Abstract
The quest for new and improved therapies for Parkinson’s disease (PD) remains of paramount importance, despite previous trial failures. There is a current debate regarding the potential of stem cell research as a therapeutic approach for PD. The studies of dopaminergic fetal stem cells for PD treatment, their design, and the results of the initial surgical placebo-controlled trials were reviewed in this study. Some of the fundamental methodological challenges and possible strategies to resolve them were proposed. In this article, we argue that the most important impact lies in the proof-of-principle demonstrated by clinical trials for cell replacement strategies in reconstructing the human brain. While some researchers argue that the considerable technical challenges associated with cell therapies for PD warrant the discontinuation of further development using stem cells, we believe that the opposing viewpoints are instrumental in identifying a series of methodological misunderstandings. Here, we propose to expose key challenges to ensure the advancement of the field and unlock the potential of stem cell therapies in PD treatment. Overall, this review underscores the need for further research and innovation to overcome the hurdles in realizing the potential of stem cell–based therapies for PD.
Keywords
Introduction
The prevalence of Parkinson’s disease (PD) is on the rise, leading to increased years lived with disability 1 . The global PD community emphasizes the need for research to understand the disease’s mechanisms and explore the potential benefits of new reconstructive therapies 2 . The conceptually straightforward goal of dopamine cell replacement in PD serves as an attractive starting point for the development of more complex cell replacement therapies to repair the central nervous system (CNS). Despite the absence of a current therapeutic product, clinical benefits are evident in many patients with PD transplanted with human fetal dopaminergic cells in the context of clinical trials 3 . Researchers have put forth compelling evidence-based arguments for proceeding with the project of translating dopaminergic cell transplantation into an effective treatment for PD 4 .
Developments in the field of stem cell technologies have been viewed as a game changer, offering an alternative to the logistical and ethical challenges associated with fetal cells. Stem cells hold the potential to provide a consistent and controlled supply of suitable cells for transplantation, produced in accordance with good manufacturing practices 5 . It has been proposed that the most suitable stem cells for reconstructing the nigrostriatal dopaminergic system are those which closely approximate the authentic isogenic human midbrain dopamine-releasing neurons affected by PD neuropathology 6 .
Currently, extensive international projects of preclinical studies are underway7,8, to uncover the mechanism of action of candidate stem cell lines and also phase 1 clinical trials to identify the optimal parameters such as patients’ age, disease duration, and symptom presentation to ensure the likelihood of significant and clinically meaningful outcomes in future trials 9 (https://www.clinicaltrialsregister.eu/; https://clinicaltrials.gov/). While progress is being made in translating cell technology into an effective treatment of PD and a range of CNS disorders, a recent negative commentary has emerged, questioning the utility of neural reconstruction and warning against the potential risks to the lives of patients 10 . To advance stem cell–based therapies in PD, it is crucial to address the methodological challenges that hinder progress in this field. Considering the disruption to the funding and progress by the previous negative commentaries targeting fetal cell transplantation11,12, this article aims to critically evaluate the methodological challenges associated with the implementation and interpretation of previous research and suggest ways of resolving these challenges by identifying methodological strategies best suited to developing safe and effective reconstructive therapies for individuals with PD.
Historical Evidence for the Efficacy of Dopaminergic Cell Transplantation
The field of dopaminergic cell transplantation has seen significant developments over the years. Initial attempts at using autologous adrenal cells for transplantation proved unsuccessful. However, subsequent phase 1, open-label clinical trials employing human fetal dopaminergic cells 13 – 15 showed very strong levels of efficacy. For example, a study of five patients demonstrated both clinically meaningful and statistically significant improvements of 38.9% on the motor component of the Unified Parkinson’s Disease Rating Scale under no medication conditions (UPDRS motor, off), and a 61% increase in 18F-fluorodopa neural imaging from baseline, indicating both increased dopamine turnover and functional recovery following cell transplantation 13 .
