Abstract
Adult glioblastoma (GBM) is a highly malignant tumor with a poor prognosis and high mortality rate. As versatile 3D culture systems in vitro, organoid models are emerging as a promising new tool for GBM research and combat. However, Matrigel, the most used extracellular matrix, is animal-derived with a complex composition and significant batch-to-batch variability, requiring further optimization for GBM organoid construction. Hydrogels, high-affinity polymers, have been widely employed in organoid construction for their customizable properties. In the present study, we selected and tested several commonly used hydrogel materials—hyaluronic acid methacryloyl (HAMA), chitosan methacryloyl (CSMA), and gelatin methacryloyl (GelMA)—for the construction of GBM organoids. To address the limitations of a single Matrigel, we combined Matrigel with different hydrogels and found that hydrogels influenced glioblastoma stem cells and organoid formation in distinct ways. Matrigel-HAMA (MH) promoted the formation of independent spherical clones but with a significantly lower glioblastoma stem cell (GSC) proliferation rate. GelMA-HAMA(GH) could replace Matrigel preserving the characteristics and proliferative capacity of GSCs and supported the formation of more compact spherical clones than MH did. Further experimentation with ribosomal inhibitor CX5461 and CX5461 + IFNβ indicated that GH-based GBM organoid model constituted an efficient system for GBM drug testing, discovery, and precision medicine.
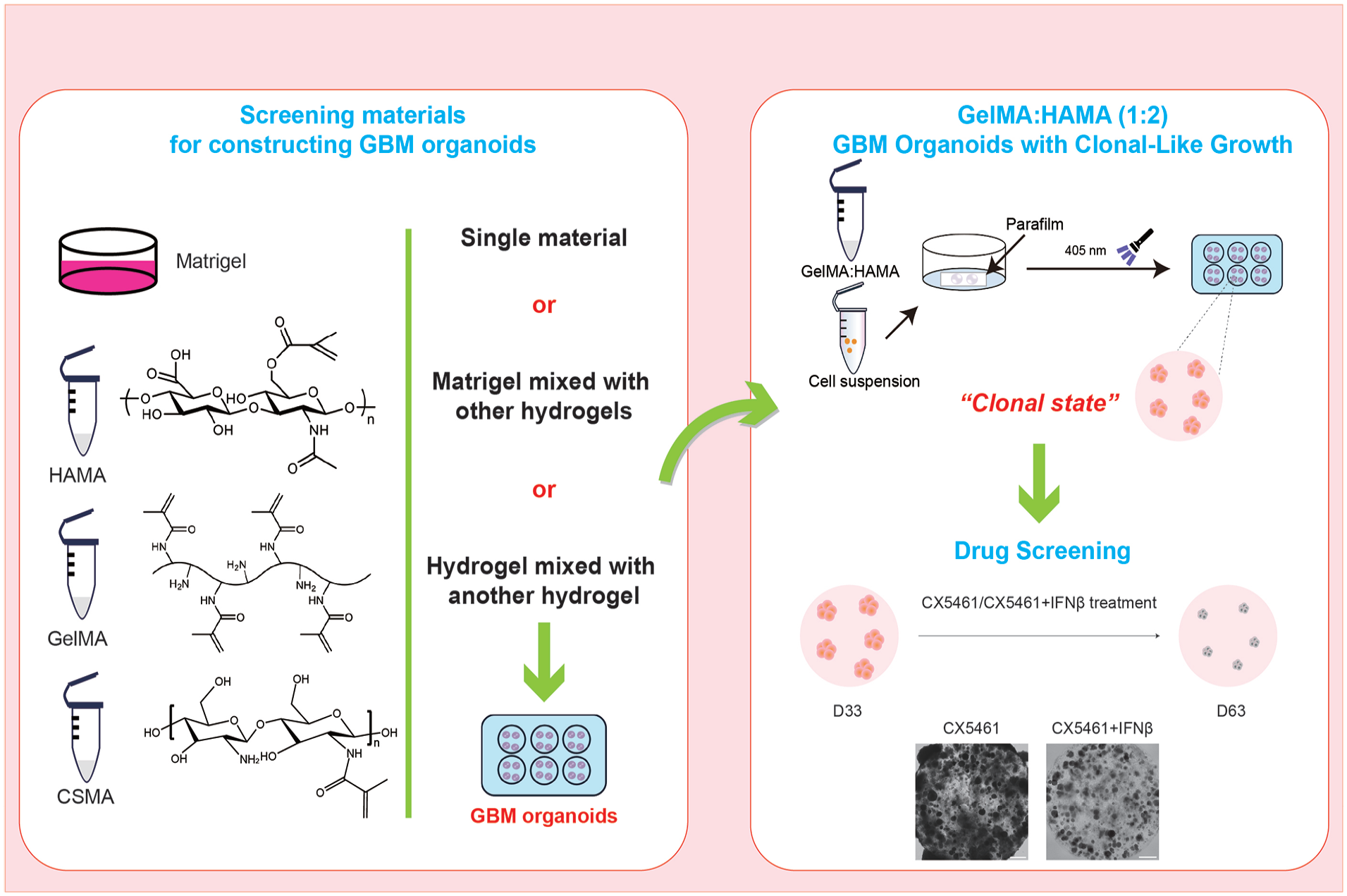
Introduction
Adult glioblastoma (GBM) is a highly malignant, diffuse glioma and represents one of the most aggressive types of brain tumor, characterized by poor prognosis and limited therapeutic effectiveness1,2. GBM has strong intertumoral and intratumoral heterogeneity, a hallmark significantly contributing to its therapeutic resistance and clinical management challenges3–5. Glioblastoma stem cells (GSCs) are widely recognized as the primary cause of GBM initiation and progression6,7. The growth of GBM tumors is inherently dependent on the three-dimensional (3D) in vivo environment, rendering conventional two-dimensional (2D) cell culture models unable to capture the complexity of GBM biology8,9. In recent years, organoid technology has emerged as a promising approach, offering new opportunities and insights into GBM research10,11.
