Abstract
Enterocutaneous fistula (ECF) traditionally relies on surgical treatment and lacks effective therapies to promote tissue regeneration. This study constructed a composite system based on hypoxia-preconditioned human umbilical cord mesenchymal stem cell–derived exosomes loaded onto a gelatin sponge (GS Hypo-Exos), aiming to explore its potential application in ECF treatment. First, systematic characterization of exosomes was performed: transmission electron microscopy, nanoparticle tracking analysis, western blot, and flow cytometry confirmed successful exosome extraction. Material evaluation indicated that the gelatin sponge exhibited high porosity and good biocompatibility. Scanning electron microscopy confirmed successful loading of exosomes onto the sponge surface, and
Keywords
Introduction
An enterocutaneous fistula (ECF) is an abnormal connection between the gastrointestinal tract and skin, leading to the leakage of enteric contents1,2. Its causes are diverse, including surgical complications, trauma, and local invasion by malignancies3–5. Malignancy-associated ECF often indicates locally advanced disease with poor prognosis 3 . Current clinical management primarily focuses on infection control, nutritional support, and definitive surgical repair6–8. However, surgical interventions are often complex and carry significant risks 1 . A major gap in ECF treatment is the lack of effective, non-surgical therapies that can actively promote tissue regeneration and fistula healing, highlighting the need for novel regenerative strategies.
In recent years, mesenchymal stem cells (MSCs) and their secreted exosomes (Exos) have made groundbreaking advances in the field of regenerative medicine and have shown promise for treating ECF, owing to their exceptional capabilities in immune regulation, anti-inflammation, pro-angiogenesis, and tissue repair9–12. Human umbilical cord mesenchymal stem cells (hUCMSCs) are an ideal source of cells for research and therapy because of their easy accessibility, low immunogenicity, and strong regenerative potential 13 . Furthermore, their exosomes (hUCMSC-Exos) circumvent the risks associated with cell transplantation, emerging as a promising alternative strategy14,15. Studies have indicated that hypoxic preconditioning can significantly enhance the bioactivity of MSCs and their Exos, enriching them with pro-repair factors and improving their adaptability to inflammatory microenvironments16,17. However, a key limitation of direct exosome application is their rapid clearance from injury sites and difficulty in maintaining effective concentrations 18 . Therefore, developing a biomaterial carrier capable of loading, protecting, and enabling the sustained release of Exos at the fistula site is crucial. Gelatin sponge (GS), a clinically established and widely used absorbable hemostatic material, offers excellent biocompatibility, biodegradability, a three-dimensional structure with high porosity, and efficient biomolecule loading and sustained-release capabilities. This makes GS an ideal platform for the efficient local delivery of Exos19,20.
Building on this foundation, this study proposes the construction of a composite system consisting of a GS loaded with hypoxia-preconditioned hUCMSC-derived Exos. Utilizing an experimental animal model of colocutaneous fistula, we will conduct investigations aimed at exploring the efficacy and functional value of this composite system in fistula healing, thereby providing a novel therapeutic strategy for the treatment of colocutaneous fistulas.
Materials and methods
Isolation and identification of hUCMSC-exos
The hUCMSCs were purchased from Guangzhou Saliai Stemcell Co., Ltd. They were cultured in DMEM/F12 medium supplemented with 10% Fetal Bovine Serum (FBS) under humidified air at 37°C containing 5% CO2. hUCMSCs were passaged when cell confluency reached 70%–80%. Passages 2–5 were used for exosome-extraction experiments. Cells were cultured in serum-free basal medium for 24 h, and the supernatant was then collected for exosome extraction. To obtain hUCMSC-Exos under hypoxic conditions, hUCMSCs were cultured in a hypoxic (5% O2) environment, and the supernatant was collected for exosome extraction. Exos were isolated using an exosome-extraction kit (Thermo Fisher, Shanghai, China). Exosomes under hypoxic conditions were characterized by Western Blot (WB) to detect the exosome surface marker protein Alix. Conventional Transmission Electron Microscope (TEM), Nanoparticle Tracking Analysis (NTA), and flow cytometry were used to characterize hUCMSC-Exos under hypoxic conditions (hUCMSC-Hypo-Exos), and the expression of their markers (CD63, CD81, and CD9) was analyzed. Subsequently, hUCMSC-Exos were stored at −80°C.
