Abstract
Xenografts of human skeletal muscle generated in mice can be used to study muscle pathology and to test drugs designed to treat myopathies and muscular dystrophies for their efficacy and specificity in human tissue. We previously developed methods to generate mature human skeletal muscles in immunocompromised mice starting with human myogenic precursor cells (hMPCs) from healthy individuals and individuals with facioscapulohumeral muscular dystrophy (FSHD). Here, we examine a series of alternative treatments at each stage in order to optimize engraftment. We show that (i) X-irradiation at 25Gy is optimal in preventing regeneration of murine muscle while supporting robust engraftment and the formation of human fibers without significant murine contamination; (ii) hMPC lines differ in their capacity to engraft; (iii) some hMPC lines yield grafts that respond better to intermittent neuromuscular electrical stimulation (iNMES) than others; (iv) some lines engraft better in male than in female mice; (v) coinjection of hMPCs with laminin, gelatin, Matrigel, or Growdex does not improve engraftment; (vi) BaCl2 is an acceptable replacement for cardiotoxin, but other snake venom preparations and toxins, including the major component of cardiotoxin, cytotoxin 5, are not; and (vii) generating grafts in both hindlimbs followed by iNMES of each limb yields more robust grafts than housing mice in cages with running wheels. Our results suggest that replacing cardiotoxin with BaCl2 and engrafting both tibialis anterior muscles generates robust grafts of adult human muscle tissue in mice.
Introduction
Generating grafts of human skeletal muscle cells in mice, or “xenografting,” provides an important preclinical tool for studying the in vivo biology of skeletal myopathies in a human context and for testing potential therapeutics. Xenografts may be especially useful in studies of genetic diseases for which murine models do not fully replicate the human condition, or if potential therapeutics have activities specific to human muscle. Several approaches to xenografting have been undertaken, from injecting suspensions of human myoblasts into healthy murine muscle to suturing large bundles of human myofibers into the mouse hindlimb1–10 (reviewed in previous studies2,11–16). Previously, we developed a method that involves damaging a significant portion of the tibialis anterior muscles of immunodeficient mice with a myotoxin, preventing murine muscle regeneration by X-irradiation, injecting immortalized human myogenic precursor cells (hMPCs) into the damaged muscle, and then promoting engraftment and maturation of the engrafted tissue with intermittent neuromuscular electrical stimulation (iNMES) 10 . These grafts typically contain hundreds of well-organized, mature muscle fibers of almost exclusively human origin that replicate the molecular and cellular features of the tissues from which the hMPCs were originally derived. In particular, we have used this approach to create mature human skeletal muscle tissue in mice that replicates the molecular and epigenetic profiles of muscles from individuals with facioscapulohumeral muscular dystrophy (FSHD) and from healthy controls10,16.
In the course of developing these methods, we tested a wide range of parameters, including the dosage of X-irradiation, the lines of immortalized hMPCs injected, how the cells were cultured prior to injection, what we injected together with the hMPCs, the sex of the engrafted mice, whether one or both hindlimbs were engrafted, whether mice carrying the xenografts were exposed to running wheels instead of iNMES, the frequency of iNMES, and the timing of several of these steps relative to one another and to the final collection of engrafted tissue. Because cardiotoxin from Naja mossambica, which we had previously adapted for our purposes10,16, ceased to be commercially available, we also tested other myotoxins as alternatives. Here, we report our findings, consider the advantages and disadvantages of our approach, and suggest additional steps that might further enhance engraftment.
Materials and Methods
Mice
Methods were as described by Sakellariou et al and Mueller and Bloch10,16. Immunocompromised, NOD. Cg-Rag1tm1Moml2rgtm-Wjl/SzJ mice (NOD-Rag, or NRG, originally from Jackson Laboratories, Bar Harbor, ME, and later bred at the University of Maryland, Baltimore). Mice were anesthetized with avertin (250 mg/kg) prior to X-irradiation. For all subsequent steps, mice were anesthetized with 2.5%–4.5% isofluorane and, for injections, treated with 5 mg/kg carprofen. All procedures were approved by the Institutional Animal Care and Use Committee, University of Maryland, Baltimore.
