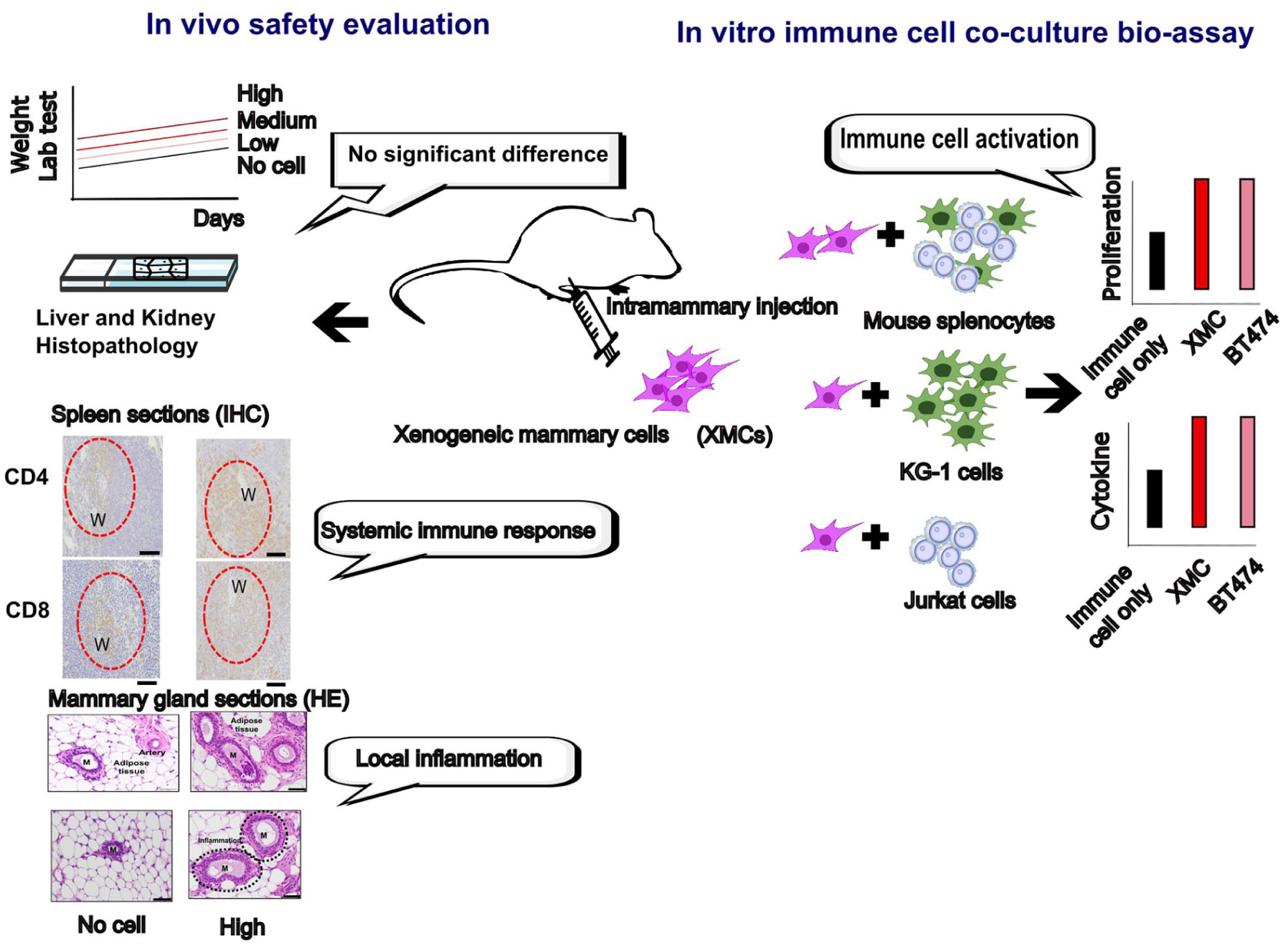
Abstract
Xenogeneic cells isolated from safe animal tissues and expanded
Keywords
Introduction
Cancer immunotherapy, which harnesses the body’s immune system to fight cancer, has reemerged as a breakthrough therapeutic strategy for treating malignancies in recent years. This resurgence follows the early use of bacteria as an immunotherapeutic agent by Dr. William Coley, who successfully treated sarcoma patients by stimulating the immune system to target and eliminate infected or tumor cells 1 . Currently, immune checkpoint inhibitors and chimeric antigen receptor (CAR) T-cell therapies are the primary forms of cancer immunotherapy. However, tumor cells often develop resistance and escape mechanisms, resulting in limited long-term efficacy for both treatments2,3. Therefore, new cancer immunotherapy approaches are needed to more comprehensively engage the immune system and achieve durable effects that are resistant. As bacterial therapy was once proposed to redirect anti-bacteria immunity into antitumor immunity, xenogeneic tissue cells have similarly been proposed as a cancer immunotherapy agent. This approach aims to transform immune rejection into antitumor immunity, exploiting the biological similarities between animal tissue cells and human cancer cells to trigger immune activation. The use of animal cells, tissues, or organs in human medicine is known as xenotransplantation. Pigs, in particular, are considered ideal donor species due to their ease of breeding, rapid maturation, and physiological similarities to humans 4 . Recent clinical trials explore the transplantation of genetically edited pig hearts and kidneys into patients with heart or kidney failure. In these studies, certain pig genes responsible for hyperacute rejection (HAR) were removed, and human genes that reduce inflammation were introduced. Despite these modifications, patients still required immunosuppression therapy to prevent rejection5,6, indicating that immune incompatibility remains a significant challenge to successful xenotransplantation. In contrast, the xenogeneic tissue cell immunotherapy approach seeks to convert xenorejection—traditionally a barrier—into an immune-stimulating mechanism that can help the body fight cancer.