It is important to note that phase 1 trials, which typically involve small patient cohorts, raise concerns about the external validity of the findings. To address this issue, meta-analyses have been conducted to integrate the multiple and diverse results, indicating strong overall weighted mean improvements in published studies, such as a 39.4% improvement from baseline on UPDRS (motor, off) under no medication conditions and an overall 40.3% increase in 18F-fluorodopa uptake 3 .
One crucial finding from the open-label studies was the significant positive correlation between improvements in motor and dopaminergic functioning. For instance, a review of key reports reported a correlation (rho) of 0.66, with P < 0.05, between increases in 18F-fluorodopa uptake and UPDRS (motor, off) improvements in 10 patients participating in two open-label studies14,15. These promising results provide a solid foundation for further advancing the project of translating cellular transplantation into effective reconstructive treatments for people with PD. It is understood, however, that the changes observed in open-label studies may not accurately represent the causal effects of the transplanted cells, given that the results of open-label trials can be influenced by confounding extraneous variables, such as placebo effects in patients or observer bias by the researchers.
Randomized controlled trials (RCTs) are considered the “gold standard” designs for establishing causal effects and evaluating the safety and efficacy of developing novel therapies16,17. When a developing novel treatment involves surgical procedures, most researchers argue that the implementation of confirmatory surgical placebo-controlled trials is a necessary design feature which requires sham (or imitation) surgical procedures to create the control groups. Even though relying upon sham surgery controls is ethically controversial 18 – 20 , they have been accepted as necessary to control for placebo effects and observer bias associated with evaluating the efficacy of experimental treatments of regenerative therapies requiring surgical procedures19,21.
In the mid-1990s, the National Institutes of Health (NIH) funded two surgical placebo-controlled trials to confirm whether the transplantation of human fetal dopaminergic cells actually caused the benefits found in open-label studies or if the benefits were influenced by placebo effects and observer bias22,23. Despite the therapeutic success seen in many patients participating in open-label trials, the apparent failure of two double-blind, randomized trials to report significant benefits led some researchers to believe that the transplantation approach had reached a dead end 10 . This perception, along with the costs, ethical and methodological challenges, and sustained commitment, required by researchers to complete projects for the transplantation of dopaminergic cells to achieve clear therapeutic benefits, turned the attention of many researchers’ toward exploring other promising alternate approaches to reconstructive therapies for PD 9 . It is essential to note that before the above two randomized trials were commenced, concerns were expressed that the research program of neural transplantation for PD had not progressed to a point at which implementation of randomized sham-controlled trials for confirming safety and efficacy was justified 24 . There were crucial questions regarding the optimal parameters for implementing these trials that remained unanswered in the mid-1990s. With hindsight, waiting until the results of preliminary safety efficacy trials were available might have improved the outcomes of the surgical placebo-controlled trials and generated valid decisions regarding efficacy, critical for the further development of cell transplantation for PD13,14.
Consistent with the issues raised above, the designs of the first two surgical placebo-controlled trials addressed research questions which are arguably more suitable for preliminary phase 1 safety and efficacy investigations 24 , rather than for designing phase 2 confirmatory trials. It would have been advantageous to have preliminary information for answering questions regarding key parameters, such as the ages of patients best suited for transplantation 22 or the quantity of cells required for ensuring optimal benefits 23 , rather than to attempt to answer these questions within the framework of the two confirmatory surgical placebo-controlled trials, as shown in Table 1.
The Design and Results of the First Two Surgical Placebo-Controlled Trials.

Although the two surgical placebo-controlled trials failed to demonstrate at the designated trial end points statistically or clinically significant benefits on the primary outcomes selected by the researchers for the transplantation of dopaminergic fetal cells, their results do not necessarily constitute valid evidence for the failure of the entire research program. It was recognized by the researchers that it was a mistake to rely on the subjective “Global Rating Scale” as the primary outcome measure for evaluating treatment efficacy 25 . When data were analyzed on UPDRS (motor, off), statistically significant differences (P = 0.0003) were found for the transplanted group with patients 60 years or younger as compared with the sham-operated controls 25 . Also a significant effect size was evident in the second trial 23 , within a group of patients receiving four fetal tissue transplants per side up to 6–9 months following transplantation, although this benefit gradually faded to no effect from baseline at the 2-year endpoint of the trial, possibly due to the premature cessation of immunotherapy.
