Abstract
The biopsychosocial model (BPSM) has guided psychological science for decades by emphasizing the interplay of biological, psychological, and social factors in health. Despite its influence, the model has been critiqued for lacking mechanistic specificity, particularly in clarifying how psychosocial factors translate into cellular and molecular processes. Mitochondria, once viewed mainly as energy producers, are now understood to shape immune signaling, stress responses, and neural functioning. Emerging evidence shows that mitochondrial processes are sensitive to psychosocial influences such as chronic stress, trauma, and social connection, with downstream effects on inflammation and disease risk. We highlight findings that support these links while noting areas in which evidence remains preliminary, including possible feedback from mitochondrial changes to psychosocial processes. We also review interventions such as physical activity, mindfulness, and social support that may enhance mitochondrial function. This article positions mitochondria as one promising biological bridge between mind and body, offering a testable extension of the BPSM and identifying opportunities for psychologists to advance this emerging field.
Psychology has long asked how psychological processes shape physiological functioning. The biopsychosocial model (BPSM), formalized by George Engel in 1977, provided a framework for linking biological, psychological, and social levels of analysis. Drawing on earlier foundations such as Claude Bernard’s concept of the “milieu intérieur,” Hans Selye’s work on stress physiology and Robert Ader’s discovery of neuroimmune conditioning reshaped health psychology, behavioral medicine, and psychosomatic science and have influenced research agendas, clinical practice, and health policy (Ader, 2003; Engel, 1977; Selye, 1936; Taylor, 2022). Yet critics note that the BPSM offers limited mechanistic specificity, particularly in explaining how social and psychological experiences are translated into cellular and molecular change (Bolton & Gillett, 2019). In practice, the BPSM is most informative when paired with explicit biological pathways that translate psychosocial experience into cellular and molecular change, which allows falsifiable predictions and direct tests.
Recent advances are beginning to close this gap. New methods allow researchers to identify biological systems that convert psychosocial experiences into physiological outcomes. One promising candidate is mitochondria, the small structures within cells best known as the “powerhouses” that generate energy for nearly all cellular activity. Beyond energy production, mitochondria regulate immune signaling, hormone release, and stress adaptation. They are also sensitive to environmental and psychosocial conditions, with early human studies and animal models suggesting that stress, trauma, and social connection can shape mitochondrial function (Marchi et al., 2023; Picard, McEwen, et al., 2018).
Work in psychoneuroimmunology has already demonstrated that immune cells carry receptors for stress hormones and that both the central and peripheral nervous systems respond to immune-derived signals. These systems secrete their messengers such as hormones, neurotransmitters, and cytokines, creating a dynamic two-way communication network among the brain, endocrine system, and immune system (Blalock, 1989; Kusnecov & Anisman, 2013). Building on these advances, mitochondria emerge as another integrative hub. They sit at the crossroads of stress physiology, immune regulation, and cellular energy balance. Their sensitivity to psychosocial factors makes them a tractable biological pathway for extending the BPSM into measurable mechanisms (Kramer & Bressan, 2018; Marchi et al., 2023).
In this article, we review evidence that mitochondrial biology offers a meaningful extension of the biopsychosocial model. We outline key mitochondrial functions relevant to psychology, summarize research on how psychosocial experiences influence mitochondrial processes, and highlight interventions that may promote mitochondrial resilience and mitigate age-related cognitive decline. Our goal is to synthesize these insights for a broad audience of psychological scientists, students, and science communicators. To support accessibility, we also include a glossary of key terms (Table 1) and a summary of commonly used mitochondrial measures (Table 2).