Xenogeneic tissue cells express orthologous antigens similar to those found in human cancer cells of the same tissue type. These xenoantigens, including glycans, proteins, and lipids, can activate both innate and adaptive immune responses through mechanisms similar to those triggered by tumor neoantigens. In advanced cancers, tumor-associated carbohydrates and lipids are often suppressed by cancer cells through the immune escape phase of immunoediting. Previous studies have explored xenogeneic vaccines as an immunotherapeutic approach—for instance, plasmid DNA vaccines expressing human tyrosinase were used as adjunctive therapy for oral malignant melanoma (MM) in dogs 7 , and a Listeria vaccine expressing chimeric human HER2 was employed to prevent metastatic disease in dogs with osteosarcoma 8 . Given that xenogeneic tissue cells express thousands of xenoantigens capable of eliciting multiple immune responses, the resulting cross-reactivity against the human forms of these antigens leads to the targeted destruction of neoplastic cancer cells. Thus, the use of xenogeneic tissue cells represents a promising and innovative future direction for cancer immunotherapy.
In our previous study, intratumoral administration of xenogeneic mammary cells (XMCs) of porcine origin was shown to prevent tumor development and induce antitumor immunity by increasing T-cell and natural killer (NK) cell infiltration, immune cell proliferation, cytotoxicity, and cytokine secretion in a preclinical mouse breast cancer model 9 . Furthermore, intratumoral XMC treatment also reduced tumor growth at non-injected sites in a bilateral tumor model, suggesting the induction of a systemic immune response 9 . These findings indicate that XMCs may serve as a novel immunotherapeutic agent for breast cancer, with the potential to activate potent and widespread immune responses. Despite the availability of different therapeutic modalities for breast cancer, the disease causes an estimated 42,250 deaths in the United States, and the 5-year survival rate for advanced cases has improved only modestly—from 24% to 31%—over the past decade10,11. Therefore, more innovative and therapeutic approaches are urgently needed to improve patient outcomes.
To support the development of a new cell-based therapy derived from xenogeneic tissue cells for breast cancer, this study conducted an
Materials and Methods
Animals
All adult female mice were purchased from the National Laboratory Animal Center (Taipei, Taiwan). Animal experiments were conducted in accordance with institutional ethical approval (IACUC No. 113061). During the experiment, animals were housed under a 12-hour light/dark cycle with free access to food and water. The ambient temperature was maintained at 25°C ± 2°C. All animal studies were designed and performed according to the 3Rs principle (Replacement, Reduction, and Refinement). Every effort was made to minimize both the number of animals used and any potential discomfort. Tissues from euthanized animals were collected for a subsequent analysis.
Preparation of Porcine XMC and Cell Culture
XMCs were isolated and expanded from specific-pathogen-free (SPF) pigs obtained from the National Agricultural Technology Research Institute (Miaoli, Taiwan). Porcine mammary gland tissues were dissected, minced into small fragments, and incubated in a cell-isolation solution containing type VI collagenase (Worthington, Lakewood, NJ, USA) in Hanks’ balanced salt solution (100 U/ml) to facilitate tissue dissociation. Following isolation, XMCs were maintained in DMEM (Dulbecco’s Modified Eagle Medium)/Ham’s F12 medium supplemented with 10% fetal bovine serum (FBS), 10 mM HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid), and antibiotics (100 U/mL penicillin, 100 µg/mL streptomycin, and 5 µg/mL amphotericin B) at 37°C in a humidified atmosphere containing 5% CO₂. Cell morphology was assessed using a Nikon Diaphot inverted microscope at 40× magnification.
Mouse splenocytes were isolated from adult C57BL/6 mice (National Laboratory Animal Center, Taiwan). Mice were euthanized with isoflurane, and spleens were aseptically collected into 1.5-mL Eppendorf tubes containing 1 mL ice-cold RPMI-1640 medium supplemented with 10% FBS and 1% antibiotic-antimycotic. To single-cell suspensions, spleens were gently pressed through a 70-µm sterile cell strainer placed in a 50-mL tube and rinsed with 5-mL ice-cold complete RPMI medium. Using the plunger of a 1-mL syringe, spleens were mechanically dissociated into fine particles. The cell suspension was centrifuged at 3000 rpm for 5 min at 4°C, and red blood cells were lysed with 2 mL of RBC lysis buffer for 5 min at room temperature. After a second centrifugation, splenocytes were resuspended in warm complete RPMI medium and used for further experiments. The human Jurkat and KG-1 cell lines were obtained from the Bioresource Collection and Research Center (BCRC, Hsinchu, Taiwan), also confirmed their identities. Both cell lines were maintained in RPMI-1640 medium (Gibco, 22400071) supplemented with 10% heat-inactivated FBS (Gibco, A38401), 1% penicillin-streptomycin (Gibco, 15140122), and 1% antibiotic-antimycotic (Gibco, 15240062) under the same incubation conditions (37°C, 5% CO2).
