Abstract
Background:
Asthma is a chronic inflammatory disease that affects millions of people worldwide. Chronic asthma is commonly resistant to steroid therapy. Acupuncture has an anti-inflammatory effect and has been widely used as an add-on therapy for asthma.
Objective:
To evaluate the effects of acupuncture on the inflammatory response and airway remodelling in the bronchioles of an asthma mouse model.
Methods:
A chronic asthma model was produced in female BALB/c mice by ovalbumin (OVA) sensitisation. 32 mice were randomised into four groups: control; asthma (OVA); OVA+BL13; and OVA+BL13+ST36. OVA was administered by intraperitoneal injection on days 0 and 14 followed by aerosol exposure of 1% OVA three times a week for 6 weeks. Manual acupuncture (MA) was performed three times a week for 6 weeks at BL13 alone, or BL13 in combination with ST36, in the two MA-treated groups. At the end of the experiment, blood samples were collected to determine eosinophil and neutrophil counts and lung tissue was prepared for histological examination.
Results:
A pronounced reduction in the neutrophil count was achieved after MA at BL13+ST36 (P=0.005) while the eosinophil count was lowered after MA both at BL13 (P=0.007) and BL13+ST36 (P=0.006). Reduction in the bronchiolar epithelial and smooth muscle thickness and the number of goblet cells was observed after MA at BL13 (P=0.001, P=0.001 and P=0.002, respectively) and BL13+ST36 (P=0.001, P=0.002 and P=0.001, respectively).
Conclusion:
Acupuncture can reduce the inflammatory response and prevent airway remodelling in a chronic asthma mouse model.
Keywords
Introduction
Asthma is a common health problem worldwide, with an estimated 235 million people currently suffering from the condition. 1 Asthma is a chronic inflammatory disease of the airway with heterogeneous causes. 2 Studies in humans mostly use tissues from biopsies that are limited to the mucosal layer and thus cannot evaluate deeper structures. More thorough examinations can only be performed using postmortem samples from asthma patients. Therefore, the use of an appropriate animal model is essential to study asthma. 3
The asthma syndrome occurs as a result of an immune system disorder and autonomic nervous system dysfunction such as hyperactivity of the parasympathetic nervous system as well as blockade of β-adrenergic receptors of the sympathetic nervous system. 4 Bronchial asthma is a chronic inflammatory disease characterised by an increased production of eosinophils, mast cells and immunoglobulin E (IgE), mucous hypersecretion, subepithelial fibrosis and airway hyperresponsiveness (AHR). 5 An increased number of neutrophils in the airway was found during acute asthma exacerbation, 6 leading to various degrees of structural changes in the airway that finally resulted in airway remodelling.5,7 Eosinophils, interleukin 17 (IL-17) and neutrophils can cause inflammation of the airway, mucous hypersecretion, bronchoconstriction and airway remodelling. Structural changes observed in airway remodelling include epithelial hyperplasia and metaplasia, sub-epithelial fibrosis, smooth muscle hyperplasia and hypertrophy, increased angiogenesis and increased numbers of goblet cells.3,8
Through local inflammatory reactions, intercellular transduction, cutaneo/somato-visceral reflexes and neural transmission, 9 acupuncture decreases the production of Th-2 cytokines, stimulates Th-1 cytokines and keeps them in equilibrium. Acupuncture modulates the balance of Th-1/Th-2, 10 and reduces the number of eosinophils and inflammatory cells in the ovalbumin (OVA)-induced mouse model of asthma. Acupuncture is believe to promote balance of the immune system and maintain homeostasis. 11
The traditional acupuncture point BL13 (Feishu) is located at the level of the third thoracic vertebrae,12,13 in areas innervated by sensory nerve fibres of the thoracic segment of the spinal cord. Thus, acupuncture at this location can stimulate the sympathetic nerves arising from the thoracic segments, in particular segments 1–4, which innervate the bronchus. Stimulation of the sympathetic nerves leads to the release of neurotransmitters, which activate β2-adrenergic receptors located in the smooth muscle of the bronchus and its branches, giving rise to bronchodilation. Stimulation of the receptors in the bronchial mucous glands can inhibit mucous secretion, while degranulation of mast cells is blocked. 14 Acupuncture at ST36 (Zusanli) can maintain the homeostasis of the immune system due to its immunomodulatory effects on Th-1 as well as Th-2 dominant pathological conditions. 15 We aimed to evaluate whether acupuncture at BL13 and ST36 can reduce the inflammatory response and prevent airway remodelling of the bronchioles in a chronic asthma mouse model.
Methods
Animals
Thirty-two female BALB/c mice, provided by the Integrated Research and Testing Laboratory (LPPT) unit IV of Gadjah Mada University (UGM) in Yogyakarta, Indonesia, were used in the study. The mice were around 6–8 weeks of age and weighed approximately 20–25 g. Only female mice were used due to their better response to allergen (OVA). 16 The mice were housed in standard cages with controlled lighting and temperature (25–28°C). Food and water were given ad libitum. The work was conducted in accordance with animal care and use guidelines of Gadjah Mada University and all experimental procedures were approved by the Medical Research Ethics Committee of the Faculty of Medicine, Sebelas Maret University, Surakarta, Indonesia (ref. 518/VIII/HREC/2014). All efforts were made to minimise the suffering of the animals.
