Abstract
Immune rejection presents a significant challenge in xenogenic meniscal transplantation. Pigs are widely regarded as an advantageous tissue source for such transplants, with porcine GGTA1, CMAH, and B4GALNT2 being among the most common xenoreactive antigen (Ag) genes. While some studies have suggested that allogeneic meniscus (AM) transplants may exhibit immunoprivileged properties, our study observed slight immunological rejection has been observed following contact between human meniscal cells (HMCs) and human peripheral blood mononuclear cells (PBMCs). Given the limited systematic research on immune responses following xenograft meniscus transplantation, we established porcine meniscus transplantation (PMT) models to comprehensively assess the immunogenicity of porcine meniscus (PM) from both innate and adaptive immune perspectives. Our investigations confirmed that PMT beneath the epidermis led to innate cell infiltration into the xenografts and T-cell activation in local lymph nodes. T-cell activation upregulated the interleukin (IL)-17 signaling pathway, disrupting collagen organization and metabolic processes, thereby hindering PM regeneration. Using freeze-thaw treatment on PM alleviated T-cell activation post-transplantation by eliminating xenogenic DNA. In vitro findings demonstrated that gene editing in porcine meniscal cells (PMCs) suppressed human T-cell activation by downregulating the expression of xenoreactive Ag genes. These results suggest that GGTA1/CMAH/B4GALNT2 knockout (KO) pigs hold significant promise for advancing the field of meniscal transplantation.
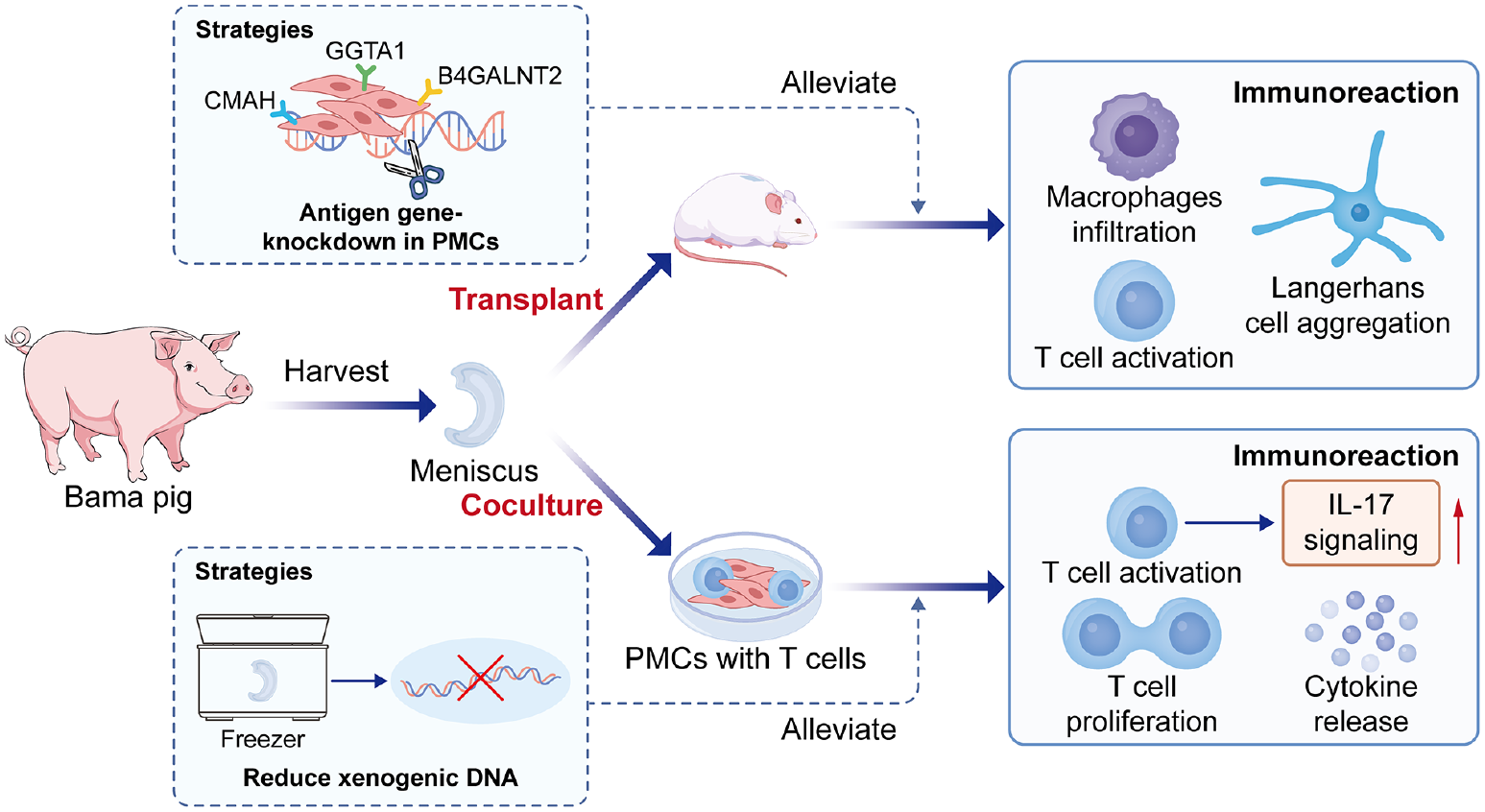
Introduction
The meniscus, a fibrous cartilage within the knee joint, plays crucial roles in load transmission, stress distribution, and joint stability 1 . Damage to the meniscus often results in the loss of these functions, subsequently leading to osteoarthritis2,3. Furthermore, most meniscal injuries, except for those at the peripheral edges, have limited natural healing capacity, necessitating the resection of the damaged portion 4 . Meniscus replacement becomes imperative to restore its functional integrity and prevent the onset of early degenerative joint diseases 5 . Notably, allogeneic meniscus transplantation (AMT) has been performed, with freeze-thawed menisci being the most commonly utilized in this procedure6,7. However, due to the scarcity of available allografts and the technical intricacies involved in AMT, the procedure has been performed at an estimated rate of only 1 per million of the population 8 .
Freeze-thawed xenogenic menisci were anticipated to be among the most promising xenografts for meniscal repair due to their ability to mitigate graft immunogenicity through a relatively straightforward method by removing the xenogeneic DNA and potential pathogens from the xenogeneic tissues9,10. However, the residual Ags from xenografts still provoke immunological rejection, posing a challenge to meniscus regeneration 2 . Effectively mitigating immune and inflammatory responses is vital for enhancing graft survival 11 . Given the anatomical and physiological similarities between pigs and humans, pigs have been selected as ideal donors for xenotransplantation research. Research indicates that the GGTA1 gene, which exists in pigs but is absent in humans, triggers the production of anti-α-Gal IgG in humans11,12. Addressing the aforementioned challenge necessitates exploring strategies to reduce xenoantigen-mediated rejection.
In our study, we used meniscus transplantation models to assess antigen-specific T-cell responses post-transplantation and to explore model-based strategies for immune regulation. The porcine meniscus (PM) was trimmed and transplanted beneath the epidermis of mice, following the animal models established by Yoon et al. 13 and Ochi et al. 14 . Our objective was to analyze the immune reaction following PMT from the perspectives of both innate and adaptive immune responses. Specifically, T-cell activation was evaluated by examining draining secondary lymphoid tissues (SLTs), which are in close proximity to the PMT site 15 . Recognizing that the glycoprotein CD69 is among the earliest markers to be upregulated following T-cell activation16,17, we closely monitored changes in CD69 expression to determine whether PMT induced T-cell activation. We found that freeze-thaw treatment was an effective method for mitigating T-cell activation post-PMT.
