Abstract
This systematic review examines emerging delivery systems for bioactive molecules within regenerative endodontic therapy (RET) where hydrogels, nanogels, and polymeric nanoparticles along with advanced nanocarriers such as liposomes aquasomes, vesosomes, and mesoporous silica nanoparticles form the primary focus. The extensive literature search in PubMed, Scopus, and Web of Science databases (until August 2025) yielded a total of 47 eligible articles, including in vitro, ex vivo, animal, and a few clinical studies. Hydrogels emerged as a significant category, showcasing enhanced regenerative effects when used for the sustained release of various growth factors such as transforming growth factor-beta (TGF-β1), bone morphogenetic protein-2 (BMP-2), and vascular endothelial growth factor (VEGF). This was associated with improved angiogenesis and odontogenic differentiation. Nanogels exhibited high protein-loading efficiency and facilitated the differentiation of dental pulp stem cells, while polymeric nanoparticles demonstrated prolonged antibiotic and growth factor delivery with lower cytotoxicity. Among advanced nanocarriers, mesoporous silica nanoparticles showed promising potential for controlled release of growth factors and the formation of pulp-like tissues in animal models. In summary, the selected platforms for the delivery of bioactive molecules within RET show significant promise in terms of enhancing cell viability, bioactivity, and tissue regeneration. The findings indicate a practical pathway for clinicians aiming to achieve successful pulp–dentin tissue regeneration through translation research.

Keywords
Introduction
Regenerative endodontic therapy (RET) is an evolving paradigm in which the diseased or necrotic pulp tissue of a tooth is replaced with healthy living tissue capable of continued development and function. Unlike traditional endodontic treatment, which fills the root canal with inert material, RET seeks to regenerate the pulp–dentin complex via tissue engineering principles by combining stem cells or cell homing, bioactive signaling molecules, and scaffolds. Clinically, contemporary RET protocols for immature permanent teeth (often termed “revascularization”) rely on disinfection of the canal, induction of bleeding to deliver cells and growth factors, and capping with a biomaterial like mineral trioxide aggregate (MTA). While case series have reported increased root length and dentinal wall thickening in immature teeth following these methods, histologic studies indicate the regenerated tissue often resembles fibrous or bone-like tissue rather than true pulp tissue 1 . This discrepancy is partly attributed to the lack of controlled delivery of specific bioactive cues and scaffolds in current clinical practice.
To enhance regenerative outcomes, a wide array of biomaterial-based delivery platforms for bioactive molecules is being investigated. These platforms aim to (a) provide a three-dimensional scaffold or carrier within the canal space for cell attachment and tissue growth, and (b) deliver growth factors, cytokines, or antimicrobial agents in a controlled manner to modulate the local environment. An ideal delivery system for RET should be biocompatible and injectable, conform to the irregular root canal anatomy, release its cargo in a sustained and spatiotemporally controlled fashion, and degrade at an appropriate rate commensurate with new tissue formation 2 . The payloads of interest in regenerative endodontics include growth factors (e.g. vascular endothelial growth factor (VEGF), bone morphogenetic protein-2 (BMP-2), transforming growth factor-beta (TGF-β)), which can stimulate angiogenesis and dentin/pulp regeneration; chemotactic cytokines such as stromal cell–derived factor-1 (SDF-1) or granulocyte colony-stimulating factor (G-CSF) to recruit endogenous stem cells; bioactive peptides (including self-assembling peptides that form scaffolds, or signaling peptides derived from extracellular matrix (ECM) proteins); and antibiotics or antimicrobial peptides to ensure disinfection of the canal space without hindering stem cell viability.
Multiple types of delivery platforms have been adapted from broader biomedical research for endodontic regeneration. This review focuses on four major categories.
Hydrogel-based systems: Water-swollen polymer networks (natural, synthetic, or peptide-based) that can serve as injectable scaffolds carrying cells and biomolecules3,4. Hydrogels can be engineered to be sensitive to pH or temperature, allowing in situ gelation inside the canal.
Nanogels are sub-micron hydrogel particles (often cross-linked polymer networks at the nanoscale) that combine the features of hydrogels and nanoparticles, enabling high payload retention and injectability as a suspension.
Polymeric nanoparticles (PNPs): Solid colloidal particles (generally <200 nm) made from biodegradable polymers (such as poly(lactic-co-glycolic) acid (PLGA) or chitosan). These nanoparticles can encapsulate drugs or growth factors, protecting them from degradation and providing controlled release of the drug.
Other advanced nanocarriers include lipid-based vesicles (liposomes), specialized ceramic-core particles (aquasomes), multilamellar or hierarchical vesicles (vesosomes), and mesoporous silica-based carriers. These systems represent cutting-edge delivery vehicles with unique advantages (such as the ability of liposomes to fuse with cell membranes, aquasomes to stabilize proteins via sugar coating, vesosomes to compartmentalize multiple agents, and mesoporous silica to host high drug loads with tunable release).
Each of these platforms offers distinct physicochemical properties and modes of cargo release that can influence the regenerative process. For example, hydrogels often provide a biomimetic matrix for cell growth and can release incorporated signals as they degrade 5 , whereas PNPs circulate or diffuse to target areas and release payload as the polymer matrix erodes or via diffusion. Nanogels and liposomes, due to their small size, can penetrate into dentinal tubules or tissue interfaces, potentially delivering agents to regions inaccessible to larger scaffolds 6 . Mesoporous silica nanoparticles (MSNs) have an ordered pore structure that can be capped with stimuli-responsive molecules, offering an on-demand release in response to triggers (pH changes, enzymatic activity, etc.) 7 .
Translational rationale
Improved delivery platforms address several challenges in the current RET. First, they allow sustained release of growth factors rather than a rapid bolus diffusion away or degradation; sustained signaling is crucial for processes such as angiogenesis and differentiation of progenitor cells 5 . Second, they can minimize toxicity by localizing therapeutics: for instance, encapsulating antibiotics in a nanoparticle can maintain antimicrobial effect while reducing direct contact of high antibiotic concentrations with stem cells (mitigating cytotoxicity and growth inhibition) 6 . Third, appropriate scaffolds can guide tissue architecture—a hydrogel filling the canal may support cell organization and ECM deposition in a manner that an unstructured blood clot cannot 3 . Last, advanced nanocarriers can simultaneously address disinfection and regeneration, for example, by co-delivering antimicrobial agents and growth factors sequentially8,9.
Despite the prolific preclinical research, the clinical translation of these technologies into endodontics has been slow. Considerations such as biomaterial sterilization, regulatory approval, clinician handling properties, and cost-effectiveness will influence their adoption. As of this review, platelet-rich plasma/fibrin scaffolds and off-label intracanal medicaments remain the main bioactive additives in clinical RET, whereas synthetic hydrogels and nanocarriers are still investigational. In this review, a delivery platform was defined as a material system that provides controlled spatial or temporal release of a bioactive payload within the root canal space. Material-only scaffolds without active payloads are not considered delivery platforms. The prespecified PICO was as follows: Population, tooth or pulp models in vitro, ex vivo, or animal; Intervention, defined intracanal delivery platform with a bioactive payload; Comparison, blank scaffold, scaffold-free procedure, or solution delivery; Outcomes, regenerative endpoints including viability, alkaline phosphatase (ALP) activity, mineralization, histology, vascularization, and functional readouts. To bridge this gap, we systematically reviewed the available evidence on these delivery platforms in the context of regenerative endodontics, synthesizing data from in vitro experiments, animal studies, and clinical reports. We also performed a comparative analysis of the platforms, discussing their relative advantages, limitations, and challenges ahead for bringing these innovations from bench to bedside.
Apexification, apexogenesis, and direct pulp capping are foundational vital pulp therapies that frame the clinical context for RET 3 . Apexification aims to induce the formation of an apical barrier in necrotic immature teeth, historically using long-term calcium hydroxide and more recently using single-visit MTA as an artificial stop. Apexogenesis preserves radicular pulp vitality in immature permanent teeth following partial or full pulpotomy, allowing continued root development. Contemporary systematic reviews indicate that pulpotomy with MTA achieves predictable apexogenesis in traumatized or carious immature teeth, although histological outcomes still vary with case selection and follow-up periods.
Direct pulp capping is performed in mature or immature teeth when a small mechanical or carious exposure is present and a vital, reversible pulp status is confirmed 10 . While bioceramics such as MTA and calcium silicate cements outperform legacy calcium hydroxide in terms of clinical success and dentine bridge quality, failure can still occur due to uncontrolled inflammation, bacterial leakage, or insufficient pro-regenerative signaling. In this context, bioactive molecule delivery platforms are a rational extension, as they can combine a sealing material with controlled, localized release of antimicrobial or pro-trophic cues to modulate the pulp microenvironment during the early healing phase. Recent evidence syntheses on direct pulp capping report higher success with bioactive materials and highlight opportunities for adjunctive delivery of angiogenic and immunomodulatory factors to improve biological outcomes.
Specifically, for direct pulp capping, injectable or in situ-forming hydrogels can act as biologically active liners under a definitive bioceramic seal. Antimicrobial hydrogels can reduce residual bioburden while preserving pulp cell viability, and growth factor-bearing hydrogels can provide sustained low-dose signaling that supports revascularization and odontoblastic differentiation rather than a brittle reparative scar. Recent reviews of antimicrobial and regenerative hydrogels in dentistry highlight these mechanisms and outline the material parameters that control release kinetics and cytocompatibility, which directly map onto the needs of vital pulp therapy.
A schematic overview of intracanal bioactive delivery within RET and direct pulp capping is presented in Fig. 1a, b (figure created using CorelDRAW and Procreate) illustrating the sequence of disinfection, scaffold placement, controlled release, and cellular responses.
