Abstract
Coronary artery bypass grafting (CABG) involves creating new conduits to restore blood flow to the heart. Using autologous vascular grafts presents challenges, including infection risk after vein harvesting, and scarcity of viable vein tissue. Researchers are exploring tissue-engineered vascular grafts (TEVGs), aiming to create optimal small-diameter vascular graft (SDVG) (<6 mm) for CABG surgery. TEVGs with suitable long-term patency could offer a promising alternative, potentially reduce patient morbidity and improve surgical outcomes. However, no SDVG is commercially available for CABG surgery in Europe, and no new clinical trials of coronary TEVGs have been reported since 2018. This narrative review summarizes the development and use of SDVG (<6 mm) for CABG, focusing on TEVGs and cell-seeding strategies. We outline the main types of grafts tested in preclinical and early clinical studies and highlight how different cell sources and seeding approaches aim to enhance graft functionality, patency, and long-term outcome.
Keywords
Introduction
Coronary artery disease (CAD) is the most common type of cardiovascular disease, responsible for approximately 20% of all deaths in Europe. According to the World Health Organization, an estimated 23.4 million individuals worldwide will die from cardiovascular-related diseases in 20301–3.
CAD is caused by narrowing or blockage of the coronary arteries. Partial or complete obstruction restricts the blood flow that provides oxygen to the heart muscle and can lead to angina, myocardial infarction, silent myocardial ischemia, and sudden cardiac death 4 . The main cause of CAD is atherosclerosis, which influences the vessel structure and function. Atherosclerosis is a chronic inflammatory disease that arises from accumulation of lipids in the inner layer (tunica intima), forming a plaque mainly comprising fat, cholesterol, and calcium5,6. This process occurs over time and is influenced by different risk factors, some of which are non-modifiable, for example, aging, gene disposition, and family history. The other factors are associated with a sedentary lifestyle and bad habits (fast food, smoking, and obesity), which cause diabetes, hypercholesterolemia, hypertension, and hyperlipidemia7–11. Atherosclerosis starts with a disturbance of the normal functioning of the endothelium inside the arterial wall. This disruption leads to the accumulation of lipoprotein droplets within the intima of the vessels, provoking an inflammatory–immunomodulatory reaction12,13. Vessel enlargement gives rise to atheroma and, eventually, to vessel calcification due to the invasion of exogenous cells and recruitment of smooth muscle cells (SMCs) from the tunica media to the intima, which promotes thickening and increases the deposition of calcium salts and fibrotic components11,14,15. This atheroma leads to stenosis and thrombosis, which increases the risk of myocardial infarction when localized in the coronary arteries 8 . Stenotic lesions, or those causing clinical symptoms, typically require treatment with a percutaneous or surgical revascularization procedure. Non-stenotic lesions may remain asymptomatic for an extended period; however, their disruption can trigger episodes of unstable angina or myocardial infarction9,16,17.
CAD therapies and prognosis are determined based on the patient’s history, including the symptoms of the disease, age, and the distribution of the disease in the coronary arteries. Cases of arterial occlusion are mainly treated using two different therapeutic strategies: percutaneous coronary intervention (PCI) or coronary artery bypass grafting (CABG). While the CABG procedure requires autograft extraction to bypass blocked or narrowed coronary arteries, and thereby restore blood flow to the heart 12 , PCI is a minimally invasive procedure in which the narrowed vessel is reopened and blood flow is recovered through stent implantation13,18,19.
CABG is the preferred strategy for those patients with a more complex CAD, involving the left main or triple vessel disease, and is one of the most frequently performed major surgical interventions 19 . The procedure typically involves harvesting autologous blood vessels from other parts of the body and grafting them onto coronary arteries beyond the blockage site 20 . Autologous veins and arteries constitute a suitable graft due to their excellent mechanical stability, natural antithrombogenicity, and no potential for immune rejection 19 . CABG is generally a safe and effective procedure, but is associated with some morbidities and problems, which are largely related to the extraction of autologous blood vessels 20 .
The most common autologous grafts used in CABG are saphenous veins and internal mammary arteries. Vein graft failure rates of up to 50% have been reported within a 10-year period after CABG. Long-term patency is hindered by bypass graft failure and the evolution of native vessel coronary disease18,19.
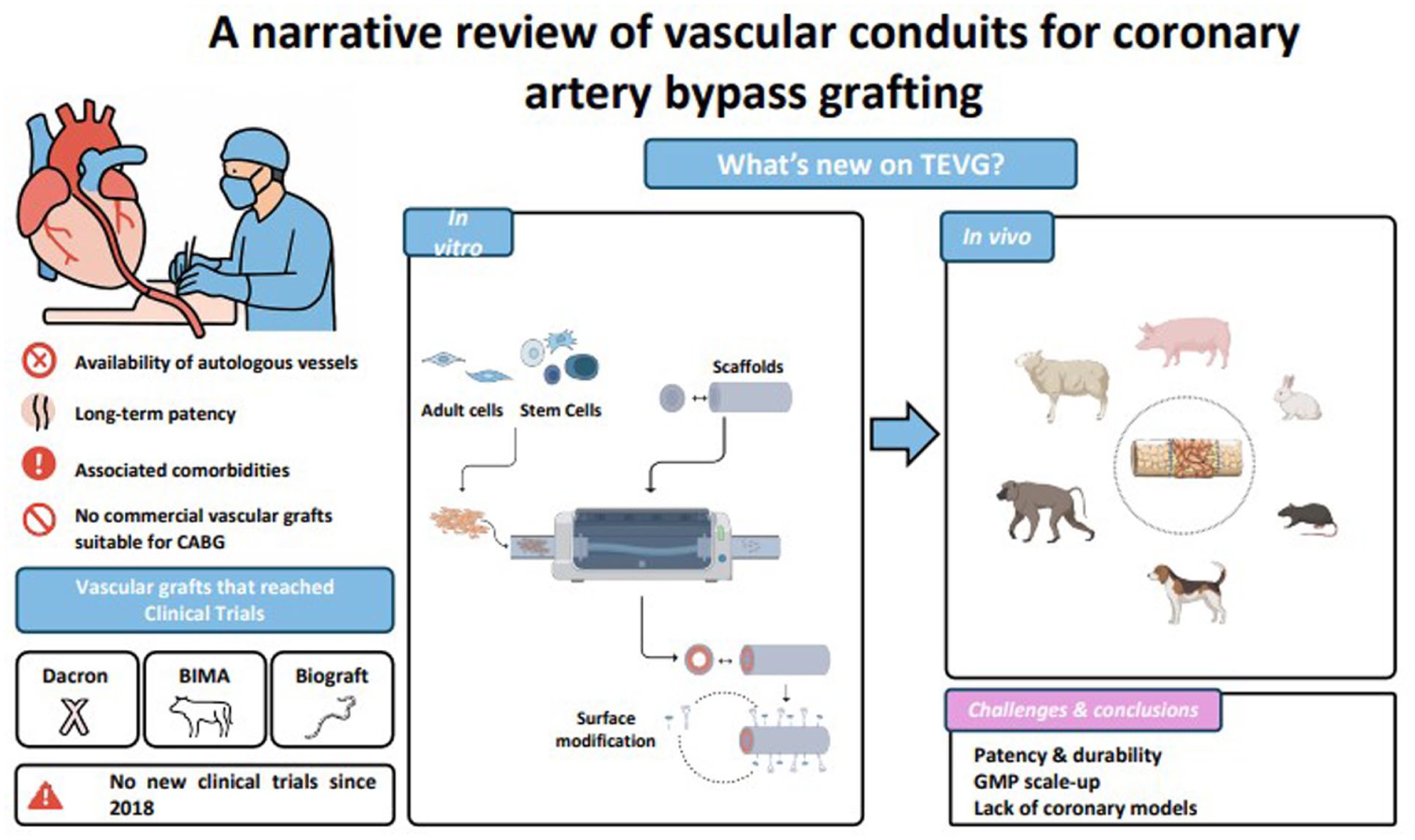
To overcome these limitations, researchers have been investigating the use of tissue-engineered vascular grafts (TEVGs) for the creation of an optimal SDVG (<6 mm) for CABG surgery21–24. Several such grafts, made from synthetic materials, are commercially available in some countries—including Dacron, which is made of woven polyethylene terephthalate (PET), and the Perma-Flow graft, manufactured from modified expanded polytetrafluoroethylene (ePTFE). Vascular grafts derived from biological materials are also commercially available—including allografts based on umbilical veins (e.g. HUV Biograft) and xenografts (e.g. Bioflow)25,26. These grafts have been utilized in vascular bypass surgery and as vascular access conduits; however, their use has not yet been adopted on a large scale. Therefore, no vascular graft is commercially available for CABG surgery in Europe, and no new clinical trial with coronary TEVGs have been reported since 201827,28.
