Abstract
Diabetic foot ulcers (DFUs) are associated with a high risk of amputations and a 50% 5-year survival rate due at least in part to the limited angiogenic and wound healing capacity of patients with diabetes. Cell therapy via intramuscular injection of peripheral blood mononuclear cells showed encouraging but limited results. Such limitations may arise from the limited ability of therapeutic cells to adhere to the target tissue. The development of a methodology able to support the targeted delivery of viable angiogenic cells would improve cell therapy outcomes in DFU. Here, we optimized a protocol for the production of autologous extracellular matrix (ECM)-rich pericyte-based cell sheets for cell delivery. Pericytes were isolated from skeletal muscle biopsies of DFU patients and non-diabetic controls and characterized by flow cytometry and immunofluorescence. Human umbilical vein endothelial cells used for the optimization of collagen IV deposition showed a positive correlation with seeding density and a negative one with sub-culture passaging (P < 0.05). Macromolecular crowding significantly increased collagen IV deposition both in human umbilical vein endothelial cells and in patient-derived pericytes (P < 0.01) without affecting proliferation (P > 0.05). Finally, DFU patient-derived pericytes effectively deposited ECM supporting their use for autologous cell sheet production.

Introduction
Wound healing and angiogenesis are impaired in diabetic patients. Wound healing is composed of four phases in which endothelial cells (ECs) and pericytes play a central role: homeostasis, inflammation, proliferation, and remodeling1,2. Following vessel rupture, resident cells react to the reduced oxygen tension by synthesizing pro-angiogenic factors (e.g., vascular endothelial growth factor-A, VEGF-A), which in turn stimulate ECs proliferation and migration 3 . The hypoxic environment recruits leukocytes to the site of injury, with neutrophils being the first responders, followed by macrophages in the acute phase of inflammation 4 . At the end of the inflammatory phase, characterized by macrophage invasion, a disorganized network of newly formed capillaries is produced by the VEGF-driven proliferation of ECs 5 . The restored oxygen tension drives a switch from pro- to anti-angiogenic signals (e.g., Ang-2), which resolves in apoptosis of ECs in excess capillaries 6 . During this remodeling process, pericytes migrate after platelet-derived growth factor-β (PDGF-β) gradients toward the well-formed capillaries and prevent neighbor ECs from undergoing apoptosis 7 . As previously shown by our group and others, diabetes is associated with several dysfunctions affecting angiogenic potential 8 . Such impairments include an impaired amount of functionally compromised circulating pro-angiogenic CD34+ endothelial progenitor cells, a decreased CD34+ homing capacity at the wound site, a loss in pericytes number and function, and a misregulated production of angiogenic factors, contributing to peripheral artery disease and critical limb threatening ischemia (CLTI), which sets the basis for the development of diabetic foot ulcers (DFUs)9–12. Current therapies failed to restore the angiogenic potential in diabetic patients; however, most recent approaches relying on cell delivery in situ with or without the support of a scaffold show promise 13 . Autologous CD34+ cell delivery, a matter of study in our hosting institute, showed positive results in the treatment of CLTI 14 . Therefore, the development of a biological, xenogenic-free platform for the delivery of autologous angiogenic cells is a pivotal step in the development of a strategy for the treatment of CLTI. A recent approach to tissue engineering is cell sheet technology (CST), which exploits the innate ability of cells to produce their own extracellular matrix (ECM), relying on thermoresponsive polymers for the growth and collection of the cell sheets 15 . CST has been applied so far to the regeneration of several tissues, as well as to wound healing16,17. However, the optimal cell type and cell culture conditions to produce an efficient and autologous cell sheet to stimulate angiogenesis in diabetic patients still need to be elucidated. Pericytes play a pivotal role in supporting angiogenesis in vivo, orchestrating the activation and migration of ECs and endothelial progenitor cells, besides retaining elevated plasticity and viability in vitro. Moreover, pericytes co-operate with ECs in basal lamina deposition in vivo, are easier than ECs to isolate and expand in vitro, and have mesenchymal origin which confers them a lower immunogenicity 18 . Therefore, we hypothesized that pericytes would represent the cell type of choice as a functional support for endothelial progenitor cells. A central player in the efficiency of a cell sheet is its ECM, which also plays a critical role in angiogenesis, as it provides fundamental mechanical and biochemical cues for the neo-vessels to sprout 19 : Macromolecular crowding (MMC) is emerging as a tool to sustain and