Recently, gene editing has emerged as a promising approach to address graft rejection stemming from xenografts. Among the genes implicated in xenoreactive antigenicity, GGTA1, CMAH, and B4GALNT2 are widely recognized18–20. In our study, human lymphocytes were employed as the responsive cells in the mixed lymphocyte reaction (MLR), wherein T-cell proliferation and activation were confirmed upon stimulation with porcine meniscal cells (PMCs). Subsequently, RNA-seq analysis was conducted to delineate the cascade of immune responses triggered by T-cell activation. Finally, we individually knocked down the aforementioned three genes in PMCs and used MLR to assess variations in immunogenicity. The findings suggest that downregulating these three genes in PMCs contributes to mitigating T-cell-mediated rejection within the human immune system.
Materials and Methods
Meniscus
All experiments were conducted in strict accordance with the guidelines of the Institutional Animal Care and Use Committee for the Care and Use of Experimental Animals at Beijing Medical University. Twenty 3-month-old male Bama pig were included in the study. We harvested the bilateral meniscus from the Bama pigs (Vital River Co., Ltd.) under sterile conditions and subsequently rinsed them with phosphate-buffered saline (PBS) containing 1% antibiotics to remove any residual blood. The menisci were then divided into two groups for preservation: in the native meniscus (NM) group, transplantation occurred within 24 hours (hr), while in the freeze-thawed meniscus (FM) group, the tissue was stored at −80°C for 14 days (D) before thawing.
Biochemical Analyses
We recorded both the initial weight and the dry weight of the samples, calculating the percent water content by dividing the difference between them by the initial weight. The meniscus patch was then digested using a Genomic DNA Extraction Kit (Tiangen Biotech Co., Ltd), with centrifugal columns employed for genomic DNA extraction. DNA content was quantified using a NanoDrop 2000 spectrophotometer, and the quantity of DNA was determined by normalizing the total DNA content to the initial dry weight of the sample, expressed as ng/mg. Collagen content was assessed based on hydroxyproline (HYP) content. The HYP content in the samples was measured using an HYP assay kit (Sigma–Aldrich), with a HYP-to-collagen ratio of 1:7.2. The final collagen content value was then divided by the dry weight of the sample. The mean and standard deviation of all biochemical analysis data were calculated using IBM SPSS Statistics 24. Comparisons between groups were conducted using the two-tailed independent t-test.
Scanning Electron Microscopy
Scanning electron microscopy (SEM) was conducted to assess the extracellular matrix (ECM) of the meniscus in the NM and FM groups. Meniscal sections were initially fixed with 2.5% (v/v) glutaraldehyde in PBS for 24 hours, followed by dehydration using a graded alcohol series, and complete drying using a vacuum drying apparatus. Subsequently, the dried samples were sputter-coated with gold-palladium and observed using a JSM-7900F microscope (JEOL Ltd, Japan). The porosity of each group was analyzed using ImageJ.
Xenogenic Meniscus Transplantation
Before transplantation, each meniscus was dissected into patches measuring approximately 3 × 3 × 2 mm³. Male BALB/c mice were selected as recipients, and the xenografts were divided into three groups: NM (n = 12), FM (n = 12), and sham (n = 6). Under respiratory anesthesia induced by isoflurane, a posterior incision was made using surgical tools, and one meniscus patch was transplanted beneath the epidermis on the upper back, followed by suturing of the skin. Throughout the study period, mice were housed with ad libitum access to food and water. In the sham group, the same surgical procedure was performed as in the other groups, excluding xenograft transplantation. Three patchs of transplanted meniscus per group, along with the skin and subcutaneous tissues, were harvested at post-transplantation D 7 and D 14, respectively. Graft survival was monitored daily by assessing whether the grafts remained present and unabsorbed. To compare the survival rate of FM and NM groups, we observed 6 transplantation models in each group until D 28. A surival curve was drawn using GraphPad Prism 9, and the log-rank test was used for statistical analysis.
Draining Lymph Node Harvest
The bilateral brachial lymph nodes of the recipient mice were harvested at post-transplantation D 7 and D 14 and promptly placed into Eppendorf tubes containing ice-cold PBS on ice. We used a distal syringe for grinding and icy PBS for rinsing to isolate lymph node cells. Single cells were then filtered through 70-µm cell strainers and subsequently labeled with antibodies (Abs) for flow cytometry. Detailed information on the antibodies is provided in Supplemental Table 1.
Histological Staining
After excising the dorsal skin of the mice with the transplanted PM, the tissues were promptly collected and embedded in optimal cutting temperature (OCT) compound (Sakura), then rapidly frozen in liquid nitrogen with acetone neutralization. Samples were sliced to a thickness of 8 µm, transferred onto slides, and fixed using acetone. Each slide was subsequently stained with H&E and scanned using a Nano Zoomer (Hamamatsu).
Immunofluorescence Staining
After fixation with acetone, the slides were washed with PBS and Fc receptors were blocked using 5% goat serum. Subsequently, the slides were stained with diluted antibodys (Abs): Allophycocyanin (APC)-conjugated anti-CD11b (1:50 dilution), Fluorescein (FITC)-conjugated CD16 (1:50 dilution), anti-α-Gal epitope (1:100 dilution), and phycoerythrin (PE)-conjugated anti-CD207 (1:50 dilution). 4,6-diamino-2-phenyl indole (DAPI) was applied to all slides for nuclear staining. Images were captured using a confocal microscope (Zeiss, China).
Isolation of Meniscal Cells
Informed consent for the use of excised meniscus in medical research was obtained from three human patients before a partial meniscectomy. The inner sides of the porcine and human menisci from different donors were dissected under sterile conditions and minced into small pieces. Following approximately 4 hours of digestion in 1% collagenase (Sigma–Aldrich), Dulbecco’s Modified Eagle’s Medium (DMEM) with 10% fetal bovine serum (FBS) was employed to halt the digestion process. The cells were subsequently rinsed and cultured in a 75 cm2 flask. The culture medium comprised DMEM with 4.5 g/L glucose, 1% penicillin/streptomycin (BD Biosciences), and 10% FBS. Cells were seeded at a density of 2.5 × 104 cells/cm2 and allowed to proliferate until reaching confluence. Expansion continued until passage 2 was achieved.
MLR
Blood samples were collected from a single healthy human subject with informed consent. Peripheral blood mononuclear cells (PBMCs) were isolated from the blood samples using Ficoll density gradient centrifugation (Ficoll-Paque™ PREMIUM sterile solution, GE Healthcare). The MLR test was conducted following a previously described protocol 21 . Six-well plates were used to culture passage 2 PMCs or human meniscal cell (HMCs) with 2 ml of medium. Upon reaching 80% confluence, the cells were treated with 25 mg/ml mitomycin-C (Sigma–Aldrich) for 45 minutes to arrest proliferation and rinsed three times with medium. Subsequently, 2 × 106 PBMCs were added per well to the cell culture plate. As a negative control (NC), the same quantity of PBMCs was added to blank wells with an equivalent volume of medium. Reactive PBMCs were collected on D 0, 1, and 3 for analysis via flow cytometry. The PMCs and HMCs used at the same point were from three different donors. For each sample, 1 × 104 live PBMCs without contaminating PMCs were collected for flow cytometric analysis.