This narrative review summarizes the development and use of small-diameter vascular grafts (SDVGs) (<6 mm) for CABG, focusing on TEVGs and cell-seeding strategies. We outline the main types of grafts tested in preclinical and early clinical studies and highlight how different cell sources and seeding approaches aim to enhance graft functionality, patency, and long-term outcome.
The clinical standard for CABG
The 2018 European Society of Cardiology / European Association for Cardio-Thoracic Surgery (ESC/EACTS) guidelines 29 recommend CABG for the relief of symptoms (mainly angina) that are refractory to medical treatment or PCI, and in cases with substantial risk of unfavorable events, such as recurrent angina, myocardial infarction, and death30,31. CABG surgery results in the creation of a new connection between two vascular segments, thereby restoring blood flow distal to the arterial obstruction, and improving the perfusion of tissue that may have become hypoperfused. The long-term success of the procedure is evaluated based on graft patency, the prevention of major adverse cardiovascular events, and overall patient survival32,33.
Whether the patient’s disease is acute or chronic, the suitability of myocardial revascularization is determined by the severity of the affected arteries, and the presence of comorbidities, such as diabetes, peripheral or cerebrovascular disease, and left ventricular systolic dysfunction26,34. In addition, myocardial viability and myocardial ischemia should be considered before performing CABG 35 . The Surgical Treatment for Ischemic Heart Failure (STICH) trial highlighted the importance of myocardial viability for determining the benefit of CABG for patients with ischemic heart failure, revealing that revascularization could improve symptoms and quality of life, particularly in patients with viable myocardium35,36.
Like any other surgical procedure, CABG is associated with risks and potential complications. Morbidity and mortality rates can vary depending on factors, such as patient characteristics and conduit choice. For example, age and previous cardiovascular events are directly related to CABG morbidity and mortality37,38. According to EuroSCORE, the mortality risk is generally low, with reported mortality rates ranging from 0.18% in low-risk patients (EuroSCORE < 4%) to 0.78% in high-risk patients (EuroSCORE > 8%)39,40. Notably, beyond the immediate postoperative period, mortality rates depend on the patient’s overall health and lifestyle, and the severity of their underlying heart disease. Long-term survival rates are generally favorable after CABG, especially in patients who adhere to the recommended lifestyle changes and follow-up care41–43.
Postoperative morbidities are also influenced by the diseases present before surgery (diabetes, renal disease, etc.), and can include infection, bleeding, arrhythmias, stroke, cognitive decline, deep vascular thrombosis, and wound healing issues44,45. Moreover, 30%–40% of patients experience depression and anxiety after CABG surgery, according to the review by Jannati and Aslani 46 . However, most discomforts that patients experience after surgery are related to the graft donor site—including the risk of sternal dehiscence and mediastinitis associated with dissection of the internal mammary artery 47 and the risk of infection at the saphenous vein harvest site26,41,48.
Overall, despite the positive impacts of CABG on improving quality of life and long-term survival, the procedure is still associated with some significant challenges15,49.
Clinical standard autologous vascular grafts
Autologous veins and arteries have favorable characteristics that make them suitable graft options for CABG. Importantly, these grafts possess excellent mechanical stability that allows them to withstand the hemodynamic forces within the cardiovascular system, and natural antithrombogenic properties that reduce the risk of blood clot formation within the graft. Moreover, since these grafts are taken from the patient’s own body, there is no possibility of immune rejection, thus eliminating the need for immunosuppressive medications and reducing the likelihood of graft failure 6 .
The autologous grafts most used in CABG include saphenous veins, internal mammary arteries (internal thoracic arteries), radial arteries, and others (as gastroepiploic artery or the inferior epigastric artery). The choice depends on the number and locations of the blockages, vessel availability, patient characteristics, and surgeon preference and expertise33,50,51. The availability of suitable autologous arteries is limited by the scarcity of redundant arteries and veins with sufficient length in the human body 52 .
Venous grafts
The saphenous vein is considered an optimal graft for CABG due to its absence of atheromatous plaques, easy accessibility, and suitable length and diameter. During the procedure, the saphenous graft is positioned in a reversed manner, to enable unobstructed blood flow through the venous valves. Notably, maintaining the long-term patency of this graft can present a significant challenge. Over time, the graft can be compromised by histological changes; notably, intimal hyperplasia can lead to narrowing of the vessel lumen, reducing blood flow32,49.