Previous studies mainly employed mouse-derived Matrigel to construct GBM organoids12,13. However, Matrigel is primarily composed of laminin, which creates an environment more conducive to the growth of epithelial tumors14–16. In contrast, the brain’s native extracellular matrix is rich in hyaluronic acid (HA) and proteoglycans 17 . As a result, the environment provided by Matrigel is not optimal for the physiological conditions of the brain. Matrigel also has rather different biochemical and biophysical properties with the brain extracellular matrix 18 . In addition, Matrigel has considerable batch-to-batch variability, significantly impacting the consistency and reproducibility of experimental outcomes 19 .
Hydrogels are hydrophilic polymers with standard materials such as HA, alginate, gelatin, and fibrin 20 . HA is critical in the brain’s extracellular matrix, providing essential hyperelasticity and strength 21 . Gelatin, a hydrolyzed derivative of collagen, offers cell adhesion sites 22 , while chitosan, a polysaccharide-based hydrogel, demonstrates excellent stability and high swelling capacity 23 . For their biocompatibility, degradability, non-toxicity, cost-effectiveness, high availability, and ease of modification, these materials are extensively used in tissue engineering, regenerative medicine, and cosmetic applications 24 . Hydrogels are increasingly used as extracellular matrices to provide 3D support for various cell and tissue cultures in biological research 25 . However, hydrogels alone do not accurately recapitulate the complex compositions and physiological properties of cancer tissue extracellular matrix (ECM). Therefore, combining various materials to better mimic physiological conditions is a crucial research direction.
In the present study, we investigated three biological hydrogel materials—hyaluronic acid methacryloyl (HAMA), chitosan methacryloyl (CSMA), and gelatin methacryloyl (GelMA)—along with Matrigel. We examined the morphology and proliferation of GSCs in these materials and characterized the mechanical properties of GSCs cultured in each. This approach enabled us to screen reliable hydrogel materials for constructing GBM organoids and explore optimal material ratios to create more accurate models.
Materials and methods
Preparation of hydrogel materials
The photocured GelMA, CSMA, HAMA hydrogel materials, and lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) were purchased from Guangzhou Tanshtech Company (LAP, 81000720; GelMA, 81000598; CSMA, 81000610; HAMA, 81000352). Following the manufacturer’s instructions, the selected mass concentration was 5%–-15% (w/v) for GelMA, 1%–2% (w/v) for CSMA, and 1%–2% (w/v) for HAMA. Photocuring of the hydrogels required the LAP dissolved in PBS (VivaCell, C3580-0500) (pH 7.4). The reaction mixture was then placed in a water bath at 40°C, with manual oscillation for 30 s every 5 min to ensure complete dissolution. When clear and transparent, the solution was filtered through a 0.22 μm filter for sterilization, yielding a 0.25% (w/v) LAP solution. The filtered LAP solution was stored in sterile Eppendorf tubes at 4°C, protected from light, for future use.
Preparation of GelMA solution: A 5% (w/v) GelMA solution was prepared by dissolving GelMA in LAP solution. The mixture was then placed in a shaker at 1500 rpm and 65°C for 30 min, or until the solution became clear and transparent while avoiding light exposure.
Preparation of CSMA solution: A 1% (w/v) CSMA solution was prepared by dissolving CSMA in LAP solution. The mixture was placed in a shaker at 1500 rpm and 50°C for 5 h, or until the solution became clear and transparent while avoiding light exposure.
Preparation of HAMA solution: A 1% (w/v) HAMA solution was prepared by dissolving HAMA in LAP solution. The mixture was then continuously vortexed at room temperature, away from light, for 30 min or until the solution became clear and transparent.
The HAMA, GelMA, and CSMA solutions were pasteurized to prepare them for cell culture. They were placed in a constant-temperature shaker, heated to 80°C, and maintained at that temperature for 30 min. Following this, they were rapidly transferred to an ice-water mixture and soaked for 5 min. This process was repeated once.
Preparation of composite hydrogel
Matrigel (Corning, growth factor reduced Matrigel, 354230) should be thawed overnight at 4°C and stored in separate aliquots for future use. For mixing Matrigel with hydrogels, the operation should be performed on ice. First, calculate the total volume needed to construct the organoids and then mix the hydrogel with Matrigel according to the required ratio. Gently pipette the mixture several times to avoid bubble formation until the components are thoroughly mixed. The phenol red indicator in Matrigel can help confirm that the material is properly mixed. In this research, Matrigel was mixed with HAMA, GelMA, and CSMA in 1:2 ratios, respectively. To form gel spheres, 15 µL of the mixed solution was dropped onto the parafilm in a Petri dish. The dish was left at room temperature for 5 min, followed by 30 s of irradiation with a 405 nm light source. The Petri dish was then inverted, and gravity was used to help the gel droplets form into spheres. The gel spheres were then solidified by placing the dish in an incubator for 5 min.