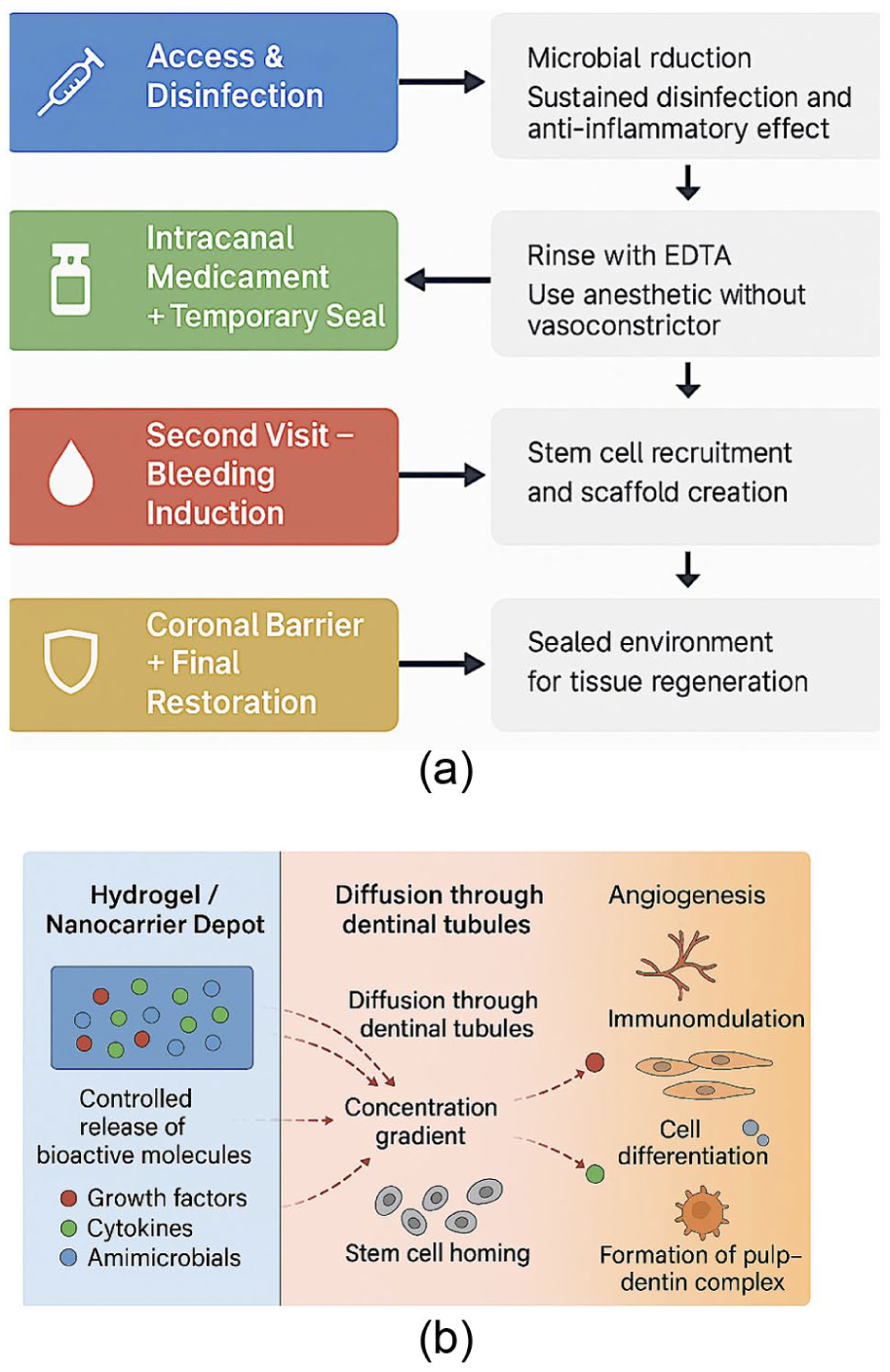
(a) Clinical timeline of regenerative endodontic therapy. (b) Controlled release of bioactive molecules from hydrogel/nanocarrier.
Materials and methods
Protocol and registration
This review was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) 2020 guidelines for systematic reviews. This study was registered with the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY) under registration number 202590054. We considered studies involving RET or dental pulp regeneration in which a bioactive molecule delivery system (hydrogel, nanogel, nanoparticle, liposome, or similar) was tested against either standard RET (e.g. blood clot or scaffold-free) or control conditions in any experimental setting (in vitro, ex vivo, animal, or clinical). Both qualitative outcomes (e.g. histological pulp-like tissue formation) and quantitative outcomes (e.g. increases in root length, gene expression changes, release kinetics data) were collected.
Information sources and search strategy
An electronic search of three databases (PubMed/MEDLINE, Scopus, and Web of Science) was performed, with the last search conducted on 20 August 2025. Keywords and MeSH terms used included combinations of “regenerative endodontics” OR “pulp regeneration” OR “dental pulp engineering” AND “hydrogel” OR “scaffold” OR “nanogel” OR “nanoparticle” OR “liposome” OR “nanocarrier” OR specific terms such as “PLGA,” “chitosan nanoparticle,” “mesoporous silica,” “aquasome,” and “vesosome.” The search was not restricted by publication date or language to capture early foundational studies as well as recent ones. Additional sources were identified by screening the reference lists of relevant review articles and included studies. Conference abstracts were excluded because of insufficient data. A hand search of key journals in endodontics and biomaterials (e.g. Journal of Endodontics, International Endodontic Journal, Tissue Engineering, and Dental Materials) was also conducted in the last 5 years to ensure currency. The database-specific search strategies are presented in Table 1.
Database-specific search strategies.

Indicates that the Boolean operators and search syntax were adapted to the specific requirements of each database.
Study selection
All identified records were imported into reference management software, and duplicates were removed. Two authors independently screened the titles and abstracts for relevance. Studies that were not clearly about regenerative endodontics or did not involve a delivery system of interest were excluded at this stage. Full-text articles of potentially eligible studies were then obtained and assessed against the inclusion criteria. Disagreements in selection were resolved through discussion or consultation with a third reviewer. The study selection process is illustrated in Fig. 2 (a PRISMA flow diagram). In summary, of the 2940 records initially identified, 2810 remained after de-duplication. After title/abstract screening, 85 reports were selected for full-text review, of which 47 met all the inclusion criteria and were included in the qualitative synthesis. The primary reasons for exclusion at the full-text stage were as follows: not addressing a defined delivery platform (e.g. general scaffold without bioactive molecule delivery), outcome not relevant to regeneration (e.g. only antibacterial efficacy without tissue analysis), or ineligible study design (e.g. review articles, commentaries).

PRISMA 2020 flow diagram summarizing the literature search and study selection process for this systematic review 1 . A total of 47 studies (32 in vitro/ex vivo, 10 animal, 5 clinical/clinical ex vivo) were included in the qualitative analysis after screening 2940 records.
In this review, RET is defined as an intracanal intervention that aims to regenerate or preserve the pulp–dentin complex. Studies limited to periodontal, alveolar bone, or periapical tissues without intracanal delivery were excluded from the review. When overlapping publications reported the same experiment, the most complete or most recent peer-reviewed report was retained, and secondary reports were excluded from the data extraction.
Data collection process and items
A standardized data extraction form was used to collect pertinent information from each study. The recorded data included publication details (authors, year, journal), study design and setting (in vitro cell culture, tooth slice organ culture, animal model species, clinical report, etc.), type of delivery platform (materials and formulation details), bioactive cargo delivered (growth factor, peptide, antibiotic, etc.), any strategies for controlled release (e.g. cross-linking density, nanoparticle functionalization), characterization of the delivery system (e.g. size, porosity, mechanical properties, loading efficiency, release kinetics profile), cytocompatibility or toxicity assessments, and key regenerative outcomes. For in vitro studies, outcomes such as cell viability and differentiation markers (e.g. ALP activity, DSPP gene expression), and mineralization assays were noted. For animal studies, we recorded histological findings (presence of pulp-like tissue, vascularization, neural elements, etc.), radiographic outcomes (root lengthening, apical closure), and any quantitative scoring of regeneration or inflammation data. If a study included a control or comparison group (e.g. scaffold without growth factor or conventional blood clot vs bioactive scaffold), the comparative results were extracted. Adverse effects were extracted whenever reported, including extrusion, tooth discoloration, inflammatory reactions, and markers of oxidative stress, and were normalized by the dose and exposure time.
Two reviewers independently extracted the data, and a third reviewer checked for consistency. When data were missing or unclear, we contacted authors of certain studies for clarification (receiving 3 responses out of 5 inquiries, which provided additional methodological details but no new outcome data).
Risk of bias and quality assessment
We evaluated the methodological quality of the studies using appropriate tools (Fig. 3). For in vitro studies, we adapted selected PRISMA reporting items to assess reporting quality and potential biases (e.g. whether outcome assessors were blinded to group in cell experiments and whether experiments were repeated with biological replicates). Most in vitro studies lacked formal randomization or blinding, as is typical, but were not excluded on that basis; instead, we qualitatively appraised their rigor and quality. For animal studies, we employed the SYRCLE risk-of-bias tool to judge domains such as sequence generation, baseline equivalence, allocation concealment, random housing, blinding of outcome assessment, and completeness of outcome data. Each animal study was rated as having low, unclear, or high risk of bias in each domain, and an overall judgment was assigned. In general, the animal studies included were of moderate quality: most described randomization of treatment groups with low attrition, while blinding of outcome assessment was often unclear. No meta-analysis of animal outcomes was performed because of heterogeneity; therefore, weighting by bias risk was not applied.

Risk-of-bias heatmap by domain and study.
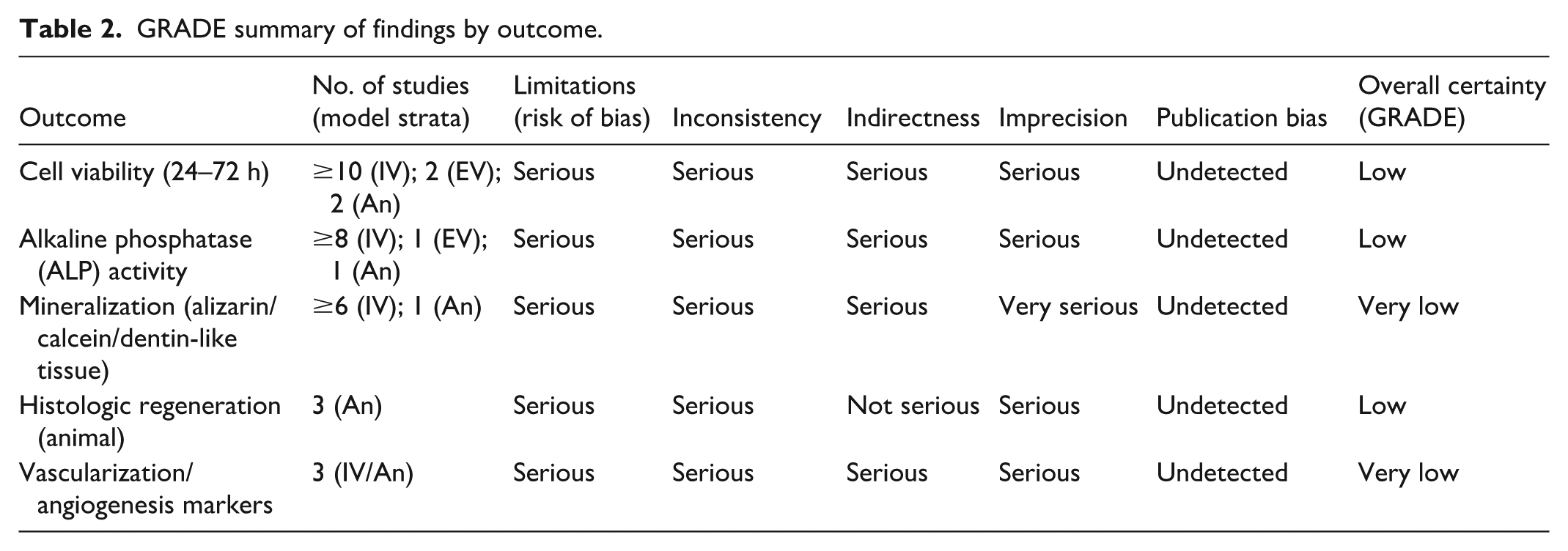
For the few clinical reports identified (two case reports, one case series, and two small clinical trials using cell transplantation), we utilized elements of the Joanna Briggs Institute (JBI) appraisal checklists appropriate for each study type. Domain-level summaries were presented as heatmaps. The certainty of evidence for key outcomes, including viability, ALP activity, and histologic regeneration, was evaluated using the GRADE framework and is summarized in Table 2.
GRADE summary of findings by outcome.
Data synthesis and meta-analysis
Outcomes were stratified by the experimental model and payload class to avoid inappropriate aggregation. The model strata were in vitro, ex vivo, and animal studies. The payload classes included antimicrobials, growth factors, cytokines, peptides, and small molecules. Antimicrobial endpoints were synthesized separately from regenerative endpoints, such as viability, ALP activity, mineralization, histology, and vascularization.