Once implanted, around 10%–25% of these grafts become occluded within the first year after CABG; subsequently, 1%–2% of grafts become blocked each year up to 5 years after surgery, and 4%–5% become occluded during each year from 6 to 10 years post-operation 51 . Overall, up to 40%–50% of saphenous veins used in CABG exhibit graft failure within 10 years50,53. Moreover, among the 50%–60% of saphenous veins grafts that remain patent at 10 years after CABG, only half of them show no angiographic evidence of atherosclerosis 51 .
To enhance venous graft durability, an alternative technique, known as “no-touch” saphenous vein grafting, has been introduced. This method is an atraumatic approach in which the saphenous vein is removed along with its surrounding cushion of tissue, thereby avoiding any direct manipulation of the vessel. Preserving the perivascular environment reduces mechanical stress and minimizes vasospasm, which reportedly improves patency rates compared with standard harvesting approaches. However, concerns have been raised about a higher incidence of complications at the harvest site33,54,55.
Arterial grafts
Arterial grafts have the advantage that they have already been functioning under arterial flow conditions, unlike veins which must undergo a process of “arterialization” following exposure to arterial flow52,56.
Currently, the internal mammary artery is considered the best arterial graft option and shows the highest patency rate (>90% after 10 years) 53 . The left internal mammary artery (LITA) is the preferred graft choice for CABG, particularly for anastomosis to the left anterior descending artery, due to its superior longevity and reduced rate of major adverse cardiovascular events. The widespread consensus among surgeons reinforces its position as the standard of care for coronary revascularization 33 . When more arterial grafts are needed, the right internal mammary artery (RITA) may be considered. The reported long-term patency rates of the RITA are variable, with some studies suggesting similar or even better outcomes compared with the LITA. Importantly, bilateral internal mammary artery harvesting may be associated with higher risks of deep sternal wound infection, dehiscence, and mediastinitis, and should thus be avoided in high-risk patients, especially in elderly, obese, or insulin-dependent diabetic patients19,33.
The radial artery can be easily harvested by endoscopic or open surgery, is larger than other arteries, and shows good correspondence and size matching to most recipient coronary vessels. The thick muscular wall of the radial artery is suitable for coronary and aortic anastomoses and wound infections are uncommon. Radial artery grafts show the highest patency when they are attached to a left-sided coronary artery with a high-grade stenosis. Conversely, their performance is less favorable when attached to the lower-pressure right coronary artery 51 . In addition, use of the radial artery should be avoided in patients with a positive Allen test, diffuse arteriosclerosis, medial calcification, renal dysfunction, upper limb trauma, Raynaud’s disease, or recent transradial coronary angiography19,57.
The gastroepiploic artery—specifically the inferior epigastric artery—has only rarely been used to bypass the right coronary artery or its branches. Its use has significantly declined due to its challenging dissection, which requires a superior midline laparotomy and expertise in mobilizing the stomach, as well as the high spasticity of this artery, which makes it difficult to manage. Other concerns are related to the potentially inadequate flow in the presence of coronary flow competition 48 . The patency rates for the gastroepiploic artery are approximately 91% at 1 year, 80% at 5 years, and 62% at 10 years 51 .
To overcome the drawbacks of autologous grafts—such as limited availability, functional limitations, and associated adverse effects—substantial efforts have been made to develop alternative small-caliber grafts. Several commercially available alternatives aim to effectively replace traditional grafts, while demonstrating satisfactory rates of patency15,49. However, such grafts have not yet demonstrated adequate patency rates, and cannot yet be used in the aortocoronary position (Table 1)34,58.
Commercialized SDVG and implanted patients in aortocoronary position.

Vascular grafts that reached clinical trials
Clinical studies on non-autologous vascular grafts for CABG are limited, mostly old, and largely anecdotal. These early trials provide only preliminary insights into the feasibility and safety of TEVGs in humans, and their use is still restricted due to complications such as stenosis, thrombosis, and limited long-term patency. Within the field of vascular substitutes for CABG, grafts can be broadly divided into three categories: synthetic, biological, and TEVG.
Synthetic grafts
Synthetic grafts have been described since the 1950s 68 . The main materials used to create synthetic grafts for peripheral vascular reconstructions include woven PET69,70, polytetrafluoroethylene (PTFE), expanded PTFE (ePTFE), and modified ePTFE.
PET, commonly known as “Dacron,” is utilized in the form of multiple filaments that are woven or knitted into vascular grafts. Dacron was introduced in 1939 and patented in 1950 by DuPont6,52,69,70. Such grafts have been used as an alternative to autologous grafts in certain cases. The first reported applications of Dacron as a coronary graft were in two children—one in 1965 and the other in 1966 59 : one connecting the right ventricular section of the coronary artery to the aorta 71 , and another was anastomosed to the ascending aorta and to the end of the coronary artery, and the graft remained patent at 1 month post-surgery 72 . In another case, Dacron was successfully interposed between the aorta and the right coronary, in a 65-year-old patient, and the graft showed good patency after 16 months of follow-up 59 .
PTFE, known as Teflon, was patented in 1937 by DuPont, but was not used as a vascular graft until 1960. PTFE is widely used in the medical field. Its excellent biocompatibility and inertness make it suitable for various medical devices and implants, in areas such as blood vessels, the heart, jaw bones, nasal structures, eyes, and the abdominal wall58,69,70.
The ePTFE, also known as Gore-Tex, offers improved mechanical properties, increasing its range of medical applications. This non-biodegradable polymer exhibits an electronegative luminal surface with anti-thrombotic properties. Currently, ePTFE is commonly used in lower-limb bypass grafts51,58,70.
In 1974, Molina et al. 73 reported the first coronary bypass using ePTFE, which showed short-term patency but was not recommended for routine use. Between 1974 and 1978, 16 patients received ePTFE grafts; although they were considered a feasible alternative when veins were unavailable, long-term patency remained unconclusive. Similar conclusions were drawn from a 1984 case in which two ePTFE grafts remained patent at 53 months74,75. In a series of eight patients (1982–1983), patency was 64% at 28 weeks, consistent with earlier short-term results. Rare long-term reports exist, with grafts patent for up to 7 and 12 years, but these remain isolated findings. Overall, the evidence consistently indicates that ePTFE grafts should only be considered when autologous conduits are unavailable, given their limited and unpredictable long-term patency60,76.