T-Cell Proliferation Assay
T-cell proliferation was assessed in naive T-cells following established protocols. Naive T-cells were isolated from human PBMCs through negative selection using a human T-cell isolation kit (STEMCELL, 17951), adhering strictly to the manufacturer’s instructions. Subsequently, the isolated naive T-cells were labeled with 1 μM CellTrace™ Far Red (CFR) cell proliferation Kit (Thermo Scientific, C34564). These CFR-stained naive T-cells (2 × 105 cells/well) were then co-cultured with PMCs in a 48-well round-bottom plate using complete RPMI 1640 medium supplemented with 10% (v/v) FBS, 100 U/ml penicillin, and 100 μg/ml streptomycin at 37°C with 5% CO2 for a duration of 5 days. As a control, untreated naive T-cells were included. The assessment of T-cell proliferation was conducted by measuring the mean fluorescence intensity (MFI) of the far red dye through flow cytometry analysis.
B-Cell Proliferation Assay
B cells were isolated from human PBMCs using a human B-cell isolation kit (STEMCELL, 17254), following the manufacturer’s instructions. Subsequently, the purified B cells were labeled with CFR dye (1 μM). The labeled B cells were then stimulated with PMCs. The cells were cultured in 48-well tissue-culture plates at a concentration of 2 × 105 cells per well in 1 ml of culture medium and maintained at 37°C in a humidified chamber with 5% CO2 for a period of 7 days. Purified B-cells that were not stimulated with PMCs were included as a control group for comparison.
Small Interfering RNA Transfection
A small interfering RNA (siRNA) transfection kit was obtained from RiboBio Co., Ltd. (Guangzhou, China) for the transfection of PMCs. The siRNA transfection (siTF) procedure was conducted following the supplier’s protocol. Briefly, the working concentration of siRNA was set at 100 nM, and the incubation period lasted for 72 hours. Three different single siTF genes were used in this study, targeting specific sequences: CTGTCGGAAGATACATTGA for the GGTA1 gene, GAACTCCTCATAAGATAAG for the B4GALNT2 gene, and CTGAGTTACCCAACACT GA for the CMAH gene. An NC siTF was included in the experiment. Following the completion of transfection, the cells were washed with fresh culture medium and subsequently subjected to the MLR for 3 days. Finally, PBMCs were collected for flow cytometry analysis, and cellular supernatant was collected for enzyme-linked immunosorbent assays (ELISAs).
Flow Cytometry Analysis
Fresh cells were suspended in staining buffer (Solarbio) and subsequently incubated with CD16/32 Ab to minimize nonspecific Ab binding. Lymph node cells were stained with FITC-CD19, APC-CD3, PerCP-Cy5.5-CD80, and PE-CD69 Abs along with fixable viability dye (FVD) for 30 minutes at 4°C. For PBMCs, FITC-CD3, APC-CD20, PE-CD69, and PE-Cy7-CD80 Abs along with FVD were used for staining. After staining, the cells were washed with staining buffer and fixed with 4% paraformaldehyde (PFA). Flow cytometry analysis (BD Biosciences) was conducted to assess the stained cells, and the data were analyzed using FlowJo V 10.6.2 software. Statistical analysis was carried out using the two-tailed independent t-test.
Real-Time qPCR
We employed TRIzol reagent (Invitrogen) for the extraction of total RNA and synthesized complementary DNA using a reverse transcription system (Vazyme) as per the manufacturer’s instructions. Quantitative real-time polymerase chain reaction (qRT–PCR) was conducted using the iQ5 optical system software (Bio-Rad) with FastStart Universal SYBR Green Master Mix (Recho, Basel, Switzerland) to quantify mRNA levels of all specified genes. Relative expression levels were determined using the 2−ΔΔCt method and normalized to GAPDH. The primer sequences used were as follows: GAPDH: forward 5′-GTCGGAGTGAACGGATTTGGC-3,’ reverse 5′-CACCCCATTTGATGTTGGCG-3′; GGTA1: forward 5′-GCTTCTGCCGATCAGGAGTT-3′, reverse 5′-ACCTTCTGGGCTGTTGATGT-3′; CMAH: forward 5′-CTTGCTGAGAGAAGAC CAGATGT-3′, reverse 5′-GAACGCCATCAAGATCATG-3′; B4GALNT2: forward 5′-GCTT TTGGTCCTGAGCGTTG-3′, reverse 5′-AACAGCCCGTTTTCACCGTA-3′; a-Gal: forward 5′-CCAATGACCTCCGACACATCA-3,’ reverse 5′-GTTTACCATAGCCACAGCCCA-3′; Neu5GC: forward 5′-TCACCAGCGACAGTTTGCTT-3′, reverse 5′-GCCTCCAACATCTGGTTTGT-3′; and Sda: forward 5′-GGCGTCCTTAGAAGCAAGAG-3,’ reverse 5′-TCACCAGCGACAGTT TGCTT-3′.
RNA Sequencing of Human T-Cells
Naive T-cells were co-cultured with 80% confluent PMCs in a 6-well plate for 72 hours, serving as the control group. For the experimental group, T-cell seeding was performed with culture medium supplemented with 25 μl/ml human CD3/CD28/CD2 T-cell activator (STEMCELL, 10990) and 50 IU interleukin (IL)-2 (NovoProtein, C013). The activated T-cells were then co-cultured with an equal amount of PMCs for 72 hours. Supernatants were collected without disturbing the bottom and centrifuged at 500 g for 5 min to remove cellular debris. The resulting pellet was treated with TRIzol reagent (Invitrogen) to extract total RNA. After confirming the integrity of the RNA, it was reverse-transcribed to generate a cDNA library for subsequent sequencing.
Cytokine Assay
The supernatant from the co-cultured cells was centrifuged at 1700 rpm for 5 minutes at 4°C to remove cellular debris. The levels of IL-2 and interferone (IFN)-γ in the serum were determined using ELISA kits.
Results
Biochemical Analyses and DAPI Staining of the Meniscus
Compared to the NM group, the DNA and water contents within the meniscus were notably lower in the FM group (Fig. 1A, B), the expression of xenogenic genes of PM was downregulated after freeze-thawing, including GGTA1, CMAH, and B4GALNT2 (Fig. 1D, E, F). In addition, the collagen content within the meniscus significantly increased following freeze-thawing (Fig. 1C). Upon staining with DAPI, the images displayed a significant decrease in the numbers and area of nuclei in the FM group compared to the NM group (Fig. 1G, H), indicating a degree of cell removal after freeze-thaw treatment.