The implication which may be drawn from the above two confirmatory trials when their methodological limitations are taken into account is that although the transplantation of fetal cells may not work for all subgroups of patients with PD under all circumstances, the overall results from phase 1 and phase 2 studies clearly provide “proof of concept” for the potential benefit of cell transplantation. It is unfortunate that following the mistaken inference of “futility,” no new RCTs have been published for the last two decades, resulting in a dearth of new trial results for making valid decisions regarding the safety and efficacy of the implantation of dopaminergic fetal cells 26 .
Evidence for the Magnitude of Placebo Effects and Responses in Surgical Placebo-Controlled Trials
The perceived failure of cellular therapies under surgical placebo-controlled trials designs has been attributed to the powerful influences of observer bias and placebo effects, inflating the apparent benefits of treatment outcomes17,27. It has been suggested that given the perceived demands associated with participating in an experimental neurosurgical procedure and the strong expectations of treatment benefits, it follows that, “. . . consenting to undergo experimental brain surgery elicits the largest and most durable placebo response and therefore must be controlled for with sham procedures” 10 .
Distinguishing between the concepts of placebo response and placebo effect is important; the response refers to the measured changes on specific outcome measures in the control group, while the effect represents inferences drawn regarding the true therapeutic benefits of the sham procedures 28 . It is important to note that no evidence emerged in the two ground-breaking surgical placebo-controlled trials for improvements on either the primary or the secondary outcome measures for clinical improvements in the sham-controlled groups22,23 (see Table 1). Further, a later meta-analysis reported that a combined weighted average placebo responses of the sham-controlled groups across nine published surgical placebo-controlled trials were found to be 4.3 units, with 95% confidence interval (CI) of 3.1–5.6 on UPDRS (motor, off) 29 . Because this magnitude of improvement in PD patients has been established as a modest to moderate response 30 , we suggest that it probably would not exert a decisive influence on the large, clinically significant improvements from baseline in successfully transplanted groups postulated as approximately 30%–50% or >20 points on UPDRS (motor, off) 29 .
There is convincing evidence that placebo effects are causally associated with not only increased activity of dopaminergic neurons regulating motor functioning but are also involved in mediating the research participants’ expectations of reward31,32. However, the evidence for the association between improved motor functioning and neural imaging indicating increased dopamine activity in placebo-controlled groups has been obtained in the context of pharmacological trials 31 . The aims and the timelines at the endpoints of the symptomatic pharmacological treatments are quite different from those of confirmatory trials of surgical cell transplantation. Consistent with the earlier findings, the results of a recent meta-analysis of sham surgical procedures have not shown statistically significant and clinically meaningful benefits based on positron emission tomography (PET) scan assessments indicating the absence of structural reconstruction 29 .
RCTs are implemented to subtract the results of the control sham-operated group from active group to identify the true unbiased value of the efficacy of the intervention. A relevant question is why is it that in sham surgery-controlled trials, there are variable outcomes, some of which are successful in showing therapeutic benefit and others not? 11 . The reduced overall performance of some of the patients under sham surgery-controlled double-blind designs 31 may be attributed to the diminished potential contribution of patients in the transplanted groups to the effective integration of the dopaminergic grafts compared with open-label trials 33 .
A question which emerges from the above analysis is why the transplanted fetal cells generated marked functional benefits in some patients, while in other cases, the cells survived but there were no clinical improvements. To address this question, more evidence is needed from well-defined stem cell lines (good manufacturing practice) in well-characterized subpopulation of people living with PD. Recently, a number of new biomarkers for PD have been established, for example, an olfactory test combined with CSF-derived alpha-synuclein seed amplification assay that may help select a more well-defined group of patients with PD 34 .