Modified ePTFE, also known as Perma-Flow, is a 5-mm synthetic vascular prosthesis with a restriction made from silicone rubber and a Venturi shape in the distal part to improve its patency61,77,78. Between 1992 and 1995, Perma-Flow grafts were used in 15 patients who needed coronary surgery and lacked sufficient autologous vessels. In each case, the synthetic graft was placed between the ascending aorta and either the right atrium or the superior vena cava, with coronary vessels connected in a side-to-side fashion. A smooth stenosis at the distal end of the prosthesis restricted arteriovenous shunting, helping to maintain stable coronary flow, which theoretically improved graft patency. The study was conducted under the United States Food and Drug Administration (FDA), and 76.9% of the constructed bypasses were functional after 1 year 79 . The Perma-Flow graft is the only synthetic graft that is currently approved in the United States, with an humanitarian device exemption58,74.
Lack of functionality of synthetic grafts
Synthetic grafts—such as those made from Dacron and ePTFE—have demonstrated limited functionality, particularly in small-diameter vessels and low-flow environments. When used under these conditions, Dacron grafts exhibit poor patency rates due to inadequate radial compliance, which is more important than mechanical strength for vascular conduits. To address this issue, grafts have been fabricated using polyurethane, which offers higher elasticity19,80,81.
When used for CABG, synthetic prosthetic grafts are typically rejected by the immune system within months52,82. The pattern of rejection is similar for both Dacron and ePTFE grafts—including the formation of amorphous deposits, fibrin coagulum, thrombosis, calcification, aneurysm, or neointimal hyperplasia, and ultimately resulting in graft failure. Such failure primarily occurs due to the absence of an endothelial cell (EC) layer, which is particularly problematic in vessels with diameters smaller than 6 mm52,58,69,83. Overall, synthetic coronary grafts have been largely unsuccessful for coronary applications, with long-term patency reported only in rare anecdotal cases19,84.
Biological grafts
Attempts have been made to use biological grafts as substitutes for bypass operations in patients without sufficient autologous grafts. Such procedures have included the application of human vessels as allografts and different types of animal vessels (bovine, canine, porcine, and ovine) as xenografts19,85,86.
Allografts
CABG with allografts refers to the use of grafts or blood vessels from a same-species donor, usually obtained from a deceased donor. The main allograft preservation techniques are cryopreservation and glutaraldehyde (GA) fixation.
Allografts maintain the tissue’s natural architecture and mechanical properties, thereby facilitating better integration and function. Because of being fixed in GA or cryopreserved to reduce antigenicity, allografts are still considered a foreign material and have the potential to trigger an immune response. In particular, the ECs are considered the most immunogenic part. Therefore, allograft recipients may still require immunosuppressive medications67,87,88. The most used cryopreserved grafts are the umbilical vein, saphenous vein, and internal mammary artery.
Umbilical veins are easily accessible, can be stored without requiring patient dissection, and have ability to withstand wall tension and arterial flow, due to its elasticity. A GA-preserved human umbilical vein (HUV), known as HUV Biograft, was approved by the FDA in 1979 89 . The HUV Biograft had a 4-mm internal diameter and showed an angiography patency rate of ~46% at 3–13 months after implantation in a coronary artery position. Other studies67,90 reported similar short-term patency rates where the HUV Biograft was used in the same position and found that intimal dissection was a common occurrence62,91.
In addition, clinical trials have investigated the use of cryopreserved saphenous veins and internal mammary arteries. Although the venous grafts showed good patency after surgery, the patency rate decreased to 60% within 1–8 weeks and eventually dropped to 0% (0 out of 9) at 6–30 months post-surgery. The internal mammary artery grafts remained functional for 2–18 months after surgery67,92–94.
To date, no long-term outcomes or additional patient data have been documented or disclosed.
Xenografts
Xenografts have demonstrated success for applications in large-diameter vessels, such as arteriovenous fistulas and lower extremity bypasses; however, their use in small-diameter vascular applications has been limited 88 . Although some commercially available xenografts have been used as bypass vessels in the aortocoronary position in humans, they are not yet approved.
In Europe, the no-react bovine internal mammary artery (NRIMA) has been available for restricted clinical use in CABG surgery. Despite the manufacturer indicates that it has a patency rate of 60%–80% when used as a coronary graft 63 , clinical trials who have undergone surgery with NRIMA have yielded conflicting results, such as Reddy et al. 95 that showed a patency rate of 57% and Englberger et al. 63 that found an overall graft patency rate of 23.1% unrelated to the intraoperative flow measurement. Based on these unsatisfactory results, NRIMA should only be considered as a last resort conduit 58 .
The Bioflow xenograft is a bovine internal thoracic artery with a 3-mm diameter, which is treated with dialdehyde starch. It was evaluated in three different studies in the 1990s58,63,65,66. The only study that assessed long-term patency found a patency rate of around 15% and concluded that it was not a suitable graft 58 .
Biocor BIMA Biograft is a treated bovine internal mammary artery with a diameter of 4–5 mm, which is treated with GA. Its implantation in the coronary position has been reported in 20 patients. Only two patients were followed up at 6 months, and the graft patency rate was less than 50%34,67,96.
Lack of functionality of biological grafts
Despite attempts to use non-autologous biological grafts in vascular surgery, both allogeneic and xenogeneic grafts have shown significant limitations. Despite their use in peripheral artery bypass procedures for years, allografts have exhibited poor long-term patency in CABG surgery97–99.
Similarly, substantial challenges are faced in the clinical use of xenogeneic grafts. These xenografts (NRIMA, Bioflow, Biocor BIMA Biograft) are chemically fixed—not decellularized—using treatments such as GA, dialdehyde starch, or No-React, which reduce antigenicity but lead to complications such as calcification, stiffness, and poor long-term patency.
Most tested xenografts have failed for CABG purposes—which can be attributed to rejection due to immunogenicity and the manufacturing process100,101.
Tissue-engineered vascular grafts
There remains a need to develop an ideal non-thrombogenic vascular graft with prolonged patency, and there are not yet any feasible commercial grafts for CABG. TEVGs are being developed with the aim of overcoming the limitations of traditional grafts25,26,102. As illustrated in Figure 1, TEVGs, including decellularized grafts, synthetic polymer-based grafts, and hybrid grafts, incorporate natural and synthetic materials and cellular components to achieve enhanced functionality.