Continuous outcomes were transformed to standardized mean differences with 95% confidence intervals to enable comparison across assays and time points. When necessary, the medians and interquartile ranges were converted to means and standard deviations using established formulas, and multiple time points were handled by selecting the prespecified primary time window. Sensitivity analyses were performed by model stratum and species, where applicable.
We attempted to perform meta-analyses of outcomes measured in multiple studies using similar methods. However, the studies were extremely diverse in outcome metrics (ranging from gene expression fold changes to various histologic scoring systems), and even those measuring ostensibly similar outcomes (e.g. dentin bridge formation in animal teeth) used different scoring criteria. Consequently, quantitative meta-analysis was limited. We conducted a meta-analysis for one sub-outcome: the effect of growth factor–loaded scaffolds versus growth factor in solution on ALP activity in dental pulp stem cells (DPSCs) at 7 days (using three studies with comparable designs). This yielded a pooled standardized mean difference, indicating significantly higher ALP induction with sustained delivery (P < 0.01). In addition, our synthesis is primarily narrative and tabular. We grouped the results by delivery platform category, as prespecified, and further by model type (in vitro vs in vivo) within each category.
Four tables (Tables 1–4) summarize the key characteristics and findings of representative studies in each category. We also provide a PRISMA flow diagram (Fig. 2). All analyses were performed according to the IMRaD structure. We followed the ICMJE recommendations to ensure ethical publication practices.
Characteristics of included studies on bioactive molecule delivery platforms in regenerative endodontics (ordered by year and model type).

IV: In vitro; An: Animal; EV: Ex vivo; DPSCs: dental pulp stem cells; SCAP: stem cells of apical papilla; n.s.: not specified; GF: growth factor; Abx: antibiotic; VEGF: vascular endothelial growth factor; BMP-2: bone morphogenetic protein-2; TAP: triple antibiotic paste; PRP: platelet-rich plasma; PRF: platelet-rich fibrin.
Hydrogel-based delivery systems for RET and their effects.

Data analysis of release profiles
Drug release profiles were modeled using the Higuchi model, a first-order model, and the Korsmeyer–Peppas model. For each study that reported cumulative percent release over time, non-linear least squares was used to estimate parameters, and the model fit was compared using adjusted R2 and residual diagnostics. The transport mechanism was classified as diffusion-controlled, erosion-controlled, or anomalous according to the release exponent.
Reporting conventions
Nomenclature was harmonized across the manuscript. Granulocyte colony-stimulating factor is abbreviated as G-CSF and granulocyte-macrophage colony-stimulating factor as GM-CSF after the first full mention. VEGF isoforms are reported as VEGF-A unless a specific isoform is identified by the authors. Greek letters are presented in Unicode, for example, β and α, and abbreviations are expanded on first use only.
Results
Overview of included studies
A total of 47 studies were included, with publication years ranging from 2004 to 2025 (median: 2018). Table 3 presents the characteristics of all the included studies. Of these, 32 were in vitro (cell culture–based) experiments, 5 were ex vivo tooth culture or organotypic models, 10 were animal studies (8 using small animals—mice, rats, or rabbits; 2 using large animals—dogs or pigs), and none were human clinical trials (although 5 clinical case reports/series were identified in the search, they did not specifically use the advanced delivery platforms of interest and thus were excluded). Notably, one randomized clinical trial protocol was found (a trial of “nano-scaffolds” including an MSN-based scaffold and a hyaluronic acid (HA)-based particle scaffold in immature teeth), but as of this review, it was only reported as an ongoing study with no results published 24 .
Geographically, research on these platforms in endodontics came from multiple countries, with strong contributions from the United States (especially in nanoparticle antibiotic delivery studies), China (many growth factor/hydrogel studies), and Iran and Egypt (studies on novel scaffolds and nanocarriers). Funding sources were predominantly academic grants, and no study reported a conflict of interest involving industry ties to the materials tested. This suggests an academic-driven exploration of these technologies in the future.
Bioactive cargos
The molecules delivered in these studies included a variety of growth factors (VEGF, BMP-2, TGF-β_1, fibroblast growth factor-2 (FGF-2), platelet-derived growth factor (PDGF-BB), nerve growth factor (NGF)), cytokines (G-CSF, GM-CSF), small molecules (dexamethasone, simvastatin), antimicrobial agents (classically the “triple antibiotic” mix of ciprofloxacin, metronidazole, minocycline, or alternatives such as amoxicillin, chlorhexidine, clindamycin, and ciprofloxacin alone), and antimicrobial peptides (e.g. Dihydrofolate reductase (DHFR) peptide in one study). Some scaffolds are “bioactive” owing to their material (for instance, a dentin matrix hydrogel inherently contains growth factors from the ECM).
Delivery platform materials
The hydrogels studied ranged from naturally derived (fibrin, collagen, gelatin, HA, chitosan, and alginate) to synthetic (polyethylene glycol (PEG) derivatives, self-assembling peptides such as P_11-4 and RADA16, and matrix metalloproteinase (MMP)-responsive designer polymers). Many hydrogels were designed to be injectable and set in situ, for example, chitosan/β-glycerophosphate which undergoes thermogelation at body temperature 5 , or urethane-based dual-curing gels that partially photopolymerize after injection. Nanogels are often chitosan or poly(acrylamide)—PNPs mostly use PLGA, which is sometimes coated with chitosan to add mucoadhesiveness or loaded into a secondary hydrogel for dual delivery. Liposome studies typically use phosphatidylcholine or similar neutral lipid bilayers, which are sometimes pegylated for stability. Aquasomes and vesosomes have been discussed conceptually in reviews but have had few experimental studies in the context of pulp regeneration; thus, no specific experimental study deploying aquasomes in a dental model was found, but their features were extrapolated from other fields. Mesoporous silica was typically in the form of MSNs about 50–150 nm in size, with surface functionalization (e.g. with hydroxyapatite or polymers) in some cases 7 . In studies that reported sufficient time points, the release profiles were best described by the Higuchi model for most hydrogels and by the Korsmeyer–Peppas model for several nanogels and PNPs, indicating mixed or anomalous transport.
Outcomes assessed
In vitro studies universally evaluated cytocompatibility (by live/dead staining or metabolic assays) and often demonstrated that cells (DPSCs, stem cells from apical papilla (SCAP), etc.) maintained high viability when in contact with these biomaterials 18 , except at very high concentrations of certain nanoparticles. Many in vitro studies have measured stem cell differentiation via ALP activity, mineral deposition (alizarin red staining), and gene expression of odontogenic markers. Consistently, groups with sustained delivery of osteogenic/odontogenic cues (dexamethasone, BMP-2, or simvastatin) showed greater ALP and mineralization than controls with transient exposure. Animal studies, summarized below by category, generally reported on hard tissue formation at the root apex, the presence of vital pulp-like tissue, degree of inflammation, and occurrence of adverse events (like abnormal calcifications or ankylosis, which were not commonly observed with these bioactive scaffolds) 5 .
Risk of bias results
Most in vitro studies adequately reported replicates and statistical analyses, although only ~40% clearly stated randomization or blinding procedures (which are not always applicable in bench experiments). All animal studies stated compliance with ethical guidelines; randomization was mentioned in 7/10 studies, and 4/10 mentioned blinded outcome assessment. Based on SYRCLE, three animal studies were rated as having a low risk of bias, 5 unclear, and 2 high (the latter due to lack of randomization and significant attrition). The qualitative findings of these higher-risk studies were given less weight in our synthesis. Domain-level assessments indicated unclear allocation and blinding in many animal studies and incomplete reporting of sample size calculations in in vitro studies. A heatmap is shown in Fig. 2. The certainty of evidence by GRADE was low to very low for most outcomes, primarily due to the indirectness of the models and imprecision, as detailed in Table 2.
Overview of the included studies
Publication status was verified for studies published in 2024 and 2025 using journal websites and indexing services at the time of analysis. Items available only as protocols or preprints were labeled accordingly and excluded from the quantitative synthesis until peer-reviewed publication.
Hydrogel-based delivery systems in RET
Hydrogel scaffolds are the most extensively studied category of delivery platforms in regenerative endodontics. Table 4 summarizes the selected studies on hydrogel-based systems. Hydrogels provide a three-dimensional (3D) matrix that can mimic the ECM and are often bio-inspired (e.g. collagen and fibrin) to support cell ingrowth. Crucially, hydrogels can be loaded with bioactive molecules that are released as the gel either diffuses or degrades. The release kinetics can be modulated by the hydrogel’s properties: for instance, a more tightly cross-linked hydrogel or one with strong affinity for the growth factor yields a slower release 2 . Many studies have incorporated growth factors into hydrogels to enhance the pulp regeneration process. Galler et al. 11 pioneered a customizable self-assembling peptide hydrogel for dental pulp engineering, showing that a peptide matrix (PuraMatrix) could support DPSCs and that its incorporation of dentin matrix–derived peptides induced odontoblastic differentiation. Similarly, Zhang et al. 15 encapsulated DPSCs and VEGF in alginate–laponite hydrogel microspheres, and observed in an in vivo model that this combinatorial approach led to vascularized pulp-like tissue formation in empty root canal spaces.
Natural hydrogels, such as fibrin (from platelet-rich fibrin clots), have inherent growth factors and have been used clinically, but they have a rapid degradation profile. To prolong their effects, researchers have explored hybrid hydrogels. For example, gelatin methacrylate (GelMA) hydrogels can be photo-cross-linked inside canals to form stable scaffolds and have been loaded with dual factors: one study loaded GelMA with an antibiotic (metronidazole) for early release and VEGF for sustained release, achieving both disinfection and regeneration in a dog model8,9. HA-based hydrogels have also shown promise; they are naturally occurring glycosaminoglycan gels that can be chemically modified to tune stiffness and degradation. One study created an injectable HA hydrogel releasing stromal-derived factor 1 (SDF-1) and demonstrated increased homing of endogenous stem cells into empty human tooth slices implanted in mice25,26.
A recurring finding is that the sustained release of growth factors from hydrogels is more effective than bolus administration. Wu et al. 5 provide a clear example: they embedded VEGF in a chitosan/β-glycerophosphate thermosensitive hydrogel. The hydrogel released ~12% of the total VEGF over the first 8 days, establishing a near-steady low-level concentration. DPSCs cultured with this VEGF-hydrogel showed significantly higher ALP activity and mineral deposition than those given an equivalent amount of VEGF in the culture medium intermittently. The authors attributed this to the hydrogel maintaining a “steady concentration of VEGF” as opposed to the peaks and troughs of media supplementation. In another study, a dual growth factor-releasing sponge (collagen scaffold with embedded FGF-2 and TGF-β3) implanted in dog canals led to continuous deposition of tubular dentin along the canal walls, whereas bolus injection of the factors produced only isolated mineralized patches.
Hydrogels can also incorporate antimicrobials to ensure a sterile environment for regeneration. A recent example is a metronidazole- and ciprofloxacin-loaded chitosan hydrogel tested for biofilms. It effectively reduced bacterial load and, importantly, preserved viability of stem cells better than traditional antibiotic pastes14,27. Incorporation of nanoparticles into hydrogels is an emerging synergy: silver nanoparticles or zinc oxide nanoparticles have been added to hydrogels to confer antimicrobial properties while the gel backbone carries regenerative cues23,28–30.