X-irradiation
The left hindlimbs or, when both hindlimbs were used for engraftment, both hindlimbs of young adult mice (8 weeks old) were subjected to a single, localized dose of X-irradiation, as described17,18. Briefly, mice were anesthetized with avertin (250 mg/kg) and placed within a lead restraint. One or both hindlimbs were extended beyond the lead container and exposed to X-irradiation at a single dose, which varied from 15 to 25Gy at 2.3Gy/min. Ionizing radiation was delivered with a Pantak-Seifert 250KpV X-ray Irradiator (bipolar series model HF 320, East Haven, CT) or an X-RAD 320 biological irradiation system (Precision X-ray, North Branford, CT). The two irradiators gave results that were statistically indistinguishable. The radiation beam was focused onto the lower hindlimb(s). Ion chamber dosimetry (PTW model 31006, Freiburg, Germany) was performed outside the collimator to ensure delivery of the exact dosage to the hindlimb, as well as inside the collimator (lead shielding), to monitor backscatter of radiation.
Cells (hMPCs) and Cell Injection
Immortalized hMPCs were prepared from biopsy samples of biceps muscle of patients carrying the FSHD-permissive 4A allele, their first-degree unaffected relatives or other healthy donors19,20,21, immortalized as described 22 , and expanded in culture prior to injection. Single clones of control and FSHD hMPCs were used in these studies. The 15A and 16A lines were derived from FSHD patients; 15V was derived from an unaffected sibling of the donor of 15A; 15B was from another sibling of the 15A donor who carried the same mutation but who did not show signs of disease (“non-manifesting”) 19 . A4, C6, and C12 lines were from a patient chimeric for FSHD 20 . Unless otherwise noted (see Results for details), cells were cultured in 75-mm tissue culture flasks in RoosterBasalTM-MSC medium (SU-005) with RoosterBoosterTM-MSC (SU-003), 15% fetal bovine serum, and 10-mM sodium pyruvate on 75-mm2 tissue culture flasks (Sarstedt, Germany). Lines 15A, 15V, and 16A were grown in flasks pre-coated with 0.1% gelatin. A4, C6, and C12 lines were grown in uncoated flasks. Cells were grown to 85% confluency, treated with trypsin, collected by brief centrifugation, resuspended in 60-µl medium or medium supplemented with extracellular materials (see text), and injected along the length of the tibialis anterior muscle (TA). Aliquots of 2 × 106 cells were injected per muscle. Additional details are provided in reference. 10
Intermittent Neuromuscular Electrical Stimulation
iNMES was performed 12 times as described 10 , except when 9 or 15 stimulation sessions were introduced, as in Fig. 7. In brief, mice were anesthetized with isoflurane, and their common peroneal nerves were stimulated electrically through the skin to induce contraction of the ankle dorsiflexors, in 4 sets of 10 contractions each. Each contraction lasted 500 ms (150-Hz pulse frequency), followed by a 500-ms rest. Each set of 10 was followed by rest times of 2 min. This procedure was performed three times/week for 4 weeks. Most samples were analyzed between 4 and 5 weeks after injection of hMPCs.
Tissue Collection
TAs carrying xenografts were collected at ≥4 weeks after engraftment. Mice were anesthetized and euthanized by cervical dislocation. TAs were dissected, weighed, flash frozen in liquid N2, and stored at −80°C. Tissue sections, 12- to 16-µm thick, were mounted on glass micro-slides. Cross-sections of the entire engrafted muscles were studied.
Immunofluorescent Labeling
To identify and count human myofibers and myonuclei, we used human-specific antibodies to h-β-spectrin and h-lamin A/C to label the sarcolemmae and nuclear envelopes of human fibers and myonuclei, respectively 10 . Sources of the antibodies have been reported 10 . Human myonuclei were identified as those that labeled with both 4’,6-diamidino-2-phenylindole (DAPI) and anti-h-lamin A/C. Murine myonuclei were identified as those that labeled with DAPI but not with anti-h-lamin A/C. Murine myofibers were identified as those that failed to label for human β-spectrin and human lamin A/C but that were outlined by labeling with fluorescent wheat germ or with antibodies to laminin. The failure of the human-specific antibodies to label murine myofibers and murine myonuclei serves as an internal negative control for the specificity of their labeling.
Imaging, Morphometry, and Quantification
Imaging was performed in the UMB Confocal Microscopy Core on an LSM-DUO (Carl Zeiss, Jena, Germany), Nikon CSU-W1 spinning disk (Nikon Instruments, Melville, NY) or an LSM-Meta (Carl Zeiss) confocal microscope. We used ImageJ or FIJI to measure the relative areas of each cross-section that were occupied by either mouse or human muscle fibers, as well as to quantify human fiber number, minimum Feret’s diameter, compaction (measured as fiber-to-fiber distances), and central nucleation manually, as described 10 .