Glossary of Common Biological Terms Used in the Article
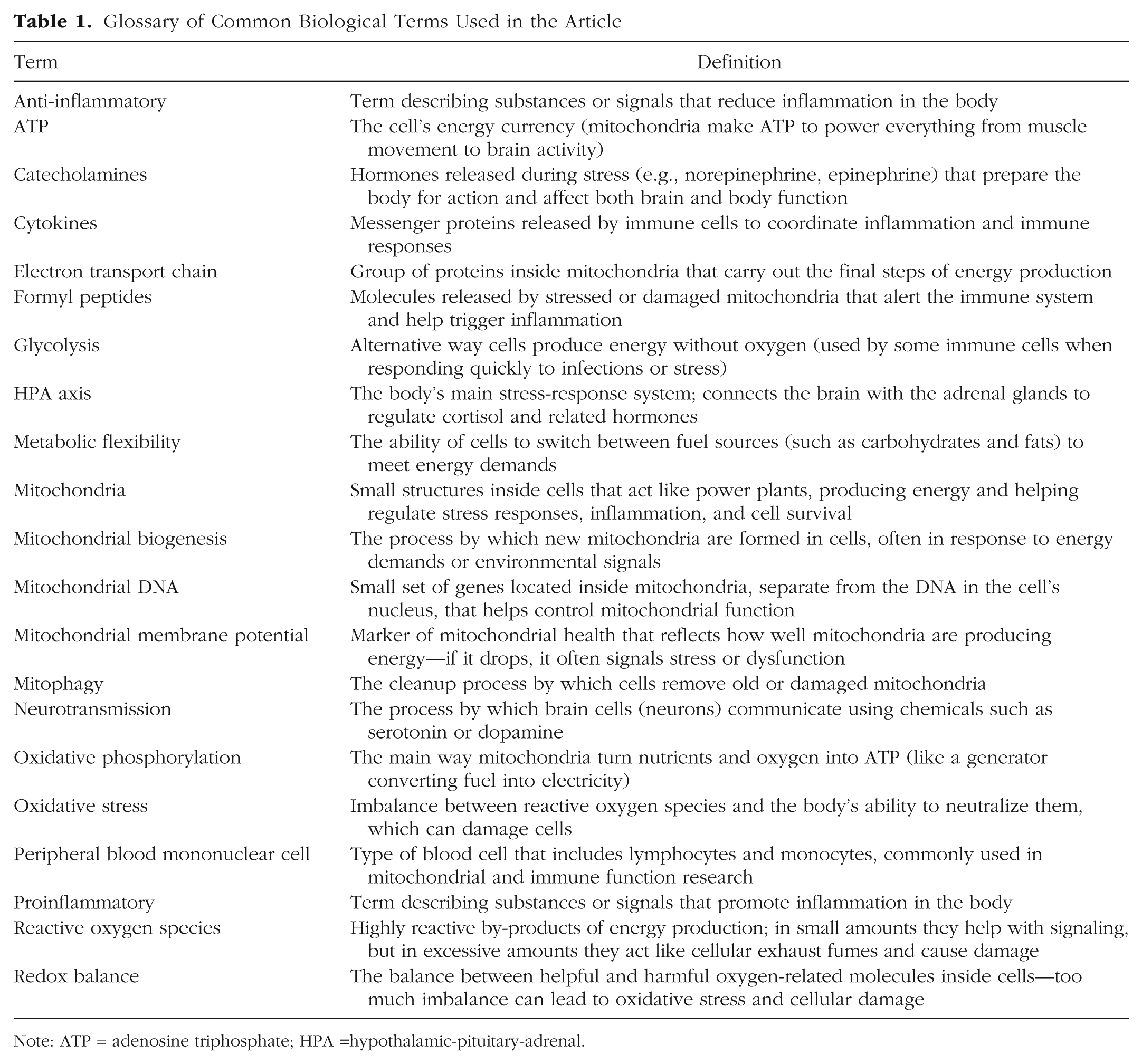
Note: ATP = adenosine triphosphate; HPA =hypothalamic-pituitary-adrenal.
Standard Measures of Mitochondrial Function: Biological Roles, Psychological Relevance, and Feasibility

Note: mtDNA = mitochondrial DNA; PBMC = peripheral blood mononuclear cell; PTSD = posttraumatic stress disorder; ATP = adenosine triphosphate; ROS = reactive oxygen species; 8-oxo-dG = 8-oxo-2’-deoxyguanosine; PINK1 = PTEN-induced kinase 1.