Asthma model
OVA (Nacalai, Catalogue 01 205–42, Japan) was used to sensitise the mice. Intraperitoneal injections of 10 µg OVA and 1 mg aluminium hydroxide in 0.5 mL of 0.9% sodium chloride (NaCl) were administered to the mice on day 0 and 14 followed by aerosol exposure of 1% OVA in 0.9% NaCl for 30 min. This procedure was performed three times a week for 6 weeks, starting from day 21 and ending on day 63. 17
Grouping
This was an experimental study with a post-test only group design. The mice were randomised into four groups: control group; untreated asthma group sensitised with OVA; asthma group treated with manual acupuncture (MA) at BL13 (OVA+BL13); and asthma group treated with MA at BL13 and ST36 (OVA+BL13+ST36). The mice were identified by a number, and a randomisation table was generated using Microsoft Excel 2007 to allocate the mice to one of the four groups, each comprising eight mice. An a priori power calculation incorporating fixed effects, omnibus and one-way analysis of variance (ANOVA) was performed using G*Power 3.1 software with neutrophil count as the primary outcome. Based on our pilot study, we estimated that seven animals per group would be required to elicit a difference of 0.86 in neutrophil count at 95% power and α level of 0.05 assuming an SD of 8.93. To compensate for the anticipated mortality during the study, we allocated eight animals to each group. Outcome measurements were performed by two observers who were independent and blinded to experimental group allocation.
Acupuncture
The traditional acupuncture points used in the study were ST36, located approximately 5 mm inferolateral to the anterior tibial tubercle, 18 and BL13, located at the depression lateral to the lower border of the spinous process of the third thoracic vertebra (around 0.3 cm from the midline in mice). 12 Disposable stainless steel acupuncture needles measuring 0.18×7 mm (Huanqiu, China) were bilaterally inserted to a depth of 3 mm and retained for 15 min without any further stimulation. MA was given three times a week for 6 weeks starting on the 21st day by a well-trained acupuncturist. In total, each mouse received 18 treatments during the study. The study protocol is described in Figure 1.
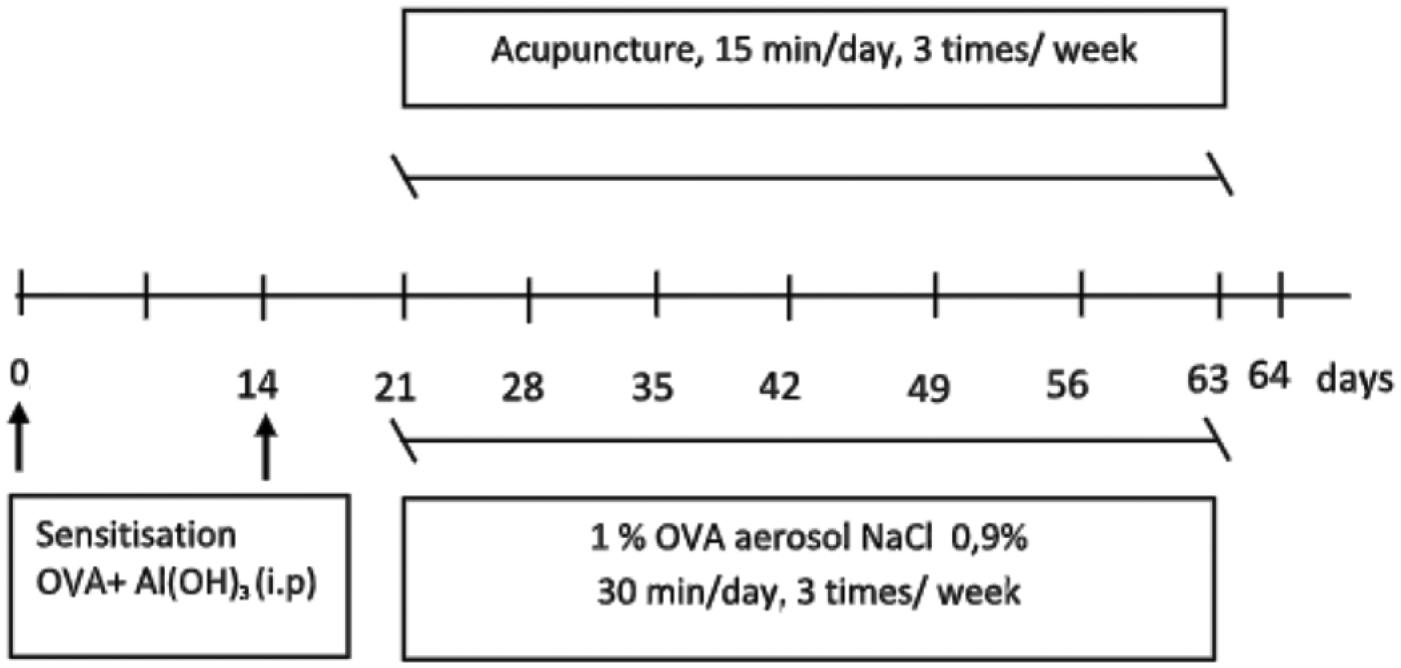
Schematic diagram of the study protocol. The chronic asthma model was created by intraperitoneal injection of 10 µg ovalbumin (OVA) and 1 mg of aluminium hydroxide (Al(OH)3) in 0.5mL of 0.9% sodium chloride (NaCl) on days 0 and 14 followed by nebulisation of 1% (weight/volume) OVA in 0.9% NaCl for 30 min three times/week for 6 weeks, from day 21 to day 63. Starting on day 21, manual acupuncture was given for 15 min, three times/week for 6 weeks. One day after all the procedures were completed (day 64), blood samples were collected to determine eosinophil and neutrophil counts and lung tissue was prepared for histological examination.