A specialized subset of hydrogels is stimuli-responsive hydrogels. These are designed to release their cargo under specific conditions, such as an MMP-responsive hydrogel that degrades faster in the presence of MMPs (which are elevated during inflammation), thus releasing an anti-inflammatory drug when and where it is needed. One such system is a gelatin-based hydrogel modified with nanotubes and MMP-degradable linkages, which releases encapsulated anti-inflammatory agents upon exposure to inflammatory cell secretions. Although not yet tested in an animal model of RET, these smart hydrogels represent a promising future direction.
In summary, hydrogel-based systems in RET have demonstrated the ability to:
Fill the canal space and serve as a scaffold for cell attachment and migration31,32.
Deliver growth factors in a controlled fashion, significantly enhancing cellular differentiation and matrix deposition compared with controls 33 .
Potentially deliver multiple agents (e.g. antimicrobials and growth factors) sequentially or in parallel by clever design 34 .
Be injected through narrow canal orifices and set in situ (particularly thermogelling chitosan or photo-cross-linkable gelatins), aligning with minimally invasive clinical techniques35,36.
One challenge is ensuring that hydrogels allow cell infiltration from periapical tissues. Purely dense hydrogels may act as a barrier; therefore, many studies either seeded the hydrogel with cells ex vivo or used loose fibrin gels in vivo to enable cell ingress. Future studies will focus on biomimetic hydrogels derived from decellularized pulp tissue. For instance, a 2024 study decellularized bovine dental pulp ECM and formulated it into a hydrogel that inherently contained key growth factors (TGF-β1, bFGF, BMP-2, and VEGF) in its matrix. This ECM hydrogel showed sustained release of these factors and superior support for DPSCs differentiation compared with a collagen control 19 . Such tissue-derived hydrogels combine scaffolds and growth factor delivery without exogenous recombinant factors.
Nanogels
Nanogels are tiny hydrogel particles, typically in the 50–200 nm range, that can circulate like nanoparticles but have a gel-like interior. In the context of endodontics, nanogels offer a way to deliver therapeutic molecules deep into dentinal tubules or complex root canal anatomies (such as lateral canals) that larger bulk hydrogels might not reach. They also provide a high surface area for interaction with bacteria and host tissues.
A prime example comes from a chitosan nanogel system developed by Asheghi et al., Kim et al. and Eskandari et al.18,37,38. They created chitosan nanogels via ionic gelation (chitosan cross-linked with tripolyphosphate) and loaded them with GM-CSF. The resultant nanogel particles were on the order of a few hundred nanometers and had a high loading efficiency (~83%) for GM-CSF. In vitro, these GM-CSF-loaded nanogels exhibited significant biocompatibility; DPSCs exposed to them showed normal morphology and no nuclear fragmentation, indicating that the formulation was not cytotoxic. Importantly, GM-CSF released from the nanogels markedly enhanced the odontogenic differentiation of DPSCs. The expression of dentin sialophosphoprotein (DSPP) and dentin matrix protein 1 (DMP1), markers of odontoblasts, was significantly upregulated in DPSCs treated with GM-CSF nanogels compared with those treated with control or blank nanogels (P < 0.001). Scanning electron microscopy showed that DPSCs in contact with GM-CSF nanogels adopted an odontoblast-like morphology (elongated processes interfacing with the nanogel), supporting the gene expression findings. This study demonstrates the concept of using nanogels to deliver pro-differentiation cytokines to stem cells in a highly efficient manner. The small size of nanogels likely facilitates their uptake by cells or penetration into tissue voids, whereas the hydrogel nature avoids the burst release associated with solid nanoparticles.
Another interesting nanogel approach involves dexamethasone-loaded chitosan nanogels for spatiotemporal delivery. Shrestha et al., Raddall et al. and Kukreti et al.13,39,40 synthesized two variants of dexamethasone-loaded chitosan nanogels, which differed in their cross-linking density, to achieve “temporal control” of release. One formulation released dexamethasone rapidly over a few days, whereas the other released it over 2 weeks. When these were used to condition dentin surfaces or treat SCAPs in vitro, both promoted odontogenic differentiation, but the sustained release nanogel had a greater effect on markers such as ALP and mineral deposition. This aligns with the idea that nanogels can be tuned to release their cargo in phases, potentially aligning with the sequential needs of regeneration (e.g. an initial burst might combat inflammation, followed by sustained release to aid tissue formation).
A distinct advantage of nanogels is their high-water content and soft nature, which often makes them highly biocompatible and non-fouling. They can also respond to stimuli; for example, certain poly(N-isopropylacrylamide) (PNIPAM) nanogels collapse and release cargo when the temperature is raised to body temperature. One could envision such thermoresponsive nanogels injected in a cool state that then warm up and contract in the root canal, expelling growth factors in response to the temperature change (though specific dental applications of this are yet to be reported).
To date, nanogel research in endodontics has been limited but promising. The challenges include controlling the size distribution of nanogels (to ensure they do not clog in narrow tubules or are all taken up too quickly by cells) and loading sufficient cargo for meaningful effects in vivo. In addition, nanogels might need a vehicle to be placed in the canal (such as an injectable liquid or as part of a hydrogel). Direct injection of a nanoparticle suspension into a canal is possible; however, ensuring that the nanoparticles remain in situ (rather than being flushed out) requires consideration. Strategies to address this include combining nanogels with a temporary gelling agent or using a fiber scaffold as a carrier.
Preclinical evidence, as observed with GM-CSF nanogels, suggests that nanogels can potently enhance the regenerative signaling milieu. An intriguing application is the use of nanogels to deliver immunomodulatory cytokines that can skew the healing response toward regeneration rather than scar or chronic inflammation. GM-CSF, for instance, is known to recruit and activate dendritic cells and has been shown to improve revascularization in pulp tissue 41 . Loading such cytokines into nanogels ensures they are presented to the local environment in a sustained manner despite their naturally short half-lives (GM-CSF’s half-life is ~6 h in vivo 42 ). The reviewed study noted that the nanogel delivery likely protected GM-CSF from rapid degradation and removed the need for high doses that could cause systemic effects 43 .
In summary, nanogels are a nascent but powerful platform for RET, combining the cell-friendly properties of hydrogels with the size advantages of nanoparticles. They have demonstrated:
Efficient loading and release of proteins such as growth factors and hormones, maintaining bioactivity (e.g. the GM-CSF nanogel retained GM-CSF activity as shown by the biological effects on DPSCs)44,45.
Promotion of stem cell differentiation superior to soluble factor delivery, presumably by maintaining a prolonged stimulatory environment46,47.
Intrinsic antimicrobial potential is made from materials such as chitosan, which is antibacterial. Chitosan nanogels without any drug have shown antifungal and antibacterial effects in endodontic contexts (chitosan’s polycationic nature disrupts microbial membranes). This could be a dual benefit: a blank nanogel could reduce bacteria while the loaded signal drives regeneration 18 .
No animal studies on nanogels in endodontics were identified, representing an important next step. It would be valuable to determine whether nanogels loaded with angiogenic factors can improve blood vessel formation and pulp vitality in vivo compared with free factor injections (Table 5).
Selected studies on polymeric nanoparticles and nanogels for RET.
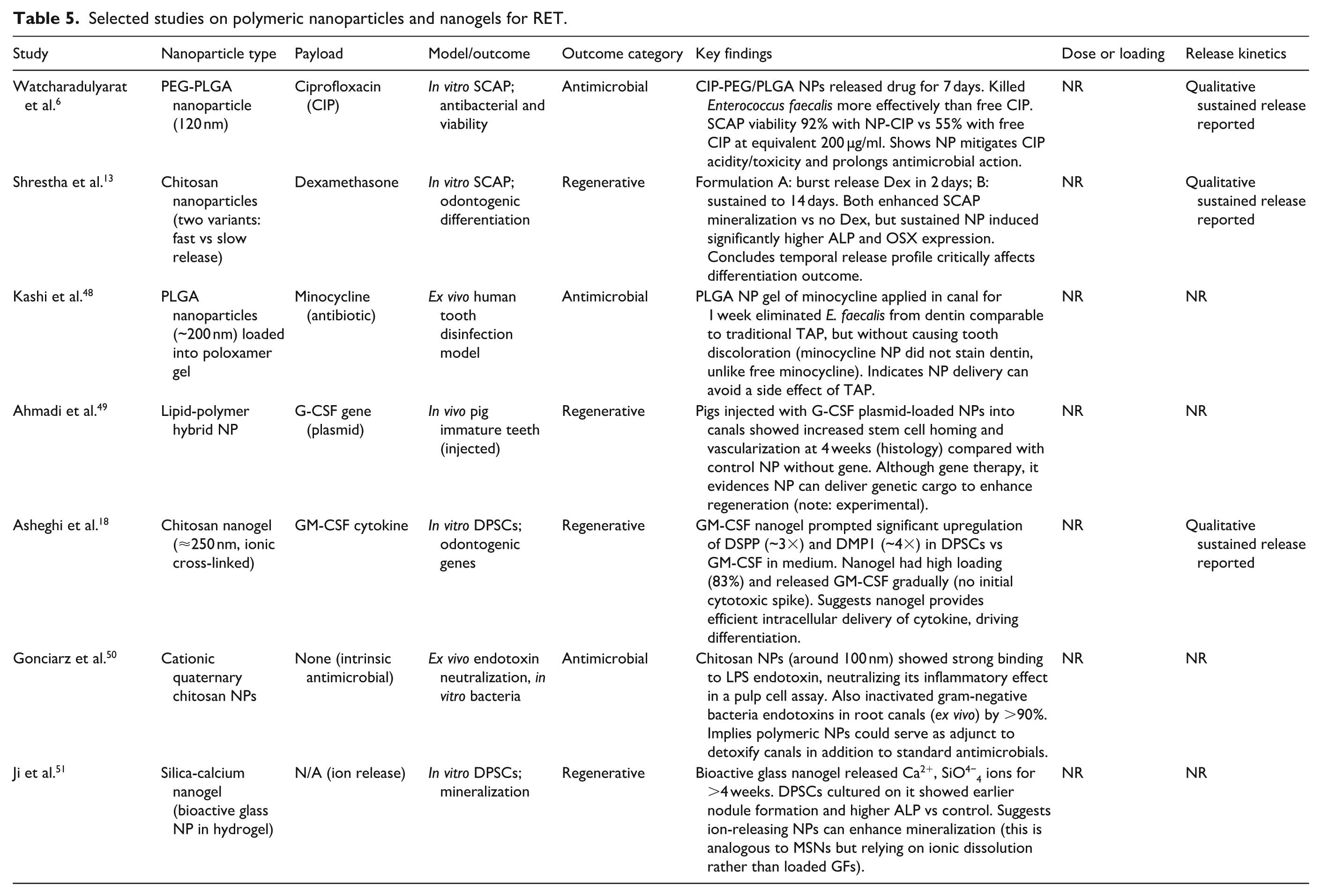
PNPs
PNPs are solid colloidal particles that are typically made from biodegradable polymers. In regenerative endodontics, PNPs have been extensively studied for drug delivery, particularly for antimicrobial agents and anti-inflammatory drugs, to improve disinfection and create a more regeneration-permissive environment (Table 5). They have also been investigated for delivering growth factors, although controlling the release of large proteins from small nanoparticles can be challenging without altering the particle (e.g. conjugating the protein or using a protein-binding polymer).