Mitochondria, Stress, and Cellular Adaptation
Mitochondria are central to cellular and overall body function. They are critical in energy-demanding cells such as neurons and immune cells, where they help the body respond to psychological and physical challenges (Marchi et al., 2023). Mitochondria generate adenosine triphosphate (ATP) and adjust their energy output to meet changing demands. This flexibility makes them critical for maintaining balance and supporting resilience under stress (Picard & McEwen, 2018).
Beyond producing energy, mitochondria serve as hubs that connect cellular activity with larger regulatory systems. They integrate signals from nutrients, hormones, and stress mediators to shape how cells allocate resources. This adaptability, known as metabolic flexibility, allows cells to shift between fuel sources depending on current needs (Chandel, 2015). Mitochondria also interact with the hypothalamic-pituitary-adrenal axis and the immune system, helping the body mount immediate defenses while maintaining longer-term stability (Picard, McEwen, et al., 2018). Their sensitivity to environmental and psychosocial signals positions them at the intersection of mind and body.
Acute stress shows how mitochondria rapidly adjust to meet immediate demands. Preclinical studies (i.e., research in animal models or cell systems) show that acute stress hormones, including cortisol and catecholamines, act directly on mitochondria to increase energy production, stimulate mitochondrial biogenesis, and enhance enzyme activity (Picard & McEwen, 2018). These rapid adjustments provide the energy needed for short-term adaptation, while safeguards limit the buildup of reactive oxygen species (ROS). In this way, mitochondria support resilience by sustaining balance during acute challenges.
Chronic stress, in contrast, gradually erodes mitochondrial efficiency and resilience. Preclinical work shows that prolonged exposure to cortisol and inflammatory signals reduces ATP production, increases ROS, and accelerates mitochondrial DNA damage (Lapp et al., 2019; Picard & McEwen, 2018). Over time, these changes disrupt energy balance, amplify inflammation, and impair brain signaling. Such processes provide a cellular basis for fatigue, cognitive difficulties, and mood disturbances often linked to prolonged stress (Picard & McEwen, 2018).
Although mechanistic detail comes primarily from preclinical models, human studies reveal parallel patterns. Experimental research shows that acute psychosocial stress triggers rapid increases in circulating cell-free mitochondrial DNA (mtDNA) along with cortisol elevations (Trumpff et al., 2019). Other work suggests that mitochondrial function in circulating immune cells reflects broader physiological states, including brain energy metabolism (Bernier et al., 2020). These findings indicate that mitochondria not only respond to stress in measurable ways but also may serve as accessible biomarkers of how stress becomes biologically embedded.
Together, these findings position mitochondria as a biological link between stress exposure and health outcomes. They help the body adapt to short-term challenges, but also reveal how chronic stress undermines cellular efficiency and contributes to long-term health risks. Figure 1 illustrates this central role, showing how psychosocial stressors activate neuroendocrine mediators that alter mitochondrial function, leading to downstream changes in oxidative stress, inflammation, and energy metabolism. These processes, in turn, influence emotional, cognitive, and physical health. Solid arrows depict well-established pathways, whereas dotted arrows highlight emerging or speculative links, including possible feedback loops from mitochondria back to psychosocial processes.