For the experiment involving mixing hydrogels, sterilized GelMA, CSMA, and HAMA solutions were combined in ratios of 1:0, 2:1, 1:1, 1:2, and 0:1, respectively. The mixture was gently pipetted several times to ensure complete blending. Drops of the mixed solution (15 µL per drop) were then placed onto the sealing film in a Petri dish and irradiated with a 405 nm light source for 30 s.
Characterization of hydrogel materials
1. Swelling test: The swelling rate was measured to characterize the swelling properties of different materials. The hydrogel was prepared in the required volume and weighed, and its initial mass before swelling was recorded as M₀. The hydrogel was then soaked in PBS (pH = 7.4) and incubated at 37°C for 24 h. Afterward, the surface was gently cleaned with filter paper to remove excess PBS, and the hydrogel was weighed again, with the final weight denoted as Mₜ. The swelling rate was then calculated using the formula:
where S represents the swelling rate. Each group of hydrogels was tested in triplicate.
2. Storage modulus: Prepare 200 µL of different materials for testing the storage modulus using a rheometer (ARES-G2). The constant strain was set at 1%, and the angular frequency range was 0.1–100 rad/s. The test was conducted at a constant temperature of 37°C, with measurements taken at multiple angles.
Culture of GSCs
The GSCs used in this experiment were obtained from the cell bank in our laboratory26,27. These cells require culturing on Poly-
Thawed GSCs were seeded onto the coated plates, and the GSCs culture medium used was DMEM/F12 (Gibco, 11330-032) supplemented with 1× N-2 (Gibco, 17502-048), 1× B-27 (Gibco, 17504-044), 1× Pen-Strep (Gibco, 15140-122), and 1× GlutaMAX (Gibco, 35050-061). The medium was freshly supplemented with 25 ng/ml rhEGF (R&D Systems, 236-EG-01M), 25 ng/ml bFGF (HumanZyme, HZ-1272), and 25 ng/ml heparin (Sigma, H3149) before use. Every other day, the GSCs were refreshed with a new medium.
Organoid construction and culture
The amount of Matrigel and hydrogel required was calculated based on the system of embedding 4000 GSCs in 15 μL droplets. Different hydrogel materials or Matrigel were selected and mixed in varying proportions, followed by gentle mixing several times to ensure uniformity. The required number of cells was then added and mixed again to avoid air bubbles. It is important to note that experiments involving Matrigel should be performed on ice to prevent solidification. Parafilm was placed on a Petri dish, and the mixture was dispensed onto the Parafilm, with 15 μL per droplet. For the MH, MC, and MG models, the Petri dish was first placed in a biosafety cabinet for 5 min, followed by irradiation with a 405 nm light source for 30 s to solidify the mixture. It was then transferred to an incubator for another 5 min to ensure complete solidification of the Matrigel components. After solidification, the droplets were gently transferred from the Parafilm to the culture plate with the medium and cultured further in the GSCs medium. For GH and CH models, the Petri dish was irradiated with the 405 nm light source for 30 s to solidify the mixture, after which the droplets were transferred from the Parafilm to the culture plate using the GSCs medium and cultured further.
Comparison of MH models constructed with HAMA of different molecular weights
Prepare 1% (w/v) HAMA with molecular weights of 150 kDa (Engineering for life, EFL-HAMA-150K), 300 kDa (Guangzhou Tanshtech Company, 81000352), and 400 kDa (Engineering for life, EFL-HAMA-400K). Construct MH models following the established protocol and perform long-term observations.
Organoid fixation and immunofluorescence
The organoids were carefully aspirated from the culture plate into a new 24-well plate using a 1 ml wide-bore pipette tip (a 1 ml standard pipette tip, cut and disinfected). The residual medium in the well was aspirated and discarded, and the organoids were washed once with 1× PBS. They were then fixed in 4% PFA for 15 min, followed by three washes with 1× PBS, each for 10 min. To perform gradient dehydration, the samples were immersed for 3 days in a series of sucrose solutions: 10% sucrose, 20% sucrose, and 30% sucrose. For embedding, a solution of PBS with 10% sucrose and 7.5% gelatin was prepared. The samples were incubated in the embedding solution at 4°C for 10 min and then quickly frozen on dry ice for 20 min. The samples were sectioned at a thickness of 10 μm using a cryostat (Leica, CM1950) and mounted onto slides. Slides were stored at −20°C until staining.