Preparation of GS-Exos complexes
The Exos extracted under the aforementioned different conditions were dissolved in phosphate buffered saline (PBS; 50 μg Exos in 50 μL PBS), followed by the addition of trehalose (final concentration: 25 mM). The GS were cut into 5 mm3 pieces and divided into three groups. These were then infused with PBS, hypoxic Exos complex, and normoxic Exos complex, followed by lyophilization for 48 h. The morphology of the GS loaded with PBS/exo complexes was observed using Scanning Electron Microscope (SEM). The three types of lyophilized complexes were used in subsequent animal and cell experiments.
Detection of the controlled-release ability of GS-Exos complex
The obtained lyophilized GS hypoxic exosome complex (GS Hypo-Exos) and GS-normoxic exosome complex (GS-Norm-Exos) were immersed in 500 μL of PBS (37°C, 5% CO2). At designated time points (0.5, 1, 2, 3, 5, 7, 10, 14, and 21 days), half of the PBS (250 μL) was collected and replaced with an equal volume of fresh PBS. The concentration of VEGF released into the medium was measured using a human VEGF ELISA kit (Mlbio, Shanghai, China), and the concentration of IGF-1 released was measured using a human IGF-1 ELISA kit (Mlbio, Shanghai, China). All measurements were performed in triplicates for each sample. Three independent experiments were conducted.
Animal model and treatment protocol
Twenty-four male Sprague–Dawley rats (SPF grade), weighing 200 ± 20 g, were used in this study. After 1 week of adaptive feeding, a colonocutaneous fistula model was surgically established in all rats (n = 6 per group). Briefly, a 3-cm midline laparotomy incision was made. The distal cecum was exteriorized through an offset opening in the abdominal musculature and subcutaneous space, creating a 10-mm-long tract. The cecum was secured to the skin at the incision site on the right flank of the rat using four sutures (Vicryl 4/0), establishing ECF. The rats were allowed free access to food and water. Model establishment was considered successful if the external fistula orifice diameter was ≥4.5 mm and exhibited fecal discharge. Following successful fistula establishment, the rats were randomly divided into four groups (n = 6 per group A)—group A (control): received no implant treatment; group B: implantation of a lyophilized GS-PBS complex into the fistula tract; group C: implantation of lyophilized GS Hypo-Exos into the fistula tract; group D: implantation of lyophilized GS-Norm-Exos into the fistula tract. The external fistula orifice and tract were assessed every 3 days. Retention of the Exos within the fistula tract was monitored using a small-animal
Histological staining
Hematoxylin and eosin (H&E) and Masson’s trichrome staining were performed for histological evaluation to examine the gross morphology, inflammatory infiltration, and collagen distribution in the fistula tissue. The collagen volume fraction (CVF), defined as the ratio of the collagen-positive blue area to the total tissue area, was quantified using Masson’s trichrome staining. The CVF was calculated using the ImageJ software according to the following formula: CVF (%) = (collagen area / total tissue area) × 100%. Three randomly selected fields were analyzed per pathological section, and the mean value of these fields was recorded as the fibrosis ratio for each section. Three independent experiments were conducted.
Quantitative PCR analysis of Ki67 and CASP-3 expression
Fistula tract tissues from rats in each group were collected, minced, and thoroughly mixed with TRIzol reagent (Thermo Fisher, Shanghai, China), followed by homogenization. Total RNA was extracted using an RNA extraction kit. After determining the concentration and purity of the total RNA using a Nanodrop spectrophotometer, the RNA samples were reverse-transcribed into cDNA using the FastKing RT Kit (Tiangen, Beijing, China). Reaction mixtures were prepared containing a Taq Pro Universal SYBR qPCR Master Mix (Vazyme, Nanjing, China), gene-specific primers, cDNA template, and ddH2O (double-distilled water). Real-time quantitative polymerase chain reaction (RT-qPCR) was performed using a LightCycler 96 system. The primer sequences are listed in Table 1. Three independent experiments were conducted.
Primer sequence information.

Statistics
Statistical analyses and data graphing were performed using GraphPad Prism 9.0 and IBM SPSS Statistics (Version 26) for data management and advanced statistical modeling. Data are presented as mean ± SD. Comparisons among multiple groups were analyzed using one-way analysis of variance followed by Tukey’s post-hoc test. Statistical significance was defined as
Results
Comprehensive characterization of exosomal
The isolated Exos exhibited a homogeneous distribution within the TEM field of view (Fig. 1a, b). These vesicles displayed distinct, intact membrane boundaries with a characteristic cup-shaped morphology, consistent with classical exosomal ultrastructural features. NTA quantified the preparations with a median particle diameter of 108.1 nm and a concentration of 5.4 × 1012 particles/mL (Fig. 1c). The expression of the exosomal marker Alix was confirmed by WB analysis (Fig. 1d). The exosomal surface markers CD63 (mean ± SD: 73.79 ± 3.33), CD81 (mean ± SD: 76.54 ± 3.28), and CD9 (mean ± SD: 41.68 ± 2.30) were detected in the exosome samples using flow cytometry (Fig. 1e).