enhance the innate capability of cells to deposit their own ECM, therefore providing a unique approach to produce autologous, self-assembled constructs for tissue engineering applications20,21. MMC aims to improve cell culture conditions by reducing the gap between in vivo crowded and in vitro diluted environments21–23. Its mechanism of action relies on the excluded volume effect (EVE), namely the reduction in the medium volume through which the cell-secreted molecules are free to diffuse 24 . Coupled with the constant formation and disaggregation of transient, delimitated micro-environments in the conditioned medium, EVE increases the reaction rate of the proteases involved in the processing of fibrillar procollagens (e.g., BMP-1, ADAMTS-2, MMPs) by increasing their effective concentrations and favoring the association with their substrates. Such general characteristics are influenced by the nature of the molecule chosen as a crowder. Negatively charged molecules such as carrageenans or dextran sulfate are more effective than neutral crowders like ficoll or polyvinylpyrrolidone (PVP) at lower concentrations on account of their larger hydrodynamic radius25,26. Polydispersity index, generally larger in biological molecules such as hyaluronic acid (HA) or carrageenan, has also been shown to positively affect ECM deposition 27 . By combining the innate ability of cells to deposit their matrix with the activity of MMC and the innovation of CST, we aim to produce an efficient and xenogeneic-free scaffold for the treatment of chronic DFU.
Based on these concepts, we hence hypothesized that the application of an optimized MMC preparation to the culture of patient-derived pericytes will lead to the increased deposition of an ECM capable of homing angiogenic cells, offering an important implementation to the autologous cell delivery–based approach in the treatment of CLTI. To select the most efficient combination of MMC to enhance the deposition of the peculiar ECM of pericytes, several molecules known to have crowding effects were tested. Finally, in view of a future clinical application on DFU patients, the ECM deposited by diabetic patient–derived pericytes was compared to the ECM deposited by non-diabetic counterparts in terms of expression and quantification of typical basal membrane molecules.
Materials and Methods
Donor Patients
After approval by the ethics committee of MultiMedica (protocol number 452.2020, approval report no. CE-119.2020 of December 16, 2020), and in conformity with the Declaration of Helsinki, 27 patients (mean age 68.23 ± 12.61 years, 27.27% females) attending the diabetic foot unit and 28 patients (mean age 45.68 ± 13.69 years, 53.57% females) from the hand surgery and rehabilitation units at the MultiMedica hospital in Sesto San Giovanni (Milan) were enrolled upon signing of the informed consent. From each patient, a skeletal muscle biopsy was sent to the research laboratory from which pericytes were isolated.
Isolation of Vascular Cells From Muscle Tissue
Muscle tissue was weighed and then minced with the help of a scalpel before being incubated with a cocktail of bacterial collagenase and dispase (Merck, #11097113001) at a concentration of 1 mg/ml for 2 h at 37°C with shaking. The resulting digested tissue was then filtered with a cell strainer with a pore size of 70 μm and washed in phosphate-buffered saline (PBS). Cells contained in the pellet were selected with anti-CD31 magnetic beads (Miltenyi, #130-091-935), to eliminate ECs. CD31− cells were further selected with anti-CD146 magnetic beads (Miltenyi, #130-093-596). The resulting CD31−CD146+ cells were seeded as pericyte candidates. Cells were grown with a culture medium for vascular cells (ATCC, #PCS-100-030) supplemented (ATCC, #PCS-100-041) with VEGF, epidermal growth factor (EGF), insulin-like growth factor-1 (IGF-1), basic fibroblasts growth factor (bFGF), heparan sulfate,
Characterization of the Isolated Cell Populations by Immunofluorescence Assay
Upon reaching the right level of confluence (70%–80%), the cell cultures specifically grown in chamber slides for immunofluorescence were washed with PBS and fixed by incubating for 20 min in 4% paraformaldehyde (Sigma, #P6148) in PBS. After three washes in PBS, they were permeabilized with a 0.1% Triton X-100 (Sigma, #T8787) solution in PBS for 10 min, washed three times in PBS, incubated with 10% goat serum (abcam, # ab7481) solution for 30 min to cover the non-specific binding sites for the antibodies, and subsequently incubated with the immunofluorescence antibodies listed in Table 1 at the relevant dilutions. After O/N incubation, cells were washed three times in PBS and incubated for 45 min with secondary antibodies (Thermo Fisher #A-21428, #A-11001) diluted 1:500 in PBS. At the end of the incubation with secondary antibodies, cells were washed three times in PBS and the chambers were disassembled and coverslipped with Vectashield mountant with DAPI (Vector laboratories, # H-1200-10) and examined with an inverted fluorescence microscope (Leica, DM IL Fluo).