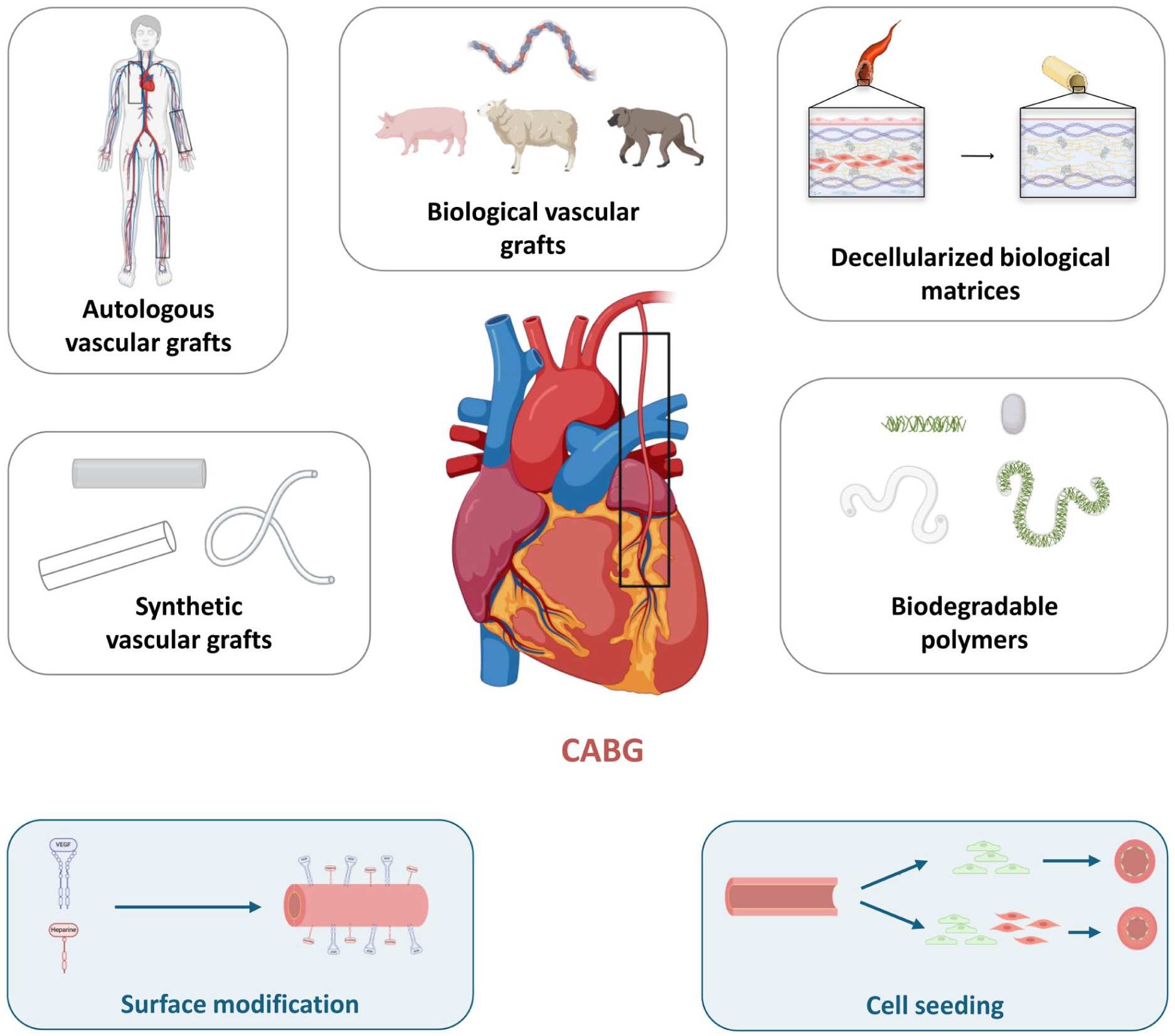
Summary of the different vascular grafts that exist for CABG surgery (
Vascular tissue engineering technology encompasses a range of techniques that involve combinations of technology with physical, chemical, and biological approaches. The main objective is to create an organized neotissue that can be seamlessly incorporated into the patient’s existing tissue, leading to restoration or enhancement of physiological function. Theoretically, such TEVGs would have an endothelium with mechanical functionality similar to native vessels26,102–104. These approaches primarily involve three components: scaffolds (polymer-based/decellularized vessels), signals (chemotactic factors and growth factors), and cells (differentiated cells and stem cells) 104 .
Scaffolds
Biodegradable polymer-based
To avoid the aneurysm problems encountered with synthetic grafts, biodegradable polymers have been developed, with biostable characteristics and properties conducive to retaining the seeded cells 19 . A biodegradable scaffold ends up degraded and being replaced by extracellular matrix (ECM). When an appropriate balance of degradation kinetics is achieved, a scaffold can provide initial mechanical support and guide tissue ingrowth, while ultimately promoting complete cellular integration and replacement by functional neotissue. These features are critical for successful tissue engineering applications and the development of biodegradable scaffolds that promote optimal healing and regeneration85,94,105,106.
A polymer’s biocompatibility is determined by its degradation products and is related to pore size 84 . In recent years, polyurethanes have been commonly used in this field, due to their adjustable thickness and porosity 83 .
Compounds currently used as vascular grafts include polylactic acid (PLA), polyglycolic acid (PGA), polyhydroxyalkanoate, polydioxanone (PDO), poly-4-hydroxybutyrate, and their copolymers83–85. Under biological conditions, such grafts are fully absorbed after 5–6 months. Investigators have attempted to adjust the properties of these grafts by creating copolymers, adding proteins, or seeding the scaffolds. In addition, new advances have been made in this field by combining nanotechnology and applying electrospinning to create scaffolds with controlled higher porosity, to facilitate cell recruitment, adhesion, and proliferation to the scaffold107,108.
Natural polymers, such as chitosan and fibroin silk, can also be used to create vascular grafts and to improve the overall structural integrity of grafts
24
. Chitosan is derived from a deacetylated derivative of polysaccharide chitin. Chitin is a substance found in the shells of crustaceans, such as shrimp and crabs. As a biocompatible and biodegradable material, chitosan is suitable for use in biomedical applications. Silk is a natural biopolymer produced by insects. Silk fibroin (SF), obtained from the silkworm (
Hybrid polymeric biodegradables, natural otherwise synthetic or a combination of both, can be blended with synthetic polymers (non-degradable) or copolymers, or chemically modified. These scaffolds are designed to exhibit enhanced compatibility with the body and to facilitate the infiltration of cells. By incorporating components found in the body’s ECM—such as collagen, gelatine, elastin, and fibrin—these natural materials promote biocompatibility, stimulating cellular integration, proliferation, and differentiation83,85. Moreover, these hybrid grafts show strength similar to that of human intrathoracic arteries and low thrombogenicity24,83,86.
