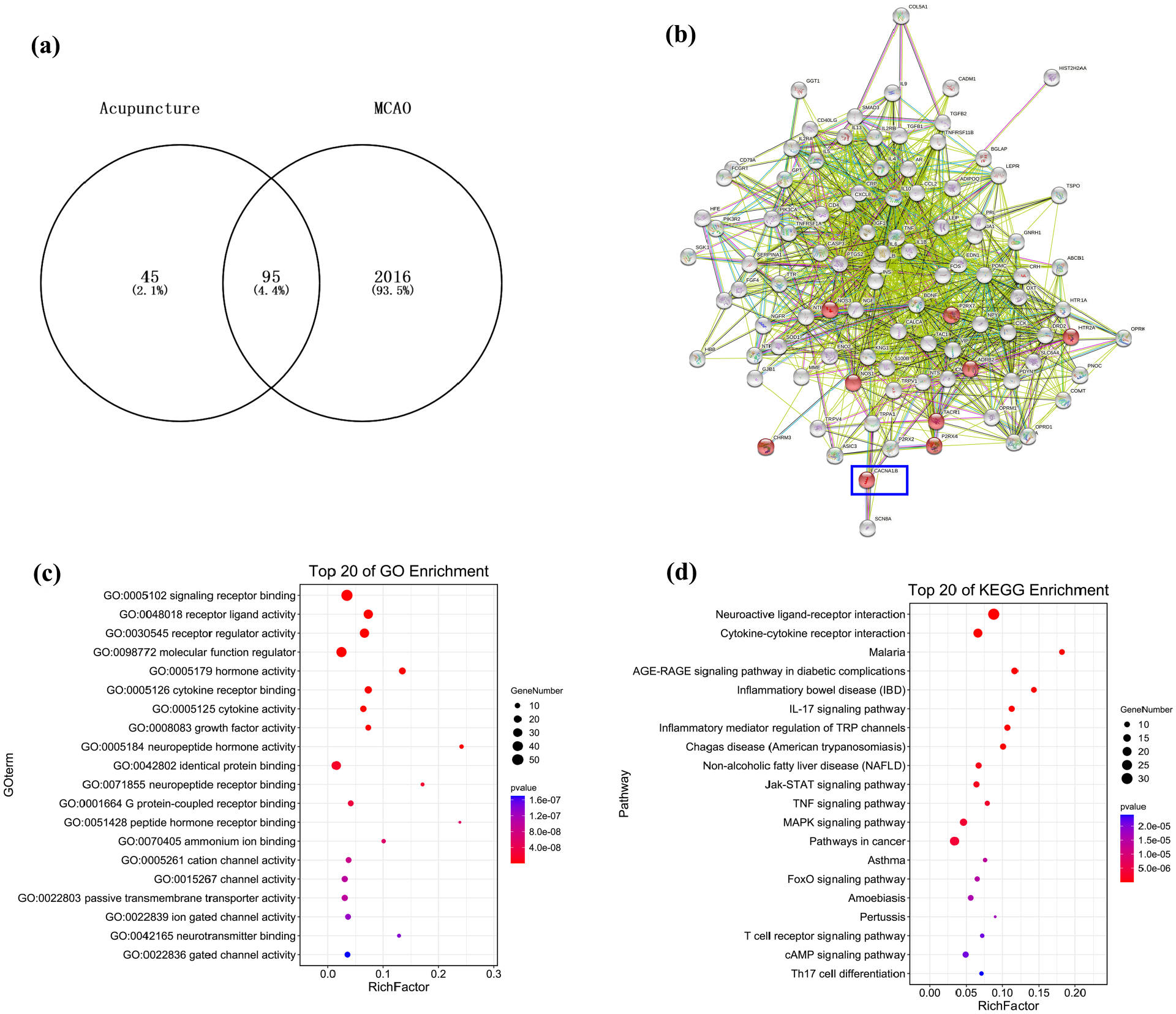
CACNA1B appeared to be a target gene of electroacupuncture (EA) in middle cerebral artery occlusion (MCAO) rats. (a) Overlap of 95 target genes in bioinformatic prediction algorithms. (b) PPI network of EA against MCAO. (c) Gene Ontology (GO) functional enrichment analysis. (d) Histogram of Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment result.