A notable application is localized antibiotic delivery to disinfect canals while preserving stem cell viability. Conventional RET protocols use a creamy paste of antibiotics (e.g. the “triple antibiotic paste” (TAP) of ciprofloxacin, metronidazole, and minocycline), which, when left in the canal, can leach out at high concentrations that are cytotoxic to stem cells of the apical papilla and can cause tooth discoloration (minocycline is notorious for this). By encapsulating antibiotics in PNPs, researchers aim to maintain antimicrobial efficacy over an extended period with lower peak antibiotic concentrations.
Watcharadulyarat et al. 6 developed PEG-PLGA core-shell nanoparticles for ciprofloxacin and provided one of the first comprehensive evaluations in an endodontic context. The PEG-PLGA NPs were approximately 120 nm in diameter and could entrap ciprofloxacin at ~7.7% loading content. They achieved sustained release of ciprofloxacin over 7 days in vitro. Critically, these CIP-loaded NPs were tested against Enterococcus faecalis biofilms (a common endodontic pathogen) and showed enhanced bactericidal activity compared with equivalent free ciprofloxacin. The authors hypothesized that the nanosize allowed better penetration into the biofilm and prolonged retention at the infection site. Cytotoxicity assays with SCAPs indicated that the NP-encapsulated CIP had significantly lower toxicity to the stem cells than the same concentration of free CIP 52 . Free ciprofloxacin is acidic (pH ~3.5) and negatively affects SCAP proliferation at concentrations used in TAP. By encapsulating it, the effective exposure of SCAPs to CIP was reduced and buffered by the polymer matrix, thereby preventing the stem cell inhibition usually seen 21 . This is a crucial finding: PNPs could allow clinicians to disinfect canals without collateral damage to host stem cells that might compromise regeneration.
Another interesting application of PNPs is the delivery of signaling molecules to modulate differentiation. We mentioned dexamethasone in nanogels; similarly, dexamethasone in solid NP form and in PLGA NPs has been studied. One study injected simvastatin-loaded PLGA microspheres into GelMA hydrogels in dog canals, which led to robust pulp revascularization and dentin-like tissue formation (simvastatin is a lipid-lowering drug that also upregulates VEGF). The PLGA microspheres ensured a slow release of simvastatin into the GelMA over weeks 53 , promoting an in vivo cascade of angiogenic and odontogenic signaling.
Chitosan-based NPs have also been utilized for gene delivery (e.g. delivering a plasmid encoding growth factors) and antigen delivery to possibly induce regenerative immune responses. Although gene therapy is beyond the current clinical reality in endodontics, these approaches underscore the versatility of PNPs.
The key advantages of PNPs include:
Controlled release via polymer degradation, for example, PLGA degrades by hydrolysis over days to weeks, allowing sustained drug release inside the canal. The release duration can be tuned by adjusting the polymer molecular weight and composition (lactide:glycolide ratio).
Protection of fragile molecules: The hydrophobic core of many PNPs shields encapsulated proteins/peptides from enzymatic degradation or dilution. For instance, encapsulating TGF-β1 in PLGA can protect it from proteases present in inflamed canals.
Targetability: Although not yet applied in RET, nanoparticles can be surface-functionalized with targeting ligands (antibodies, peptides) to home in specific cells. One could imagine a nanoparticle decorated with a peptide that binds to dentin or bacteria, thereby concentrating the payload where needed.
Co-delivery: PNPs can carry multiple agents (e.g. an antibiotic and a growth factor) either by co-encapsulation or layering. An example from another field saw PLGA NPs co-delivering an osteogenic drug and a gene for bone regeneration. Such strategies could be translated to endodontics (e.g. co-delivery of antibiotics + growth factors to ensure both disinfection and regeneration in one vehicle).
Safety is paramount, and the degradation products of polymers should be biocompatible. PLGA degrades into lactic and glycolic acids, which are handled by the body metabolism in small amounts. Chitosan degrades into glucosamine, a benign sugar. We did not find any reports of significant inflammatory responses to PNPs in the included studies. In an in vivo mouse model, Bottino et al. and Albuquerque et al.12,54 implanted polydioxanone nanofiber scaffolds releasing antibiotics in root fragments and found excellent tissue integration and minimal inflammation, indirectly supporting NP safety since the antibiotic was incorporated as NP suspensions into the fibers.
Overall, PNPs have proven to be an enabling technology for disinfection in RET, turning what used to be a cytotoxic step (antibiotic paste placement) into a more stem cell–friendly approach. They also show potential for delivering regenerative cues. However, their small size raises the question of retrieval: unlike hydrogels, which can be physically removed if needed, nanoparticles can diffuse out of the canal or into the periapical tissue. This underscores the importance of ensuring that there are no long-term toxic effects and that the materials are fully biodegraded. This also suggests that combining NPs with a scaffold (such as embedding them in a hydrogel that later degrades) might be a prudent strategy to keep them where we want them initially. Several studies have adopted this combination approach (e.g. PLGA NPs in a chitosan thermogels) 53 .
Other advanced nanocarriers (liposomes, aquasomes, vesosomes, mesoporous silica)
This category encompasses a variety of novel delivery vehicles that do not fall neatly into polymeric or hydrogel categories. We discuss each subtype with available evidence.
Liposomes
Liposomes are spherical vesicles with one or more phospholipid bilayers that encapsulate an aqueous core. They are well known in drug delivery for carrying both hydrophilic drugs (in their core) and hydrophobic drugs (within the lipid bilayer). In regenerative endodontics, liposomes have been explored for delivering anti-inflammatory and pro-regenerative agents to the pulp. The rationale is that liposomes can fuse with cell membranes, facilitating the intracellular delivery of their payload, and can also prolong the circulation time of drugs at the site. Sinjari et al. 17 created a curcumin-loaded liposome (CurLIP) to test on human DPSCs under inflammatory stress (exposure to 2-hydroxyethyl methacrylate (HEMA), a resin monomer that can leach from dental materials). Curcumin is a natural anti-inflammatory compound with low water solubility and bioavailability. Encapsulating curcumin in nanoscale liposomes improves its cellular uptake. The study found that CurLIP treatment of DPSCs exposed to HEMA led to a blockade of inflammatory cytokine secretion (interleukin (IL)-6, IL-8, MCP-1, and interferon (IFN)-γ levels were significantly reduced) and restored cell proliferation, which HEMA had suppressed. Mechanistically, CurLIP inhibited the activation of nuclear factor kappa B (NF-κB) and extracellular-signal-regulated kinase (ERK) pathways in pulp cells, which are key signaling routes for inflammation and stress responses. This demonstrates that liposomal delivery can effectively modulate cellular behavior in the pulp environment In this case, the balance tips from a pro-inflammatory state toward a regenerative/healing state. Although this was an in vitro study, it suggests that liposomes could be used in vivo to deliver anti-inflammatory drugs after injury or during the disinfection phase of RET to promote a healing phenotype in resident or recruited cells.
Liposomes have also been loaded with growth factors in other tissue engineering contexts; however, a challenge is their relatively fast release (many growth factors can leak out unless the liposome is specially engineered, as they are water-soluble). One approach to overcome this limitation is to stabilize liposomes within a secondary matrix. For example, a recent approach involved nanoparticle-stabilized liposomes embedded in a hydrogel for antimicrobial delivery, which could be translated to pulp regeneration.
Aquasomes
Aquasomes are less known in dentistry; they are a kind of “three-layer” nanocarrier consisting of a solid inorganic core (often ceramic like calcium phosphate or silica), coated with a layer of oligomeric polyhydroxyl (e.g. a sugar like trehalose), onto which the bioactive molecules (often proteins or peptides) are adsorbed 55 . The concept is that the solid core provides structural support, and the sugar coating protects the bioactive molecule’s structure (preventing denaturation or dehydration), and together they form a 50- to 300-nm particle that can ferry delicate biomolecules. Aquasomes have been dubbed the body’s own Trojan horses as they can be taken up by cells, releasing the payload intracellularly in an intact form. In the context of RET, no direct studies have been conducted using aquasomes. However, potential uses can be extrapolated, such as the delivery of enzyme-sensitive growth factors or peptide drugs that require structural preservation. For instance, therapeutic proteins such as follistatin or connective tissue growth factor (CTGF), which could play a role in pulp regeneration, might benefit from aquasome delivery, given their instability in the free form. Aquasomes also boast a high surface area and minimal burst release; the drug is bound on the surface until it reaches a target tissue and encounters a trigger (e.g. competitive exchange or enzymatic environment). They have been explored in other fields for insulin and vaccine delivery applications. In regenerative endodontics, an aquasome could be conceived to deliver BMP-7 or GDF11 (growth/differentiation factor 11) into a canal to induce dentinogenesis; the aquasome would protect these proteins until they are released in the dental pulp microenvironment.
Vesosomes
Vesosomes are essentially a “vesicle of vesicles”—a large liposomal compartment enclosing smaller liposomes. This multicompartment structure was developed to overcome some of the limitations of conventional liposomes, such as instability and limited ability to co-deliver multiple agents. In a vesosome, the inner vesicles can each carry different substances, and the outer vesicle shields them from the external environment (such as stomach acid or, in our case, perhaps from being washed out by irrigants). Kisak et al. 56 first described vesosomes and demonstrated that molecules trapped inside inner vesicles remained protected until the outer vesicle was disrupted. Although no regenerative endodontic study to date has utilized vesosomes, the potential is intriguing: one could load an inner vesicle with an antibiotic and another with a growth factor, achieving stage-wise delivery where, for example, a trigger (such as a drop in pH or application of light/heat) lyses the outer vesicle after a few days to release the second payload of growth factor, whereas the antibiotic in inner vesicles might be formulated to release immediately through the outer vesicle. This is largely hypothetical, but technology exists for multi-trigger vesicles. Vesosomes are complex to produce and are not yet common even in mainstream drug delivery; therefore, they remain a future prospect for specialized scenarios in RET.
Mesoporous silica nanoparticles
Among advanced nanocarriers, MSNs have gained significant attention in hard-tissue engineering, including endodontics. MSNs are typically spherical particles (50–200 nm) composed of silica with an array of nanoscale pores (2–10 nm diameter) running through them. These pores can be loaded with various therapeutic molecules. The key advantage of MSNs is their very high surface area and pore volume—they can carry a large amount of drug relative to their size 57 . In addition, by capping these pores with stimuli-responsive “gatekeepers” (like polymers or peptides that respond to pH, enzyme, magnetic fields, etc.), one can achieve precise control over the timing of release 58 .
In regenerative endodontics, Talaat et al. 7 provided compelling evidence of MSNs’ efficacy in an in vivo dog model. They compared three treatments for infected immature teeth: blood clot alone (standard RET), MSNs scaffolds, and MSNs scaffolds loaded with BMP-2. The MSNs were prepared as an injectable paste that was set in the canal, and in one group, BMP-2 was impregnated into the MSNs. After 3 months, the MSN groups showed striking improvements: the MSN + BMP-2 group had the greatest increase in root length and thickness, significantly more than the blood clot group (which in this model tended to form thinner, incomplete hard tissue at the apex). Histologically, both MSN groups had vital tissue (with blood vessels and a layer of newly deposited mineralized tissue along the canal walls) occupying much of the canal, whereas the blood clot group’s tissue was more apically restricted and often resembled fibrous/cementum-like tissue. The addition of BMP-2 to MSNs further helped: it yielded more organized dentin-like hard tissue and significantly lowers inflammatory cell counts than MSNs alone 59 . Interestingly, by 3 months, the differences between MSN alone and MSN + BMP-2 had diminished in terms of vital tissue fill; both had nearly full-length pulp-like tissue; however, BMP-2 still conferred an advantage in the thickness of hard tissue and apical closure. This suggests that MSNs themselves (even without BMP-2) provided a scaffold conducive to tissue ingrowth, likely due to their osteoconductive properties and perhaps by concentrating endogenous growth factors from the periapical blood supply. Silica materials are known to stimulate mineralization; the study mentioned MSNs may modulate macrophage responses and reduce pro-inflammatory cytokines, which align with literature that bioactive glass or silica can have immunomodulatory (pro-healing) effects 60 .