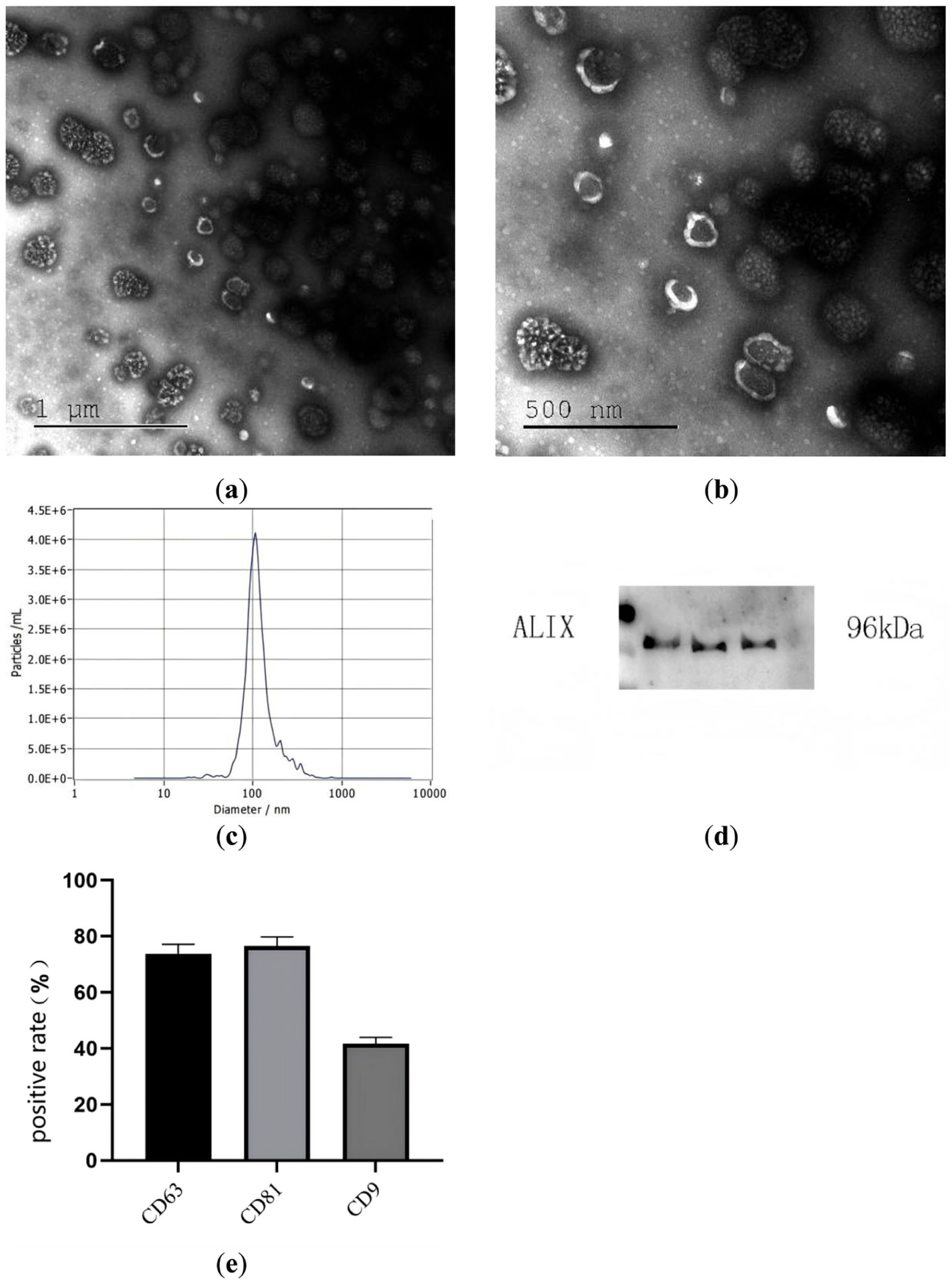
Characterization of Exos derived from hUCMSCs under hypoxic conditions (n = 3). (a–b) The morphology of Exos observed under a Transmission Electron Microscope (TEM); (c) particle size distribution of Exos; (d) Western Blot (WB) of Exos marker ALIX; (e) Exos surface markers. CD63 (mean ± SD: 73.79 ± 3.33), CD81 (mean ± SD: 76.54 ± 3.28), and CD9 (mean ± SD: 41.68 ± 2.30). hUCMSC: Human umbilical cord mesenchymal stem cells.