Inflammatory response
Assessment of the inflammatory response was carried out by calculating the number of eosinophils and neutrophils per mm3 in peripheral blood using a haemato-analyser (Sysmex-XS-800i, Japan).
Airway remodelling
Airway remodelling was examined by assessing the effects of chronic inflammation on epithelial thickness, smooth muscle thickness and the number of goblet cells in the bronchioles. Before necropsy, the mice were euthanised by cervical dislocation. Afterwards, lung tissues were collected and prepared for histological examination. Haematoxylin and eosin was used to stain the epithelial and smooth muscle layers while periodic acid-Schiff was used to stain the goblet cells. Images were obtained using light microscopy (Olympus CX21) at 400x magnification and assessed using Image Optilab Pro 6.1 software.
Statistical analysis
Data were analysed using ANOVA followed by the least significant difference (LSD) post hoc test with SPSS for Windows version 22.0 (SPSS Inc., Chicago, IL, USA). The significance level was set at P<0.05.
Results
Inflammatory response
The inflammatory response was assessed by counting the number of eosinophils and neutrophils in peripheral blood. The eosinophil count was significantly higher in the OVA group than the control group (P=0.039). The number of eosinophils significantly decreased after MA both at BL13 (P=0.007) alone and BL13+ST36 (P=0.006) as compared with the asthma group. However, there was no difference in the reduction of the eosinophil count between MA at BL13 alone and the combination of BL13 and ST36 (P=0.961). The number of neutrophils in the control group (19.75±6.04/mm3) differed significantly (P=0.009) when compared with the OVA group (38.00±14.00/mm3). The neutrophil count did not differ from the control group following MA at BL13 alone (P=0.674). However, a notable reduction (P=0.005) in the neutrophil count was achieved when MA at BL13 was combined with MA at ST36 (21.00±6.30/mm3). Data on the inflammatory response are presented in Figure 2.
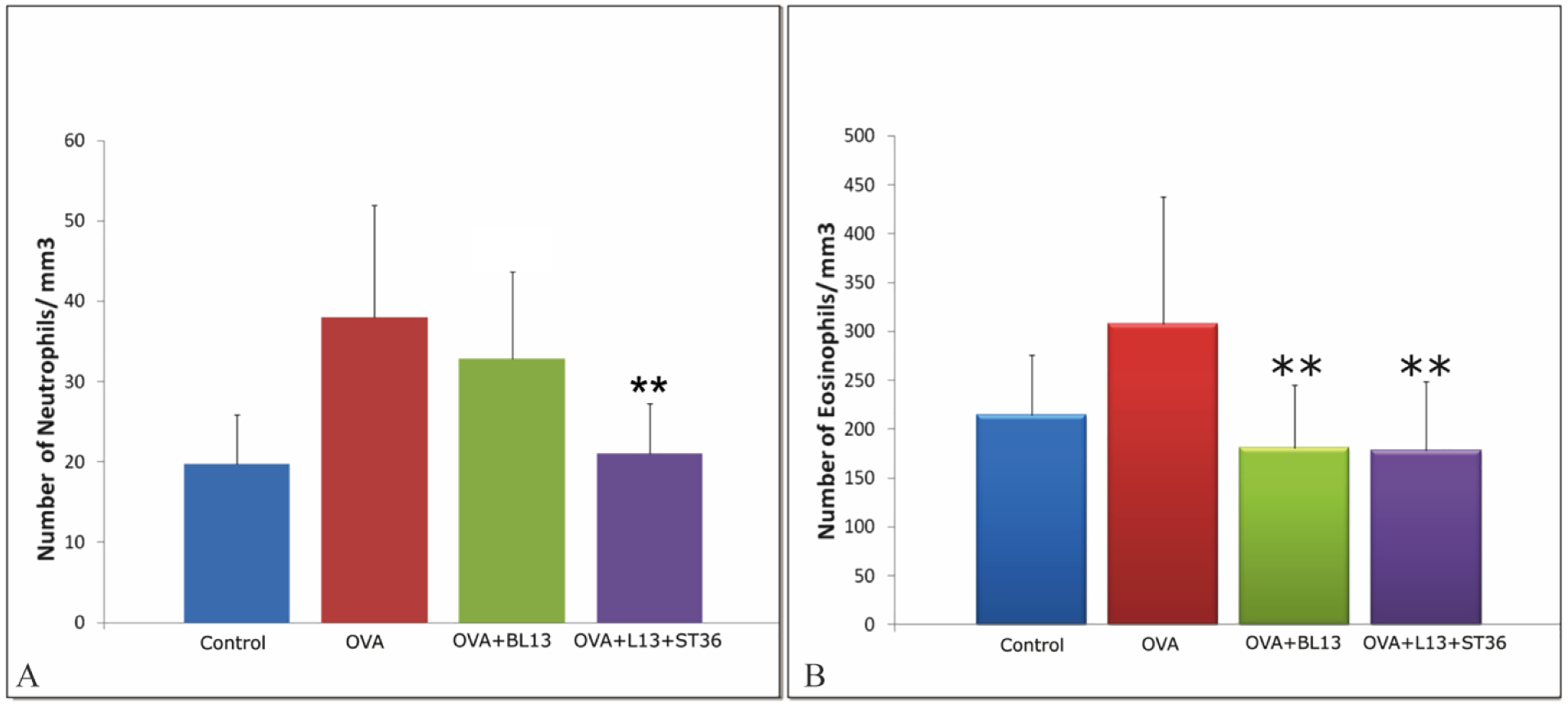
The number of neutrophils (A) and eosinophils (B) in each group. Data are presented as mean+SD. OVA, ovalbumin. ** p<0.01 versus OVA group.
Airway remodelling
OVA sensitisation significantly (P=0.001) increased epithelial thickness of the bronchioles (36.64±10.12 µm) compared with the control group (14.51±1.20 µm). A pronounced reduction of epithelial thickness was observed (P=0.001) after MA treatment at BL13, either when given individually (19.64±1.93 µm) or in combination with MA at ST36 (17.88±3.20 µm). Data on the epithelial thickness of the bronchioles are presented in Figure 3.
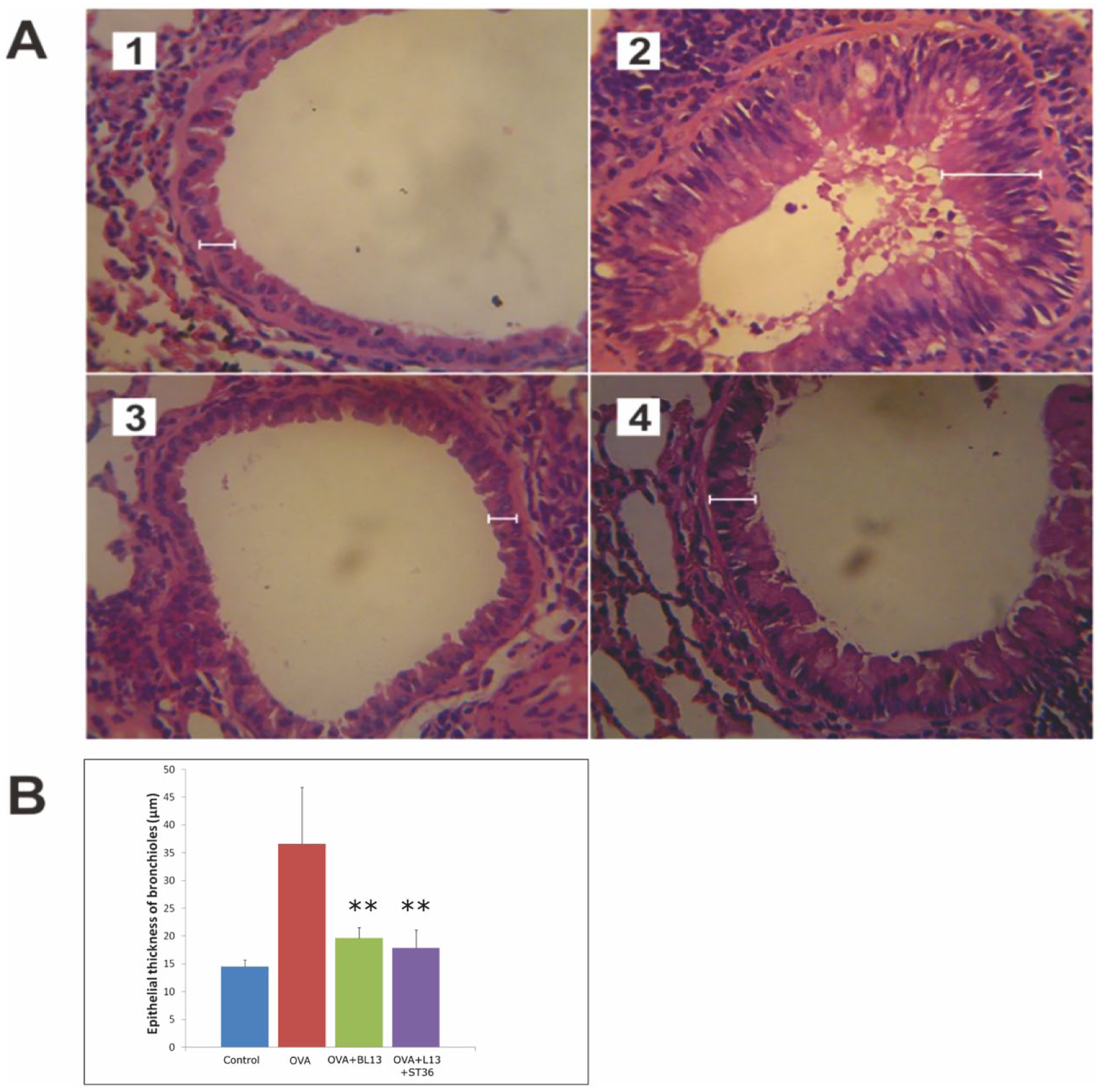
Histological features (A) and morphometric analysis (B) of the thickness of the epithelial layer of the bronchioles in each group. (1) Control group. (2) OVA group. (3) OVA+BL13 group. (4) OVA+BL13+ST36 group. Haematoxylin and eosin was used to stain the smooth muscle layer. Images were obtained using light microscopy (Olympus) CX21 at 400x magnification. Data are presented as mean+SD. OVA, ovalbumin. ** p<0.01 versus OVA group.
Smooth muscle thickness of the bronchioles in the OVA group (11.70±3.16 µm) was significantly increased (P<0.001) compared with the control group (6.34±1.29 µm). MA treatment both at BL13 alone (7.84±1.06 µm) or at a combination of BL13 and ST36 (7.89±2.49 µm) significantly decreased the smooth muscle thickness compared with the OVA group (P=0.001 and P=0.002, respectively). Data on smooth muscle thickness of the bronchioles can be seen in Figure 4.