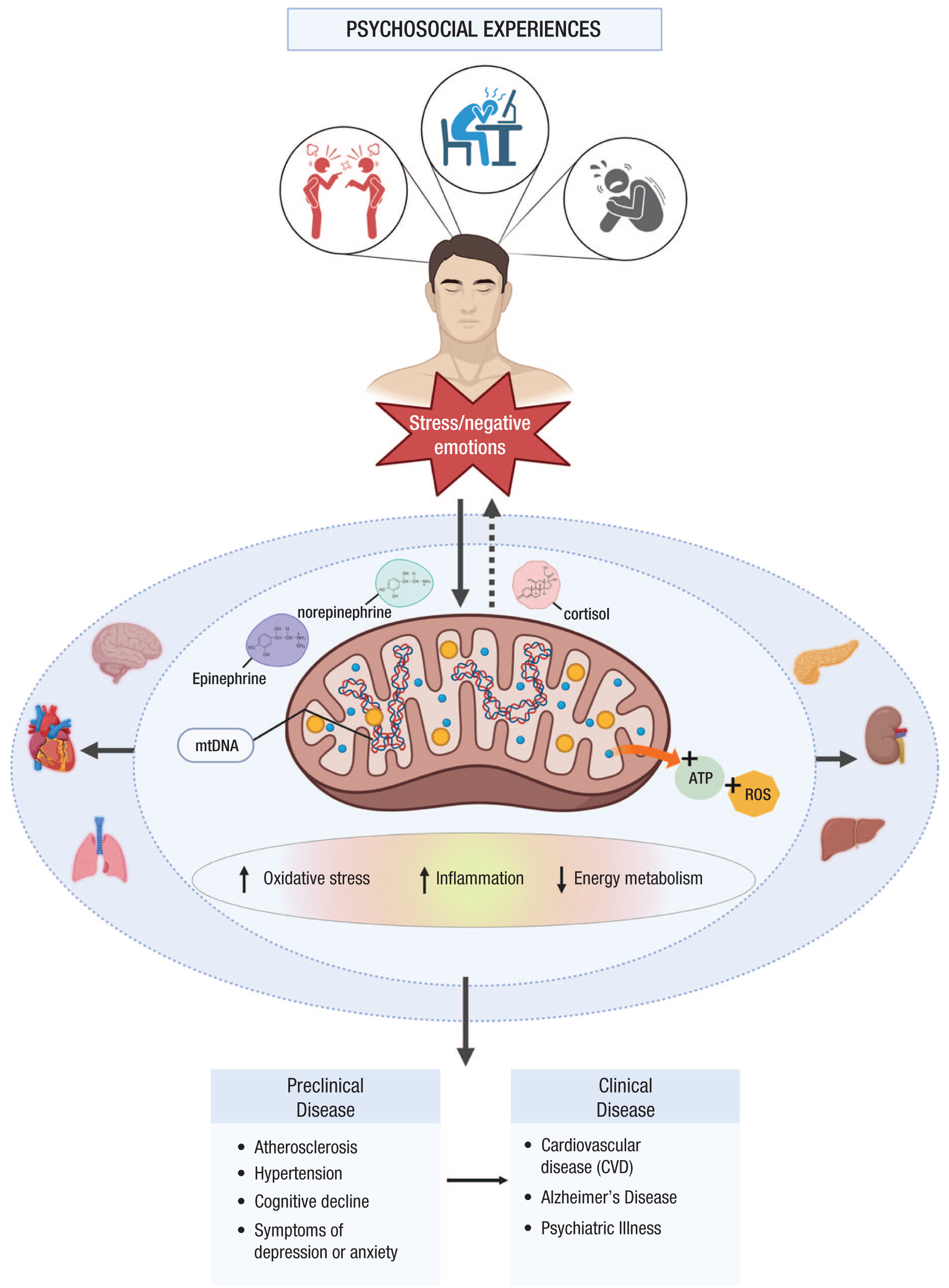
Activation of neuroendocrine mediators by psychosocial stressors. Psychosocial stressors activate pathways that release stress mediators (norepinephrine, epinephrine, cortisol), which can disrupt mitochondrial function. Altered mitochondrial activity contributes to oxidative stress, inflammation, and impaired energy metabolism, with downstream effects on physical and mental health. Solid arrows indicate well-supported pathways, whereas dotted arrows represent proposed or less-established links, including a potential feedback loop back to psychosocial processes.