Antibodies Used for Immunofluorescence and FACS Characterization of Pericytes.

Characterization of the Isolated Cells by Flow Cytometry
Magnetic bead-sorted cells were seeded in culture-treated dishes and grown until reaching approximately 80% confluence before characterization. Cells were then harvested by incubation with trypsin-EDTA (Sigma, #T2601) at 37°C, washed in PBS, and incubated with the fluorescence activated cell sorting (FACS) antibodies listed in Table 1 for 10 min at 4–8°C. Visualization and recording were then performed with the BD LSRFortessa™ X-20 Cell Analyzer using at least 150,000 cells per sample and comparing the samples to the antibody-free negative control. The gating strategy of Fig. 1 shows the selection of a morphologically defined cell population free of doublets.

Pericytes characterization by FACS and immunofluorescence. Pericytes isolated from human skeletal muscle biopsies by CD146+ magnetic beads sorting express typical pericyte/mesenchymal markers by immunofluorescence (A) and FACS (B) characterization; (C) comparison of pericyte markers expression between control and diabetic patients. Ns: nonstatistically significant, *P < 0.05, **P < 0.01, ***P < 0.001, N = 10 controls, N = 9 diabetic patients.
Treatment With Macromolecular Crowders
Cells were seeded on day 0 of the experiment between passages 2 and 4 (pericytes) or 4 and 8 for human umbilical vein endothelial cells (HUVECs) at a density of 20,000 per cm2 in chamber slides (Thermo Fischer, # 16250681) with medium containing 2% (HUVECs) or 10% (HUVECs, pericytes) FBS. On day 1, the medium was changed to medium containing the MMC (see Table 2 for a complete list of crowders and concentrations tested). For optimization experiments with HUVECs, time points were set on days 3 and 6. Based on observations made on HUVECs, day 3 was identified as the optimal time point for the following experiments.
Descriptive of the Macromolecular Crowders Tested.

Evaluation of the Expression of Individual Matrix Components
Pericytes were seeded at a density of 20,000 cells/cm2 in 8-well chamber slides with complete vascular cell culture medium with supplements, P/S, and 10% FBS. The following day (day 1) the medium was replaced with a control medium without crowders (in controls) or a medium containing 25 mg/ml ficoll 400 kDa and 37.5 mg/ml ficoll 70 kDa as MMC mix (in treated samples).
Statistical Analysis
Variables are summarized by frequency and percentage or mean and standard error of means for categorical and continuous variables, respectively. Differences between the groups were tested by Student t-test or non-parametric Wilcoxon’s test. Statistical significance was defined as *P < 0.05, **P < 0.01, and ***P < 0.001. No statistical significance was defined as P ≥ 0.05. Statistical analysis was conducted using GraphPad Prism 9 software (Prism 9.0.0).
Results
Isolation and Characterization of Pericytes
In order to evaluate the matrix deposition by human pericytes, we set up a protocol for isolating these cells from muscle tissue samples of diabetic patients affected by CLTI and non-diabetic patients by sorting the total cells with magnetic beads recognizing CD146, a typical antigen expressed by pericytes. Pericytes were then characterized by FACS and immunofluorescence using the specific antibodies listed in Table 1. Flow cytometric analysis revealed that pericytes expressed on average 59.36% ± 30.01% CD146, 85.88% ± 16.71% CD90, and finally 66.44% ± 31.52% CD140b, typical markers of the pericyte phenotype, while they showed as expected a lower expression of antigens characteristic of ECs (0.48% ± 0.72% CD31, 33.32% ± 25.76% CD34) or leukocytes (5.06% ± 12.61% CD45). Figure 1 shows the morphological gating strategy and a representative image of the results of the flow cytometric analyses, as well as the characterization by immunofluorescence, which confirms the positivity for the pericyte markers CD140b, NG2, and αSMA.