Repeated Dose Toxicity of XMCs in the Mouse Model
A total of 16 adult female BALB/c mice (6–7 weeks old) were used to evaluate the effects of intramammary administration of xenogeneic porcine mammary cells (XMCs) at varying doses. Mice were randomly assigned to four groups: a no-cell treatment group, a low-dose group, a medium-dose group, and a high-dose group, each receiving two injections. On day 1, mice in the treatment groups received intramammary injections into the mammary fat pads with or without XMCs, followed by a second injection on day 22. The administered XMC doses were 5 × 105 cells (low dose), 1 × 106 cells (medium dose), and 5 × 106 cells (high dose). Body weight was recorded on days 1, 8, 15, 22, and 28 to monitor any treatment-related changes. Mice were observed daily for clinical signs of toxicity and euthanized on day 28 for histopathological examination of the liver, kidneys, and injection sites.
Laboratory Testing: Hematology and Biochemical Analysis
Whole blood samples were collected from each mouse before treatment (day 0), on day 2 after treatment initiation, and again on days 21 and 28 (before necropsy). Blood samples were collected in EDTA-2K tubes to prevent coagulation and maintain sample integrity and subsequently stored at 4°C to minimize cellular degradation before analysis. Hematological parameters were analyzed using the ProCyte Dx hematology analyzer (IDEXX, Columbia, MO, USA), while biochemical parameters were assessed using the DRI-CHEM 4000 analyzer (Fuji, Japan).
Histopathology
On day 28, all animals were necropsied, specific organs and tissues—including the spleen, liver, gallbladder, bilateral kidneys, mammary glands, and surrounding adipose tissue—were excised and weighed. The tissue was trimmed, dehydrated through graded alcohols, cleared with xylene, and embedded in paraffin. Sections measuring 4–6 µm were cut and stained with hematoxylin and eosin (H&E) for microscopic examination. Histopathological findings were evaluated using semi-quantitative criteria as described in the study by Mann et al. 12 , a four-tier grading system. A score of 0 indicated a normal tissue structure (appropriate to age, sex, and strain), while scores of 1, 2, and 3 corresponded to minimal, mild, and moderate lesions, respectively. A standardized system that enabled consistent comparison of pathological changes across treatment groups.
Immunohistochemistry
Formalin-fixed, paraffin-embedded (FFPE) tissue sections were cut at 4 µm thickness, deparaffinized in xylene, and rehydrated through a graded ethanol series. Antigen retrieval was performed by incubating in citrate buffer (pH 6.0) at 95°C for 20 min, followed by cooling at room temperature. Endogenous peroxidase activity was blocked using 3% hydrogen peroxide in methanol for 10 min. Sections were incubated overnight at 4°C in a humidified chamber with primary antibodies against CD4 (GTX85525, GeneTex, Irvine, CA, USA) and CD8α (GTX53126, GeneTex, Irvine, CA, USA). After PBS (Phosphate-Buffered Saline) washes, an HRP (Horseradish peroxidase)-conjugated secondary antibody was applied for 1 hour at room temperature, followed by DAB (3, 3’-diaminobenzidine) detection and hematoxylin counterstaining. CD4+ and CD8α+ T cells were identified by membrane staining and quantified in spleen sections using ImageJ software (NIH).
Proliferation Co-Culture Bioassay
XMCs (5 × 105 cells/well) were seeded in a six-well plate and incubated in DMEM/F12 complete medium. After overnight adhesion, immune cells (1 × 106), including KG-1 cells, Jurkat cells, and mouse splenocytes labeled with 5 μM CFDA-SE solution (Invitrogen, V12883), were added separately for co-cultures. On day 3, immune cells were harvested by gently pipetting the medium from the plates into sterile tubes. Following centrifugation at 3000 rpm, the supernatant was collected into a separate tube for cytokine assay. Cells were resuspended in 1× PBS and centrifuged again to wash out residual medium. Immune cells in PBS were analyzed for CFDA intensity using flow cytometry (BD Biosciences, San Jose, CA, USA). CFDA-low cells were defined as proliferating cells based on fluorescence intensity gates set against non-divided controls.
Cytokine Secretion ELISA Assay
Cytokine secretion was harvested from the co-culture assay. Expression levels of interferon (IFN-γ) and IL-2 were analyzed using ELISA kits according to the manufacturer’s instructions with minor modifications. For human cytokine (Abcam, AB270883 and AB236895), the antibody–antigen complex formation was performed in a single step by mixing the samples with antibody cocktails and incubating them in coated-plate strips. For mouse cytokines (Biolegend, 430815 and 431001, San Diego, CA, USA), a 96-well plate coated with capture antibody was used 1 day before the assay. After blocking, samples were added, followed by the detection antibody to form the antibody–antigen complex. TMB (Tetramethylbenzidine) development solution and stop solution were then applied, and optical density (OD) was measured at 450 nm using a Multiskan GO plate reader (Thermo, 24899, MA, USA).