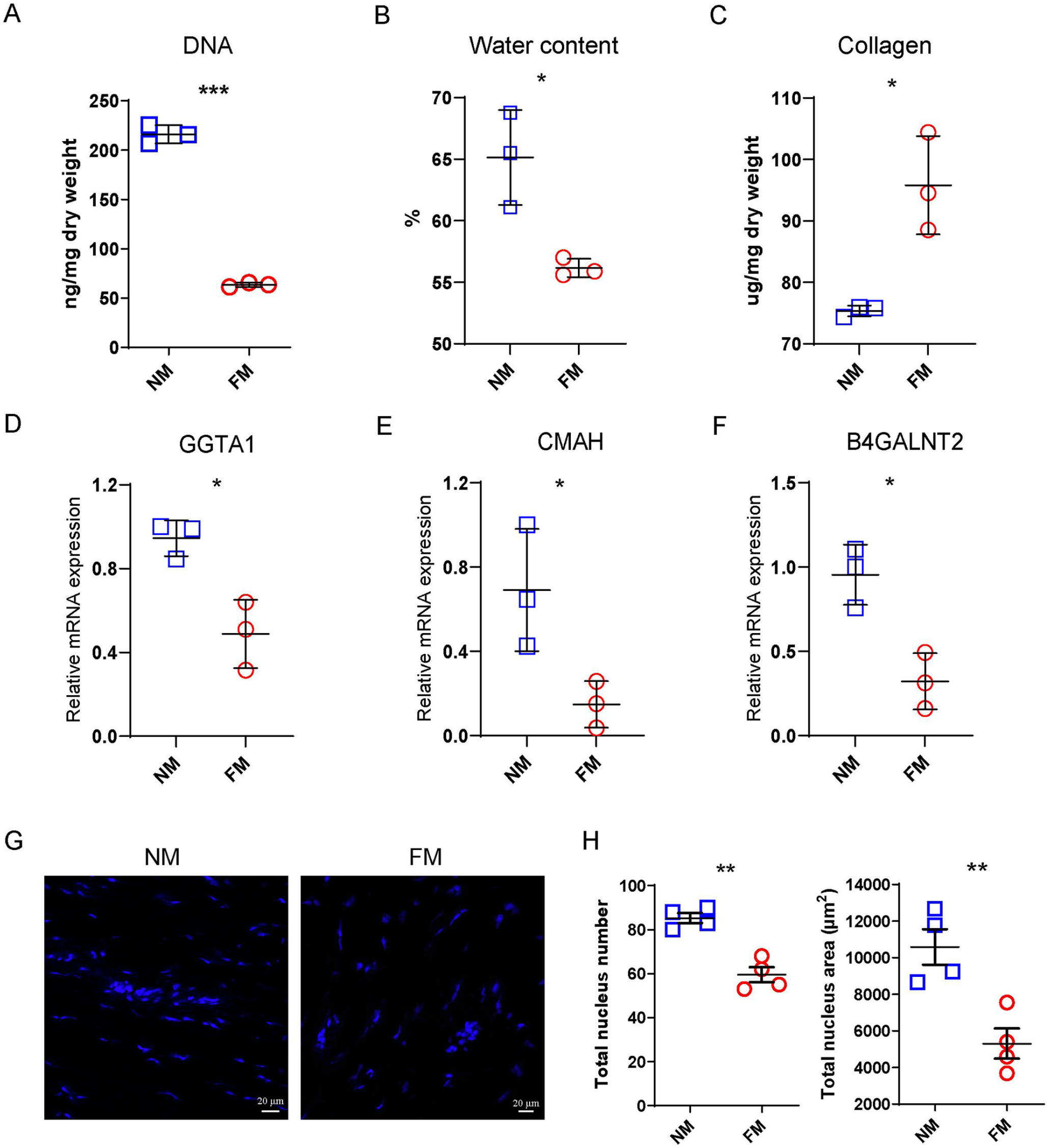
Freeze-thaw treatment of PM reduced the content of xenogenic reactive genes. Biochemical characteristics of the specimens of (A) DNA, (B) water content, and (C) collagen in the NM and FM groups (n = 3). Relative mRNA expression of (D) GGTA1, (E) CMAH, and (F) B4GALNT2 in the NM and FM groups, respectively. (G) DAPI staining of longitudinal sections from the NM and FM groups; scale bar: 20 µm. (H) Total number and area of nuclei in each section in the NM and FM groups (n = 3). NS: P > 0.05, *P < 0.05, **P < 0.01 and ***P< 0.001.
The Meniscal Xenograft Triggered the Host Innate Immune System
The ECM structures in the NM and FM groups are depicted in Fig. 2A. The FM group exhibited denser ECM, while the NM group appeared relatively loose and featured more pores, which is conducive to cell infiltration. The porosities in the NM and FM groups were 26.11% and 14.04%, respectively. Porcine meniscal sections were transplanted under the dorsal epidermis of each recipient mouse (Fig. 2B). A survival curve with a P value less than 0.01 is presented in Fig. 2C; as time elasped, the survival rate of the NM group decreased. The NM group displayed more lymphocyte infiltration across the transplantation border than the FM group after H&E staining. With the extension of transplantation time, decreased lymphocyte infiltration and hair growth were observed on D 14 (Fig. 2D). Immunofluorescence staining was employed to identify the types of infiltrated cells, confirming that numerous CD11b+ and CD16+ cells crossed the transplantation border (Fig. 2E), demonstrating that the meniscal xenograft triggered intense innate immune responses in the recipient.
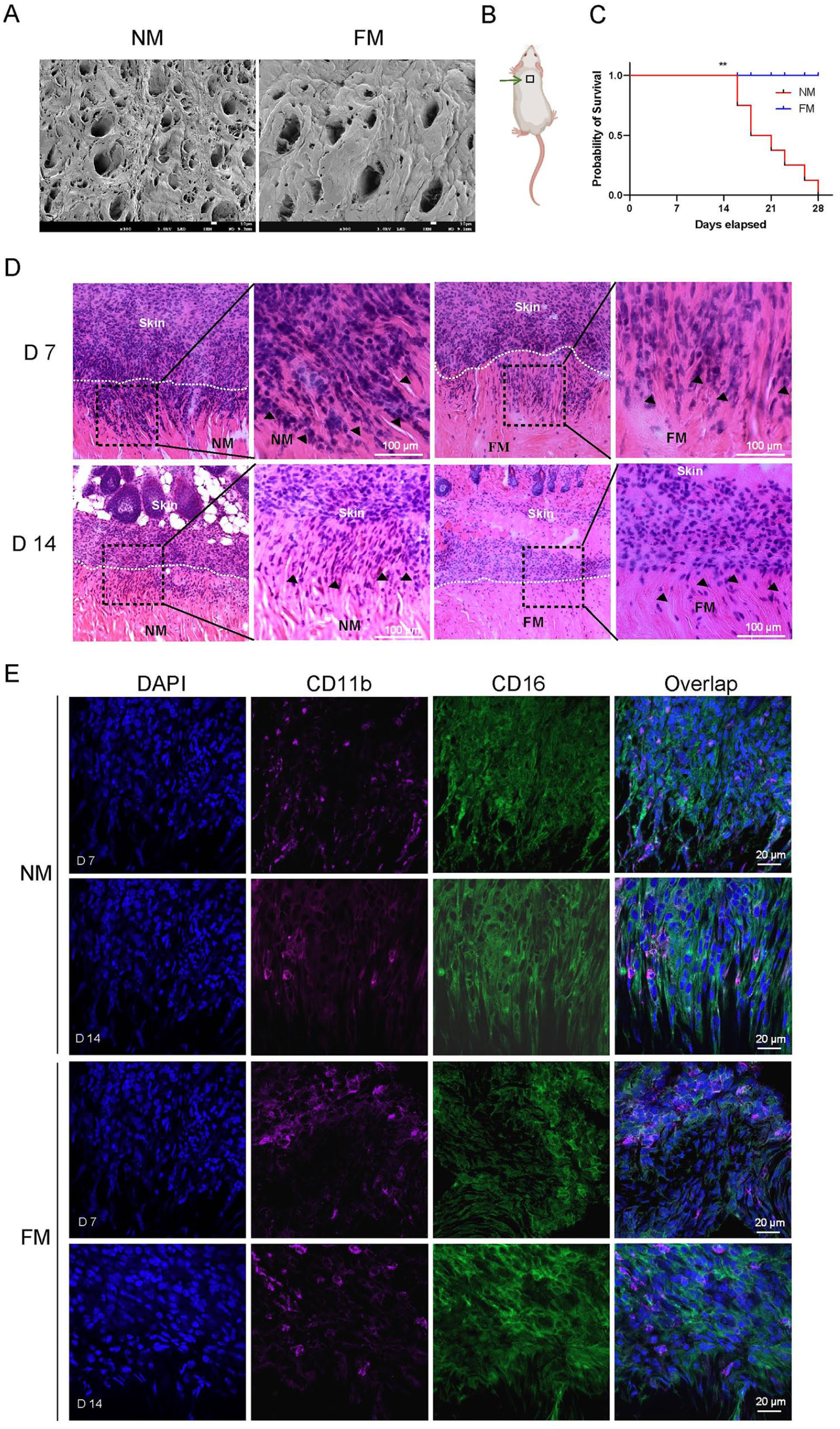
Lymphocytes infiltrated into the transplant border. (A) SEM images of FM and NM; scale bar: 10 µm. (B) The meniscus section was transplanted under the dorsal epidermis, as indicated by the green arrow. (C) The survival curve of the xenografts in the NM and FM groups. (D) H&E staining images of the transplanted NM and FM at D 7 and D 14; the infiltrated lymphocytes were marked with black arrowheads in the images; scale bar: 100 µm. (E) Immunofluorescence staining of the transplanted NM and FM at D 7 and D 14. Blue indicates DAPI, green indicates CD16, and purple indicates CD11b; scale bar: 20 µm. **P < 0.01.