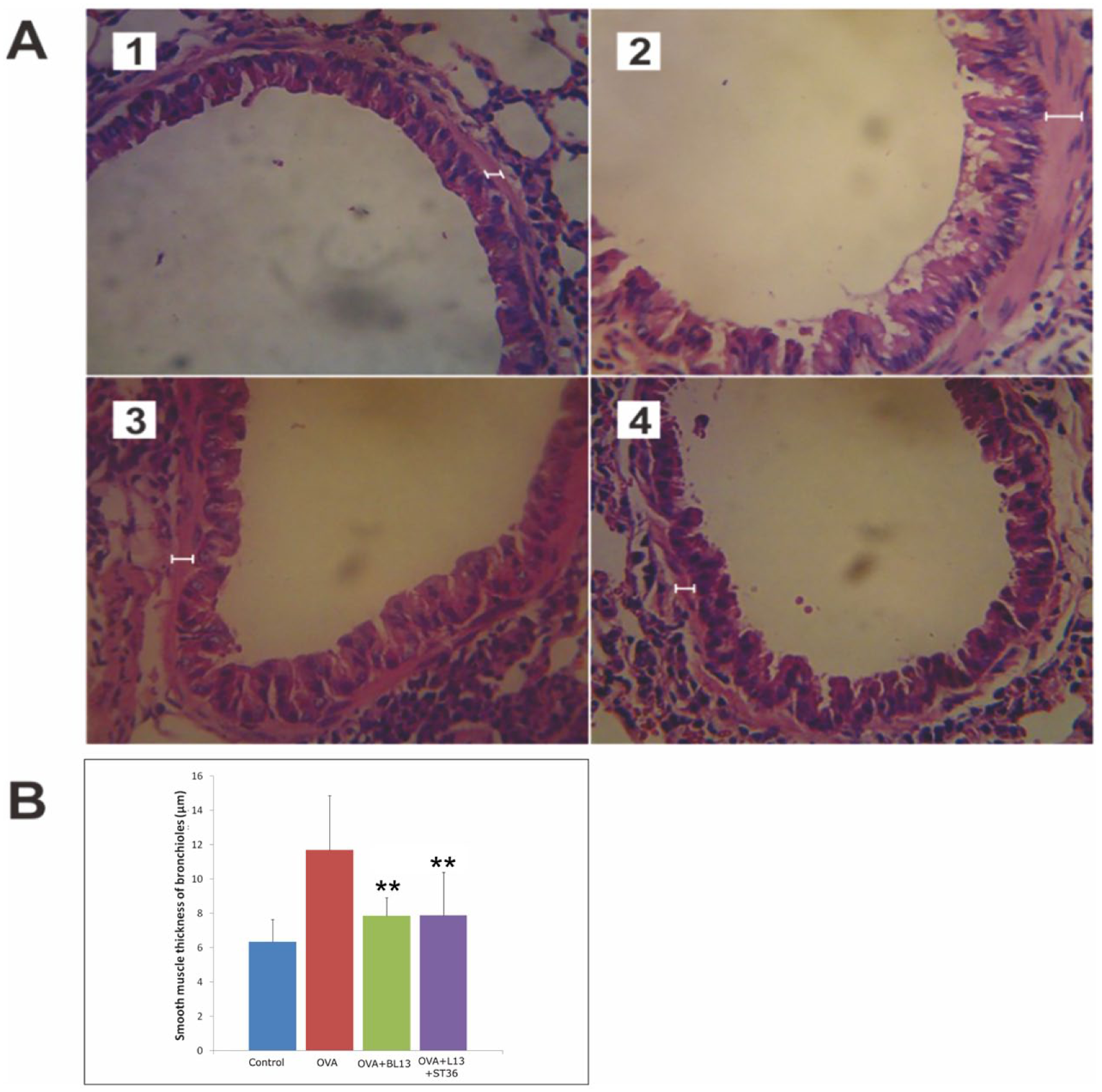
Histological features (A) and morphometric analysis (B) of the smooth muscle thickness of the bronchioles in each group. (1) Control group. (2) OVA group. (3) OVA+BL13 group. (4) OVA+BL13+ST36 group. Haematoxylin and eosin was used to stain the smooth muscle layer. Images were obtained using light microscopy (Olympus CX21) at 400x magnification. Data are presented as mean+SD. OVA, ovalbumin. ** p<0.01 versus OVA group.
The number of goblet cells in the OVA group was elevated significantly compared with the control group (P=0.001). Acupuncture at BL13 both alone and in combination with ST36 reduced the number of goblet cells significantly compared with the untreated asthma model group, with (P=0.02 and P=0.001, respectively). However, there was no pronounced difference in the reduction of the number of goblet cells when MA at BL13 was compared with MA at BL13+ST36. Data on goblet cell changes are presented in Figure 5.
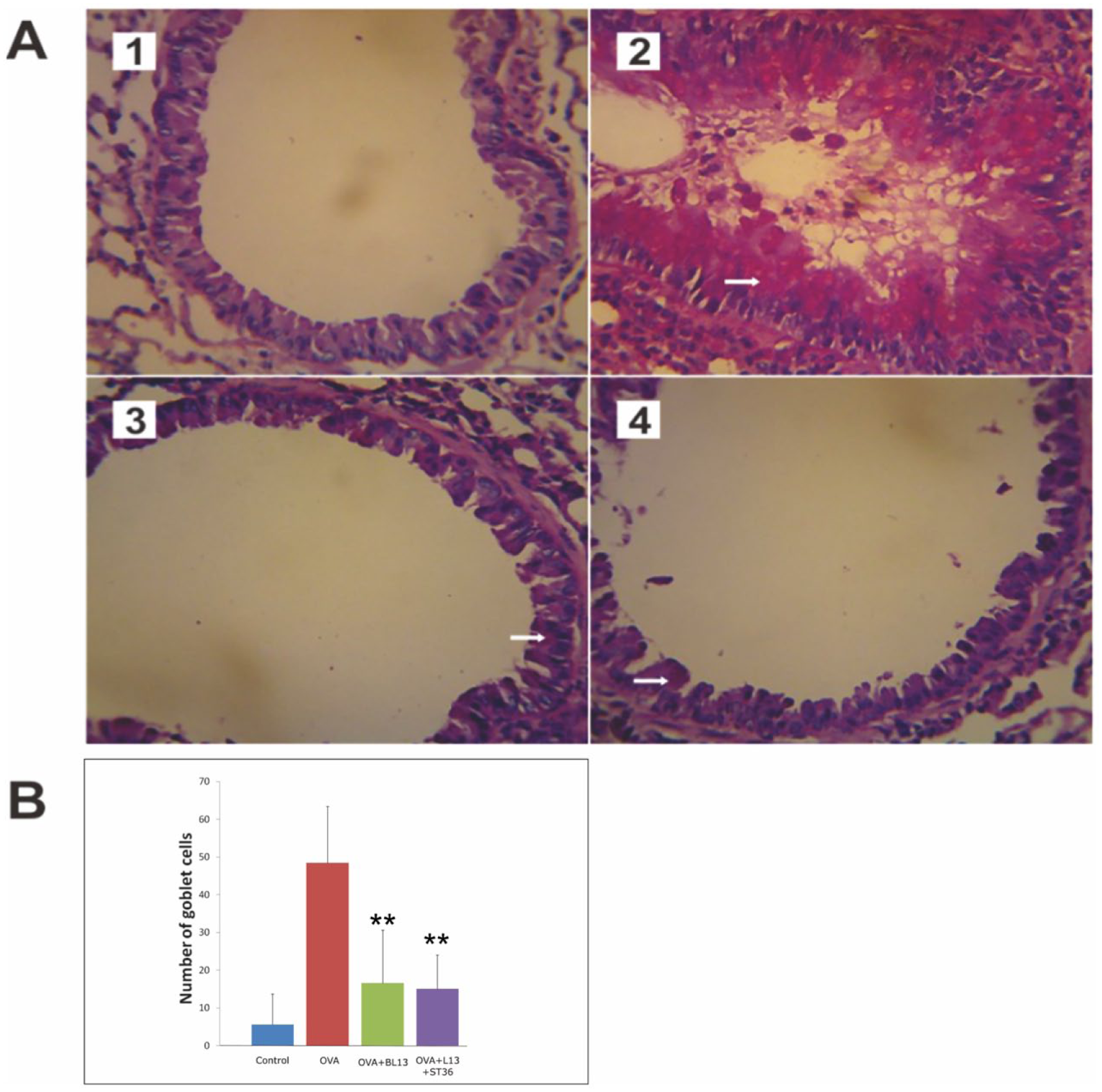
Histological features (A) and morphometric analysis (B) of goblet cells in the epithelial layer of the bronchioles in each group. (1) Control group. (2) OVA group. (3) OVA+BL13 group. (4) OVA+BL13+ST36 group. Goblet cells stained with periodic acid-Schiff are shown in pink (arrow). The OVA group showed goblet cell hyperplasia and excessive mucous production. Images were obtained using light microscopy (Olympus CX21) at 400x magnification. Data are presented as mean+SD. OVA, ovalbumin. ** p<0.01 versus OVA group.
Discussion
Inflammatory response