Mitochondria in Stress and Health
If mitochondria help translate psychosocial stress into cellular changes, the next question is how those changes manifest in health outcomes. Research indicates that differences in mitochondrial function may partly explain why some people are more vulnerable to stress whereas others remain resilient. For example, Picard and McEwen (2018) proposed that mitochondria act as a cellular “stress integrator,” adjusting energy output and signaling in ways that influence resilience or vulnerability. Converging evidence links mitochondrial function to psychological and physical health.
Genetic and daily-life evidence
Large-scale analyses reinforce this connection. Liu et al. (2023) examined the UK Biobank and found that variations in mtDNA were associated with greater risk for anxiety and depression, with associations stronger at higher levels of C-reactive protein, a marker of inflammation. Many of the implicated mtDNA variants mapped onto genes regulating oxidative phosphorylation, the process by which mitochondria turn nutrients into energy. Evidence from everyday contexts offers further support. Picard, Prather, et al. (2018) introduced a mitochondrial health index that integrates mtDNA copy number and respiratory chain enzyme activity into a single score of mitochondrial capacity (see Table 2). In mothers of children with autism, a high-stress caregiving group, this index was lower than in controls, and caregivers also reported less daily positive affect. Participants who experienced a more positive mood in the days before blood collection had higher values, suggesting that mitochondria reflect not only chronic stress but also short-term psychosocial experiences.
Caregiving and resilience
Recent work extends these findings to dementia caregiving. Wu-Chung et al. (2024) examined spousal caregivers and found that those with higher mitochondrial respiratory capacity, a measure of how efficiently mitochondria can generate energy when demand is high, reported better physical functioning and were buffered against the negative emotional effects of caregiving burden. Caregivers with lower capacity were more vulnerable, underscoring mitochondrial efficiency as a biological factor in resilience to chronic stress.
Immune pathways and stress signaling
Because mitochondria regulate both energy production and immune function, they represent a potential bridge between psychosocial stress and inflammatory outcomes. West and Shadel (2017) reviewed evidence that damaged mitochondria release mtDNA fragments and oxidative by-products that act as danger signals to the immune system, amplifying inflammation. Consistent evidence in humans comes from Lacourt et al. (2022), who found that mitochondrial metabolism in circulating immune cells was associated with fatigue and inflammatory profiles in breast cancer survivors. Overall, these findings indicate that mitochondria contribute to both cellular energetics and immune regulation, positioning them as an integrative pathway through which psychosocial stress affects health.
Mechanistic and experimental evidence
Animal and cell models clarify how stress signals reach mitochondria. For example, Lapp et al. (2019) demonstrated that glucocorticoid receptors, proteins that bind cortisol, can translocate into mitochondria and directly regulate mtDNA transcription. This alters respiratory chain activity and energy signaling, pointing to a molecular pathway by which stress hormones influence mitochondrial function in ways relevant to stress adaptation. Laboratory stress experiments in humans parallel these links. Trumpff et al. (2019) showed that acute psychosocial stress led to rapid increases in cell-free mtDNA, small fragments of mitochondrial DNA released into the bloodstream that rose in tandem with cortisol. Stress was induced with the Trier Social Stress Test (Kirschbaum et al., 1993), and cell-free mtDNA was quantified from serum using quantitative polymerase chain reaction. The study did not include a no-stress control group, which limits causal inference. In a later study, Trumpff et al. (2024) analyzed postmortem human brain tissue and found that individuals reporting greater social support and a sense of purpose had higher mitochondrial protein levels, whereas those with distress and isolation had lower levels. Together, these findings illustrate that mitochondria not only respond to stress but also track psychosocial resources, making them promising biomarkers of how stress becomes biologically embedded.
Brain vulnerability
Because the brain has exceptionally high energy demand, it appears especially vulnerable to mitochondrial dysfunction (Harris et al., 2012). Animal models link mitochondrial mutations and copy-number changes to neurodegenerative processes, and parallel human studies show similar patterns in depression, posttraumatic stress disorder (PTSD), and Alzheimer’s disease (Bersani et al., 2016; Cai et al., 2015; Pei & Wallace, 2018; Wang et al., 2017). Reduced mitochondrial efficiency limits the energy available for neurotransmission and plasticity, undermining processes that support mood regulation and memory.