Xenogenic Antigens Activate T-Cell Immunity
Fresh lymphocytes were harvested from the brachial lymph nodes of the recipients. Flow cytometric analysis revealed that the ratio of FVDnegCD3+CD69+ cells was highest in the NM transplantation group. Compared to the sham group on D 7 and 14, an increase in the FVDnegCD3+CD69+ cell ratio was observed after NM and FM transplantation (Fig. 3A, B, S1A), indicating activation of draining T cells by the Ags from the meniscal xenograft. During this process, epidermal dendritic cells might play a role in presenting xenogenic Ags. To inverstigate this, we conducted immunofluorescence staining of the PM with the contacted skin and compared the numbers of CD207+ cells between groups (Fig. 3C). The results indicated that xenoantigens from the NM group induced stronger aggregation of local epidermal Langerhans cells at the transplantation site than the FM group, facilitating the transfer of xenogenic Ags to the draining lymph nodes. The FVDnegCD3+CD69+ cell ratio and numbers of CD207 were lower in the FM group than in the NM group, suggesting that fewer xenoantigens could mitigate the triggered immune responses.
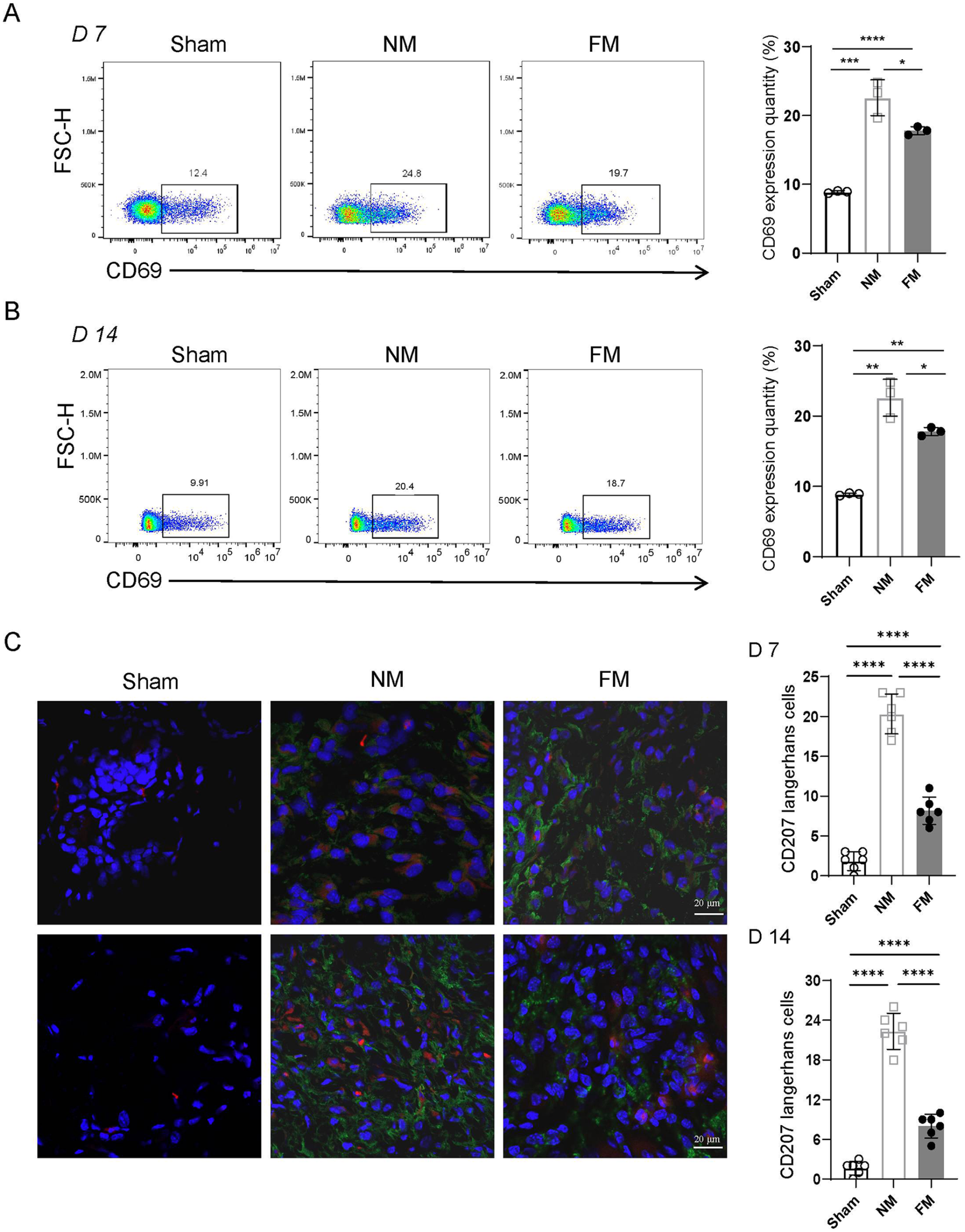
T-cell activation in the draining lymph node. (A) Flow cytometric quantification of the ratio and representative FVDnegCD3+CD69+ cells in the sham, NM and FM groups on D 7. Statistical analysis was performed using a two-tailed independent t-test for comparisons between two groups (n = 3). (B) Representative images and statistical analysis of FVDnegCD3+CD69+ cells in the sham, NM and FM groups on D 14 by flow cytometry (n = 3). (C) Immunofluorescence staining of the transplanted FM and NM, and the skin of the sham group. Blue indicates DAPI, green indicates a-Gal, and red indicates CD207; scale bar: 20 µm. Statistical analysis was performed using a two-tailed independent t-test to compare two groups (n = 6). * P < 0.05, **P < 0.01,***P < 0.001 and ****P < 0.0001.
PMCs Activate Human T Cell
Compared to the control group, the MFI values of naive T cells and naive B cells stimulated by PMCs was significantly lower, indicating that PMCs stimulated the proliferation of T cells and B cells (Fig. 4A, B). In addition, we observed a noticeable increase in the expression of CD69 with the extension of coculture time (Fig. 4C). We selected D 3 as the time point to compare the intensity of allogenic and xenogenic reactions. Flow cytometry analysis revealed that the ratios of FVDnegCD20+CD80+ cells and FVDnegCD3+CD69+ cells in the PMC group were both higher than those in the HMC group (Fig. 4D, E, S1B). Furthermore, the levels of IL-2 and IFN-γ in the cell culture supernatant were higher in the PMC group compared to the HMC group (Fig. 4F). These results demonstrate that allogenic MCs also triggered immunological rejection, albeit much milder than xenogenic rejection. HMCs highly expressed CD90 and CD146, carrying approximately 7.92% HLA-I (Fig. 4G), which could elicit a mild allogenic reaction.