This study showed that acupuncture decreased the inflammatory response manifested by a significant reduction in the number of inflammatory cells (neutrophils and eosinophils). A number of studies have suggested that the positive effect of acupuncture on pulmonary disease is achieved via down-regulation of inflammatory cells and mediators. Similar to our findings, acupuncture lowered eosinophil counts in a prior study on asthmatic rats. 19 Acupuncture also significantly reduced the level of interleukin 8 (IL-8), a neutrophil chemotactic factor, and the pro-inflammatory cytokine tumour necrosis factor α (TNFα) in a mouse model of chronic obstructive pulmonary disease (COPD). 20
In this study, acupuncture at BL13 alone did not cause a significant reduction in the number of neutrophils. However, acupuncture at BL13+ST36 significantly reduced the number of neutrophils when compared with the OVA group. It is well understood that cytokine IL-17 mobilises and recruits neutrophils. 21 Thus, this finding is consistent with our previously published data showing that MA at BL13+ST36 but not at BL13 alone notably lowered the level of IL-17. 22 It is likely that MA at BL13 alone is not sufficient to block neutrophil mobilisation, considering that Carneiro et al. used acupuncture at multiple traditional acupuncture points, such as BL13, GV14 (Dazhui), Dingchuan, LU1 (Zhongfu), CV17 (Danzhong), ST36 and SP6 (Sanyinjiao), to suppress the inflammatory response in an asthmatic mouse model. 13 Yang et al. 23 also used more traditional acupuncture points including GV14, BL12 (Fengmen) and BL13 as an add-on therapy for allergic asthma patients. Thus, it is plausible that MA at BL13+ST36 leads to a more significant reduction in the neutrophil count.