Health implications
Taken together, this body of work positions mitochondria as a key pathway, along with inflammation, autonomic regulation, and endocrine signaling, through which psychosocial factors shape health and functioning. Although most studies remain cross-sectional or correlational, the consistency of findings across diverse populations, methods, and outcomes suggests biologically meaningful links between mitochondrial biology and psychological functioning. Evidence implicates mitochondrial dysfunction in emotional symptoms such as anxiety and depression, cognitive difficulties involving memory and attention, and a range of physical-health conditions, including neuropsychiatric and neurodegenerative disorders, cardiometabolic disease such as ischemic heart disease and type 2 diabetes, and certain cancers (Lee & Han, 2017; Liu et al., 2023; Pei & Wallace, 2018; Suomalainen & Nunnari, 2024). For cardiometabolic outcomes in particular, mitochondrial and immune signaling provide a plausible mechanistic bridge to ischemic heart disease and type 2 diabetes (Geto et al., 2020).
Moving forward, embedding mitochondrial measures within biopsychosocial research will help clarify how daily experiences become biologically embedded and extend the biopsychosocial model with specific, testable molecular pathways. Doing so can move the field beyond documenting associations to identifying mechanisms that explain when, why, and for whom psychosocial stressors and resources influence health. By specifying mitochondrial pathways, psychological science gains leverage to inform intervention development, refine prevention strategies, and connect cellular biology to lived experience in ways that strengthen the translational impact of the biopsychosocial model.
Interventions
Psychological and behavioral strategies are increasingly being examined for their potential to influence mitochondria, the organelles that regulate cellular energy and stress adaptation. Although this work is still emerging, it suggests that mitochondria are not only vulnerable to psychosocial stressors but also possibly responsive to interventions that promote resilience. The evidence varies in strength across domains.
Exercise provides the strongest and most consistent evidence. Endurance training enhances mitochondrial function by boosting ATP production, stimulating mitochondrial biogenesis, and increasing the activity of enzymes involved in energy metabolism. Human studies have reported nearly twofold increases in mitochondrial enzyme activity after 12 weeks of aerobic training and roughly 30% gains in mitochondrial content after 10 weeks (San-Millán, 2023). Periods of inactivity quickly diminish these benefits, but retraining can restore much of what was lost. Collectively, this evidence suggests that mitochondrial adaptation may be one pathway through which exercise supports both physical and psychological resilience.
Mindfulness and psychotherapy show more preliminary but promising effects. In a randomized crossover trial, an intensive weekend mindfulness program reduced anxiety, increased brain-derived neurotrophic factor, and altered oxidative phosphorylation markers, although no changes were observed in cortisol, heart rate variability, or working memory (Gomutbutra et al., 2022). Similarly, in women with PTSD, 6 weeks of inpatient psychotherapy was associated with reductions in mtDNA copy-number and structural deletions, interpreted as decreased mitochondrial allostatic load. However, these biological changes were not correlated with symptom improvement, and the absence of a control group limits interpretation (Hummel et al., 2023). Together, these studies demonstrate feasibility but require replication in larger, well-controlled trials.
Social-support and stress-management programs are widely theorized to protect mitochondrial health by reducing cortisol and dampening inflammatory activity. Although animal models and indirect human research strongly support this idea (Picard & McEwen, 2018), very few studies have measured mitochondrial outcomes directly. This gap highlights a key opportunity for psychologists to test whether relational and stress-management interventions operate at the cellular level.
Taken together, the clearest evidence links exercise to improved mitochondrial health, mindfulness and psychotherapy provide proof of concept but remain preliminary, and social-support interventions are theoretically compelling yet undertested. Comparative research across these domains will help determine whether mitochondria represent a common pathway through which psychosocial interventions promote resilience.