For immunofluorescence staining, the samples were first removed from the refrigerator and allowed to reach room temperature for antigen retrieval using an antigen retrieval solution. After cooling to room temperature, the samples were immersed in 0.25% PBST (0.25% Triton-X 100 in PBS) for 20 min. The sample area was then circled with a histochemical pen and incubated with 0.125% Triton-X 100 and 5% normal donkey serum in PBS for 1 h. After blocking, the blocking solution was removed, and the primary antibody was prepared in the blocking solution. A volume of 200 μL of primary antibody was added to each slide, and the slides were incubated at 4°C for 22 h. After incubation, the slides were washed three times with 0.125% PBST. The primary antibodies used were anti-SOX2 (Mouse, Santa, sc-365823, 1:300); anti-Ki67 (Mouse, Dako, M7240, 1:500); anti-CD133 (Rabbit, Abclonal, A12711, 1:2000); anti-Nestin (Mouse, Millipore, MAB5326, 1:500); and anti-Cleaved Caspase-3 (Rabbit, CST, 1:500). The secondary antibody was prepared in 0.125% PBST, and the samples were incubated with the secondary antibody at room temperature for 2 h after the primary antibody was washed away. DAPI staining was performed after incubation with the secondary antibody, and the slides were sealed following three washes with PBS at room temperature for 20 min. The second antibody and DAPI information is as follows: Alexa Flour 488-conjugated AffiniPure Donkey anti-mouse IgG (H+L) (1:1000, Jackson, 715-545-150); Alexa Flour Cy3-conjugated AffiniPure Donkey anti-rabbit IgG (H+L) (1:1000, Jackson, 711-165-152); Alexa Flour 488-conjugated AffiniPure Donkey anti-rabbit IgG (H+L) (1:1000, Jackson, 017-000-121); Cy3-AffiniPure Donkey Anti-Mouse IgG (H+L) (1:1000, Jackson, 715-165-151); and DAPI (1:1000, Biotium, 40043).
CX5461 and IFNβ treatment of GH organoids
Organoids were cultured for 33 days prior to drug treatment. CX5461 (Selleck, S2684) was dissolved in DMSO to create a stock solution at 3 mM and then added to the GSCs medium to achieve a final concentration of 1 µM, with the concentration referenced from previous studies 28 . IFNβ (Sino Biological, 10704-HNAS) was prepared according to the manufacturer’s instructions at 0.25 mg/ml using sterile water and added to the medium at a final concentration of 100 ng/ml; The IFNβ concentration was based on previous studies 29 . The organoids were cultured in medium containing either CX5461 or CX5461 + IFNβ for 30 days, with a GSCs medium change every 2 days. CX5461 and IFNβ were replenished in the medium during each fluid change until the samples were collected.
Data acquisition and statistical analysis
Bright-field images were captured using inverted microscopes (Motic AE200 and Nexcope NIB900), while fluorescence images were acquired with an inverted fluorescence microscope (Nexcope NIB900). ImageJ software was used for scale calibration and cell and clone quantification.
Statistical analyses were conducted using GraphPad Prism 9. A two-tailed Student’s t-test was applied for comparisons between the two groups. A one-way analysis of variance (ANOVA) was used for multiple comparisons (followed by Brown-Forsythe and Welch ANOVA tests).
Result
Culturing and characterization of GSCs
First, we examined the cells used to construct the organoids. We cultured GSCs and observed that the cells were in a highly proliferative state with a typical spindle shape26,27 (Fig. 1a). The GSC identities were confirmed through immunofluorescence analysis, positive for GSC markers CD133, SOX2, and Nestin (Fig. 1b). More than 90% of the cells were Ki67+, indicating a high proliferative capacity of the GSCs (Fig. 1c). Together these results demonstrate successful maintenance of GSCs, preserving their key characteristics during continuous passaging.

Culture and characterization of GSCs. (a) GSCs light image of the first 3 days after passage. Scale bar = 30 μm. (b) Immunofluorescence staining was used to identify the markers CD133, SOX2, and Nestin for GSCs. Scale bar = 100 μm; the bar chart represents the positive rates of CD133, SOX2, and Nestin. Error bar: ± SEM. (c) Immunofluorescence staining of the proliferative marker Ki67, with the bar chart representing its positive rates. Scale bar = 100 μm, error bar: ± SEM.
The stiffness of the 2D microenvironment influences the growth of GSCs
Studies have demonstrated that matrix stiffness influences the diffusion and migration capabilities of GBM 30 . To investigate the effect of stiffness on GSCs, we first selected three different concentrations of HAMA, GelMA, and CSMA within their applicable ranges, as specified in the material guidelines, and measured their storage modulus (Fig. 2a). The results indicate a significant increase in storage modulus with rising concentrations for all three materials, confirming their tunable mechanical properties.

The stiffness of different materials influences the growth of GSCs. (a) The rheometer measures the storage modulus of HAMA (1%, 1.5%, 2%), GelMA (5%, 10%, 15%), and CSMA (1%, 1.5%, 2%) at varying concentrations. Error bar: ± SD, ****P < 0.0001. (b) GSCs were cultured on cell culture plates coated with Matrigel, 1% HAMA, 5% GelMA, or 1% CSMA. Four materials were all diluted 100-fold. Scale bar = 200 μm. (c) GSCs were cultured on cell culture plates coated with Matrigel, 1% HAMA, 5% GelMA, or 1% CSMA. Four materials were all diluted 33-fold. Scale bar = 200 μm.
Next, we selected a specific concentration for each hydrogel material—HAMA (1%), GelMA (5%), CSMA (1%)—along with Matrigel to investigate the impact of 2D matrix stiffness on GSCs behavior. Standard 2D GSC cultures typically utilize low laminin concentrations (~5 µg/ml). Therefore, we diluted each hydrogel 100-fold to coat the culture plates before seeding the GSCs.