GS Hypo-Exos exhibits excellent biocompatibility and sustained-release effects
Tests on the GS showed that its average porosity was 81.34%, and its average water absorption rate was 59.37%. H&E staining revealed that after implantation into the subcutaneous space on the back of Sprague–Dawley rats, no significant pathological changes were observed, indicating that the material was safe and reliable (Supplementary Material 1). SEM of the GS-Exos complex showed that the exo-loaded GS had a rough surface with visible spherical and cup-shaped extracellular vesicles attached, whereas the exo-free GS surface appeared smooth (Fig. 2a–c). Controlled release testing of the composite materials revealed that the lyophilized GS Hypo-Exos complex achieved 100% IGF-1 release by day 10 (mean ± SD: 0.999 ± 0.0006), while the GS-Norm-Exos reached complete IGF-1 release by day 7 (mean ± SD: 1 ± 0) (Supplementary Material 2). For VEGF release, the GS Hypo-Exos complex attained 100% release by day 21 (mean ± SD: 1 ± 0), compared to day 14 for the GS-Norm-Exos complex (mean ± SD: 0.999 ± 0.001) (Supplementary Material 3). Furthermore, cumulative concentration measurements demonstrated that the lyophilized GS Hypo-Exos maintained significantly higher concentrations of both IGF-1 and VEGF throughout the release period (Fig. 2d, e).

GS-Exos scanning electron microscope and slow-release capacity (n = 3). (a) GS-PBS; (b) GS-Norm-Exos; (c) GS Hypo-Exos; (d) cumulative concentration of IGF-1 detected by ELISA; (e) cumulative concentration of VEGF detected by ELISA. GS: gelatin sponge; PBS: Phosphate Buffered Saline; Exos: exosomes; ELISA: enzyme-linked immunosorbent assay; VEGF: Vascular Endothelial Growth Factor; IGF-1: Insulin-like Growth Factor 1.
GS-Exos fluorescence declines gradually in fistula tract

hUCMSCs-Exo promotes ECF healing