Future Directions
The next phase of research on mitochondria and psychosocial processes requires moving beyond correlational evidence toward designs that can establish causal pathways. Longitudinal studies with repeated mitochondrial assessments will clarify how chronic stress and resilience shape bioenergetic adaptation over time. Experimental designs that manipulate stress or resilience in controlled settings while embedding mitochondrial endpoints will provide further leverage for identifying mechanisms. Together, these approaches can determine whether changes in mitochondrial biology serve as pathways through which psychosocial experiences exert long-term effects on health (Wu-Chung et al., 2024).
Methodological advances are also critical. Although measures such as ATP production efficiency, spare respiratory capacity, mtDNA copy number, and cell-free mitochondrial DNA are validated in laboratory and clinical contexts, their use in large-scale psychological studies remains limited. Standardized assays and reporting guidelines will be essential for ensuring comparability across studies and for scaling mitochondrial research to larger, more representative cohorts that reflect cultural and psychosocial diversity (Kelly et al., 2024; San-Millán, 2023). Developing accessible, cost-effective methods for assessing mitochondrial health will be particularly important for embedding these measures in population-level studies.
Last, mitochondria should not be studied in isolation but as part of broader networks that include brain, immune, metabolic, and stress-response systems. Integrating mitochondrial measures into these multilevel models will help clarify how lived experience becomes biologically embedded across the lifespan. To date, no single longitudinal human study has tested the complete sequence from psychosocial exposure to mitochondrial change to clinical outcomes within the same cohort. By examining mitochondria within the context of these interacting systems, psychological science can move closer to identifying the cellular pathways through which psychosocial factors promote resilience or increase vulnerability (Marchi et al., 2023; Suomalainen & Nunnari, 2024).
Conclusion
Mitochondria provide one promising pathway for refining the biopsychosocial model into a more mechanistically precise framework. Current evidence, although still limited and often correlational, suggests that mitochondrial function is sensitive to psychosocial stress and protective factors, with implications for cellular energetics, immune signaling, and neural processes. These findings highlight mitochondria as tractable mediators through which lived experience can influence health, but they should be understood as part of a larger network that also includes inflammation, autonomic regulation, and neuroendocrine function. Future research must clarify causal pathways and delineate the conditions under which mitochondria contribute uniquely versus redundantly to these processes. Embedding mitochondrial measures within multilevel models that include brain, immune, and metabolic systems will allow psychologists to trace how subjective experience becomes biologically embedded. In doing so, the field can expand its methodological reach, deepen its mechanistic precision, and clarify the cellular foundations of resilience and disease, advancing both psychological theory and public health.
Recommended Reading
Kelly, C., Trumpff, C., Acosta, C., Assuras, S., Baker, J., Basarrate, S., Behnke, A., Bo, K., Bobba-Alves, N., Champagne, F. A., Conklin, Q., Cross, M., De Jager, P., Engelstad, K., Epel, E., Franklin, S. G., Hirano, M., Huang, Q., Junker, A., . . . The MiSBIE Study Group. (2024). (See References). National Institutes of Health-supported platform/biobank standardizing assays and infrastructure to scale mind–mitochondria research.
Picard, M., & McEwen, B. S. (2018). (See References). Clear conceptual bridge between stress physiology and mitochondria; great on mechanisms and testable ideas.
Tyrka, A. R., Burgers, D. E., Philip, N. S., Price, L. H., & Carpenter, L. L. (2020). Stress and psychiatric disorders: The role of mitochondria. Annual Review of Clinical Psychology, 16, 165–186. Comprehensive multilevel review linking adversity to mitochondrial dysfunction in clinical contexts.
Wu-Chung, E. L., Medina, L. D., Paoletti-Hatcher, J., Lai, V., Stinson, J. M., Mahant, I., Schulz, P. E., Heijnen, C. J., & Fagundes, C. P. (2024). (See References). Connects bioenergetics to daily affect and physical functioning in human caregivers.