Statistical Analysis
Statistical analyses were performed using GraphPad Prism (GraphPad Software, Inc., Boston, USA). Data are presented as mean ± standard deviation (SD). Comparisons between two groups were analyzed using an unpaired
Results
Body and Organ Weights, Hematology, and Biochemical Analysis of Mice Treated With Intramammary Injection of XMCs
Body weight and organ weight are fundamental parameters in toxicology studies for assessing systemic toxicity. Changes in body weight may indicate reduced food intake, metabolic disturbances, or deterioration in general health. In contrast, alterations in organ weights can reflect target organ toxicity, such as hepatomegaly, nephropathy, or reproductive toxicity13,14. As shown in Fig. 1a, all mice, with or without XMC injection, exhibited a significant increase in body weightrelative to day 0, which remained relatively stable during the first 3 weeks. On day 28, organ weights, including spleen, liver, gallbladder, left and right kidneys, and mammary glands, were collected (Fig. 1b). The statistical analysis revealed that only the mammary gland weights in the medium-dose and high-dose groups were significantly different from those of the no-cell treatment group (

Body and organ weight changes, hematology, and biochemical analysis of mice treated with xenogeneic porcine mammary cells (XMC). Body weights were measured at baseline (day 0) and throughout the 28-day observation period following two injections into the mammary tissue at varying doses. (a) Body weight development in mice receiving different doses injected into the mammary fat pad. (b) Organ weights. Whole blood samples were collected at 0, 2, 21, and 28 days post treatment and analyzed using an automated hematology analyzer (c). The levels of red blood cells (RBC), white blood cells (WBC), hemoglobin (HGB), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC) were quantified. Serum levels of glutamic oxaloacetic transaminase (GOT), glutamate-pyruvate transaminase (GPT), blood urea nitrogen (BUN), and creatinine were also measured in mice treated with different doses (d). All data above are presented as mean ± SD (no-cell treatment group: n = 3; low dose: n = 3; medium dose: n = 5; high dose: n = 5). All results were compared with the no-cell treatment group. *
Hematological and biochemical analyses are essential for evaluating the safety of novel drug candidates. Hematological parameters, including red and white blood cell counts and platelet levels, provide insights into bone marrow function, immune status, and potential hematotoxicity. Clinical chemistry profiles, such as liver enzymes (glutamic oxaloacetic transaminase [GOT] and glutamate-pyruvate transaminase [GPT]), renal biomarkers (blood urea nitrogen [BUN] and creatinine), and electrolytes, help detect early signs of organ-specific toxicity. These blood-based evaluations serve as sensitive, minimally invasive indicators of systemic toxicity, allowing for dose-response assessments and reversibility evaluations 15 . Hematological results are summarized in Fig. 1c. Whole serum biochemical analyses are presented in Fig. 1d. Although some parameters fluctuated at different time points, all values remained within reference limits 16 .
Histopathological Analysis of Liver and Renal Toxicity Assessment in Mice Treated With Intramammary Injection of XMCs