We used OVA sensitisation to create an asthma model in mice. OVA exposure three times a week for 6 weeks induced a significant increase in the number of neutrophils in the OVA group, suggesting that our model replicated chronic asthma with acute exacerbation. OVA exposure also significantly increased the number of eosinophils. This is consistent with the results of previous studies that used OVA to induce asthma in Balb/c mice. 17 This elevation in eosinophil count was blocked after MA was given either at BL13 alone or in combination with ST36. This finding is supported by Yang et al., 23 who reported that acupuncture at GV14, BL12 and BL13 notably reduced the number of eosinophils in peripheral blood.
Stimulation of BL13, which is located at the lower border of the spinous process of the third thoracic vertebrae, will activate the sympathetic nerves—the thoracic segments 1–4—that innervate the bronchus. Stimulation of the sympathetic nerves causes the adrenal medulla to release catecholamines, which can stimulate adenylyl cyclase resulting in the formation of cyclic adenosine monophosphate (cAMP). Increased levels of cAMP give rise to bronchodilation, inhibition of mucous production in the bronchial glands, and blockage of mast cell degranulation. 14 Inhibition of mast cell degranulation reduces the release of inflammatory cells, including eosinophils. In addition, acupuncture at ST36 has been shown to maintain the homeostasis of the immune system due to its immunomodulatory effects on Th-1 as well as Th-2 dominant pathological conditions. 15 Kwon et al. 11 also reported that electroacupuncture at ST36 could alleviate the OVA-induced allergic asthma reaction by modulating CD4+ CD25+ T regulatory cells.