Hypoxic hUCMSCs-Exo demonstrated superior therapeutic efficacy in promoting intestinal fistula healing. A comparative analysis of rat body weights revealed a general increasing trend across all groups under identical feeding conditions prior to day 7. However, from days 7 to 21, group C (treated with GS Hypo-Exos) exhibited greater weight gain than all other groups (Fig. 4a). Measurements of the fistula orifice area on days 14 and 21 showed a reduction in size in groups C and D compared to the control groups (Fig. 4b). Furthermore, when assessed by the combined fistula orifice perimeter, group C displayed the smallest perimeter on day 21 (Fig. 4c), indicating the optimal therapeutic outcome in the hypoxic exosome–treated group.
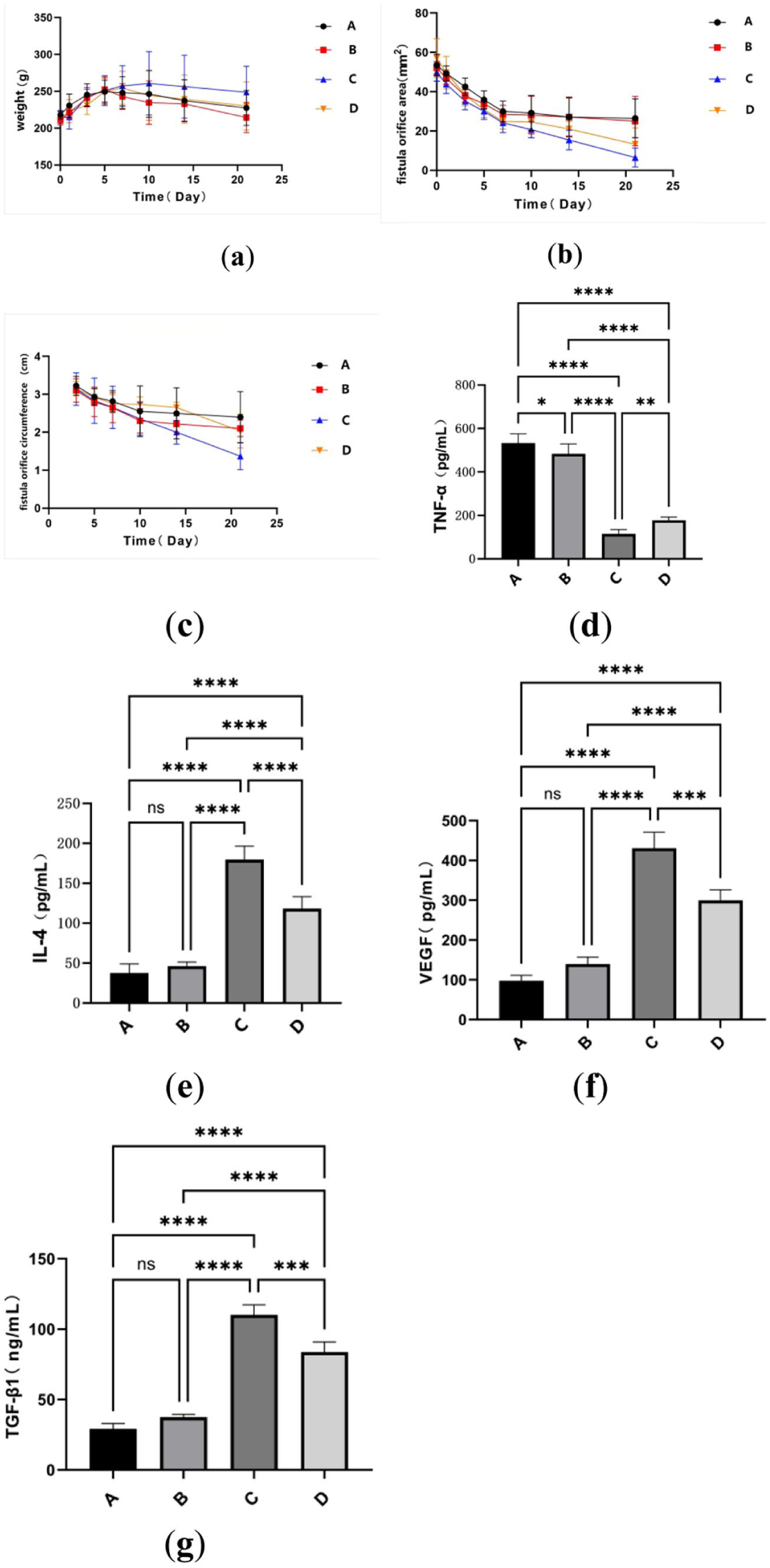
GS Hypo-Exos promotes tissue healing in rats and regulates inflammatory factors (n = 3). (a) Changes in rat weight during the treatment period; (b) changes in rat fistula orifice area during the treatment period; (c) changes in rat fistula orifice circumference during the treatment period; (d) ELISA was used to measure TNF-α levels (mean ± SD, A: 532.69 ± 42.70; B: 483.140 ± 45.634; C: 114.959 ± 20.075; D: 178.011 ± 14.376; C vs D:
Significant differences were also observed in the serum inflammatory cytokine concentrations among the groups. Compared with groups A and B, TNF-α levels were significantly reduced in both groups C and D, with group C showing a further significant decrease relative to group D (
VEGF and TGF-β1 levels in the fistula tissues were measured using ELISA. The results showed that the concentrations of VEGF and TGF-β1 in groups C and D were significantly higher than those in groups A and B (
Histology validates efficacy of hUCMSCs-Exos therapy for ECF repair
Histopathological analysis using H&E staining revealed distinct therapeutic outcomes. Groups A and B exhibited substantial inflammatory cell infiltration, with inflammatory infiltration areas of 84.754% and 71.17%, respectively (Fig. 5a, b). In contrast, group C showed reduced inflammation, with the smallest inflammatory area at 37.928% (Fig. 5c). Similarly, group D also demonstrated a decrease in the inflammatory infiltration area (56.089%) when compared to groups A and B (Fig. 5d). Compared with groups A and B, fistula tissues in groups C and D exhibited abundant blue-stained collagen fibers with a dense and organized arrangement, suggesting active collagen matrix synthesis and favorable tissue repair progression. Among these, collagen fiber deposition was most prominent in group C, and its CVF was significantly higher than that of all other groups (n = 3,