Histological examination of the liver and kidney is essential in toxicology studies, as these organs are primary sites of drug metabolism and excretion and are often the earliest targets of systemic toxicity17,18. Histopathological examination of liver tissues (Fig. 2a) revealed no treatment-related abnormalities. The renal cortex and medulla (Fig. 2b) appeared intact, with no evidence of glomerular damage. In addition, no degeneration or necrosis was observed in the renal tubules or collecting ducts. No liver or renal abnormalities were detected, indicating the absence of hepatic or renal toxicity. Collectively, these findings suggest that administration of varying doses of XMCs does not cause detectable toxicity to the liver or kidney. Overall, results showed no significant or consistent dose-related effects, supporting the conclusion that XMC administration does not affect hematological, hepatic, or renal functions.
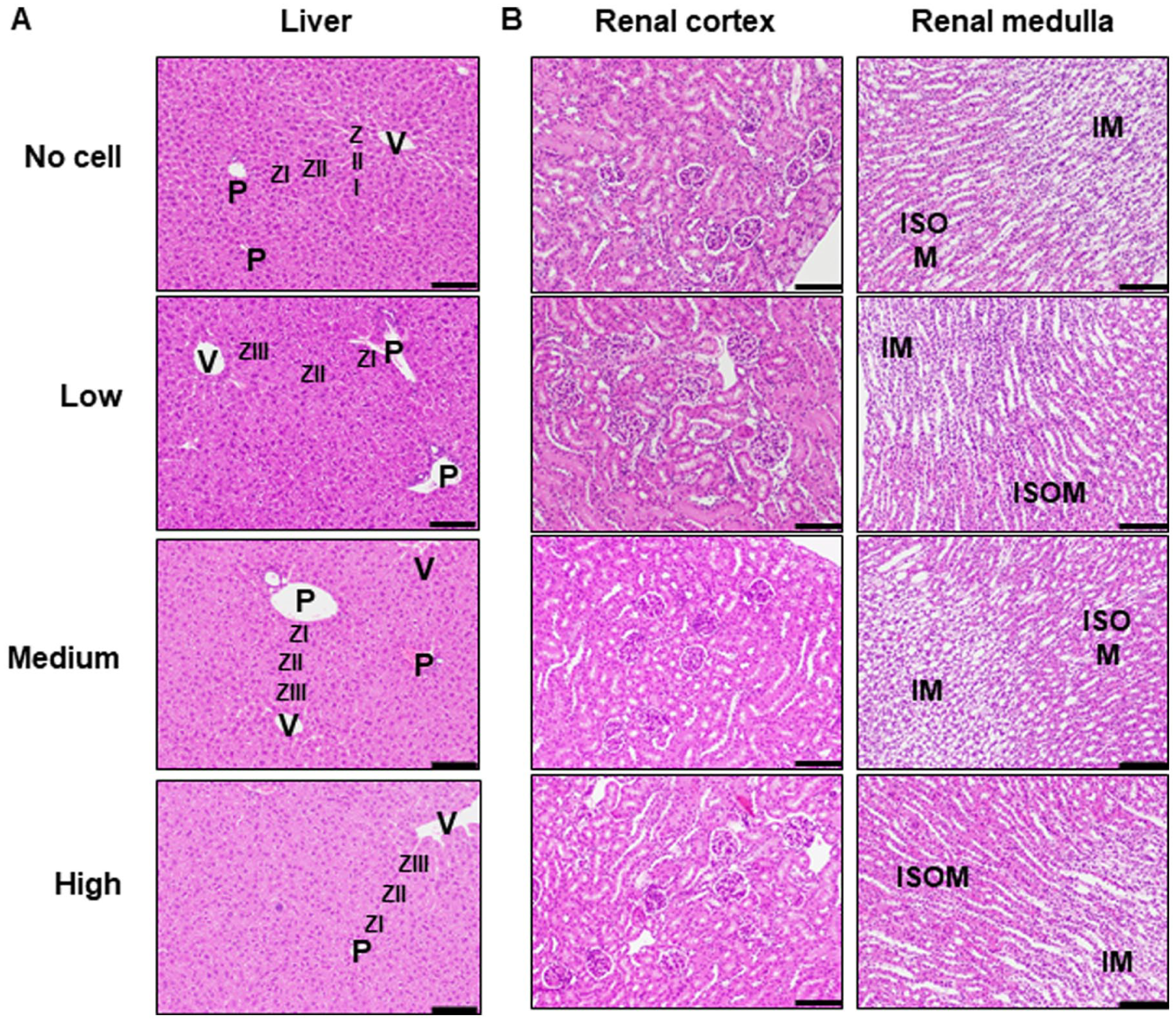
Histopathological evaluation of livers and kidneys from mice treated with different doses of XMCs. Liver and kidney histology were assessed in the no-cell, low-dose, medium-dose, and high-dose treatment groups. Hematoxylin and eosin (H&E) staining was performed to evaluate tissue morphology. (a) Liver sections from each treatment group showed no significant abnormalities in most samples. Key anatomical structures include the portal vein (P), central vein (V), and hepatocytes from the three hepatic zones: Zone I (ZI), Zone II (ZII), and Zone III (ZIII). (b) Kidney sections of both the cortex and medulla regions showed no morphological abnormalities in any samples. Key structures identified include the inner stripe of the outer medulla (ISOM) and the inner medulla (IM). Scale bar: 100 μm.
Mammary Gland Fat-Pad Histopathology of Mice Treated With XMCs
Histopathological examination of mammary gland ducts is for detecting early local toxicological responses, particularly in the context of xenogeneic cell administration or exposure to bioactive materials. The mammary ductal epithelium and surrounding stromal regions may exhibit cellular aggregation, inflammatory cell infiltration, or formation of immune-related structures, localized immune activation, or tissue irritation. Observations such as clustering of immune cells or inflammasome-like structures provide important insights into the biocompatibility and potential adverse effects of experimental treatments on mammary tissue integrity19,20. As shown in Fig. 3a, aggregated cell clusters (10–20 cells per cluster) were observed within mammary ductal or small arterial lumens, predominantly near the injection site. Under high magnification, these clusters contain two distinct cell types: (1) large round cells with pale nuclei and a nuclear-to-cytoplasmic ratio of approximately 1:2, suspected to be stem cells or detached mammary epithelial cells, and (2) smaller round cells with condensed nuclei, likely lymphocytes. Subacute inflammation surrounding the mammary ducts was observed in the low-, medium-, and high-dose groups, characterized by predominant neutrophilic infiltration, with lymphocytes and macrophages as secondary components. Samples from the low-dose group also exhibited multinucleated giant cells. Both the medium- and high-dose groups showed post-injection infiltration of neutrophils, lymphocytes, and macrophages, along with connective tissue proliferation, indicating a mild inflammatory response following intramammary administration of XMCs (Fig. 3b). Although the statistical analysis did not reveal significant differences for cell aggregates, the limited sample size suggests that these observations should be considered a potential risk factor, as animals receiving local XMC injections may exhibit a local lymphoid response, including robust lymphoid follicle development. In summary, intramammary administration of XMCs induced localized subacute inflammation, characterized by inflammatory cell infiltration, multinucleated giant cells, lymphoid hyperplasia, and transient cell aggregation within ducts, suggesting a physiologic immune response to xenogeneic cell exposure rather than overt pathological toxicity.