Seeding Density and Sub-Culture Passaging Affect Collagen Type IV Deposition by HUVECs
While optimizing the protocol for pericyte isolation, to fine-tune the ideal MMC condition for increasing collagen IV deposition, HUVECs were used. ECs were chosen as they secrete collagen IV as a matrix component, similar to pericytes. To characterize our study model, we first verified the effect of the number of sub-culture passages on the intrinsic capacity of HUVECs to deposit ECM, observing that this is an influential variable: both in the presence of 2% and 10% fetal bovine serum, collagen IV depositions, measured as % of the area covered by collagen IV/number of cells, decreases significantly at passage 8 compared to passage 4 (0.071 ± 0.03 vs 0.012 ± 0.009 with 2% serum and 0.059 ± 0.01 vs 0.015 ± 0.004 with 10% serum) (Fig. 2A, B). Second, we evaluated the seeding density and serum concentration in order to maximize collagen IV deposition: in terms of absolute values, the seeding density of 20,000 cells per cm2 proved to be significantly more efficient than the others in both serum concentrations (41.96 ± 8.87 vs 9.93 ± 5.31 at 5000 cells/cm2) (Fig. 2D, E), while no significant differences were found in the use of 2% or 10% fetal bovine serum in HUVEC (P = 0.0528, Fig. 2C). For these reasons, in the following experiments, we used cells at a passage in culture not exceeding 4 and seeded at a density not lower than 15,000/cm2.

Effect of culture conditions on collagen IV deposition by HUVECs. Subculture passaging effect on collagen deposition by HUVECs with 10% (A) and 2% (B) FBS supplementation; effect of serum concentration (C) and cell seeding density in the presence of 10% (D) and 2% (E) FBS on collagen deposition. Ns: nonstatistically significant, *P < 0.05, **P < 0.01, N = 3.
Macromolecular Crowding Enhances Collagen Type IV Deposition by HUVECs
Having set the basic culture conditions for collagen IV synthesis by HUVECs, we ventured into assessing the effect of several MMCs on HUVECs in terms of collagen type IV deposition. When collagen IV positive area % was normalized on cell number, carrageenan (day 3, 90 μg/ml, P < 0.05), dextran-40 kDa (day 3, 200 mg/ml, P < 0.01), and PVP-40 kDa (day 3, 100 mg/ml, P < 0.01) significantly increased collagen type IV deposition compared to the untreated controls in the presence of 2% FBS supplementation (Fig. 3). In the presence of 10% FBS supplementation, ficoll mix (day 6, P < 0.05), dextran-40 kDa (day 3, 200 mg/ml, P < 0.01), and PVP-40 kDa (day 3, 200 mg/ml, P < 0.05) significantly enhanced collagen IV deposition. However, dextran-40 kDa (day 3, 200 mg/ml, P < 0.01) and PVP-360 kDa (2% FBS, day 3, 200 mg/ml, P < 0.05 and 10% FBS, day 6, 200 mg/ml, P < 0.01) significantly affected cell proliferation, PVP-40 also reduced cell density though not significantly, while carrageenan (2% FBS, 90 μg/ml, P < 0.05) and ficoll did not affect cell density in the presence of 10% FBS and were therefore selected to be tested on primary pericytes (Fig. 4). Figure 5 shows that only carrageenan (2% FBS, day 3, 90 μg/ml and 10% FBS, day 6, 30 μg/ml) and ficoll (day 6, 10% FBS) medium supplementation significantly increase collagen type IV deposition independently of cell density.

Effect of macromolecular crowding on collagen IV deposition by HUVECs normalized on cell density. Mean collagen IV covered area % normalized on HUVECs density after treatment with ficoll mixture (A, F), k-carrageenan (B, G), dextran-40 kDa (C, H), polyvinylpyrrolidone-40 (D, I) and polyvinylpyrrolidone-360 kDa (E, J) in the presence of 2% (A-E) or 10% (F-J) FBS. Fc: ficoll; kCR: k-carrageenan; Dx: dextran; PVP: polyvinylpyrrolidone; Ns: nonstatistically significant; *P < 0.05, **P < 0.01, N = 3.