Decellularized biological matrices
As biopolymers, decellularized biological matrices—derived from allogenic or xenogeneic sources—have emerged as potential options in the search for an ideal graft for CABG. Tissue engineering leverages advanced technologies to create vascular grafts that possess excellent biological activity and biocompatibility. When creating decellularized biological matrices, the primary objective is to develop an acellular vascular graft suitable for use as a vascular prosthesis 84 . The structure of decellularized vascular matrix stands out for showing exceptional biocompatibility and non-immunogenicity, with an integrity almost identical to that of the native vessels113–115.
The decellularized matrix is composed of ECM and its components which contribute to the vessel’s mechanical and functional properties100,101. Conservation of the ECM in an acellular scaffold is essential for maintaining the functions and properties that enable natural vessels to resist blood pressure changes. These properties should include elasticity and facilitate the tissue regeneration process with cell migration and differentiation24,115–117.
One of the most significant challenges related to biodegradable scaffolds is aneurysmal dilation, a frequent cause of graft failure. This complication often results from inconsistent mechanical properties between the graft and the native vessel, and from inflammatory responses that weaken the structure 118 . Compliance mismatches can lead to excessive graft expansion under physiological pressures or unintended dilation of the native vessel near the anastomosis site119,120. These issues contribute to turbulent blood flow, increased thrombosis risk, and heightened wall stress, ultimately raising the likelihood of vessel rupture.
To address these risks, scaffold fabrication strategies focus on preserving the mechanical attributes of the native vessel, to ensure optimal compatibility with the recipient’s vascular system. In cases of minor compliance mismatches, the gradual process of ECM remodeling and cellular repopulation can help equilibrate the mechanical properties over time 121 .
Researchers have developed decellularized scaffolds using both allografts and xenografts. Various methods exist for achieving decellularization, with two primary approaches: the cell-seeding technique and the cell-free approach. The cell-seeding technique involves pre-implantation seeding of recipient or stem cells onto the scaffold to enhance biocompatibility and integration. Here, the cell extraction stage is a crucial step. Donor cell content directly affects the host immunogenicity and can compromise the vessel’s properties due to thrombogenicity and degenerative changes84,85,113,114,121.
The cell-free approach relies on
Signals
Modification of the scaffold surface is an additional technique that can suppress thrombogenicity and improve the long-term patency of vascular grafts 122 . When the graft materials contact fibrin in the blood, a series of changes occurs, leading to platelet activation and aggregation, and substantial platelet adherence can lead to thrombosis 123 . Bioactive molecules with anticoagulant functions have been explored, such as heparin, which is common in clinical use.
Cells
Studies of
Various strategies have been used for the cell seeding of scaffolds. Researchers have attempted to re-endothelialize acellular scaffolds with vascular lines that express low immunogenicity and have optimal functionality.
As shown in Figure 2, different cell sources present distinct advantages and disadvantages for TEVG fabrication. Cell sources include autologous differentiated cells ECs and SMCs, two key cell types for TEVG, and stem cells. A variety of stem cells (multipotent and pluripotent) are being investigated for their abilities to differentiate into ECs or SMCs, the two principal cell lines of blood vessels. Multipotent sources include mesenchymal stem cells (MSCs) as well as endothelial progenitor cells (EPCs). Pluripotent sources include embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), which can give rise to both ECs and SMCs for TEVG fabrication129,130–132.

TEVGs in the clinical setting
Only one type of TEVG approach has been tested inhuman CABG to date, the endothelialized cryopreserved allograft, created using a cryopreserved decellularized vein, employed in CABG in 1998 58 . ECs from the patient’s own saphenous vein were cultured and seeded onto the allograft before implantation. After 7 days, the graft was successfully implanted, restoring blood flow. Six months later, the endothelialized allograft remained patent 85 .
Between 1997 and 2000, cryopreserved autologous saphenous veins seeded with human autologous ECs were implanted into 12 patients who underwent CABG. All patients were chosen due to a lack of functional autologous grafts for the bypass. Seeding with autologous human venous ECs was performed according to the protocol of Jaffe et al. 133 This procedure lasted 6 days and was followed by implantation. The graft patency rate was 80% after 6 months, 50% after 9 months, and 0% after 32 months, with failure occurring due to occlusion or stenosis26,85,132,133.
Although endothelialized cryopreserved allografts were designed to restore patency in CABG, clinical results have shown limited long-term functionality. Initial implantation restores blood flow, but grafts often fail within months due to occlusion, stenosis, or thrombosis15,85,132,133. These failures are associated with incomplete endothelial coverage, loss of cellular viability over time, and insufficient integration with host tissue. Consequently, long-term patency remains a major challenge for these TEVGs, despite their early success in small patient cohorts58,132.
What’s new on TEVG?
To date, clinical trials involving SDVG (<6 mm) for coronary applications remain anecdotal, with only a handful of reports published. Consequently, no vascular graft is commercially available for CABG surgery in Europe, and no new clinical trial with coronary TEVGs has been reported since 201826,27. As a result, current progress in the field relies mainly on
In vitro studies of TEVGs
To achieve native vessel-like properties, researchers have created synthetic/natural/hybrid polymer scaffolds using approaches that include electrospinning, weaving, molding, and three-dimensional (3D) printing. The decellularization techniques encompass different physical, chemical, and biological treatments, alone or in combinations. Other utilized techniques include freeze–thaw cycle, high hydrostatic pressure, electroporation, immersion, perfusion, surfactants, enzymatic approaches, and more 101 . Recent studies have tended to combine two or more techniques for the fabrication of SDVG136–139.
Electrospinning the polymers remains one of the most studied techniques for SDVGs. Lu et al. 138 developed a three-layer poly-caprolactone (PCL)/collagen/gelatine graft, achieving 2.63 ± 0.12 MPa tensile strength and optimized human umbilical vein endothelial cell (HUVEC) proliferation at a 2:1 PCL: collagen ratio. In a similar approach, Zheng et al. 139 electrospun PCL nanofibers functionalized with dopamine and recombinant hirudin (rH), achieving a tensile strength of 4 MPa and maintained nanoscale structure for 1 month, while supporting endothelial, despite degradation initiating at day 7. King and Bowlin 140 focused on fibber orientation in PDO grafts (~3.1 ± 0.6 MPa), showing that aligned versus random fibers can tune permeability and mechanical behavior, as well as improve hemocompatibility. Expanding beyond electrospinning, Kabirian et al. 141 used 3D printing to fabricate PCL grafts coated with polyethylene glycol and functionalized with nitric oxide, promoting HUVECs proliferation and endothelialization. Compared with electrospun grafts, 3D printing offers greater control over scaffold geometry and customization, while still supporting mechanical integrity and cytocompatibility.