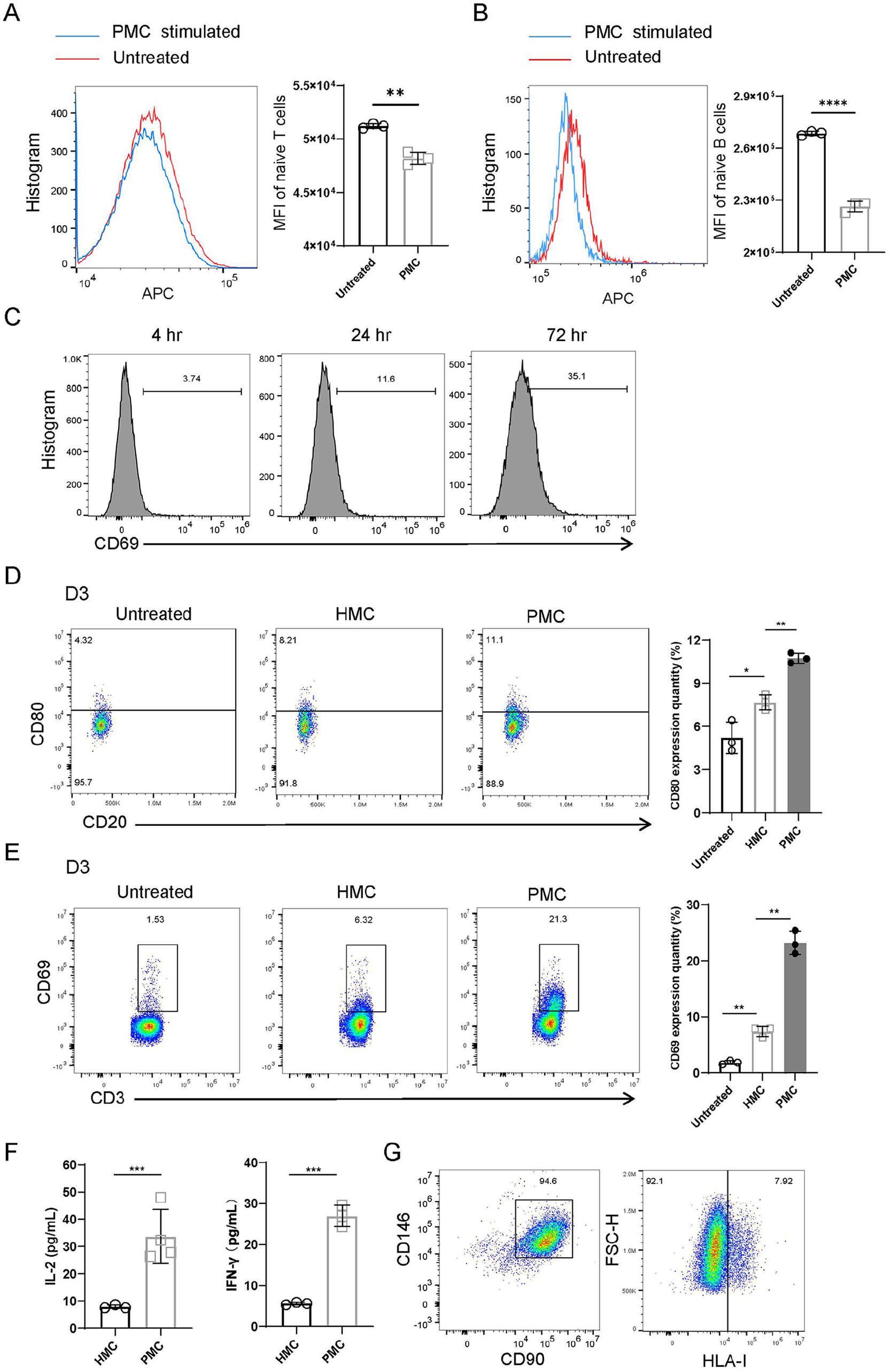
PMCs triggered human T-cell activation. (A) Flow cytometric analysis and statistical comparison of the MFI value of untreated and PMC-stimulated naive T-cells. (B) Flow cytometric analysis and statistical comparison of the MFI value of untreated and PMC-stimulated naive B cells. (C) Representative flow cytometry images indicating the CD69 expression level at different time points. (D) Flow cytometric quantification of CD80 expression levels in CD20+ cells on D3. (E) Flow cytometric quantification and statistical analysis of CD69 expression levels in CD3+ cells according to the stimulated cells in the MLR on D3 (n = 3). (F) IL-2 (left, n = 3) and IFN-γ (right, n = 3) concentrations in PBMC supernatants measured using ELISA. (G) Determination of the expression of CD146, CD90, and HLA-I in HMCs by flow cytometry. *P < 0.05, **P < 0.01 and ***P < 0.001.
Multiple Gene Expression Variation Trend after Human T-Cell Activation
In our RNA sequencing analysis of co-cultured PMCs and T-cells, we used the control group as a reference and identified 5306 upregulated genes and 4958 downregulated genes in the activated T-cell group (fold change ≥ 2; q value < 0.05) (Fig. 5A). The comparisons of expression differences of key genes between the naive and activated T-cell groups were demonstrated in a heatmap (Fig. 5B), showing a downregulation in genes related to ECM formation, collagen fibril organization and metabolism, wound healing, and cell adhesion. Subsequent gene ontology (GO) analysis revealed that activated T-cells significantly facilitated inflammatory and immune responses (Fig. 5C). Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis demonstrated that T-cell activation affected pathways including cytokine–cytokine receptor interaction, IL-17 signaling pathway, PI3K-Akt signaling, JAK-STAT signaling, and others (Fig. 5D). Notably, the cytokine–cytokine receptor interaction exhibited the best Q value. Furthermore, gene set enrichment analysis (GSEA) results indicated that activated T-cells fostered cytokine–cytokine receptor interaction (Fig. 5E) and upregulated the IL-17 signaling pathway (Fig. 5F).