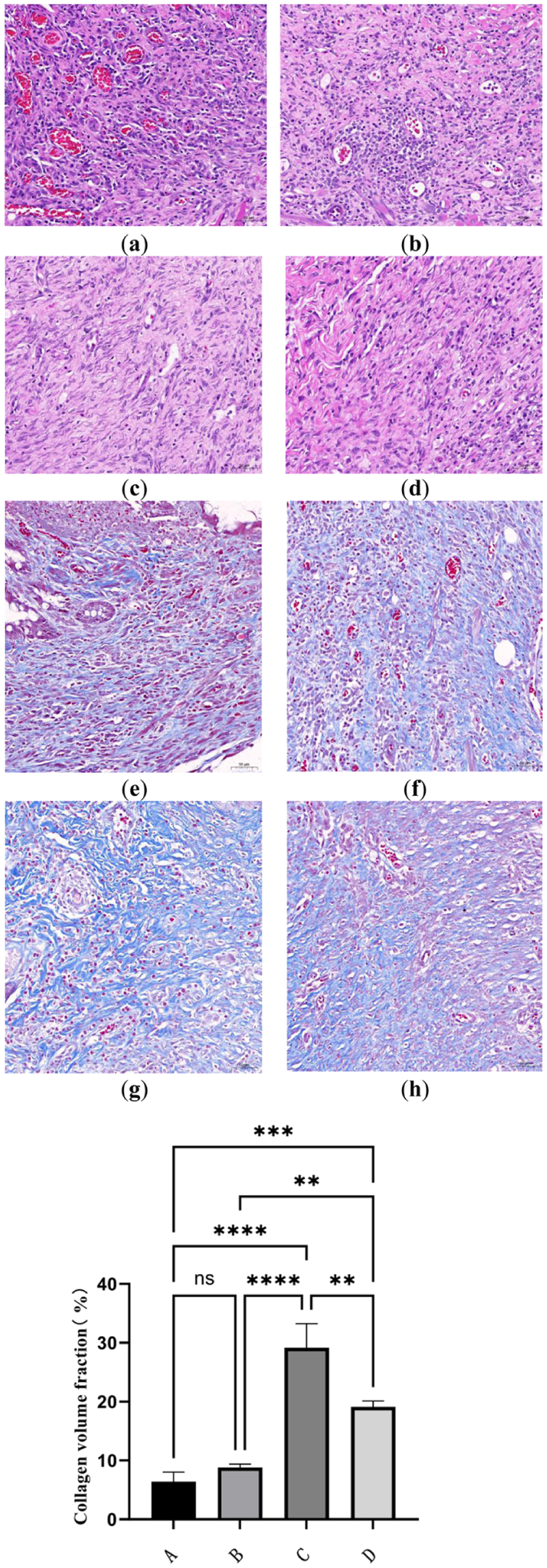
Histological analysis after GS-Exos treatment of ECF (n = 3, magnification 200X). (a) Group A (Blank) of HE; (b) group B (GS-PBS) of HE; (c) group C (GS Hypo-Exos) of HE; (d) group D (Normoxic Exos-GS) of HE; (e) group A (Blank) of masson; (f) group B (GS-PBS) of masson; (g) group C (GS Hypo-Exos) of masson; (h) group D (Normoxic Exos-GS) of masson; (i) collagen volume fraction (mean ± SD, A: 6.426 ± 1.598; B: 8.828 ± 0.543; C: 29.152 ± 4.086; D: 19.114 ± 0.988; C vs D:
RT-qPCR/WB confirms hypoxic exosomes boost Ki67 and suppress CASP-3 in fistula tissues
RT-qPCR was used to detect differences in the mRNA expression levels of Ki67 and CASP-3 in fistula tissues from different groups of rats. The results showed that the expression levels of Ki67 in the exosome-treated groups (groups C and D) were significantly higher than those in groups A and B (

The effect of GS-Exos on the expression of Ki67 and CASP-3 in fistula tissue (n = 3). (a) Ki67 mRNA expression in fistula tissue (mean ± SD, A: 1.01 ± 0.106; B: 1.62 ± 0.357; C: 8.52 ± 1.037; D: 4.18 ± 0.824; C vs D:
Discussion
ECF is one of the most serious life-threatening complications after abdominal surgery, with a high risk of death 21 . Therefore, exploring novel therapeutic strategies is of paramount importance. Exos derived from hUCMSCs have demonstrated significant therapeutic value in regenerative medicine and wound healing. With their favorable targeting capabilities and amenability to engineering modifications, they represent a promising option for ECF treatment 22 . This study successfully constructed a novel composite material based on a GS sustained-release carrier loaded with hUCMSC Hypo-Exos, which demonstrated significant pro-healing effects and accelerated tissue regeneration. By innovatively combining the sustained-release properties of biomaterials with an exosome functional enhancement strategy, this study provides a new therapeutic approach for ECF.