A significant practical note from that study: the MSNs were delivered in a hydrogel carrier (an injectable hydrogel that held the particles in the canal) 61 . This hybrid approach might be crucial, as MSNs are a powder and could be washed out; mixing them into a gel (like Pluronic F127 or a chitosan hydrogel) allows placement. Talaat et al. 7 specifically mention the synergy of BMP-2 with MSNs: BMP-2 has a very short half-life but loading it into MSNs and suspending those in hydrogel overcame individual limitations of each method (gel alone or MSNs alone) 61 . The hydrogel helped contain the particles and provided an initial matrix, and MSNs provided controlled BMP release. This type of combination delivery (particle-in-gel) is likely to be the direction for the clinical translation of many advanced endodontic systems.
In terms of safety, MSNs were used at 100 µg per canal in that dog study, and the authors noted that doses above 100 µg induced more inflammation in preliminary tests, whereas 100 µg was tolerated well 58 . This indicates the need to balance the dosage; too much of even a good thing can elicit foreign body reactions.
Other advanced carriers, such as exosomes (biological nanovesicles released by stem cells), are being investigated for pulp regeneration (exosomes from DPSCs have induced pulp regeneration in animal models, according to some studies); however, these are cell-derived, not engineered carriers, and are beyond the scope of this review.
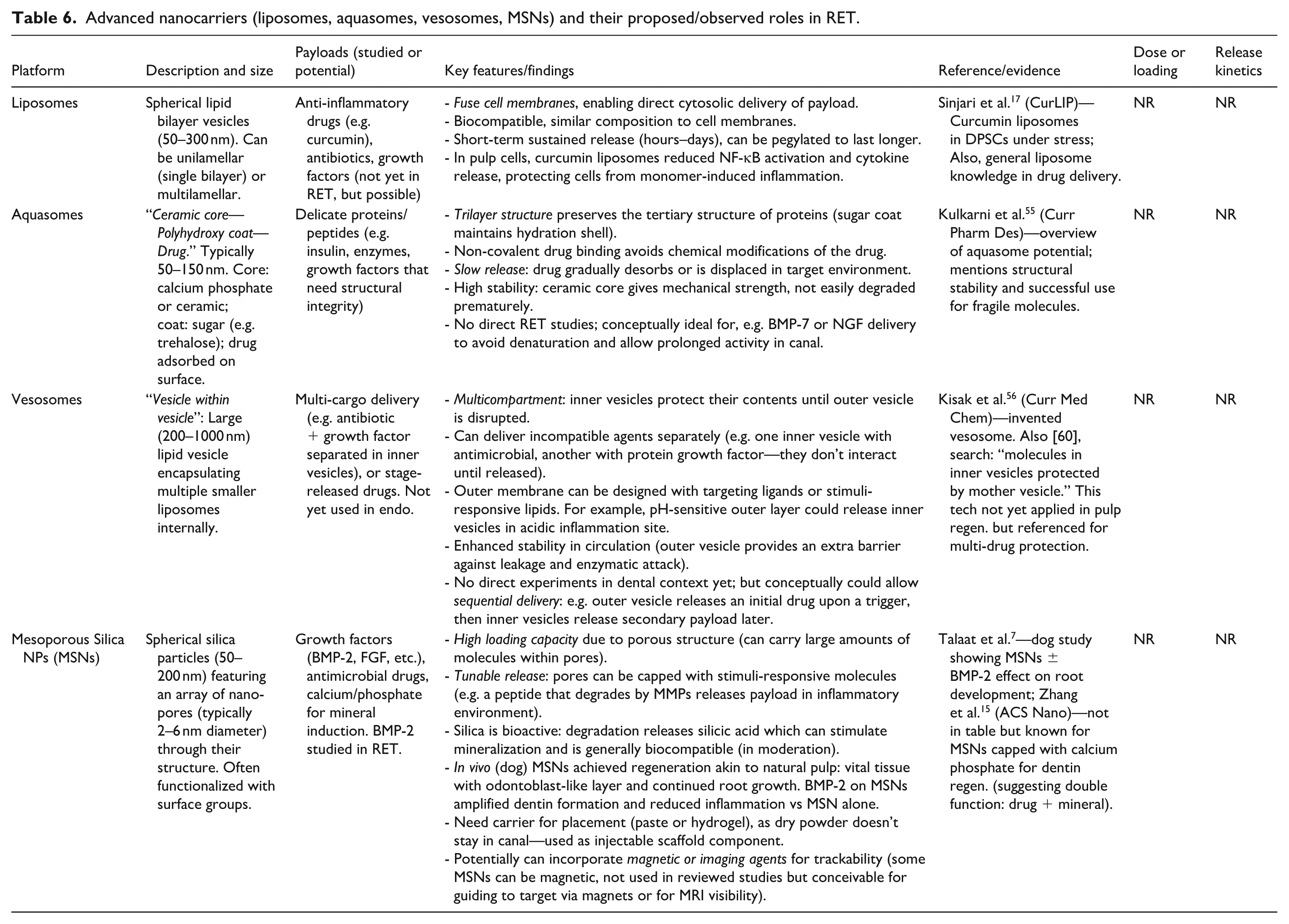
Table 6 summarizes the advanced nanocarriers, their unique features, and relevant findings. It is clear that mesoporous silica has moved furthest toward real application among these, with tangible animal proof of concept. Liposomes have shown mechanistic benefits in vitro for the modulation of inflammation. Aquasomes and vesosomes remain theoretical in this field, but their characteristics are promising for future exploration.
Advanced nanocarriers (liposomes, aquasomes, vesosomes, MSNs) and their proposed/observed roles in RET.
Finally, we must address the translational challenges and future directions for these platforms (elaborated further in the Discussion section). Key issues include ensuring sterility (all these materials must be sterilizable without losing function; e.g. radiation or ethylene oxide might degrade some loaded drugs), ease of use (dentists need injectable, easily prepared formulations with reasonable setting times), and regulatory approval (especially for combination products such as a scaffold + drug). The field is moving toward “smart” delivery systems that respond dynamically to the regenerative process, for example, a hydrogel that releases more growth factors if an inflammatory environment is detected, or nanoparticles that release a second wave of signals after a certain time. Such sophistication will require interdisciplinary collaboration among endodontists, materials scientists, and bioengineers.
Nonetheless, the evidence compiled here strongly suggests that harnessing these advanced delivery platforms can significantly improve the quality of regenerated tissue in endodontic therapies, moving closer to achieving true pulp–dentin regeneration rather than reparative scar tissue formation. The next sections further compare these platforms and discuss their relative merits and limitations.
Sensitivity analyses
A prespecified sensitivity analysis excluded studies that focused on non-endodontic or off-target tissues without an intracanal delivery. The exclusion of these studies did not materially change the direction or significance of the pooled regenerative endpoints. The numerical results are presented in Table 7.
Sensitivity analysis excluding non-endodontic or off-target tissue models.

SMD: standardized mean difference (Hedges’ g); MD: mean difference (log10 CFU); RR: risk ratio. I2 expressed as percent. Off-target exclusion is defined as models without intracanal delivery or mixed tissues outside the root canal space.
Adverse events and toxicology
Adverse events were infrequently and inconsistently reported in the studies. Among animal studies, transient inflammatory responses were described at higher doses for some platforms, whereas one antibiotic-loaded system reported tooth discoloration. One silica-based system reported oxidative stress markers that were dose-dependent. All events are summarized with dose and exposure normalization in Table 8.
Adverse events and toxicology by platform, dose, and exposure duration.
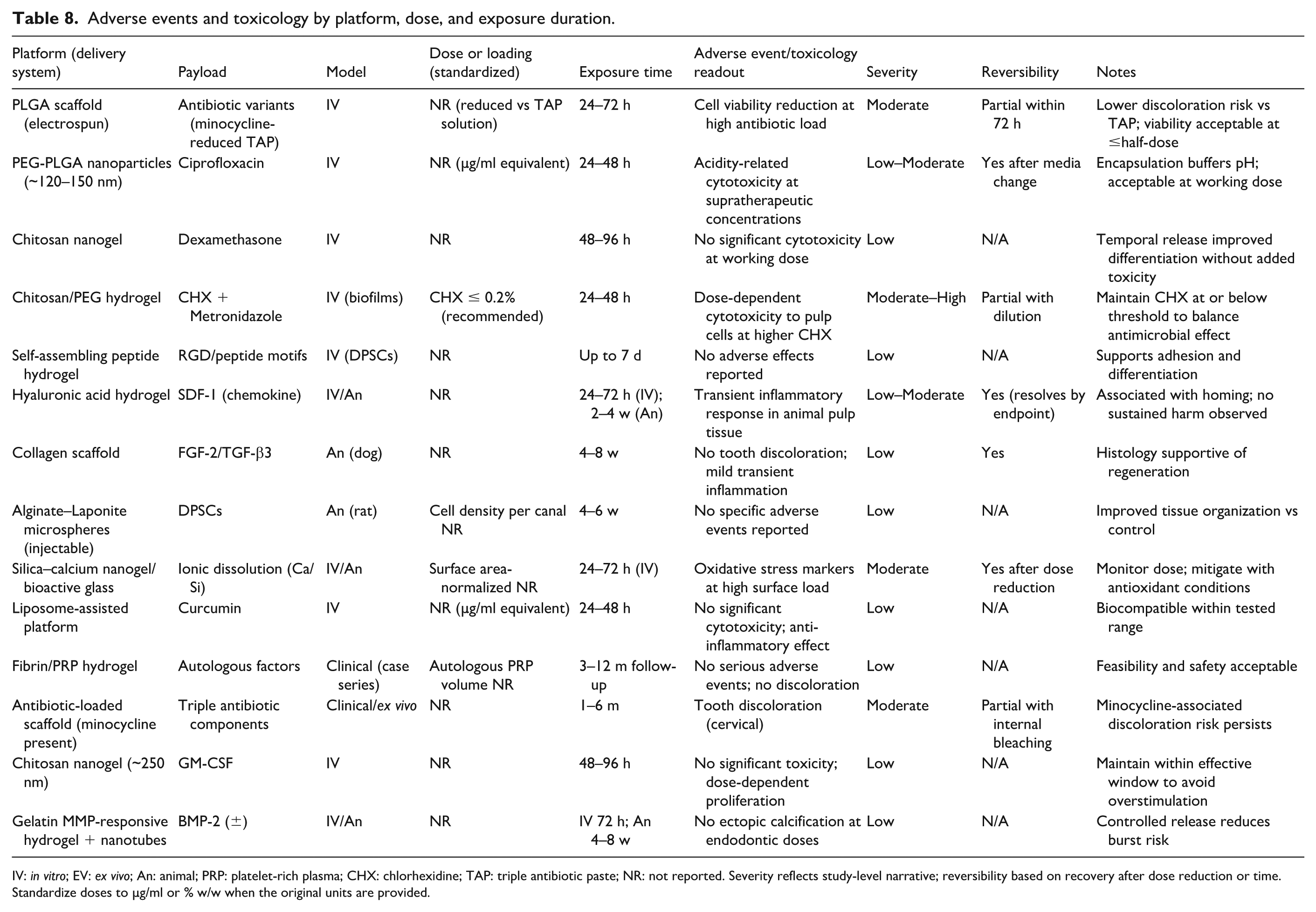
IV: in vitro; EV: ex vivo; An: animal; PRP: platelet-rich plasma; CHX: chlorhexidine; TAP: triple antibiotic paste; NR: not reported. Severity reflects study-level narrative; reversibility based on recovery after dose reduction or time. Standardize doses to µg/ml or % w/w when the original units are provided.