Effect of macromolecular crowding on HUVECs cell density. Mean cell number per field after HUVECs treatment with ficoll mixture (A, F), k-carrageenan (B, G), dextran-40 kDa (C, H), polyvinylpyrrolidone-40 (D, I) and polyvinylpyrrolidone-360 kDa (E, J) in the presence of 2% (A-E) or 10% (F-J) FBS. Fc: ficoll; kCR: k-carrageenan; Dx: dextran; PVP: polyvinylpyrrolidone; Ns: nonstatistically significant; *P < 0.05, **P < 0.01, N = 3.
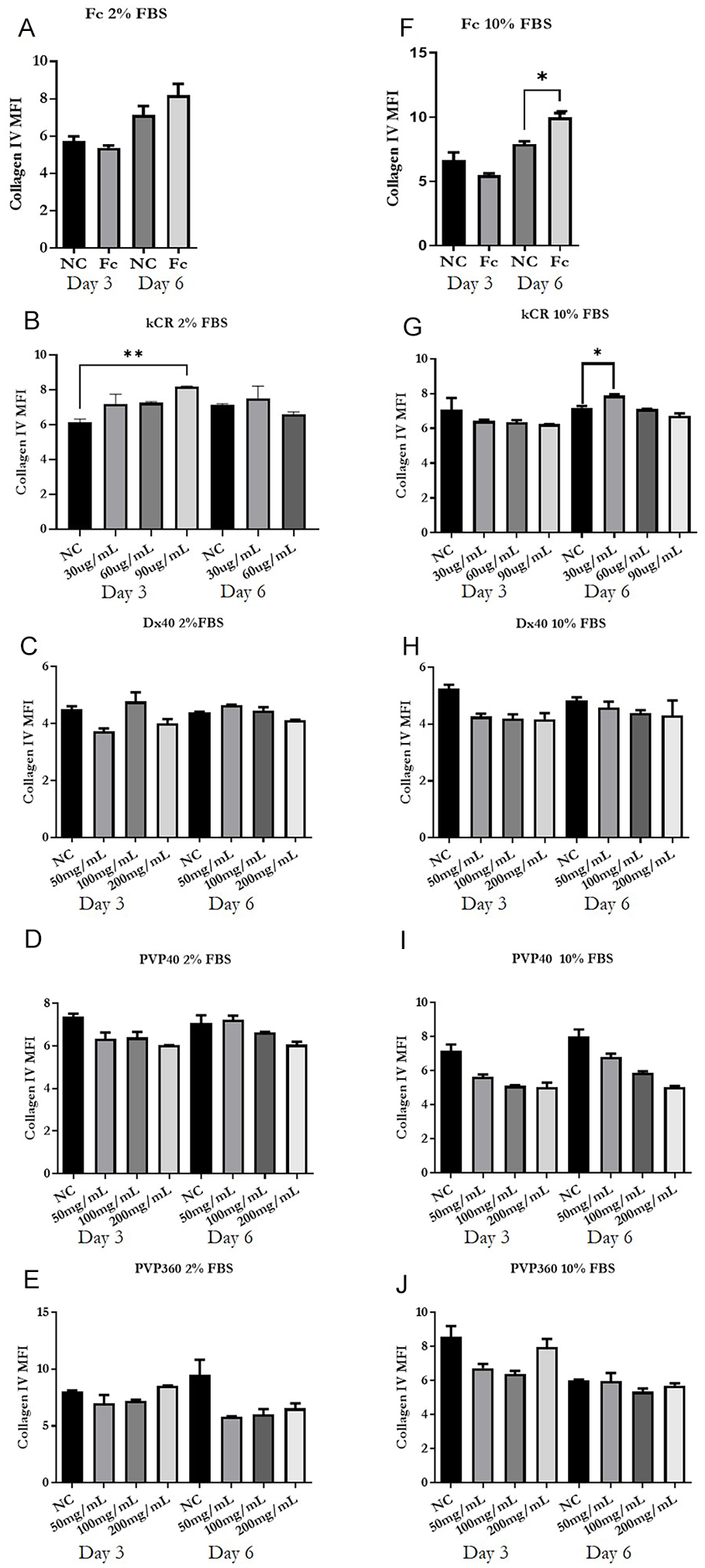
Effect of macromolecular crowding on collagen IV deposition by HUVECs. Mean collagen IV covered area % after treatment of HUVECs with ficoll mixture (A, F), k-carrageenan (B, G), dextran-40 kDa (C, H), polyvinylpyrrolidone-40 (D, I), and polyvinylpyrrolidone-360 kDa (E, J) in the presence of 2% (A-E) or 10% (F-J) FBS. Fc: ficoll; kCR: k-carrageenan; Dx: dextran; PVP: polyvinylpyrrolidone; Ns: nonstatistically significant; *P < 0.05, **P < 0.01, N = 3.