Recent reports indicate that MSCs can promote fistula closure in patients with Crohn’s. Furthermore, adipose tissue-derived MSCs (AD-MSCs) demonstrate superior efficacy in terms of lower recurrence rates compared with bone marrow-derived MSCs (BM-MSCs)23,24. MSCs facilitate tissue repair by inhibiting local immune responses and promoting the differentiation of myeloid cells toward an anti-inflammatory phenotype, thereby creating a favorable microenvironment. In addition, their paracrine effects stimulate fistula tract healing25–28. Direct injection of MSCs into the fistula tract wall or surrounding tissue remains the most common method of administration. However, definitive guidelines establishing the optimal cell dose, injection frequency, and culture conditions are currently lacking29,30. Notably, in our study, the selection of exosome dosage was based on evidence from perianal fistula, diabetic wound, and ischemic stroke models31–33. These findings collectively indicate that 10–50 μg represents an effective dosage range. Considering the administration strategy employed in this study, which involved loading Exos onto GS and implanting them into the fistula tract to achieve sustained and slow release, a dose of 50 μg was ultimately determined. Current research on MSC therapy for fistulas predominantly focuses on AD-MSCs and BM-MSCs, with relatively few reports on hUMSCs29,34. Research by Yafei Lu et al 31 demonstrated that Exos derived from hUMSCs promote healing in a rat model of complex perianal fistulas. Our research also suggests that extracellular vesicles derived from hUMSCs exhibit therapeutic effects in promoting fistula closure in a rat model of ECF.
Various preconditioning strategies have been explored to enhance the therapeutic efficacy of exosome-based treatments. Notably, Exos derived from MSCs under hypoxic conditions exhibit significantly elevated levels of immunomodulatory factors, conferring an enhanced immunoregulatory capacity 35 . Hypo-Exos demonstrate superior performance in promoting cell proliferation and migration, enhancing angiogenesis, and simultaneously inhibiting apoptosis and inflammation compared to their normoxic counterparts 36 . Hypoxic preconditioning does not alter MSC morphology, proliferation rate, or surface marker profile but significantly increases the stability of their secreted products. This enhanced stability allows Exos to maintain more consistent therapeutic properties, making them particularly advantageous for tissue regeneration 36 . In our study, Hypo-Exos demonstrated notably improved therapeutic effects in a rat model of ECF. This enhanced efficacy may be attributed to the characteristic stability and functional augmentation of Exos produced under hypoxia, although this association requires further validation.
GS-loaded Exos have demonstrated multifaceted research progress in tissue repair. In hemostasis and wound healing, studies have confirmed that GS loaded with hUCMSC-derived exosomes not only exhibit good biocompatibility but also significantly enhance coagulation efficiency and accelerate wound healing in liver defect hemostasis and full-thickness skin defect models
19
. The underlying mechanisms may involve prolonged
In the present study, we measured the levels of inflammatory cytokines in the rat serum. Within the inflammatory milieu, TNF-α serves as a canonical pro-inflammatory cytokine primarily produced by immune cells, such as macrophages. It potently amplifies the release of pro-inflammatory mediators 41 . Conversely, IL-4 significantly inhibits the production of TNF-α by macrophages and monocytes 42 . TNF-α drives inflammation by activating pro-inflammatory signaling pathways and inducing oxidative stress in cells. In contrast, IL-4 suppresses pro-inflammatory cytokines and enhances anti-inflammatory mediators through both STAT6-dependent and independent mechanisms. Thus, TNF-α and IL-4 establish a dynamic equilibrium in the regulation of inflammatory responses. In the cytokine analysis following GS Hypo-Exos treatment for ECF, serum levels of TNF-α in treated rats were significantly decreased, whereas IL-4 levels were markedly elevated. These reciprocal changes in key inflammatory cytokines indicate an alteration in the immune response of ECF rats and demonstrate the effectiveness of the current therapeutic intervention.