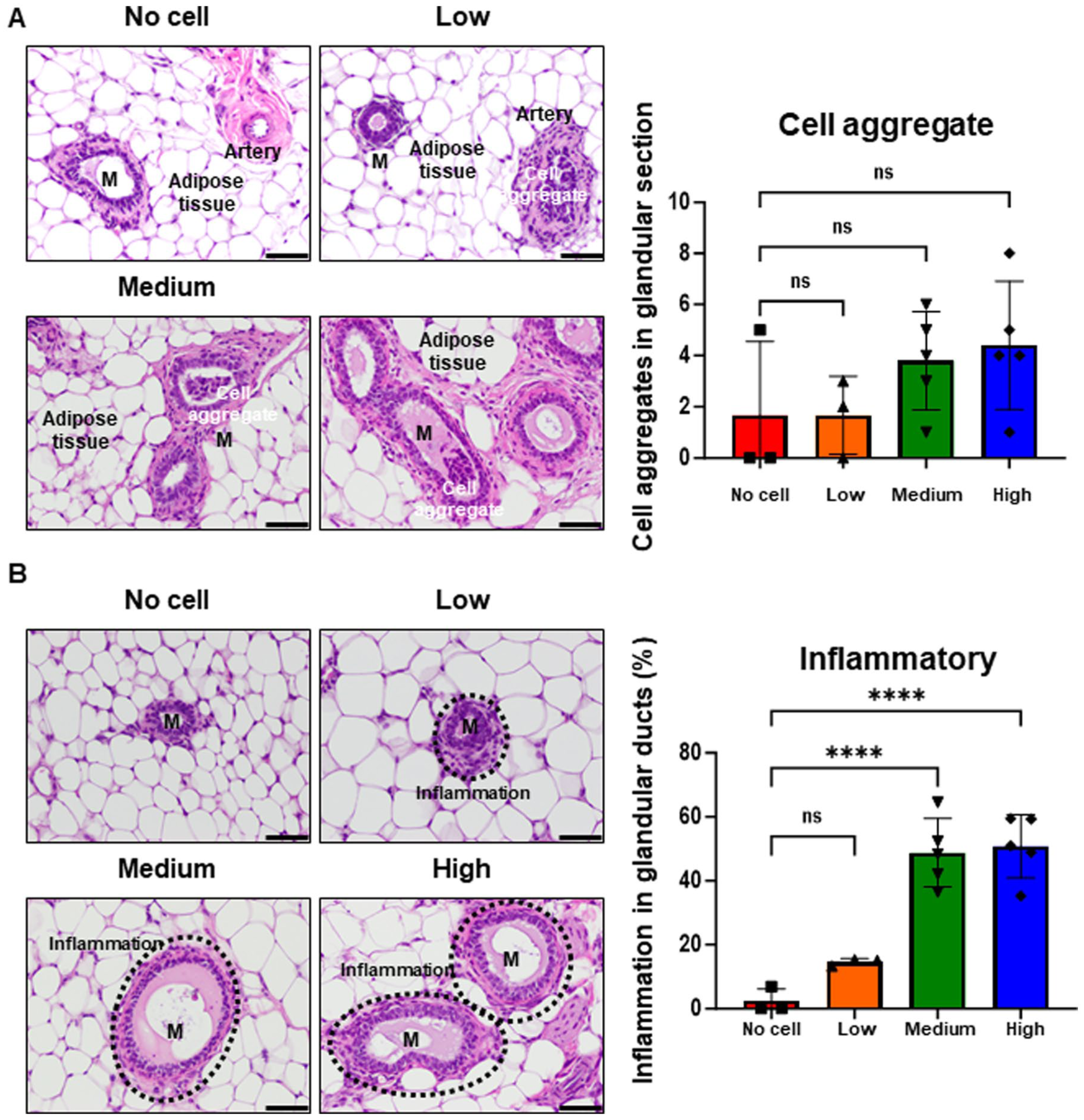
Histological analysis of mammary glands from mice treated with different doses of xenogeneic mammary cells (XMCs). Mammary gland tissues were used for hematoxylin and eosin (H&E) staining. Experimental groups included the no-cell, low-dose, medium-dose, and high-dose treatment groups. (a) Representative images show cell aggregates in mammary gland fat-pad sections from treatment groups, along with quantitative analysis. (b) Representative images show inflammation observed in the mammary gland fat pads of treated mice, with corresponding quantification. Key anatomical structures include the mammary gland (M), small artery (artery), adipose tissue (adipose tissue), and suspected stem-cell clusters (cell aggregates). Quantification of abnormal features was conducted across all mammary gland fat-pad sections, with abnormalities defined as the presence of cell aggregates or inflamed granular ducts throughout the section. All data are presented as mean ± SD. ****
Immune Cell Population in XMC-Treated Mice Spleens
To evaluate whether local XMC treatment could activate systemic immunity, immune cell populations in the spleens were analyzed by IHC staining for helper T cells (CD4⁺) and cytotoxic T cells (CD8α⁺) (Fig. 4a). For CD4⁺ T cells, the positive rate in the no-cell treatment group was 12.93 ± 0.65%. In contrast, the low-dose group increased by 58.9% (20.55 ± 0.82%), the medium-dose group increased by 81.8% (23.52 ± 7.14%), and the high-dose group increased by 116.9% (28.05 ± 3.68%,

Immunohistochemical (IHC) staining of CD4 and CD8α in xenogeneic mammary cell (XMC)-treated mouse spleens. Representative images show (a) CD4 and CD8α expression in spleen tissue from mice injected with XMC. Spleen sections were stained with antibodies specific to CD4 and CD8α, followed by DAB chromogen development (brown staining). Hematoxylin was used as a counterstain (dark-purple nuclei). Positive staining for CD4 and CD8α is indicated by brown membranous or cytoplasmic signals. (B) Quantitation of CD4 and CD8α positive cells (n = 3) in spleens from different treatment groups. Key anatomical structures identified in each section include the white pulp (W). All data are presented as the mean ± SD. **
Proliferation of Mouse Splenocytes and Human Immune Cells Induced by XMCs
Local XMC treatment elicited inflammatory responses and systemic immune effects. To further assess how XMCs modulate the immune system, cell-based assays were conducted by co-culturing XMCs with immune cells to evaluate their proliferation response. Freshly isolated mouse splenocytes, composed of T cells, B cells, NK cells, and dendritic cells (DCs) 21 , were labeled with CFDA-SE to track cell proliferation via fluorescence dilution and co-cultured with XMCs. Mouse splenocytes also displayed a marked increase in proliferation, from 14.37% ± 4.33% in control splenocytes to 67.73% ± 3.06% in the XMC co-culture group (Fig. 5a). This effect was comparable to that observed in co-cultures with BT474 cells, a human breast cancer cell line serving as a xeno-control 22 , which induced a proliferation rate of 62% ± 12.90% (Fig. 5b). The heterogeneous immune cell populations in splenocytes responding to XMCs indicate that xenogeneic cells stimulate immune cell proliferation, suggesting a potential role in modulating immune responses.