Effect of EA on expression of the CACNA1B/CaM/CaMKII/CREB signaling pathway in MCAO rats
To explore the mechanism underlying the effect of EA in MCAO rats, RT-qPCR and WB analysis were used to analyze the expression levels of CACNA1B, CaM, CaMKII and CREB mRNA and protein, respectively. mRNA levels (detected by RT-qPCR) revealed higher expression of CACNA1, CaM, CaMKII and CREB in MCAO + GB20 versus both MCAO and MCAO + NA groups (p < 0.05; Figure 4(a)). According to the WB analysis, protein levels of CaM, p-CREB and p-CaMKII were decreased by cerebral ischemia–reperfusion injury but restored by the EA intervention (p < 0.05; Figure 4(b)).
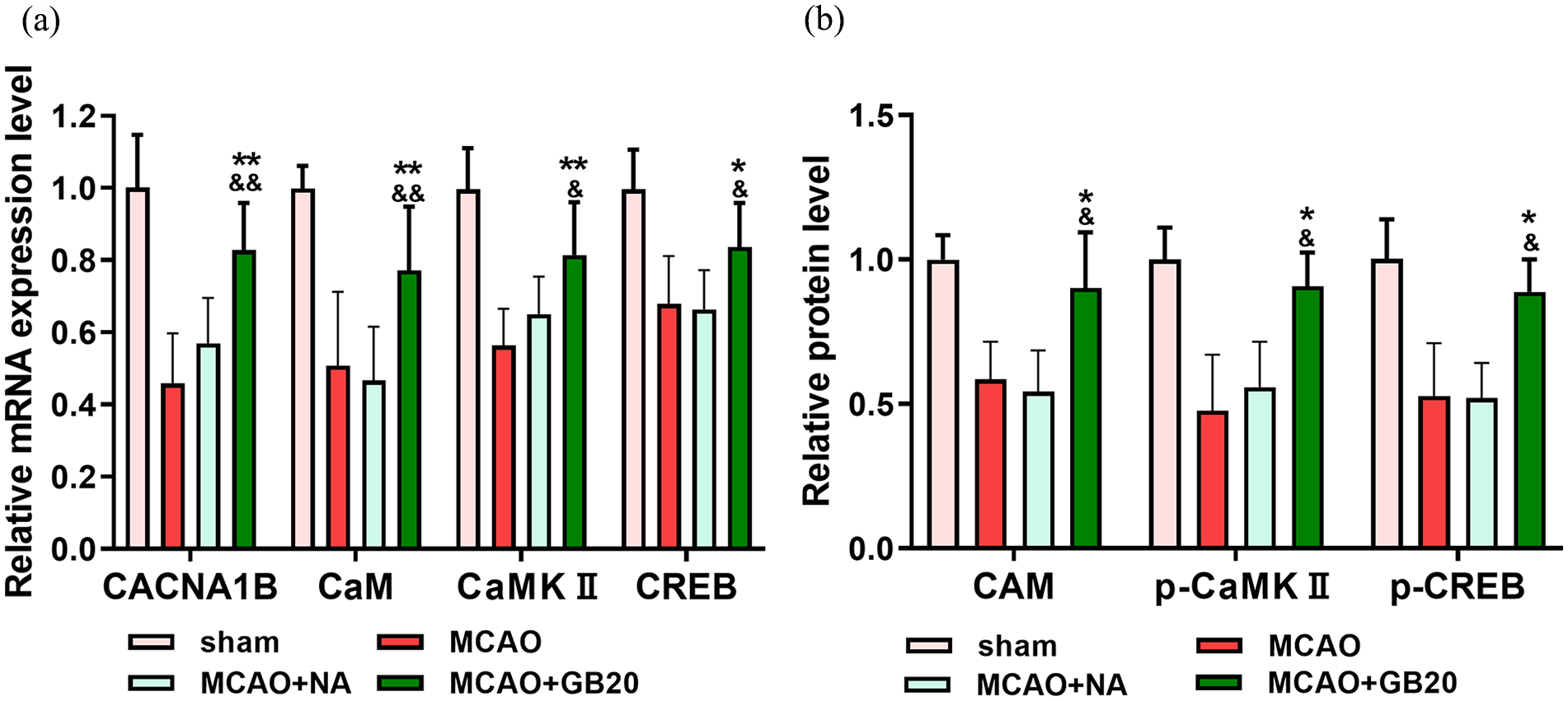
(a) mRNA levels of CACNA1B, calmodulin (CaM), Ca2+/calmodulin-dependent protein kinase type II (CaMKII) and cyclic adenosine monophosphate response element binding (CREB) in middle cerebral artery occlusion (MCAO) rats. (b) Protein expression levels of CaM, phosphorylated (p)-CaMKII and p-CREB in each group. *p < 0.05; **p < 0.01 (MCAO + GB20 vs MCAO groups). &p < 0.05; &&p < 0.01 (MCAO + GB20 vs MCAO + NA groups).