The apparent mechanical advantage biodegradable polymers have does not necessarily imply physiological suitability. Excessive stiffness can impair compliance matching with host vessels, increasing the risk of intimal hyperplasia and thrombosis, facing limitations related to long-term hemocompatibility, unpredictable degradation kinetics, and incomplete endothelial functionality. In addition, although electrospinning and 3D printing enable precise architectural control, these techniques often involve complex protocols, limited reproducibility, and scalability issues that hinder standardization for clinical use.
In contrast to these scaffolds, which rely on engineered polymers to replicate vessel mechanics, decellularized vascular matrices aim to preserve the native ECM architecture and bioactive components 142 . This approach seeks to overcome some of the biological limitations of purely synthetic grafts by providing a natural structural and biochemical environment for endothelialization. Massaro et al. developed a rapid protocol to decellularize porcine carotid arteries within 5 h using detergents, successfully preserving ECM integrity. However, endothelialization assays with HUVECs revealed that residual sodium dodecyl sulfate (SDS) strongly impaired cell survival, emphasizing the critical importance of complete detergent removal to ensure cytocompatibility 143 . Nonetheless, incomplete decellularization in xenogeneic tissues can provoke immune responses and compromise graft integration, limiting their translational potential.
Decellularized allogenic tissues represent an attractive alternative, as they combine preserved architecture with natural bioactivity while reducing the risk of immunogenicity. In 2023, Wong et al. decellularized human umbilical artery (HUA) with zwitterionic and ionic sulfate. Vessel tests showed good results regarding complete decellularization and mechanical properties. Recellularization with HUVECs yielded re-endothelialization. Notably, the burst pressures were lower than in control native vessels 144 . Other studies have used sonification to decellularize HUA offering a complementary or potentially gentler method to remove cellular components while preserving ECM integrity 145 ,146,147.
Beyond purely natural allogenic matrices, hybrid scaffold approaches, made from natural and synthetic polymers, attempt to combine the mechanical strength of synthetic polymers with the bioactivity of natural matrices. Liu et al. constructed grafts from decellularized human amniotic membrane reinforced with electrospun PCL/SF. These scaffolds preserved ECM features while gaining structural stability. Similarly, Shi et al.
124
designed a trilayer construct blending decellularized porcine thoracic aorta with poly
Despite significant progress in designing TEVGs, several critical limitations persist. Biodegradable synthetic polymers, while mechanically robust, often lack intrinsic bioactivity and can trigger thrombosis or chronic inflammation without additional surface modifications. Decellularized matrices are highly dependent on the efficiency of detergent removal and may display inferior burst pressures, limiting their reliability for coronary applications. Hybrid scaffolds are promising but remain technically complex and may face challenges regarding reproducibility, large-scale manufacturing, and regulatory approval. Importantly, most studies still rely heavily on HUVEC-based assays, which may not fully predict

In vivo studies of TEVGs
To evaluate the functionality of experimental SDVGs, these constructs are typically first implanted in anatomically accessible sites, where the surgical procedure is less demanding than CABG. Once adequate performance and patency are demonstrated, subsequent testing in the coronary position is warranted to confirm clinical relevance. The following section reviews the
Most recent works have evaluated biodegradable synthetic grafts designed to support host cell infiltration or pre-seeded endothelialization, using electrospun or 3D-printed polymers to achieve adequate mechanical integrity for small-diameter arterial implantation. The studies share several features: the scaffolds are designed to support cell infiltration and remodeling, either by relying on host cells or by pre-seeding with ECs or iPSC. For example, Obiweluozor et al. implanted a two-layer PCL/PDO scaffold co-electrospun and reinforced with 3D-printed PCL in the rat infrarenal aorta. The graft exhibited good patency, SMC infiltration, and structural integration, though ECs coverage remained lower than in native vessels 148 . Similarly, Luo et al. 151 endothelialized human iPSC-derived ECs into a PGA scaffold, maintaining mechanical stability in rats but encountering limited thrombosis at the anastomoses. Joseph et al. 149 tested a PLA nanotextile graft pre-clotted with autologous blood in pigs, showing 100% short-term patency and rapid ECs attachment, outperforming ePTFE controls. In ovine models, Ono et al. 150 evaluated an electrospun biorestorative polymer scaffold with embedded nitinol, achieving 72.7% patency at 1 year, although the small sample size and follow-up duration limited conclusions about long-term remodeling. Notably, iPSC-derived ECs grafts in rhesus monkeys maintained 100% patency after 6 months, with full integration of host cells, suggesting that iPSC cell–based strategies can enhance long-term functionality 152 . These biodegradable synthetic polymers and cell-seeded grafts show high early patency, effective endothelialization, and structural integration. Limitations include the short follow-up in most studies, limited sample sizes, and the need for long-term validation.161–163
Several researchers have tested SF or hybrid scaffolds, combining SF with synthetic polymers or bacterial cellulose. In this context, Filipe et al.
111
implanted cell-free electrospun silk grafts in rats, observing higher survival (95% vs 27% for ePTFE) and complete endothelialization at 6 weeks. Tanaka et al.152,153 tested SF grafts in mice, showing early ECs proliferation and neointimal formation, although patency was limited due to thrombosis. In dogs, glycerin-coated SF grafts achieved high patency after 3–12 months, but studies lacked controls and antiplatelet therapy
164
. Jin et al.
154
evaluated hybrid SF/poly(
Following these silk-based hybrid scaffold studies, decellularized xenogeneic vessels have been investigated to improve biocompatibility and endothelialization
These studies collectively demonstrate that decellularized and re-endothelialized vessels can reduce immune response and improve functional remodeling and the benefit of pre-implantation cellularization strategies, which appear to mitigate thrombosis and intimal hyperplasia in the short term. Nevertheless, the reliance on xenogeneic/allogenic sources and complex cell-seeding protocols still raises concerns regarding scalability, regulatory approval, and clinical translatability, limiting their immediate applicability to CABG.
These experimental vascular grafts, ranging from biodegradable polymers and silk-based hybrids to decellularized xenogeneic vessels, have demonstrated high early patency, effective endothelialization, and promising integration in large-animal models, highlighting their translational potential for CABG. However, most studies remain limited by short follow-up periods, small sample sizes, variable thrombosis rates, and incomplete long-term remodeling, underscoring the need for further optimization before clinical translation.