VEGF is primarily recognized for its role in angiogenesis. In tissue engineering, VEGF release synergizes with scaffold pore size to influence 3D cell migration and vascularization 43 . Meanwhile, TGF-β1 serves as a key mediator of fibrosis and can directly induce PD-1 expression on macrophages in chronic inflammatory contexts44,45. By detecting VEGF and TGF-β1 levels, the potential pro-angiogenic capacity and regulatory effects on the fibrotic/immune microenvironment of the composite system can be evaluated. Furthermore, assessments of the proliferation marker Ki67 and the apoptotic factor CASP-3 indicated the initiation of robust regenerative and cytoprotective mechanisms within the organism. Ki67, a nuclear protein widely employed as a marker for proliferating cells across the active phases of the cell cycle, exhibits expression levels directly correlated with fibroblast proliferative activity during tissue repair processes46,47. CASP-3, a key executioner protease in apoptosis, participates in tissue repair by regulating programmed cell death48,49. By evaluating these factors alongside HE staining results, we comprehensively assessed the tissue repair outcomes in ECF rats treated with Hypo-Exos, thereby confirming the significant therapeutic efficacy of Hypo-Exos.
While hUCMSC-Exos hold considerable promise for the treatment of various diseases, their clinical translation remains challenging. Our current study provides evidence for the potent healing effects of hypoxia-preconditioned hUCMSC-Exos on ECF in a rat model. However, this study had several limitations. First, the rat surgery-induced ECF model cannot fully encompass the complexity and heterogeneity of clinical ECF in humans. However, the model constructed in this study differs from traditional ostomy surgery, as the ECF model focuses on tissue inflammation and persistent exudates to simulate the pathological characteristics of clinical ECF. Second, the relatively limited sample size may have affected the statistical power and generalizability of the findings. Elucidating the underlying molecular mechanisms of these therapeutic effects is a critical direction for future investigations.
Conclusions
This study innovatively established an exosome-GS sustained-release carrier composite system with hypoxic pretreatment (GS Hypo-Exos) and confirmed its ability for efficient loading of Exos and sustained release of growth factors. In an ECF animal model, this composite material was able to maintain local presence for ≥21 days, significantly accelerating fistula closure and tissue regeneration. The mechanisms include inhibition of the inflammatory microenvironment, promotion of collagen deposition, upregulation of cell proliferation (Ki67), and inhibition of cell apoptosis (CASP-3). These findings present a new direction for the treatment of ECF and provide preclinical experimental data.
Supplemental Material
sj-docx-1-cll-10.1177_09636897261427905 – Supplemental material for Gelatin sponge loaded with hypoxic human umbilical cord mesenchymal stem cell exosomes promotes healing of enterocutaneous fistula
Supplemental material, sj-docx-1-cll-10.1177_09636897261427905 for Gelatin sponge loaded with hypoxic human umbilical cord mesenchymal stem cell exosomes promotes healing of enterocutaneous fistula by Yankui Liu, Wenwen Yu, Guang Wang and Hongbin Liu in Cell Transplantation
Supplemental Material
sj-docx-2-cll-10.1177_09636897261427905 – Supplemental material for Gelatin sponge loaded with hypoxic human umbilical cord mesenchymal stem cell exosomes promotes healing of enterocutaneous fistula
Supplemental material, sj-docx-2-cll-10.1177_09636897261427905 for Gelatin sponge loaded with hypoxic human umbilical cord mesenchymal stem cell exosomes promotes healing of enterocutaneous fistula by Yankui Liu, Wenwen Yu, Guang Wang and Hongbin Liu in Cell Transplantation
Supplemental Material
sj-docx-3-cll-10.1177_09636897261427905 – Supplemental material for Gelatin sponge loaded with hypoxic human umbilical cord mesenchymal stem cell exosomes promotes healing of enterocutaneous fistula
Supplemental material, sj-docx-3-cll-10.1177_09636897261427905 for Gelatin sponge loaded with hypoxic human umbilical cord mesenchymal stem cell exosomes promotes healing of enterocutaneous fistula by Yankui Liu, Wenwen Yu, Guang Wang and Hongbin Liu in Cell Transplantation
Footnotes
Acknowledgements
The authors would like to express their gratitude to The Second Clinical Medical College of Lanzhou University for providing a good research platform. The authors also appreciate the valuable feedback from Hongbin Liu during the preparation of this manuscript.
Ethical approval
The animal study protocol was approved by the Institutional Animal Care and Use Committee (IACUC) of Yunnan Luoyu Biotechnology Co., Ltd. (Protocol Code: SL20241203, Approval Date: December 16, 2024).
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
There are no human subjects in this article, and informed consent is not applicable.
Author contributions
GW: validation, data curation. HL: conceptualization, resources, validation, funding acquisition. WY: software, resources. YL: conceptualization, methodology, validation, formal analysis, writing—original draft preparation, writing—review and editing, visualization, supervision, project administration.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