CACNA1B modulates the CaM/CaMKII/CREB signaling pathway in OGD/R primary hippocampal neurons
To determine whether CACNA1B modulates the activity of the CaM/CaMKII/CREB signaling pathway during cerebral ischemia–reperfusion, we used RT-qPCR to examine mRNA expression of CaM, CaMKII and CREB in primary hippocampal neurons after OGD/R. The OGD/R group had significantly lower expression of CACNA1B than the control group, but CACNA1B pcDNA increased mRNA expression levels of CACNA1B (p < 0.001; Figure 5(a)). Compared with the control group, the levels of CaM, CaMKII and CREB mRNA were significantly downregulated in the OGD/R group, but restored by overexpression of CACNA1B (p < 0.05; Figure 5(b)). These data suggest that CACNA1B plays a role in activating the CaM/CaMKII/CREB signaling pathway in primary hippocampal neurons.
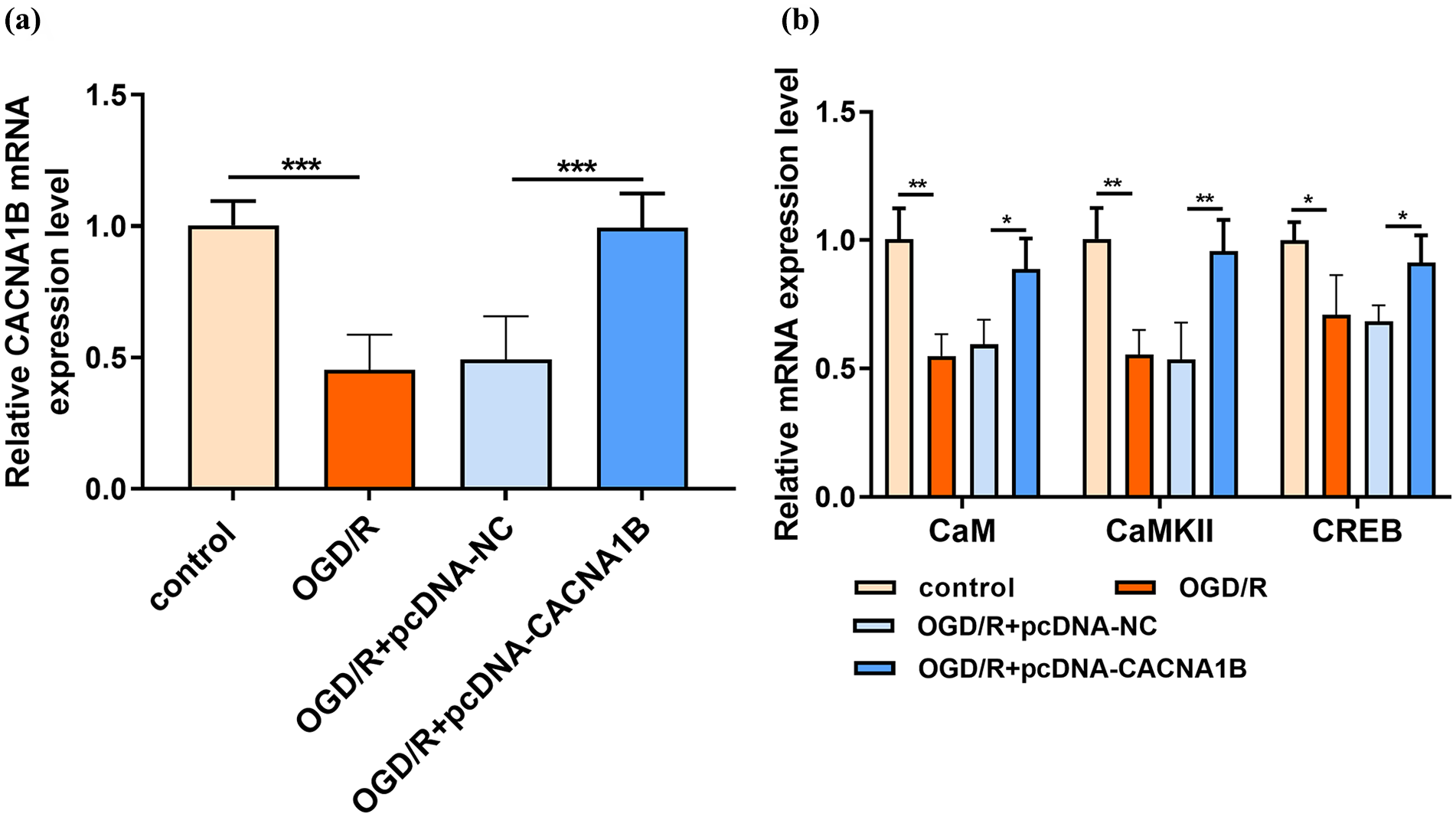
CACNA1B activated the calmodulin (CaM)—Ca2+/calmodulin-dependent protein kinase type II (CaMKII)—cyclic adenosine monophosphate response element binding (CREB) signaling pathway in primary hippocampal neurons after oxygen-glucose deprivation/reperfusion (OGD/R). (a) CACNA1B overexpression was successful. (b) CACNA1B significantly upregulated the expression of mRNAs related to the CaM–CaMKII–CREB signaling pathway. *p < 0.05; **p < 0.01; ***p < 0.001.
Discussion
EA has been widely used to treat various neurodegenerative diseases, such as Alzheimer’s disease and acute stroke.28,29 The clinical effects of EA in stroke patients have been emerging with respect to relief of spasticity, attenuation of pain and enhancement of cerebral blood flow.30,31 This study was conducted to explore the effects of EA treatment in MCAO and the underlying mechanism of action (Supplemental Figure 1). Neurological deficit scores and behavior were assessed before and during the treatment. Bioinformatics analysis was used to screen the target genes of EA against MCAO. It was hypothesized that EA adjusts the CACNA1B–CaM–CaMKII–CREB signaling function in the hippocampus and ameliorates cognitive impairment in MCAO rats. After intervention for 7 days, the expression levels of related molecules were assessed by WB and RT-qPCR. Primary hippocampal neurons with CACNA1B overexpression were used to confirm the participation of the CaM–CaMKII–CREB axis.