In light of these limitations, there remains a clear need for a clinically applicable, biologically functional graft that combines the mechanical reliability of native vessels with rapid and stable endothelialization.
To address this gap, our group has developed VasCraft, a next-generation allogeneic vascular graft designed to overcome the major shortcomings of previous constructs. Unlike previous constructs, VasCraft is based on clinically available human tissue and feasible cell sources, making it a potentially safer and more translatable option. It is generated from a decellularized saphenous vein obtained from a deceased donor, subsequently re-endothelialized with umbilical cord blood–derived ECs under continuous flow conditions in a bioreactor. This dynamic recellularization strategy promotes uniform endothelial coverage and improved maturation of the graft before implantation.
Compared with previously tested synthetic, hybrid, or xenogeneic constructs, VasCraft offers several potential advantages: it uses clinically available human tissue, eliminating xenogeneic immune risks; provides physiological compliance and native-like mechanical properties; and ensures rapid endothelialization without the need for autologous cell harvesting, facilitating off-the-shelf applicability. Preclinical evaluation in a porcine arterial model is planned in the coming months to assess patency, host cell integration, and long-term remodeling. If successful, VasCraft could represent a significant step toward clinically translatable, biologically active vascular grafts for CABG.
Discussion
CABG remains the most effective revascularization strategy for patients with severe CAD, yet long-term outcomes continue to be limited by graft availability and patency. Autologous vessels are considered the gold standard, but their use is restricted by patient comorbidities, previous interventions, or limited vessel quality. Synthetic and biological grafts, although successful in large-diameter reconstructions, have consistently failed in small-diameter coronary applications due to thrombogenicity and compliance mismatch. These limitations have driven the field of vascular tissue engineering toward the development of TEVGs.
Over the past decades, significant advances have been made in scaffold fabrication, including electrospinning, 3D printing, and decellularization techniques. These approaches aim to balance mechanical integrity with biocompatibility, supporting host cell infiltration and remodeling. Despite encouraging preclinical outcomes, clinical translation remains scarce.
Cellularization strategies have emerged as a critical factor for graft success. ECs are essential for establishing a non-thrombogenic surface, while SMCs provide structural integrity and matrix deposition. However, differentiated vascular cells are difficult to obtain in sufficient numbers, have limited proliferative potential, and are sensitive to hemodynamic stress. Stem cells therefore represent an attractive alternative, with MSCs and EPCs showing immunomodulatory and pro-angiogenic properties, and ESCs and iPSCs offering robust differentiation potential into vascular lineages. Nevertheless, clinical translation is still constrained by issues such as donor variability, cell heterogeneity, ethical concerns (ESCs), and tumorigenic risk (iPSCs).
Another major limitation is the lack of consistency across preclinical models. While small animals allow rapid proof-of-concept testing, they do not replicate the hemodynamic or immunological complexity of humans. Large-animal models such as pigs or sheep provide closer similarity but still exhibit species-specific responses such as intimal hyperplasia or hyperthrombogenicity. Moreover, most experimental grafts have been tested in non-coronary positions, making it difficult to extrapolate outcomes directly to CABG.
Taken together, the current evidence highlights that no single strategy has yet achieved the ideal balance of mechanical durability, rapid endothelialization, and long-term patency required for coronary grafts. Decellularized scaffolds seeded with autologous or stem cell–derived ECs appear particularly promising, as they combine natural ECM architecture with improved antithrombogenic properties. However, consistent
Future efforts must focus on integrating advances in biomaterials, stem cell biology, and bioreactor conditioning, while ensuring standardization under good manufacturing practices (GMPs). Long-term, multicenter preclinical studies in relevant large-animal models will be essential to establish safety and reproducibility. Only through these coordinated strategies can TEVGs progress toward clinical translation and become a viable alternative for CABG patients lacking suitable autologous conduits. Both the US FDA and the European Medicines Agency impose stringent requirements, demanding not only evidence of biological safety and efficacy but also validation of standardized and scalable manufacturing processes. This regulatory path is notably longer than that for traditional medical devices. For example, the synthetic graft Perma-Flow achieved FDA approval under the Humanitarian Device Exemption, allowing market access with limited efficacy data. In contrast, TEVGs intended as general coronary conduits face a much higher burden of proof.
Conclusion
Despite decades of investigation, the development of an ideal SDVG for CABG remains unresolved. Cellularization with differentiated ECs or stem cell–derived ECs has shown potential to improve graft patency and antithrombogenic properties, although each strategy presents inherent limitations. Among the approaches explored, decellularized scaffolds recellularized with autologous or allogeneic cells from various sources—such as MSCs, EPCs, ESCs, or iPSCs—appear the most promising. These constructs preserve ECM architecture while supporting endothelialization and vascular remodeling. However, robust long-term validation in coronary positions remains scarce, particularly concerning suturability and anastomotic sealing, both of which are critical for successful clinical translation.
Future progress will depend on coordinated advances in biomaterials science, vascular cell biology, and standardized manufacturing processes to achieve clinically feasible and reproducible TEVGs.
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
Not applicable.
Author Contributions
[Georgina Iraola-Picornell, Carolina Gálvez-Montón, Christian Muñoz-Guijosa]: Conceptualization, manuscript writing, and data analysis.
[Georgina Iraola-Picornell]: Data collection and interpretation.
[Elisabet Berastegui, Cristina Castells-Sala, Jordi Martorell, Antoni Bayes-Genis, Christian Muñoz-Guijosa, Carolina Gálvez-Montón]: Supervision and critical review of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants from MICIU (PID2022-142219OB-I00, CPP2021-008438, CPP2023-010880, CPP2023-010430), Instituto de Salud Carlos III (ICI20/00135, PI21/01703, PI22/01751, DTS22/00180), CIBERCV (CB16/11/00403) as part of the Plan Nacional de I+D+I, and was co-funded by ISCIII-Subdirección General de Evaluación y el Fondo Europeo de Desarrollo Regional (FEDER), Germans Trias i Pujol University Hospital (2021_45, 2021_44) and AGAUR (2021 SGR 01437)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
A structured literature search was conducted in PubMed and Web of Science using the following keywords: coronary artery disease, coronary artery bypass grafting, tissue-engineered vascular grafts, cell seeding, stem cells, small-diameter conduits, and clinical translation of TEVG. We included studies on vascular grafts <6 mm, implanted in the coronary/aortocoronary position in humans or large animals, as well as
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