Lessons From the Design and Implementation of Previous Clinical Trials
Currently, it is widely accepted that the neuropathology of PD extends beyond the loss of nigrostriatal projections 35 and encompasses multisystemic synucleinopathy of the human nervous system with a range of functional consequences that extend beyond the nigrostriatal system 36 . It follows that the reconstruction of the nigrostriatal system alone will not provide a cure, but if it is successful, will be of lasting clinical and life-changing benefit to people experiencing the disabling symptoms of PD.
In the past three decades, deep brain stimulation (DBS) has emerged as an effective and clinically adopted therapeutic strategy for controlling motor symptoms in PD 37 . Because DBS has emerged as an effective, evidence-based neurosurgical treatment for PD, it would serve as a more relevant comparator group than sham-operated controls when designing future trials for evaluating the real-life benefits of stem cell transplantation. A well-functioning health care system should offer a range of effective treatments that may not cure targeted chronic diseases but rather ensure the best possible quality of life for people living with disabling, progressive CNS disorders, such as PD 38 .
Most importantly, the development of a combination therapies acting in synergistic manner may ultimately yield the most favorable therapeutic outcomes 39 . For example, currently, the majority of patients undergoing successful DBS treatment may also receive drug treatment and rehabilitation as needed. There are many possible combinations, such as providing posttransplantation-targeted rehabilitation programs to facilitate the integration of grafted cells within the brain40,41. The challenge is to leverage the potential of stem cells to enhance the benefits previously observed in the transplantation of human fetal dopaminergic cells.
Rather than seeking a specific best practice treatment or a panacea, the alternative view is to develop of a range of available treatments for clinicians to address the needs of individual patients as determined by factors like age at diagnosis, severity or duration of the condition, cognitive and autonomic dysfunction, and financial resources of the patients and their communities. The optimal outcomes for the PD patient population can be best achieved by the development of treatment algorithms, similar to those used for pharmacological management of PD42,43. A realistic aim for stem cell–based research is to provide novel treatment approaches for subgroups of patients who are most likely to benefit from the long-term reversal of dopaminergic cell loss, improvement of motor symptoms, and ultimately, to provide clear, both objective and subjective evidence for enhanced quality of life 44 .
Summary and Conclusion