GB20 is a traditional acupuncture point location on the head. 32 EA treatment at GB20 can ameliorate migraine pain via inhibition of calcitonin-related gene peptide (CGRP) expression and modulation of the trigeminovascular system ascending pathway. 33 Moreover, EA can ameliorate mild cognitive impairment and has therapeutic effects on aphasia after stroke.34,35 In this study, we established the MCAO rat model and found that neurological behavior was severely impaired. Importantly, EA appeared to reverse the neurological impairment, reflected by decreased neurological deficit scores and recovery of neurobehavior. The most frequently targeted traditional acupuncture point locations in previous studies of acupuncture for stroke rehabilitation include GV20 (Baihui), ST36 (Zusanli), LI11 (Quchi), GV26 (Shuigou), GV14 (Dazhui) and LI4 (Hegu). This study provides an additional traditional acupuncture point recommendation for stroke rehabilitation.
The beneficial effects of EA are closely related to neuroprotective molecules. Growing evidence has confirmed that EA-induced reversal of ischemic brain injury caused by stroke involves a large number of molecules, among which those related to synaptic plasticity have drawn more and more attention as the potential molecular basis of learning and memory. The membrane phosphoprotein GAP-43 is associated with the formation of new synapses after ischemic injury and is a sensitive marker of axonal regeneration in the hippocampus. 36 Here, we found that expression of GAP-43 was decreased in the hippocampus after MCAO and partly restored by EA treatment. SYN and PSD-95 are two important factors associated with synaptogenesis. Previous studies have demonstrated that SYN and PSD-95 expression are downregulated in the hippocampus during cerebral ischemia. 37 Our present results suggest that EA at GB20 prevented the reduction in PSD-95 and SYN expression, thereby likely contributing to the maintenance of synaptic plasticity. Furthermore, EA increased the number of synapses in the hippocampal CA1 region after MCAO. Therefore, the effect of EA on MCAO rats may depend on the promotion of synaptic remodeling.
In this study, according to bioinformatic prediction, the overlapping genes between EA and MCAO were screened. The GO and KEGG analysis of these genes pointed to a neuro-associated signaling receptor. Among the target genes, CACNA1B is associated with cerebral infarction. 38 The CACNA1B gene encodes Cav2.2 channels that control the entry of calcium and impacts excitatory neurotransmission. 39 Previous studies have reported that the CaM–CaMKII–CREB signaling pathway participates in synaptic and cognitive function, 40 and high levels of CaMKII, CaM and CREB may be beneficial for learning and memory ability. 41 Combined with the fact that CACNA1B mRNAs are enriched in CaMKII expressing projection neurons, 39 we inferred that the therapeutic effect of EA in MCAO rats may involve moderation of the CACNA1B–CaM–CaMKII–CREB axis. To verify this, mRNA and protein expression levels of CACNA1B, CaM, CaMKII and CREB were determined. The results confirmed that EA can restore the levels of CACNA1B, CaM, CaMKII and CREB after MCAO. It has been reported that Ca2+/CaM stimulates autonomous CaMKII activity for enhancement of synaptic strength, and the CaM–CaMKII–CREB signaling pathway plays an important role in synaptic degeneration. 40 Thus, the CACNA1B–CaM–CaMKII–CREB pathway may be an important cellular signaling pathway involved in EA-induced cognitive improvement.
In conclusion, this study demonstrates that EA intervention at GB20 exhibits cognitive repair potential in MCAO rats. The mechanism underlying the effects of EA in MCAO rats was partly associated with the CACNA1B–CaM–CaMKII–CREB axis.
Supplemental Material
sj-tif-1-aim-10.1177_09645284231202805 – Supplemental material for Electroacupuncture at GB20 improves cognitive ability and synaptic plasticity via the CaM–CaMKII–CREB signaling pathway following cerebral ischemia–reperfusion injury in rats
Supplemental material, sj-tif-1-aim-10.1177_09645284231202805 for Electroacupuncture at GB20 improves cognitive ability and synaptic plasticity via the CaM–CaMKII–CREB signaling pathway following cerebral ischemia–reperfusion injury in rats by Qing Han and Feng Wang in Acupuncture in Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project was funded by the Shanghai Jinshan District Health and Family Planning Commission (JSKJ-KTZY-2019-01).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