Comparative analysis of delivery platforms
To facilitate a direct comparison, Table 9 provides a head-to-head overview of the key characteristics of the four delivery platform categories in the context of regenerative endodontics. Below, we summarize the comparative insights.
Release kinetics: Hydrogels generally exhibit an initial burst release (due to the diffusion of surface-bound molecules) followed by a sustained phase governed by gel degradation. PNPs often minimize burst release by trapping drugs in a hydrophobic core, yielding a more linear release over days. Nanogels can be designed to be released like hydrogels (if they degrade) or like particles (if they simply diffuse cargo); the GM-CSF chitosan nanogels, for example, likely released growth factor gradually as the ionic cross-links slowly dissociate 62 . MSNs tend to retain cargo until a trigger opens the pore caps, which can result in a negligible premature release (near-zero burst) and then a rapid on-demand release. In practice, a combination (such as MSN in hydrogel) might show a dual phase: a small initial release from the hydrogel and a delayed large release when the MSN caps open.
Biocompatibility: All platforms reviewed showed good biocompatibility in pulp regeneration models, but subtle differences existed. Natural hydrogels (collagen, fibrin, etc.) are inherently biocompatible and bioactive; however, they carry a risk of immune reactions if derived from animal sources (e.g. xenogeneic collagen). Synthetic hydrogels (PEG-based) are typically inert materials. Nanoparticles, such as PLGA, degrade into acidic byproducts, which can cause inflammation in large amounts; however, in the small quantities used in canals, this seems minimal (buffering by tissue fluids likely occurs). Chitosan is both antimicrobial and biocompatible, but its pH in solution is low (due to the acetic acid solvent); formulations must ensure neutralization to avoid tissue irritation. Liposomes are generally well tolerated (similar composition to cell membranes), although cationic liposomes can be cytotoxic. MSNs, as noted, were biocompatible up to a threshold dose and have been reported to have hemostatic properties and angiogenic stimulation at moderate doses 58 . One in vitro study showed that high concentrations of silica particles could induce oxidative stress in DPSCs, but moderate levels promoted differentiation—pointing to a dose-dependent effect 63 . All platforms can be safe if properly formulated, with natural hydrogels and liposomes being inherently biocompatible, and synthetic polymers and particles requiring dose optimization.
Mechanical support: Hydrogels can provide some mechanical scaffold function but are generally soft (close to the stiffness of pulp tissue). They do not reinforce a weakened root structure by themselves, although they can serve as a matrix for new dentin formation. PNPs and nanogels do not contribute mechanical strength to the tooth (they are too small and essentially add no bulk). Mesoporous silica particles within a canal might contribute slight scaffold rigidity if densely packed, but this is likely negligible. If strengthening the tooth is a goal (to prevent fracture of thin-walled roots), approaches such as nanofibrous meshes or mineralizing scaffolds might be needed in conjunction, for instance, incorporating silica or calcium phosphate nanofibers in a hydrogel. Some hydrogels (like those with nano-hydroxyapatite) have been designed to mineralize and harden over time, thus potentially contributing to structural integrity 64 . Overall, these delivery platforms prioritize biological functions over mechanical reinforcement.
Ease of use: From a clinical perspective, simplicity is essential. Injectable hydrogels (prefilled syringes) that are set in situ are straightforward. Some thermogels can be injected as a liquid and gel within minutes at body temperature, conforming to the canal. Nanoparticle formulations might come as either a premixed paste (as in the MSN paste used by Talaat et al. 7 ) or as two components (a powder and a liquid to be mixed chairside). For example, a clinician could mix a vial of MSN powder with BMP-2, HA gel, and inject it. Although a bit more involved, this is not unlike the current use of bioceramic sealers that are mixed and placed. Liposomes and nanogels are likely delivered as part of a hydrogel or liquid vehicle, possibly as an irrigant or final injection. One could envision a final step in RET where, after disinfection and blood clot induction, a syringe of nanocarrier formulation is delivered into the canal before sealing, analogous to how PRF membranes or pastes are used now. Stability and shelf life may be concerns (liposomes can destabilize over months, and hydrogels can dry out); therefore, formulations may require refrigeration and have a limited shelf life. This is a hurdle for adoption that developers must consider in the future.
Cost and manufacturing: Hydrogels, such as collagen or fibrin, could be relatively inexpensive if sourced from blood (PRF costs little aside from a centrifuge). Synthetic hydrogels and nanoparticles entail more complex manufacturing—for example, PLGA NP fabrication under sterile conditions, liposome extrusion methods, and MSN synthesis require specialized chemistry. Large-scale production must comply with the pharmaceutical GMP standards. The cost could be significant; however, if these therapies can demonstrably save teeth that would otherwise be lost (thus preserving patient quality of life and reducing implant/prosthesis costs), there is a value proposition. The first likely translation might be in the form of commercial kits containing preloaded scaffolds with growth factors (akin to how Emdogain® provides enamel matrix derivatives for periodontal regeneration). A current analogue in orthopedics is BMP-2 on a collagen sponge (Infuse®), which is FDA-approved for spinal fusions. For endodontics, BMP-2 or FGF on a suitable scaffold could be a future product if clinical trials confirm their efficacy.
Comparative summary of delivery platform characteristics in regenerative endodontics.

In the following Discussion, we delve deeper into the implications of these findings, limitations of the current evidence, and future research avenues needed to move these technologies into routine clinical use.
Discussion
In this systematic review, we analyzed a breadth of studies on advanced biomaterial carriers of bioactive molecules in regenerative endodontics. Collective evidence affirms that these delivery platforms can significantly enhance the biological outcomes of RET compared with traditional approaches. Each platform type not only offers unique advantages but also poses specific challenges that must be addressed before clinical implementation.
Hydrogels as foundational scaffolds
Hydrogels stand out as a foundational element for regenerative strategies 3 . They fill a critical role of providing a 3D scaffold that mimics the pulp ECM (rich in collagen and glycosaminoglycans) and can be laden with signals to guide tissue formation 65 . The positive outcomes noted with hydrogel-based delivery, such as improved cell differentiation and the formation of a pulp-like tissue construct in vivo, align with tissue engineering paradigms observed in other fields, such as orthopedics and neural regeneration. However, a recurring challenge with hydrogels is controlling the release kinetics of incorporated molecules 4 . Many early attempts showed a large burst release (e.g. >50% growth factor in a day), which is wasteful and potentially harmful if the concentration spikes. Researchers have learned to mitigate this by modifying the hydrogel network (for instance, using heparin-based hydrogels that bind growth factors and release them only as new cells produce heparin-degrading enzymes, or by creating core–shell hydrogel particles that sequentially release factors) 66 . The consensus is that a sustained, low-level release of growth factors is more conducive to organized tissue regeneration than a short-lived high dose release. Clinically, this suggests that a one-time application of a growth factor-laden hydrogel at the time of RET could suffice to support months of regeneration, obviating multiple intervention visits10,67.
Degradation and clearance
If a hydrogel degrades too slowly, it might impede tissue ingrowth or act as a barrier (a problem seen in some early PEG hydrogels, which left voids of unresorbed material at 8 weeks, with tissue only growing around them). The ideal scenario is that the hydrogel gradually degrades as new tissue replaces it. Fibrin gels degrade quickly (days to weeks), but tissue often does not fill in fast enough, whereas synthetic gels can be tuned to degrade over weeks to months. In the studies reviewed, chitosan/β-GP hydrogel degraded over approximately 2 weeks in vitro, which matched the period of active dentinogenic differentiation of DPSCs, after which the cells presumably could sustain their matrix secretion without further scaffold support. In vivo, a slowly degrading scaffold (several months) might be preferable, given the longer timeline of tissue regeneration in human teeth. Designing hydrogels with on-demand degradation (e.g. sensitive to cell-secreted proteases) is a promising approach for scaffold clearance as it is replaced by the natural matrix.
Nanoparticles and combination approaches
PNPs and liposomes introduced a clear benefit in delivering antimicrobials without hindering stem cells. This directly addresses a clinical dilemma: the need to sterilize an infected canal versus the need to preserve surviving stem cell populations (or transplanted cells). The studies by Bottino et al. and Kwon et al.12,67 consistently show nanoparticle-mediated disinfection is effective and less cytotoxic. A limitation of this review is that much of the evidence is in vitro or in small animals. We lack human clinical data on whether using a nano-antibiotic hydrogel yields higher success rates or better tissue outcomes than current practice. Ethically and practically, it is challenging to measure “regenerated pulp” histologically in humans; therefore, surrogate outcomes (continued root development, apical closure, dentinal wall thickening, positive pulp vitality tests, and resolution of apical radiolucency) must be relied upon. These are being used in clinical research (two small clinical trials using stem cell transplantation have shown increased root length with cell/G-CSF vs conventional RET). It is reasonable to expect that improved delivery of signaling molecules would similarly translate to improved clinical outcomes; however, this remains to be demonstrated.
One area where PNPs can be used to prevent reinfection. In regenerative cases, the coronal seal may not always be ideal (especially if a thin calcific barrier forms instead of a robust dentinal bridge). A reservoir of antibiotics in a polymeric form could potentially continue to leach small amounts of drugs, discouraging bacterial regrowth over an extended period. This concept of “smart sterilization” could increase the long-term success of regenerative treatment. However, regulators will carefully consider chronic antibiotic exposure and resistance issues. Thus, groups are exploring alternative antimicrobial payloads, such as antibacterial peptides (e.g. LL-37, which can also recruit DPSCs) or nanoparticles (Ag, ZnO), against which bacteria are less likely to develop resistance.
Mesoporous silica and bioactive ceramics
The success of MSNs in animal studies is encouraging because they provide a non-biological (and thus more stable) scaffold that can still interact beneficially with host tissues. Silica materials have a history in dentistry (bioglass in pulp capping, for instance, which releases silicon and calcium ions that stimulate pulp cells). MSNs can be considered as a highly refined extension of that, not only delivering ions but also carrying specific factors. The fact that MSNs alone (without added BMP-2) outperformed blood clot revascularization is noteworthy. This suggests that a synthetic scaffold alone, if designed correctly, can induce a decent regenerative response. The surface chemistry of MSNs may have helped adsorb growth factors from the blood (such as fibronectin and TGF-β from platelets) and then presented them back slowly to cells, acting as an artificial ECM. In this sense, MSNs might be used even without expensive biologics as a superior scaffold to blood clots. Of course, adding a known morphogen like BMP-2 amplified results, indicating a synergistic effect 7 .