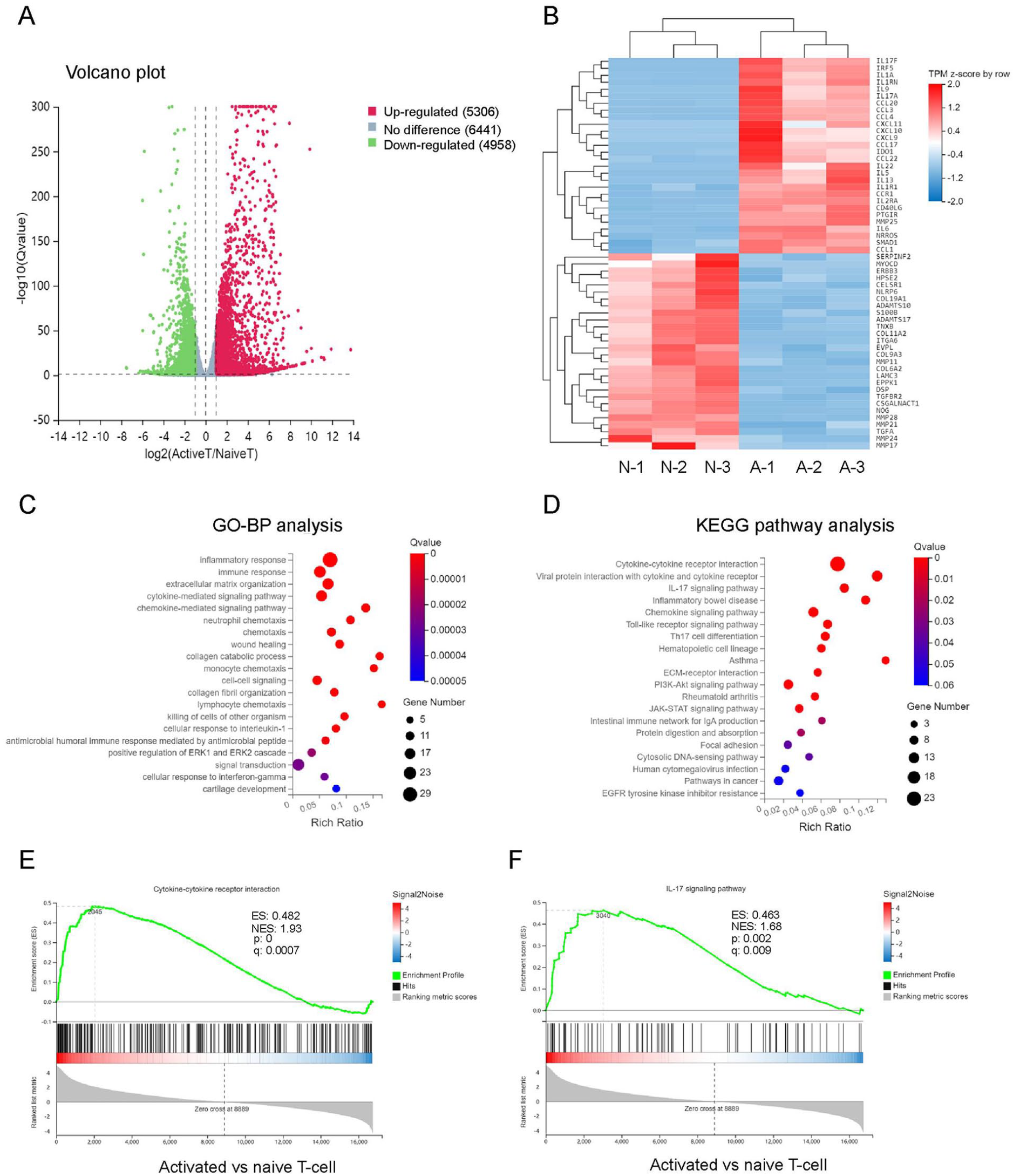
RNA-seq analysis of tendon between naive and activate T-cell groups. (A) Volcano plot of gene expression (naive versus activated; fold change ≥ 2; q value < 0.05). N, naive; A, activated. (B) Heat map of differentially expressed genes. (C) GO analysis of differentially expressed genes. (D) KEGG analysis of upregulated genes in activated T-cell group. GSEA of the genes associated with cytokine-cytokine receptor interaction (E) and the IL-17 signaling pathway (F). NES, normalized enrichment score; FDR, false discovery rate.
Porcine Gene Downregulation Alleviates Xenogenic Reactions
The confocal microscopy image revealed tight siRNA binding to the surface of PMCs (Fig. 6A). Gene expression analysis showed significant downregulation of GGTA1, CMAH, and B4GALNT2 72 hours after siTF, as confirmed by real-time qPCR (Fig. 6B). The expression of corresponding xenoantigens of α-Gal, Neu5Gc, and Sda was also significantly reduced (Fig. 6C). We then assessed the respective stimulation of PMCs on PBMCs after GGTA1-siTF, CMAH-siTF, and B4GALNT2-siTF using ELISA and flow cytometry. The NC group, treated with NC siTF, exhibited significantly higher levels of IL-2 and IFN-γ in the PBMC supernatant compared to the other siTF-treated groups (Fig. 6D). In addition, it demonstrated higher expression of CD80 in CD20+ cells and CD69 in CD3+ cells (Fig. 6E, F, G). Since CD69 serves as a T-cell activation marker and CD80 provides a costimulatory molecule for T-cell activation 22 , these in vitro results provide sufficient evidence that downregulation of porcine genes can alleviate xenogenic immunological responses.
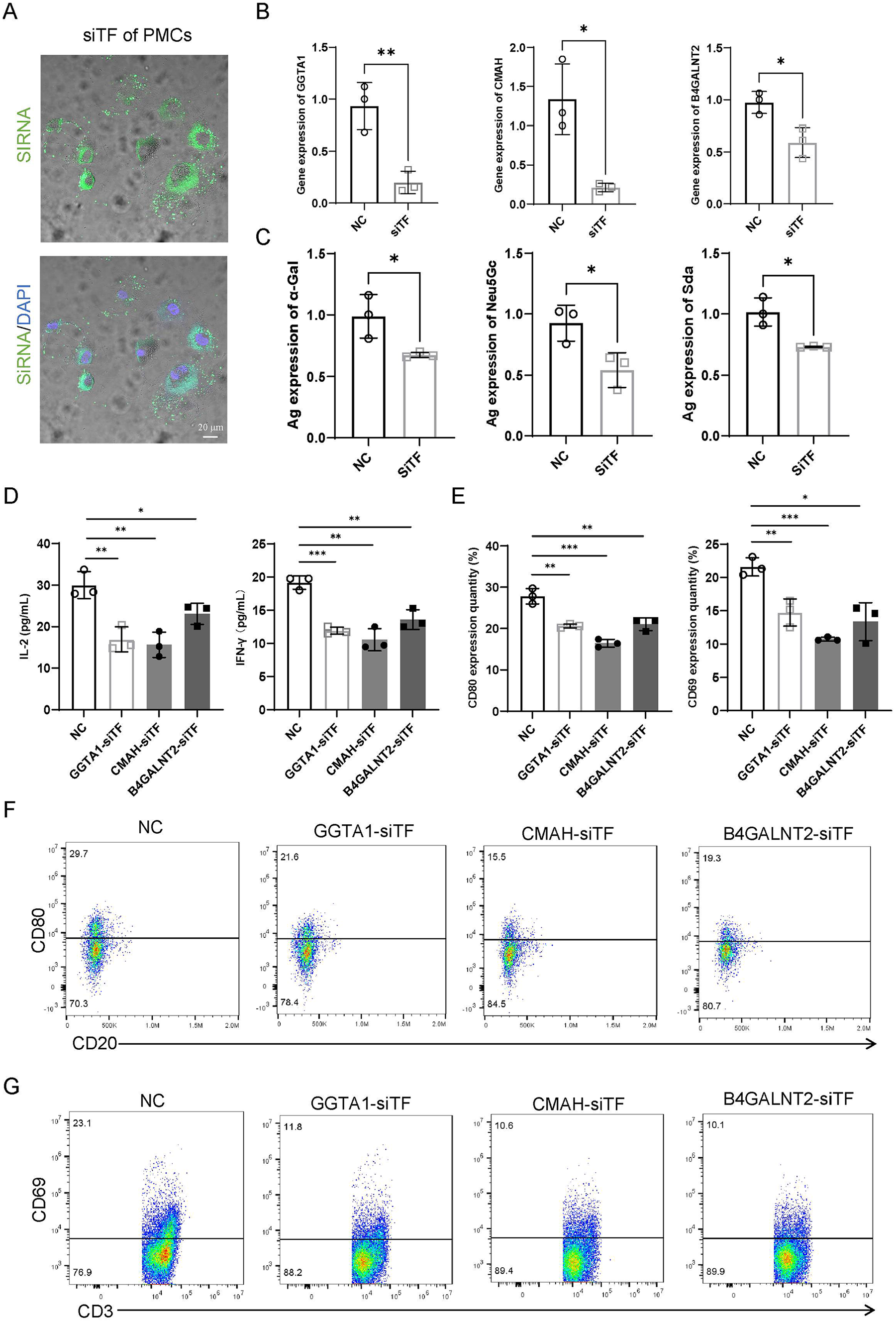
Downregulation of porcine gene alleviated immune response. (A) Confocal microscopy images of siTF-treated PMCs. Green indicates siRNA and blue indicates DAPI; scale bar: 20 µm. (B) The relative gene expression of GGTA1 (left), CMAH (middle), and B4GALNT2 (right) in the PMCs of the NC and siTF-treated groups. (C) The relative levels of Ag expression of α-Gal (left), Neu5Gc (middle) and Sda (right) in the PMCs of the NC and siTF groups. (D) IL-2 (left, n = 3) and IFN-γ (right, n = 3) concentrations in PBMC supernatants from different MLR groups. (E) Expression levels of CD80 (left, n = 3) in CD20+ cells and CD69 (right, n = 3) in CD3+ cells. Representative flow cytometry images of CD80 (F) and CD69 (G) in different groups. *P < 0.05, **P < 0.01 and ***P < 0.001.