Xenogeneic cells stimulate immune cell activation and proliferation. Freshly isolated mouse splenocytes, KG-1 dendritic cells, and Jurkat T cells were assessed for proliferation in a co-culture system with xenogeneic and allogeneic cells. Mouse splenocytes were co-cultured with (a) Xenogeneic mammary cells (XMC) and (b) BT474 human breast cancer cells. KG-1 dendritic cells co-cultured with (c) XMC and (d) BT474 human breast cancer cells (allogeneic control). Jurkat T cells were co-cultured with (e) XMC and (f) BT474 human breast cancer cells. Proliferation was measured by CFDA-SE staining detected via flow cytometry. A statistical analysis was performed using an unpaired
To evaluate the ability of xenogeneic cells to activate human immune responses, co-culture assays were performed using KG-1 dendritic-like cells, derived from a bone marrow aspirate of a 59-year-old male with erythroleukemia progressing to acute myelogenous leukemia
23
, and Jurkat cells, representing human T lymphocytes, established from the peripheral blood of a 14-year-old male with acute T-cell leukemia
24
. KG-1 cells had a baseline proliferation rate of 17.13% ± 3.69%, which increased significantly to 38.14% ± 1.80% when co-cultured with XMCs (Fig. 5c). In comparison, KG-1 cells co-cultured with BT474 cells (allo-control) exhibited a proliferation rate of 30.7% ± 6.99% (Fig. 5d), indicating a more robust stimulatory effect by XMCs. Similarly, Jurkat T cells showed enhanced proliferation in the presence of xenogeneic cells. Their baseline proliferation rate was 27.05% ± 4.41%, which increased to 71.13% ± 5.13% with XMC (Fig. 5e). Whereas co-culture with BT474 cells led to a proliferation rate of 58.6% ± 6.09% (Fig. 5f). These findings collectively demonstrate that XMCs significantly enhance the proliferation of both Jurkat and KG-1 cells, with the most pronounced effects observed in KG-1 cells. Upon
Xenogeneic Cells Promote Cytokine Secretion in Mouse Splenocytes, KG-1, and Jurkat Cells
Cytokines are signaling molecules produced and secreted by immune cells, such as DCs and T cells, during immune responses to foreign microbes or non-self-cells and communication and coordination among immune components 27 . To further examine the stimulatory effect of xenogeneic cells on immune activation, two key cytokines—IFN-γ and interleukin-2 (IL-2)—which play critical roles in antitumor immunity, were quantified in the co-cultured medium of XMCs with mouse splenocytes, KG-1 cells, and Jurkat cells. IFN-γ, secreted by DCs and T cells during immune responses, activates macrophages, DCs, and T cells to elicit antitumor activity 28 . IL-2, also by DCs and T cells, stimulates proliferation and functional activation of T cells, NK cells, and B cells 29 . After 72 hours of co-culture, cytokine levels were measured in the supernatants to assess the immunostimulatory impact of XMCs. The results revealed a significant increase in IFN-γ secretion in mouse splenocytes, with XMC co-culture yielding 31.49 pg/mL ± 6.89, compared to 26.33 pg/mL ± 0.06 in BT474 co-culture (Fig. 6a). For KG-1 dendritic-like cells, IFN-γ levels were 35.2 pg/mL ± 8.72 in the XMC co-culture and 32.8 pg/mL ± 2.64 in the BT474 group (Fig. 6b). Similarly, Jurkat T cells exhibited IFN-γ levels of 32.5 pg/mL ± 7.06 in XMC co-culture and 33.05 pg/mL ± 1.40 in BT474 co-culture (Fig. 6c). IL-2 secretion followed a similar pattern. In mouse splenocytes, IL-2 increased from 6.50 pg/mL ± 2.71 in the control to 17.42 pg/mL ± 2.65 in XMC co-culture; BT474 co-culture was 14.87 pg/mL ± 1.59 (Fig. 6d). In KG-1 cells, IL-2 level rose from 2.96 pg/mL ± 0.08 in the control to 8.07 pg/mL ± 2.33 in XMC co-culture, compared to 5.17 pg/mL ± 2.94 in BT474 co-culture (Fig. 6e). In Jurkat T cells, IL-2 increased from 4.09 pg/mL ± 0.97 in the control to 13.62 pg/mL ± 2.19 in XMC co-culture, while BT474 co-culture showed 4.95 pg/mL ± 2.63 (Fig. 6f). Collectively, these findings demonstrate that XMCs enhance IFN-γ and IL-2 secretion in both mouse and human immune cells, reflecting their immunostimulatory properties. The multiple xenoantigens (proteins, glycans, lipids) expressed by xenogeneic cells likely activate DC and T cells, leading to abundant secretion of immune mediators such as IFN-γ and IL-2. These elevated cytokine levels suggest that xenogeneic cells employ multiple immune-stimulatory mechanisms to potentiate immune activation.