On day 1, some cells in the Matrigel group adhered to the plate surface, whereas HAMA-seeded cells remained dispersed at the bottom, appearing rounded without attachment. In the GelMA group, while a few cells adhered, the majority remained suspended and scattered. Notably, cells in the CSMA group exhibited a distinct clustering pattern, with no adhesion to the plate. By day 3, cells in the Matrigel group continued to adhere and proliferate, whereas those in the HAMA and GelMA groups remained rounded and scattered at the bottom. In contrast, the CSMA group exhibited an increased tendency for cell aggregation (Fig. 2b).
The results above indicate that different materials influence GSCs growth. To further investigate the effect of substrate stiffness on GSCs proliferation, we increased the concentration of the materials. Each material was diluted to one-third of its original dilution (diluted 33-fold concentration) and left to coat the plates overnight before seeding GSCs. By day 3, a notable shift in growth patterns was observed: in response to the increased concentration, both the HAMA and GelMA groups exhibited a clear tendency toward cluster formation (Fig. 2c).
The experimental results demonstrate that GSC growth is influenced by the matrix’s composition and stiffness.
Matrigel mixed with HAMA (MH) induces GSCs in GBM to adopt a clonal state
Matrigel, a commonly used ECM scaffold for organoids, has been widely applied in constructing GBM models12,13. However, its softness does not replicate the stiffness of the glioma microenvironment, and it lacks the proteoglycan components essential for the brain environment 31 . Therefore, we aimed to optimize the culture conditions for GBM by integrating hydrogel materials with Matrigel. Specifically, we constructed three hybrid models: Matrigel mixed with CSMA (MC), Matrigel mixed with GelMA (MG), and Matrigel mixed with HAMA (MH). To explore which hydrogel composition is most conducive to GBM construction by altering stiffness, we prepared hydrogels and mixed them with Matrigel at different ratios (Fig. 3a). The concentrations of hydrogels were set within the recommended range to ensure their effectiveness. Considering that varying mixing ratios may produce different outcomes, we tested three Matrigel—hydrogel ratios—2:1, 1:1, and 1:2—and conducted storage modulus testing after mixing (Fig. 3b). Rheometer analysis revealed that the storage moduli were positively correlated with CSMA concentrations. As the proportion of CSMA increased in the MC model, the storage moduli rose. Notably, both the 1:1 and 1:2 ratios showed a significant increase compared to MC-1% 2:1 and the 1:2 ratio can increase the storage moduli even more. In contrast, the storage moduli were negatively correlated with the concentrations of GelMA. Compared to MG-5% 2:1, both the 1:1 and 1:2 ratios exhibited a significant decrease in storage moduli, which could be due to the uneven mixing of GelMA and Matrigel. For HAMA, the change in storage moduli did not follow a linear trend. Compared to that of MH-1% 2:1, the storage moduli of the 1:1 and 1:2 ratios significantly increased. Although the storage modulus of MH-1% 1:2 was lower than that of 1:1, the difference was not statistically significant. As a result, we picked the ratio of MC at 1:2 for all the following experiments. To minimize Matrigel’s influence and reduce the variability introduced by differences in Matrigel, we chose an MH model with a 1:2 ratio. For the MG model, none of the three ratios could be even mixed and hence we chose the ratio of 1:2 for the sake of consistency.

Construction and characterization of the Matrigel-hydrogel mixed model. (a) Flow chart for the construction of Matrigel-hydrogel mixed GBM organoids. (b) The storage modulus of various Matrigel-hydrogel mixtures and their corresponding mixing ratios were measured using a rheometer. Error bar: ± SEM, ***P < 0.001, ****P < 0.0001, ns = not significant (P > 0.05). (c) The morphology of Matrigel, MH, MG, and MC organoids at days 1, 7, 12, and 22 was observed by bright light microscopy. Scale bar = 500 μm. (d) Cell morphology was observed under high magnification in Matrigel, MH, MG, and MC models. Scale bar = 200 μm. (e) White light imaging of MH GBM models constructed using Matrigel mixed with 150 kDa, 300 kDa, and 400 kDa HAMA after 5, 13, and 19 days of culture. Scale bar = 500 μm or 200 μm. The chart illustrates the clone count across different molecular weights in 19 days. Error bar: ± SEM, ****P < 0.0001, ns = not significant (P > 0.05).
Next, we selected a Matrigel:hydrogel ratio of 1:2 to construct GBM organoids, with each 15 µL organoid containing 4000 GSCs. Over time (by day 7), the cells in Matrigel readily spread and proliferated, adopting a spindle-like morphology, and actively migrated toward each other, irregularly dispersed and interwoven throughout the Matrigel sphere. In contrast, the cells in MH and MC rarely spread and migrated and, as a result of active proliferation, formed spherical structures evenly dispersed within the matrix. The cells in MG did not form any uniform structures, presumably due to the uneven mixing of Matrigel and GelMA for their different temperature sensitivity (Matrigel solidified at a high temperature and GelMA at a low temperature). To distinguish the branching morphology of cells in GBM organoids cultured in Matrigel, we designated the spherical structures as “clones.” The formation of spherical clones recapitulated in vivo tumor growth and structural organization (Fig. 3c). With time, the clones in the MH group grew larger than those in the MC groups (Fig. 3d). Taken together, these results demonstrated that incorporating hydrogel can modify Matrigel, enabling GSCs to form uniform spherical clones. Among them, MH model enabled continuous proliferation and clonal expansion over long term.