Macromolecular Crowding Enhances Collagen Type IV Deposition by Patient-Derived Pericytes
Following the observations made on HUVECs and consequently to the optimization of the protocol for the isolation of pericytes, we proceeded to apply the pre-selected MMCs on pericytes isolated from the skeletal muscle of non-diabetic patients. Ficoll proved to be effective on pericytes in enhancing ECM deposition, unlike carrageenan. Densitometric analysis data shown in Fig. 6 demonstrate a significant increase in collagen IV deposition following the addition of the MMC ficoll (0.08 ± 0.05 μm2/cell with ficoll vs 0.01 ± 0.01 μm2/cell in the negative control, P < 0.05), regardless of cell density (Fig. 6).

Ficoll treatment induced an increase in collagen IV deposition by pericytes. Ficoll treatment significantly increased collagen IV deposition by muscle pericytes (A, B) without significantly altering cell proliferation (C). “*” indicates a P < 0.05 for the mean percentage of collagen IV positive area versus negative control, N = 5.
Characterization of the Composition of the Extracellular Matrix Deposited by Pericytes
After assessing that MMC enhances collagen type IV deposition by patient-derived pericytes, we compared diabetic and non-diabetic patient–derived pericytes on their ability to deposit an ECM complete with all the components of a typical basal lamina: collagen type IV, laminin, nidogen, perlecan, and fibronectin. In the presence of MMC, diabetic patient–derived pericytes showed higher levels of both fibronectin (% of covered area/cell density = 18.07 ± 0.005 in the non-diabetic and 60.903 ± 0.017 in the diabetic, P < 0.05) and laminin (0.35 ± 0.092 in the non-diabetic and 10.433 ± 3.327 in the diabetic, P < 0.01) (Fig. 7). No statistically significant difference was observed between pericytes isolated from diabetic and non-diabetic patients with regard to the deposition of collagen IV, perlecan, and nidogen in the presence of MMC, although for all the molecules examined a tendency toward an increase was observed in favor of pericytes from diabetic donors. The deposition of nidogen in the absence of MMC was significantly increased in pericytes from diabetic patients compared to controls (P < 0.05) (Fig. 7).

Composition of the matrix deposited by pericytes from diabetic and non-diabetic patients in response to ficoll treatment (Fc). Representative images and graphs showing the mean percentage + SEM of positive area for laminin 5, fibronectin, nidogen, perlecan, and collagen IV in pericyte cultures from patients with or without diabetes and in the presence or absence of ficoll treatment. In the graphs, the percentage of positive area is normalized on the number of nuclei multiplied *1,000 per field. “*” and “**” represent P < 0.05 and P < 0.01, respectively, compared to the non-diabetic control. ND = non-diabetic; D = diabetic; CN = negative control without MMCs.