Xenogeneic cells promote cytokine secretion in co-culture with mouse splenocytes, KG-1 dendritic cells, and Jurkat T cells. Cytokine secretion levels of IL-2 and IFN-γ were measured in various immune cell co-cultures under different experimental conditions. Cytokine levels in collected co-cultured media were quantified using ELISA. The production of IFN-γ in (a) mouse splenocytes, (b) KG-1 cells, and (c) Jurkat cells co-cultured with XMC or BT474 cells. The production of IL-2 in (d) mouse splenocytes, (e) KG-1, and (f) Jurkat cells co-cultured with XMC or BT474 cells. A statistical analysis was performed using one-way ANOVA and multiple comparisons. Statistical significance was defined as **
Discussion
This study demonstrated the safety profile of local XMC injection, showing no physiological, hematological, or biochemical abnormalities in treated mice. An initial NOAEL (No Observed Adverse Effect Level) was established. Furthermore,
Localized inflammatory responses characterized by increased lymphocyte infiltration were observed at injection sites. However, no evidence of tissue necrosis or fibrosis was detected, indicating that XMCs induced controlled immune activation in the surrounding tissue microenvironment rather than pathological inflammation. The appearance of increased aggregates within mammary ducts may reflect epithelial cells detaching from the basement membrane as a consequence of localized immune responses. To further evaluate these findings, immune cell activation was examined through
Clinical evidence from recent xenotransplantation studies supports this concept. In genetically engineered pig-to-human or pig-to-primate cardiac and renal transplants, the host immune responses have been shown to specifically target the xenografts without inflicting significant damage to host tissues5,6,36. Compared to organ xenotransplantation, cellular xenotransplantation is considered inherently safer because it involves only cell transfer, lacking vascular integration37–39. For cancer applications, local administration of XMCs—such as intramammary injection for breast cancer—confines their activity to the targeted tissue, minimizing systemic exposure. This localized delivery ensures that most cells remain at the injection site, reducing contact with circulating antibodies and immune cells except for those few that may enter the bloodstream. Supporting this, a biodistribution study in mice with orthotopically grafted breast tumors demonstrated that fluorescent dye-labeled XMCs remained localized within the tumor microenvironment for up to 21 days following injection 9 .
One primary concern in xenotransplantation applications is the presence of the alpha-gal epitope expressed on pig cells but absent in human cells. Consequently, humans produce natural anti-Gal antibodies. When pig organs are transplanted into a human body, these antibodies trigger HAR by activating the complement system and platelets, which result in endothelial cell damage, thrombosis, vascular occlusion, ischemic injury, and untimely organ failure 40 . This reaction can also induce alpha-gal syndrome (AGS), an allergic condition that may cause severe or even life-threatening symptoms such as anaphylaxis 41 . However, since xenogeneic cells used in our application lack vasculature, the antibody-mediated hyperacute blood clot formation observed in organ xenotransplantation is not a concern. Nonetheless, alpha-gal epitopes may still pose potential risks related to coagulation thromboembolic events or alpha-gal allergy in XMC-based clinical applications. Therefore, it is critical to assess the risk of anti-Gal-related responses in non-human primate or humanized mouse models before initiating first-in-human clinical trials. With the advancement of organ xenotransplantation6,42, genetically modified pigs have been developed to remove genes responsible for producing pig-specific carbohydrates, including alpha-gal, thereby preventing HAR and alpha-gal allergy triggered by natural antibodies in recipients. Alternatively, cells derived from such genetically modified pigs could be used in clinical applications to mitigate the risk of HAR and alpha-gal–related complications.
The immune-stimulatory activities of xenogeneic cells—collectively referred to as xenorejection—can be classified into HAR and acute humoral xenograft rejection (AHXR). These reactions rapidly target vascularized organs through host antibody and immune cell binding to the xenograft’s vascular endothelium, leading to its destruction and loss of function30,31. Xenogeneic porcine endothelial cells have been shown to stimulate CD4+ and CD8+ T cells to secrete IL-2 26 . Multiple xenoantigens (proteins, glycans, lipids) present within xenogeneic cells can activate DC cells and T cells, leading to secretion of immune mediators such as IL-2 and IFN-γ, as identified in our study. These findings suggest that xenogeneic cells employ multiple immune-stimulatory mechanisms. Both IL-2 and IFN-γ are pro-inflammatory cytokines, and IL-2 plays a critical role in promoting IFN-γ synthesis by T lymphocytes 43 . The synergy between IL-2 and IFN-γ is for the rejection of non-self xenogeneic cells or mutated cancer cells44,45. The therapeutic use of IL-2 has been approved by the US FDA for treating metastatic renal cell carcinoma and metastatic melanoma. However, its efficacy requires high doses, which can lead to severe adverse effects and limited survival benefits 46 . Similarly, IFN-γ is FDA-approved for chronic granulomatous disease and used in Japan for adult T-cell leukemia, though it has shown limited efficacy and significant side effects when used as monotherapy in other cancers 47 . The observed increase of IL-2 and IFN-γ secretion from immune cells, stimulated by xenogeneic cells in our study, supports their potential as immune-stimulatory agents. These cells may promote the endogenous production of antitumor cytokines, providing a promising immunotherapeutic approach.
Conclusion
In conclusion, the present preclinical
Footnotes
Acknowledgements
The authors are grateful to the Research and Development Office of China Medical University for supporting this research with the Amnis Image StreamX Mark II instrument.
Author Contributions
Conceptualization, L.L., C.S., C.W., W.M., and C.H.; methodology, C.S., L.L., C.W., W.M., and C.H.; writing—original draft preparation, C.S. and C.H.; writing—review and editing, C.S., L.L., C.W., W.M., and C.H.; funding acquisition, C.H. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Science and Technology Council, Taiwan, grant number MOST 110-2314-B-039-026-MY3, and China Medical University Hospital, grant number DMR-114-030.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author C.S. was employed by the company eXCELL Biotherapeutics Inc. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Statement of Human and Animal Rights
Animal experiments were conducted according to approval (IACUC No. 113061) provided by the Institutional Animal Care and Use Committee, Animal Technology Research Center/Division of Animal Technology, Agriculture Technology Research Institute on 2024/08/20.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