To further optimize the MH model, we decided to compare HAMA of three different molecular weights (150 kDa, 300 kDa, and 400 kDa). In Matrigel mixed with HAMA of different molecular weights, the cells with the 150 kDa and 400 kDa groups clustered mainly in the center, unevenly distributed within the organoids. In contrast to the 300 kDa group with evenly distributed spherical clones (Fig. 3e), not all structures were well-formed spherical clones with the appearance of filamentous cells in the 400 kDa MH organoids. As a result, there were significantly fewer clones in the 400 kDa group than in the 300 kDa group (Fig. 3e).
GelMA-HAMA (GH) 1:2 mixture forms uniform spherical clones
Matrigel, as a complex animal-derived matrix, has considerable batch-to-batch variability with relative high cost 19 . Since the addition of HAMA can modify Matrigel to support the derivation of spherical 3D clones from GSCs, we decided to test the mixing of HAMA with hydrogels. We mixed CSMA with HAMA at ratios of 1:0 (pure CSMA), 2:1, 1:1, 1:2, and 0:1 (pure HAMA). Cell proliferation and clone formation were not observed in any group except for the HAMA-only group, which successfully formed sparse GSC clones (Fig. 4a). We then tested GelMA mixed with HAMA in the following ratios: GelMA:HAMA = 1:0 (pure GelMA), 2:1, 1:1, 1:2, and 0:1 (pure HAMA). GelMA alone did support the proliferation of GSCs that exhibited a filamentous structure and failed to form spherical clones. Over time, the filamentous structures elongated and gradually filled the entire organoids by day 30, suggesting that GelMA, similar to Matrigel, supports cell spreading, migration, and proliferation (Fig. 4b). Intriguingly, as the proportion of GelMA decreased, the filamentous cells gradually decreased and the spherical clones became more prevalent. At 1:2 ratio, homogeneous, large, and well-defined spherical clones were formed with clear edges and stable structures (Fig. 4c). To further investigate the effect of the GelMA-HAMA mixture on the properties of both materials, we measured the swelling rate. The results showed that the swelling rate of pure GelMA was high, while that of pure HAMA’s was low. Mixing the two at three different ratios significantly balanced the swelling rates, with no significant difference observed between the ratios 1:1, 1:2, and 2:1 (Fig. 4d). Based on these findings, we conclude that the GH 1:2 model is better suited for GBM organoid constructions, facilitating the formation of homogeneous spherical clones.
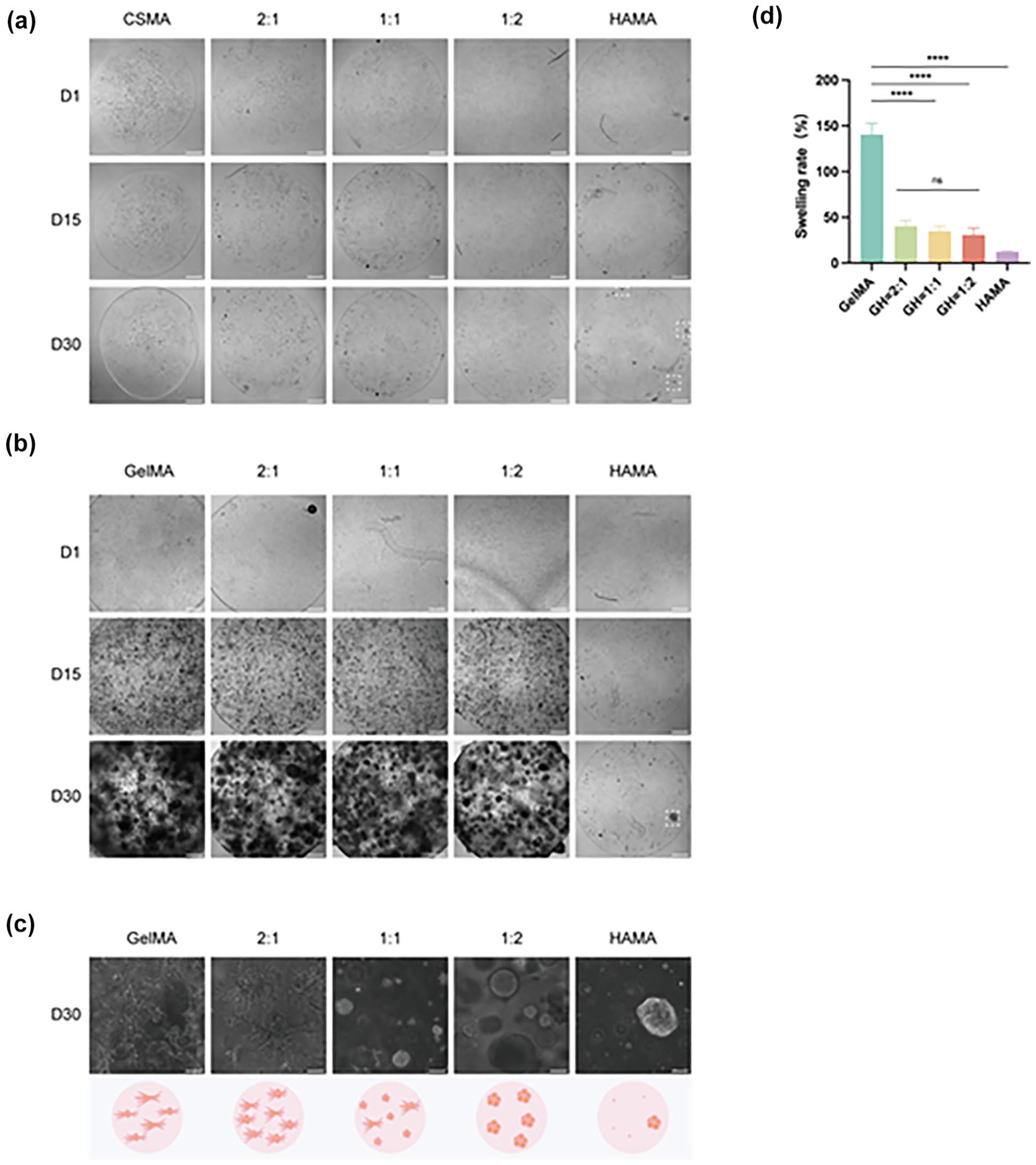
Construction of a GBM organoid using a mixed hydrogel model. (a) Bright-field images of pure CSMA, pure HAMA, and CSMA-HAMA mixtures at different proportions on days 1, 15, and 30. The dashed white box highlights the sparse clones formed in the HAMA group. Scale bar = 500 μm. (b) Bright-field images of pure GelMA, pure HAMA, and GelMA-HAMA mixtures at different proportions on days 1, 15, and 30. The dashed white box highlights the sparse clones formed in the HAMA group. Scale bar = 500 μm. (c) Cell morphology observed under high magnification in pure GelMA, pure HAMA, and GelMA-HAMA mixtures. Scale bar = 100 μm. (d) Swelling rate test results of pure GelMA, pure HAMA, and GelMA-HAMA mixtures. Error bar: ± SEM, ****P < 0.0001, ns = not significant (P > 0.05).
GelMA-HAMA (GH) 1:2 mixture GBM model exhibits tighter spherical clones