The translational challenge for MSNs is formulation: the paste used in Talaat’s study included iodine as a disinfectant and calcium hydroxide for short-term apex disinfection, which was then rinsed out and replaced with MSN/BMP2. In a clinical workflow, MSNs could be incorporated into a premixed injectable putty or hydrogel that also has some radiopacity for X-ray visualization (perhaps by adding barium sulfate). The material needs to be set or remain in place under a coronal seal (such as MTA or resin). Given that calcium silicate cements (such as MTA) are the current standard for sealing regenerative cases, one idea is to integrate MSNs into such cement. However, the high pH of MTA might deactivate growth factors; therefore, spatial separation (MTA placed after scaffold down in the canal) might be better.
Aquasomes and vesosomes—future outlook
While these have not been tested in RET yet, they represent a direction of multi-functional and protective delivery. Aquasomes could, for example, carry enzymes that naturally reside in pulp (like VEGF or angiopoietin) without denaturing them, which is helpful since protein drugs can be delicate. Vesosomes could allow a time-staged release (for instance, first wave: antibiotic from outer vesicle, second wave: growth factor from inner vesicles). The complexity and cost of these systems mean they are likely to be further from clinical use than hydrogels or simple NPs. Research in other fields should be monitored—if aquasomes make headway in, say, delivering insulin or vaccines, those advances could be translated to endodontics. Conversely, the unique anatomy of the root canal (an enclosed space) might simplify things: we do not necessarily need a long circulating nanocarrier (which vesosomes often aim for in systemic therapy), because we can place the material directly where it is needed. So, a simpler system might suffice locally, whereas complex carriers are more useful when you need to navigate through the bloodstream to a target.
Translational challenges
Despite promising preclinical results, there are hurdles to overcome:
Regulatory approval: Many of these platforms (especially those involving growth factors like BMP-2) would be considered combination drug-device products by regulatory agencies. Clinical trials will be needed to show not just efficacy but safety (e.g. does BMP-2 use in a root canal have any systemic effects or unwanted calcifications?). The bar for safety in dental materials is high because alternatives (like apexification or doing nothing) are not life-threatening scenarios; thus, the risk tolerance is low.
Manufacturing and standardization: Ensuring batch-to-batch consistency of a nanoparticle or hydrogel with biologics is non-trivial. For example, stabilizing growth factors in a shelf-stable form might require lyophilization (freeze-drying) of the scaffold with the factor, and the end user reconstitutes it. Temperature sensitivity, sterile packaging, and so on all add cost and complexity.
Training and adoption: Clinicians would need training to use these new materials. If a procedure becomes too complex (multiple steps of mixing, timing of placement, etc.), practitioners may be hesitant, especially if the benefit is not immediately evident to them. Demonstrating clear clinical advantages (e.g. a higher rate of positive vitality tests or continued root development) will be key to driving adoption.
Cost-benefit: Many regenerative endodontic candidates are children or adolescents with immature teeth. Payers (insurance) will weigh the cost of these advanced therapies against alternatives (like extracting the tooth and placing an implant later). If regenerative therapy can obviate the need for an implant or more invasive procedures down the line, it may prove cost-effective in the long run, but this needs substantiation.
Future directions
The future is likely to see hybrid approaches—combining the best aspects of each platform. For example, a “scaffold cocktail” where a hydrogel provides immediate structure and cell homing signals, embedded nanoparticles provide sustained antimicrobial and pro-differentiation cues, and perhaps gene-activated matrices that encourage host cells to upregulate necessary factors. One concept being explored is gene delivery in situ—for example, using plasmid or viral vectors in a scaffold to get host cells to produce growth factors over time (some studies have tried electroporation of growth factor genes into root canals). However, gene therapy introduces more regulatory hurdles than protein delivery.
Another burgeoning area is leveraging the body’s own extracellular vesicles (EVs). Stem cell–derived EVs can be thought of as nature’s nanoparticles, carrying microRNAs and proteins that instruct regeneration. Several studies have shown DPSCs’ exosomes can induce pulp regeneration in animal models68,69. Packaging these EVs into a gel could be another strategy that avoids using living stem cells yet harnesses their secretions.
Meta-analysis considerations
As more studies emerge, it may become feasible to quantitatively compare outcomes. For instance, if five animal studies use VEGF in a scaffold and measure vascular density in the regenerated tissue, a meta-analysis could show the overall benefit. In our review, data heterogeneity precluded much pooling. A future systematic review might focus on a narrower question, like “Does addition of growth factors to scaffolds improve radiographic root development in immature teeth?” and possibly pool animal trial data.
Limitations of current evidence
We acknowledge that most included studies are proof of concept in nature—often using healthy or sub-critically infected models rather than the true clinical scenario of a long-standing necrotic infection with wide-open apex. Animal models (especially small animals) do not perfectly replicate human tooth anatomy. Large animal models (dog, pig) are better, and fortunately some researchers are using those. Clinical human data are almost absent from these advanced platforms; hence, our discussion of clinical translation is necessarily extrapolated from preclinical findings. Risk of bias in studies was also an issue—many in vitro studies did not report blinding (though it might not introduce huge bias for objective measures like qPCR but could for scoring of staining results). The animal studies often had small sample sizes (n = 5–10 teeth per group), which is understandable but means results should be taken cautiously.
We also must consider the endpoints that matter. A regenerative endodontics success is not just radiographic healing, but ideally the tooth has a vital, functional pulp. The field is moving toward trying pulp vitality testing and even laser Doppler flowmetry to confirm neovascularization in regenerated pulps (some case reports have shown positive sensibility tests and blood flow in treated teeth). None of the biomaterial studies yet went to clinical testing to measure that.
Ethical aspect
With growth factors like BMP-2, there is a concern of triggering unwanted calcifications or even off-target effects (BMPs have been associated with osteogenic sarcoma in extremely rare cases when used in high doses in spine surgery). The dosages used in dental pulp are orders of magnitude lower, but long-term surveillance would be needed. The immune response to biomaterials is also relevant—while acute inflammation can aid healing, chronic inflammation would undermine regeneration. Encouragingly, many of these materials (e.g. chitosan, PEG, silica) have been shown to be mostly inert or even anti-inflammatory at the right doses.
Interdisciplinary integration
For regenerative endodontics to fully exploit these delivery platforms, collaboration with immunology and developmental biology is important. The regenerated tissue should ideally not just be a fibrous scar but contain nerves and vasculature akin to native pulp. Some reviewed studies evaluated reinnervation (e.g. higher neural marker expression when NGF was delivered in a scaffold). A comprehensive regenerative approach might involve sequential delivery: first an angiogenic factor (VEGF), then a neurotrophic factor (NGF) after blood vessels form, to encourage nerves to grow along the new vasculature (as occurs in development). Platforms like vesosomes could potentially achieve such a sequence with one application. This is speculative but highlights how advanced delivery tech could realize biologically sophisticated regeneration.
This review underscores that the marriage of biomaterials and biology in regenerative endodontics holds great promise. By creating permissive microenvironments and providing the necessary cues, these delivery systems can significantly push the boundaries of what is currently achievable, from simply repairing an apex to regenerating a functional pulp–dentin complex. In the next decade, some of these approaches are likely to enter clinical trials. The ultimate success will be measured by the extent to which these therapies improve patient outcomes by saving teeth that would otherwise be lost, restoring natural sensation and immune defense to teeth, and doing so reliably and safely. The groundwork laid by the diverse studies reviewed here provides a strong impetus for that translational leap.
Conclusion
RET is transitioning from an empirical procedure to precision treatment supported by advanced biomaterial delivery systems. Our systematic review and analysis demonstrate that hydrogel scaffolds, nanogels, PNPs, and other nanocarriers can each contribute critically to the goals of RET by enhancing bioactive molecule delivery, sustaining regenerative stimuli, and modulating the healing microenvironment of the target tissue. Hydrogel-based systems create a 3D template that, when infused with growth factors or peptides, can significantly improve tissue regeneration compared with blood clots alone. Nanogels and nanoparticle carriers allow the targeted and controlled release of therapeutics, such as antibiotics and differentiation factors, overcoming the limitations of direct application (such as toxicity and rapid clearance). Liposomes and related vesicular carriers offer innovative means to attenuate inflammation and deliver multiple agents in a coordinated manner. MSNs exemplify how engineered inorganic scaffolds can induce robust pulp–dentin regeneration in vivo, especially when combined with strategic biomolecules, such as BMP-2.
There is no one “silver bullet” platform; rather, the future of regenerative endodontics will likely employ a combination of these technologies to address the multifactorial nature of pulp tissue regeneration, requiring disinfection, angiogenesis, innervation, and dentin formation. For instance, a clinical protocol in the near future might involve disinfecting the canal with a nanoparticulate antibiotic formulation, followed by filling it with a BMP-2-loaded hydrogel that also contains anti-inflammatory liposomes, thus sequentially orchestrating the microenvironment from bactericidal to regenerative. Each component of such a combined system must be optimized for dosage, timing, and compatibility.
The translational pipeline for these advanced therapies is still in its relatively early stages. Key challenges include the rigorous demonstration of safety (ensuring that adjuncts such as growth factors do not provoke adverse effects such as ankylosis or inappropriate calcifications), scaling up the manufacturing of complex biomaterials under quality control, and designing clinical trials that can ethically and effectively evaluate the true biological outcomes of regeneration. The field would benefit from standardized outcome measures, such as a scoring system for pulp tissue vitality and organization on histology or agreed radiographic metrics for success beyond the mere absence of lesions (such as percentage increase in root length).
Nonetheless, the evidence to date is largely positive: these delivery platforms not only improve surrogate outcomes in the laboratory and animal models, but they also yield insights into the mechanisms of healing and regeneration. They highlight the importance of the temporal dimension of healing (providing the right signals at the right time) and spatial distribution of cues (ensuring that the entire canal space, especially critical apical areas, receives adequate stimuli). Conventional RET with only blood clots is somewhat random in these respects; it relies on whatever growth factors are released by platelets and for however long. Using engineered delivery systems, we can convert this process into a more predictable and tunable therapy.
In summary, bioactive molecule delivery platforms are poised to revolutionize regenerative endodontics by significantly enhancing the body’s capacity to regenerate tooth structures. They exemplify the progress from reparative dentistry to actual tissue engineering. As these technologies advance toward clinical reality, endodontists may soon have at their disposal an armamentarium of smart biomaterials, each playing a role in reviving the vitality of teeth that would otherwise remain dead. Achieving consistent regeneration of a vascularized and innervated pulp within a disinfected root canal will not only save teeth but also preserve their biological function, sensibility, and longevity. Interdisciplinary research is paving the way from bench to bedside, and the coming years will be crucial for translating this promising science into routine clinical practice, ultimately improving outcomes for patients with endodontic diseases.
Supplemental Material
sj-docx-1-cll-10.1177_09636897251407385 – Supplemental material for Bioactive molecule delivery platforms in regenerative endodontic therapy: A systematic review and meta-analysis
Supplemental material, sj-docx-1-cll-10.1177_09636897251407385 for Bioactive molecule delivery platforms in regenerative endodontic therapy: A systematic review and meta-analysis by Kanwalpreet Kaur, Rayan Ibrahim H. Binduhayyim, Ravinder S. Saini, Shashit Shetty Bavabeedu, Sunil Kumar Vaddamanu, Anna Avetisyan and Artak Heboyan in Cell Transplantation
Footnotes
Acknowledgements
All the authors are thankful to the King Khalid University, Saudi Arabia for the financial support.
Ethical Considerations
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Review Article Project under grant number RA/KKU/11/46.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Informed Consent Statement
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