Discussion
Clinically, AMT has been successful in restoring physiological and biomechanical functions following meniscectomy 6 . However, its widespread implementation is hindered by limited donor resources. Xenotransplantation offers a viable alternative to address the scarcity of matched allogeneic meniscal tissue, given its high cell retention and adaptability for tissue engineering 13 . Our study demonstrates that porcine meniscal transplantation (PMT) can trigger T-cell-mediated rejection, leading to pro-inflammatory responses and impacting PMC survival. Consistent with findings from other studies, we have shown that reducing xenoreactive antigens can mitigate immune rejection during the acute phase11,23. Previously, we demonstrated that freeze-thawing and irradiating a PM enhanced the long-term survival of xenografts transplanted into rabbits 10 . In this study, we further established that freeze-thawing the PM effectively reduces the expression of porcine genes without affecting collagen content, providing a straightforward approach to alleviate anti-donor responses.
A previous study suggested that allogeneic menisci in mice have immune privileges, as the host does not mount a humoral response to the allograft 14 . This implies that the Ags of the mouse meniscus are not sufficient to elicit a systemic immune response. In our study, we aimed to precisely evaluate the immunogenicity of HMCs. By coculturing HMCs with allogenic PBMCs, we demonstrated that HMCs could trigger mild T-cell-mediated immunological responses upon contact with PBMCs, indicating the necessity for the appropriate appplication of immunosuppressants before the formation of immune adaptation following AMT. However, the xenogenic reaction to PMCs was much more intense than the allogenic reaction to HMCs, consistent with the findings of previous research 24 , which demonstrates that humans are sensitized to porcine Ags.
The occurrence of local immune responses to the xenograft was confirmed in the NM and FM transplantation models. Significant infiltration of innate immune cells, such as CD11b+ macrophages, CD16+ natural killer (NK) cells, and CD207+ Langerhans cells, was observed at the transplant border. Macrophages are known sources of pro-inflammatory cytokines, including IL-1β, IL-6, IL-12, IL-23, and tumor necrosis factor (TNF)-α 13 . NK cells produce cytotoxic cytokines, such as IFN-γ, IL-1, and IL-10 25 , while Langerhans cells, as epidermal dendritic cells, play a role in Ag presentation by migrating along subcutaneous lymphatic ducts to local lymph nodes, thereby stimulating the activation of host T-cells 14 . In our study, increased expression of CD69 in T-cells within lymph nodes was detected during acute phases, suggesting activation of T-cells by xenogenic Ags. In addition, we observed increased expression of costimulatory molecules (CD80) in B-cells, providing stimulation signals for T-cell activation.
The effects of T-cell activation on PMCs were further investigated through RNA-seq analysis in our study. The results revealed up-regulation of several pro-inflammatory cytokines and chemokines in the IL-17 signaling pathway, which is known to promote autoimmune and inflammatory diseases 26 . In addition, disruptions were observed in the processes of collagen organization and metabolism in PMCs, contradicting wound healing and cartilage development. In summary, the Ags carried by PMC triggers the activation of receptor T cells, impacting graft repair. Therefore, gene editing targeting the graft-derived Ags is deemed critical.
In this study, we evaluated the immune regulation of PMCs by employing siTF targeting three genes to downregulate GGTA1, CMAH, and B4GALNT2 expression. Consequently, the activation of contacting human T-cells was significantly mitigated. While the field of meniscus transplantation has garnered less attention, considerable efforts have been directed toward generating genetically modified pigs for organ transplantation. Studies have reported successful transplantation of certain organs, such as the kidney and heart, from polygene-edited pigs into humans27,28. Therefore, polygene editing of donors for PMT is entirely feasible. Although genetically engineered pigs serve as excellent sources of xenotransplantation tissues and help alleviate anti-donor rejection post-xenograft transplantation, the cost of genetically engineered pigs escalates with the number of edited genes. Thus, targeting specific genes for KO can substantially reduce costs. Our study confirmed that the genes GGTA1, CMAH, and B4GALNT2 are xenoreactive Ag genes implicated in PMT rejection, suggesting that a meniscus from a GGTA1/CMAH/B4GALNT2 KO pig might enjoy long-term survival when transplanted into a compatible recipient.
One limitation of this study is that NM and FM were not directly transplanted into the joint. Our primary focus was to investigate the reaction triggered by PMT, but the size of the recipient mouse meniscus rendered it impractical for substitution with the PM section in the knee. Another limitation is the absence of GGTA1, CMAH, and B4GALNT2 KO pigs in the study. Nevertheless, this study demonstrated that subcutaneous transplantation of xenogenic meniscus initiated the host innate immune system, leading to T-cell-mediated rejection during the acute phase, and hindering the wound healing process. Decreasing the amount of xenoantigens can mitigate attacks by the immune system of the recipients. Techniques such as freeze-thawing and gene editing can reduce the content of xenoreactive antigens, with the aim of alleviating acute rejection.
In summary, our study contributes to the ongoing discourse on using of gene editing and freeze-thawing treatment to mitigate anti-porcine responses triggered by PMT. The findings also offer insights that could advance the application of polygene KO pigs in the field of meniscal transplantation.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241273689 – Supplemental material for Inhibiting T-Cell-Mediated Rejection of the Porcine Meniscus Through Freeze-Thawing and Downregulating Porcine Xenoreactive Antigen Genes
Supplemental material, sj-docx-1-cll-10.1177_09636897241273689 for Inhibiting T-Cell-Mediated Rejection of the Porcine Meniscus Through Freeze-Thawing and Downregulating Porcine Xenoreactive Antigen Genes by Rao Chen, Hailong Zhao, Liya Ai, Jiying Zhang and Dong Jiang in Cell Transplantation
Footnotes
Acknowledgements
The authors thank Wenqiang Yan for providing fresh PM and express their gratitude to the Department of Sports Medcine for providing research space.
Author Contributions
D.J. and R.C. designed the study, R.C. and H.Z. performed the experiments, analyzed the data, and wrote the manuscript. L.A. arranged the figures. D.J. and J.Z. confirmed the authenticity of all the raw data. All authors have read and approved the final manuscript.
Availability of Data and Materials
The analyzed data sets generated during the present study are available from the corresponding author on reasonable request.
Ethical Approval
All animal experiments complied with the ethical guidelines for animal experimentation, and experiments related with human samples were approved by the Beijing Medical University (No. PUIRB-LA2022629).
Statement of Human and Animal Rights
All animal experiments complied with the ethical guidelines for animal experimentation, and experiments related with human samples were approved by the Beijing Medical University (No. PUIRB-LA2022629).
Statement of Informed Consent
Informed consent was obtained from the human subject before blood sample collection or partial meniscectomy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the National Natural Science Foundation of China (grant nos. 82072428 and 82300460).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