Next, we sought to compare the MH 1:2 and GH 1:2 models side by side. As shown by the bright-field images, clones formed in the GH group were larger, rounder, and exhibited smoother edges than those in the MH group (Fig. 5a). Immunofluorescence identified GSC markers, and both the MH and GH models showed positive expression of CD133, SOX2, and Nestin, confirming that both models effectively supported GSC viability (Fig. 5b–d). Interestingly, we observed that the inner cells of MH 1:2 clones were less compact and more dispersed than those of the GH 1:2 clones (Fig. 5b). We then evaluated the effects of the two models on the proliferative properties of GSCs. Ki67 staining revealed high expression of Ki67 in the clones of both models. Notably, Ki67 expression varied among different clones within the same organoid, indicating proliferative heterogeneity (Fig. 5b). This observation is consistent with the intratumoral heterogeneity seen in primary glioblastoma, further validating that the organoid model could better mimic the biological characteristics of primary glioblastoma. Together these results suggest that while both MH 1:2 and GH 1:2 models maintain the characteristics of GSCs and recapitulate tumor proliferative heterogeneity, the GH 1:2 GBM model generates more uniform spherical clones.

Comparison of MH GBM organoids with GH GBM organoids. (a) Bright-field images of D30 MH and GH. Scale bar = 100 μm. (b) GBM marker CD133 and proliferation marker Ki67 were detected using immunofluorescence staining. Scale bar = 100 μm. (c) GBM marker Nestin was detected using immunofluorescence staining. Scale bar = 100 μm. (d) GBM marker SOX2 was detected using immunofluorescence staining. Scale bar = 100 μm.
GelMA-HAMA (GH) GBM model for drug screening
To assess the potential of the GH model for drug screening, we treated the organoids with drugs. Recent studies have shown that CX5461, a G-quadruplex stabilizer, can effectively target GBM 28 . IFNβ, an immunomodulatory factor, has been demonstrated to target the tumor microenvironment, thereby reducing tumor resistance to therapy 32 . Based on these findings, we hypothesized that combining CX5461 and IFNβ would have a more potent anti-GBM effect. Thus, we tested two treatment regimens: CX5461 monotherapy and CX5461+IFNβ. Drugs were given to GH model organoids cultured for 33 days, during which numerous spherical clones formed, allowing for direct observation (Fig. 6a). During the first 3 days of treatment, no significant difference was observed between the two regimens. However, after 13 days of treatment, the combined treatment group showed a marked reduction in the size of spherical clones (Fig. 6b). By day 30, the combined treatment group exhibited a significant decrease in the number of clones while the monotherapy group showed little change (Fig. 6b). To further compare the effects of the two treatments, we assessed Ki67 and Cleaved Caspase-3 (C-Cas3), a marker of apoptosis. Ki67 expression remained high in the CX5461 monotherapy group, with minimal C-Cas3 expression. In contrast, the combination treatment group showed a significant decrease in Ki67-positive cells and increase in C-Cas3 positive cells, indicating reduced GSC proliferation and the initiation of apoptosis (Fig. 6c, d). These analyses testify that the GH model can be utilized for GBM drug testing and screening.

Drug cytotoxicity assay in GH GBM organoids. (a) Process flow of CX5461 or CX5461 + IFNβ treatment. (b) Long-term bright-field observation of GH GBM organoids under drug treatment. Scale bar = 500 μm. (c) Ki67 and cleaved C-Cas3 expression in GH GBM organoids were detected by immunofluorescence. Scale bar = 100 μm. (d) Statistical analysis of the expression of Ki67 and cleaved Caspase-3 (C-Cas3) was conducted. Error bar: ± SEM, **P < 0.01, ****P < 0.001.