In addition to its effectiveness at treating pulmonary disease, the anti-inflammatory property of acupuncture has been demonstrated to have positive effects on several animal models of disease. Acupuncture alleviated the inflammatory response in a rat spinal cord injury model through its effect on M1 and M2 macrophages. 24 Acupuncture also elicited an anti-inflammatory effect in a rat model of migraine by activating the cannabinoid type 1 (CB1) receptor. 25 Our results have strengthened the evidence that acupuncture can be used to treat several pathological conditions through its anti-inflammatory effects.
Airway remodelling
Airway remodelling is believed to be the result of chronic repetitive injury caused by airway inflammation. Chronic exposure to OVA was used in this study to induce a chronic allergic inflammation and airway remodelling in Balb/C mice similar to those described in humans.3,26 Repeated OVA exposure has been shown to cause epithelial injury leading to epithelial hyperplasia and metaplasia, 3 epithelial hypertrophy, smooth muscle hyperplasia and goblet cell metaplasia. 27 In our study, a significant change in the bronchial structure, including the epithelial layer, smooth muscle thickness and goblet cells, was noticed in the OVA group compared with the control group. Locke et al. reported that OVA exposure could elicit early changes of remodelling such as goblet cell hyperplasia and epithelial thickening after just 4 days of exposure. These changes persisted with the addition of subepithelial collagen deposition in chronically exposed mice. 17 Other studies have also confirmed that epithelial and smooth muscle thickness are increased in the airway in an asthma mouse model.3,17,26
There has been debate over whether remodelling occurs due to inflammation or if it exists as a separate mechanism. Repetitive microinjuries to the airway wall due to chronic OVA exposure can cause persistent changes in the epithelium of the airway. Persistent changes in the epithelium can subsequently change the structure of the stroma, which is implicated as an important part of airway remodelling. 17 The airway epithelium responds to microinjuries by secreting various kinds of cytokines, including granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-8, transforming growth factor (TGF)α and profibrotic TGFβ, 17 and by producing large amounts of metalloproteinases (MMPs), 28 which play a role in inflammatory responses and remodelling. Infiltration of neutrophils, macrophages and T lymphocytes will further promote the release of cytokines and proteolytic enzymes, leading to airway structure destruction. 29
In our study, acupuncture at BL13 and ST36 was able to control airway remodelling as indicated by a pronounced reduction in bronchiolar epithelial and smooth muscle thickness, and the number of goblet cells. Acupuncture at BL13 and ST36 in combination also reduced the number of neutrophils significantly, which are likely to play a role in the control of airway remodelling. As previously reported, neutrophils secrete serine protease neutrophil elastase, which can destroy the elastin of airway and pulmonary components. 30 Neutrophils can cause epithelial damage by releasing reactive oxygen species, protease and cationic substances, chemokines and cytokines such as IL-1β, IL-6, IL-8 and TNFα.31,32 Neutrophils are also the main source of MMP-9, which plays a role in extracellular matrix reorganisation 28 and smooth muscle hyperplasia. 33 It has been reported that high levels of neutrophils correlate with high levels of MMP-9 in asthmatic patients. 34 Thus, it is likely that the increased level of neutrophils in our study may may have contributed to the development of airway remodelling.
Acupuncture, through activation of the sympathetic nervous system, reduces the production of Th-2 cytokines, induces the secretion of Th-1 cytokines, and creates a balanced state between these two immune cells. Acupuncture therefore modulates the equilibrium of Th-1/Th-2. 10 It also causes tissue injury leading to stimulation of Th-1 cytokine production, restoring and modulating the balance between Th-1 and Th-2 cytokines. 15 Acupuncture was also reported to regulate inflammatory cytokines and protect the lung in a rat COPD model by modulating histone deacetylase-2 (HADC2), a substance that represses the production of pro-inflammatory cytokines in alveolar macrophages. 20
Strengths and weaknesses
The OVA-induced allergic asthma model that we used in this study is well established and resembles human allergic asthma in terms of the occurrence of eosinophilic inflammation, airway hyperresponsiveness and the release of inflammatory cytokines. However, several limitations exist including the fact that we only assessed inflammatory cells in peripheral blood. Additional data from bronchoalveolar lavage fluid would provide more support for the study. Further research should also investigate the role of certain inflammatory cytokines or potential changes in CD4, interferon ϫ and T cells in this model to strengthen the use of acupuncture in clinical practice.
Conclusion
The combination of acupuncture at BL13 and ST36 can reduce the inflammatory response and control airway remodelling in the bronchioles of a chronic asthma mouse model.
Footnotes
Contributors
IN designed the research, carried out the animal studies and experimental work, analysed the data and drafted the manuscript. BP, AM, and KS conceived the study, contributed to its design and supervised data acquisition and interpretation. DHP contributed to the experimental work and data analysis. MM contributed to data analysis and interpretation, manuscript preparation and critically revised the manuscript. All authors approved the final version of the manuscript accepted for publication.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Provenance and peer review
Not commissioned; externally peer reviewed.