Discussion
The importance of pericytes as a cell type of choice in order to support ECs and endothelial progenitor cells in their angiogenic function is being backed by a growing number of studies 28 . In mice, the implantation of fibronectin/collagen I-based scaffolds seeded with HUVECs and MSCs, which are able to differentiate toward the pericytic fate, lead to the formation of a functional vascular network 29 . In bone tissue engineering, integration of an inner angiogenic layer seeded with pericytes and ECs between two nanofiber-based osteoinductive layers allowed for the subcutaneous integration of a bone-like structure in mice which was integrated with the host vascular network 30 . Finally, pericytes were applied in combination with ECs and MSCs to the generation of cell sheets–based bone constructs, corroborating the importance of using pericytes to achieve the formation of stable vascular networks. Our work so far has led us to lay the foundations for the generation of pro-angiogenic cell sheets from DFU patient-derived pericytes. Exploratory studies on HUVEC have allowed us to select the most effective MMCs to increase collagen IV deposition, namely ficoll and carrageenan, as well as to highlight the importance of some parameters such as the number of passages in culture and the seeding density to produce a confluent sheet capable of producing a homogeneous matrix. The data collected on pericytes through FACS and immunofluorescence characterization confirm the setup of a protocol to efficiently isolate and culture in vitro human skeletal muscle cells as supported by the high expression of the pericyte markers CD146, NG2, CD90, and CD140b in the populations sorted with microbeads for CD146. The analysis of the deposited matrix shows a significant effect of the MMC via ficoll on the deposition of type IV collagen: this would allow the production of a sheet of cells rich in ECM ready to home angiogenic cells to be administered to the patient a few days after the biopsy from which autologous pericytes are isolated. One limit of this study is represented by the preliminary nature of the results with respect to clinical translation. First applying the new pericyte cell sheet will be possible only after testing the capability of the cell sheet to support seeded cell function in terms of adhesion and viability. In addition, the support will need to undergo all the required testing for approval to be use in humans via the appropriate in vitro and in vivo steps. More proof-of-concept studies will be needed in the immediate future: therefore, we also plan to evaluate the pro-angiogenic effect of the cell sheets using 2D and 3D in vitro approaches as well as in vivo in appropriate animal models of DFU, and to determine the ideal administration route (injection or deposition). In doing so, also the role and added value of both the cellular and ECM components will need to be evaluated. It is important to note that the comparison with matrices deposited by pericytes of non-diabetic patients shows that also the matrices from pericytes of diabetic patients contain fundamental elements of the basal lamina such as laminin 5, nidogen, perlecan, and fibronectin and tend to deposit them in higher amounts. These data extend to pericytes some observations present in the literature, as it is known that fibronectin is highly expressed by ECs of diabetic patients in vivo, or in culture following exposure to high levels of glucose 31 . The use of autologous pericytes, while advantageous in terms of immunogenicity, might also represent a limitation to the clinical application of pro-angiogenic cell sheets for it requires technical time for its development in culture, meaning that a patient should either be hospitalized for longer periods of time, or receive pre-hospitalization care in order to donate the pericytes needed for cell sheet production in advance with respect to the surgical intervention. To overcome these potential hurdles, the efficiency of cryopreserved allogenic pericyte sheets, as well as decellularized pericyte-derived ECM, is currently under evaluation in our laboratory.
Conclusion
These results suggest that pericytes from diabetic patients are competent in the deposition of ECM when treated in culture with MMCs, making it possible to produce autologous cell sheets for the treatment of CLTI in diabetic patients. CLTI represents a major cause of DFU manifestation and recurrence, therefore the refinement of cell sheets supporting cell therapy for CLTI-dependent DFU will open the path to the clinical application of pro-angiogenic cell therapy to diabetic patients. The autologous nature of pericyte and endothelial progenitor cell–based angiogenic cell sheets should provide a novel healthcare product with very low immunogenicity. However, the use of autologous pericytes might result in longer hospitalization; besides, pericyte immunogenicity is very low. Therefore, development of allogenic cell sheets readily implantable in patients upon isolation and seeding of autologous peripheral blood–derived endothelial progenitors might represent a valuable alternative. Moreover, decellularized pericyte-derived extracellular matrices seeded with endothelial progenitors could also represent a valuable choice. Future efforts will therefore focus on evaluating the angiogenic and therapeutic potential of these alternative choices.
Footnotes
Acknowledgements
The authors thank Dr. Amelia Bertulessi for her help with patient enrollment at the diabetic foot unit in MultiMedica Hospital in Sesto San Giovanni, Milano.
Author Contributions
Andrea Rampin carried out all the experiments and wrote the first draft of the paper, Carlo Maria Ferdinando Caravaggi selected and enrolled diabetic patients with DFU from the diabetic foot unit, harvested, and sent the skeletal muscle biopsies to the laboratory to be studied, Luigi Troisi selected and enrolled non-diabetic patients from the reconstructive surgery unit, harvested, and provided the skeletal muscle biopsies, and Gaia Spinetti provided mentorship and corrected the first draft of the paper.
Ethical Approval
The study was authorized by the independent ethics committee of MultiMedica, a section of the Central Ethics Committee IRCCS Lombardy (protocol number 452.2020, approval report no. CE-119.2020 of December 16, 2020).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Consent to Participate
Patients participating in the study signed a written informed consent before donating their biopsies.
Consent for Publication
N\A.
Data Availability
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Italian Ministry of Health—Ricerca Finalizzata-Starting Grant (SG-2019-12371302) and by Ricerca Corrente to the IRCCS MultiMedica.