Discussion
With the advancement of organoid technology, researchers have increasingly sought to improve GBM organoids for refined disease modeling19,33. Patient tumor-derived organoids (PDOs) offer a more physiologically relevant microenvironment than traditional 2D cell culture systems, or a more human cancer relevance than animal models. Modeling with PDOs is particularly important for GBM because of its high intratumor heterogeneity and complex tumor microenvironment. Although it remains a widely utilized matrix-based scaffolds used for GBM organoid construction, Matrigel, as an animal-derived matrix, has a number of major caveats such as cost and batch and batch consistency, significantly limiting its utilities 19 .
In this study, we explored three hydrogel materials—GelMA, HAMA, and CSMA—that have been extensively investigated in the field of regenerative medicine. We initially combined these hydrogels with Matrigel to study whether the incorporation of hydrogels could modify the properties of Matrigel, making it more suitable for GBM organoid construction. Rheological tests confirmed that adding hydrogels did indeed alter the characteristics of Matrigel. Our findings revealed that Matrigel and GelMA could not be fully mixed, while the proliferation rate of cells in the MC group was slow during long-term culture. The MH group manifested a proliferation rate higher than the MC one but still lower than the Matrigel-based organoids. Nonetheless, GSCs in the MH group formed spherical clones, in contrast to the filamentous cell structure observed in the Matrigel model.
To develop more cost-effective matrix scaffold with high reproducibility, the above results promoted us to further explore new scaffolding strategies by mixing CSMA and GelMA with HAMA in varying proportions. However, combining CSMA and HAMA did not produce satisfactory results and the proliferation of GSCs was rather limited across all tested ratios. In contrast, the GelMA mixed well with HAMA and supported rapid cell proliferation across different mixing ratios. In particular, at a 1:2 ratio, the GH scaffold supported formation of well-defined, spherical clones characterized by robust proliferation, fewer filamentous cells, and larger spherical structures than other models. Swelling rate tests suggested that combining GelMA-HAMA altered their biophysical properties and presumably render their mixture suitable for GBM organoid construction. Further immunofluorescence staining revealed that while both MH and GH 1:2 models maintain the characteristics of GSCs and recapitulate tumor proliferative heterogeneity, the GH GBM model generates more uniform spherical clones. The composition of GH is well defined, free of animal-derived Matrigel, with improved experimental stability and reproducibility. The use of GelMA also significantly reduces experimental costs relative to that of Matrigel. GelMA promotes the survival and proliferation of glioblastoma cell lines and has been widely applied in the field of neuromedicine34,35. HA, the most abundant extracellular matrix component in brain tissue, has also been shown to facilitate GBM invasion 36 . When combined, the GH composite may represent a more suitable matrix for GBM organoid construction. Notably, we observed distinct expression patterns of Ki67 and marker proteins among different clones within the same organoids, strongly suggesting underlying clonal heterogeneity. This heterogeneity has been reported in other GBM organoid models 37 , further supporting the potential of our model to capture intratumoral heterogeneity.
To assess the potential of the GH model for drug screening in glioblastoma, we selected two drug combinations—CX5461 and CX5461 + IFNβ—based on existing literature. As expected, both treatment strategies exerted proliferation-inhibitory and cell death-enhancing effects. Compared to CX5461 alone, the combinatorial treatment exhibited enhanced cytotoxicity with significant reduction in proliferation and simultaneous increase in cell death. Taken together, these results confirm that the GH model is a viable model for glioblastoma drug testing and screening.
In summary, the present study introduces a novel cost-effective and reproducible glioblastoma organoid construction strategy based on the proper composition of GelMA and HAMA. This new system recapitulates key GBM characteristics and could be effectively applied to GBM modeling and drug screening. Nonetheless, this model’s comprehensive verification and practical application requires further experimental characterization and refinement. Expanding the study to include more patient-derived GSCs is both highly desirable and essential to confirm the model’s ability to capture intertumoral heterogeneity. Also, the exploration of hydrogel materials remains limited and further screening and evaluation of additional candidate materials are important.
Footnotes
Ethical considerations
This study was approved by our institutional review board.
Author contributions
ZG, JG, and HW conceived and designed the experiments; KY and MZ performed the experiments; KY, MZ, and JZ analyzed the data; ZL, MZ, and KY wrote the manuscript; SW, SG, YX, and WW edited the manuscript; ZG, JG, and HW reviewed and revised the manuscript; JG, HW, and ZG provided financial support. All authors approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Shanghai Pudong New Area Health System Discipline Leader Training Program (PWRd2023-05) and Scientific research project of Shanghai Municipal Health Commission (202140403) to Huan Wang; Key Disciplines Group Construction Project of Pudong Health Bureau of Shanghai (PWZxq 2022-05) to Jianrong Guo; and National Natural Science Foundation of China (No. 32370895 and No. 32070862) to Zhengliang Gao.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of human and animal rights
The GSCs used in this study were obtained from the laboratory cell bank. For further details, refer to the laboratory’s previous publications: PMID: 35501312, PMID: 34295351.
Statement of informed consent
There are no human subjects in this article, and informed consent is not applicable.
Data availability statement
All relevant data are within the manuscript, and the original data are available from the corresponding author on reasonable request.