In summary, the current state of research on reconstructive therapies using cell transplantation for PD indicates that the degree and duration of benefits vary significantly among individual patients. The mixed results for an emerging complex therapy do not indicate that we should discontinue further exploration of cell transplantation; rather, they emphasize the need for more work to be done to improve conditions that promote stable functional connections of transplanted cells45,46. Figure 1, provides a summary of the review.
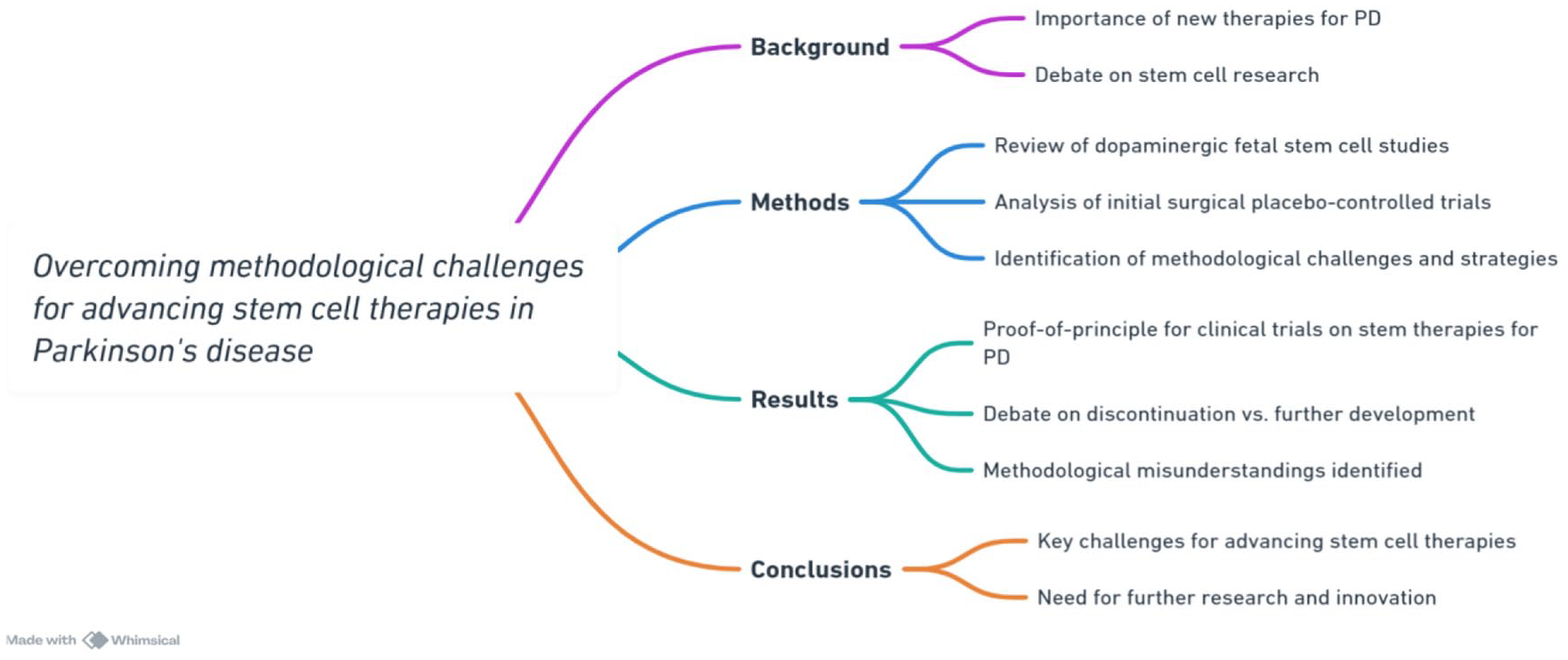
A summary fo the review.
The recent trial conducted by TRANSEURO (2019) illustrates the multiple challenges—ethical, logistical, financial, and professional dedication—confronting researchers undertaking clinical trials to evaluate the safety and efficacy of cell transplantation for PD 47 . Insufficient data and inadequate statistical power to analyze the trial results have contributed to mistaken conjectures such as calling for an end to stem cells research on the grounds that the previous trials with fetal mesencephalic cells were “futile”10,11.
While there is unequivocal evidence of placebo effects and responses in clinical trials evaluating the efficacy of pharmacological products for PD, the evidence emerging from the sham surgical arms of trials assessing the efficacy of cell transplantation remains disputed. A recent systematic review and meta-analysis of published phase 2 trials 29 found no convincing evidence for placebo effects and reported overall modest to moderate placebo responses, challenging the methodological necessity of imposing sham surgical procedures on patients with PD volunteering as trial participants.
The discussion in this review highlights the great challenges associated with the reconstruction of the human brain and suggests methodological approaches for translating the ground-breaking stem cell research discoveries into effective PD treatments. Table 2 presents some of the fundamental methodological challenges and possible strategies discussed in this article to resolve them. Overall, this review underscores the need for further research and innovation to overcome the hurdles in realizing the potential of stem cell–based therapies for PD.
Addressing the Methodological Challenges.

Footnotes
Acknowledgements
The authors are grateful for the assistance from Michele Bernshaw in editing and proofreading this article.
Authors’ Note
The authors confirm that this article has not been either previously published or submitted simultaneously for publication elsewhere.
Author Contributions
SP contributed to the literature review, framework formulation, structure and sequence of theoretical arguments, and reviewed the final form of the article. LK and DIF contributed to the sequence of theoretical arguments, the literature review, and reviewed the final form of the article.
Availability of Data and Material
All the papers reviewed in this article are publicly available.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