Liquid Chromatography-Tandem Mass Spectrometry
We subjected a commercial batch of cardiotoxin from Sigma-Aldrich (St. Louis, MO, USA), which supported xenografting effectively (eg, Mueller and Bloch 16 ) to liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis, to identity its component proteins and peptides. Briefly, the cardiotoxin samples were analyzed on a high-resolution Orbitrap Fusion Tribrid mass spectrometer (Thermo Scientific Corp., San Jose, CA, USA) coupled to a nanoACQUITY UPLC system (Waters Corporation, Milford, MA, USA). Peptides were eluted using a 3%–40% acetonitrile gradient over 165 min at 400 nL/min. The eluted peptides were interrogated with a data-dependent acquisition method using a top-speed selection mode. Acquired tandem mass spectra were searched against the cardiotoxin reference sequences using a Sequest HT algorithm in its nonspecific cleavage mode with a maximum mass error tolerance of 10 ppm. The abundances of the cardiotoxin and its degradation products were estimated from the extracted-ion chromatogram.
Cytotoxins and Synthesis of Cytotoxin 5
We purchased the following snake vena and venom products: Naja pallida (Millipore-Sigma, St. Louis, MO, USA); Naja pallida, Naja mossambica, and Notexin (Latoxan, Portes-les-Valences, France); Naja kaouthia (Sigma-Aldrich, St. Louis, MO), and Notexin (Accurate Chemical, Carle Place, NY, USA). We also tested a recombinant form of cytotoxin 5 (cyto-5) from N. mossambica from MyBioSource (San Diego, CA, USA).
Inclusion of Extracellular Materials
We initiated engraftment by introducing hMPCs with extracellular materials, as follows. Geltrex (Gibco/ThermoFisher, Waltham, MA) was diluted 1:10 with cold tissue culture medium, and 30 μl was immediately injected into the proximal end of the TA. This was followed by the cell injection at the distal end of the TA. The TA was then massaged to spread the matrix and the cell suspension. Growdex (UPM, Helsinki, Finland) was diluted with media to 0.25%, and 25 μl of the dilution was added to the cell pellet and gently mixed prior to injection into the TA. For Matrigel (Corning Life Sciences, Glendale, CA) and laminin (Sigma-Aldrich), 25 µl of 10-mg/ml solutions in phosphate buffered saline (PBS) were added to the cell pellet prior to injection into the TA. Otherwise, injections were of compositions mentioned earlier.
Statistical Analysis and Data Access
We used unpaired t tests with Welch’s correction for statistical analyses of fiber number and TA weight. Tests of fiber diameter and compaction were analyzed with one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test. For non-parametric analyses, we used the Kruskal-Wallis test followed by Dunn’s multiple comparison. Additional statistical methods are given in the text. All statistical analyses were performed with GraphPad.
Data generated in these studies are available upon request.
Materials
Materials were from Sigma-Aldrich (St. Louis, MO) and ThermoFisher (Waltham, MA) unless otherwise noted.
Results
Our published procedures for xenografting10,16 utilize an immunodeficient strain of mice, Nod-RAG or NRG, and involve several steps: (i) X-irradiation of the mouse hindlimb, to prevent subsequent regeneration of murine muscle; (ii) injection of a myotoxin to eliminate part of the TA muscle; (iii) injection of hMPCs, previously grown in tissue culture; (iv) electrical stimulation of the graft intermittently, to promote its development and maturation. Here, we examine each of these parameters in more detail.
25Gy X-Irradiation Minimizes Murine Muscle Regeneration and Murine Myonuclear Contamination of Human Fibers
Previous studies that involved X-irradiation of murine muscle to enable engraftment indicated that doses of 15 or 18Gy were required to suppress myogenesis of the endogenous murine muscle (eg, see the works of Gomez and Love; 22 Alameddine et al; 23 and Wakeford et al 24 ). As the sensitivity of myogenesis to X-irradiation has not been studied in NRG mice, we tested doses of 15, 18, 20, and 25Gy, delivered at a rate of 2.3Gy/min. We then followed our standard procedures for xenografting and, after a month, collected the engrafted tissue and counted central nuclei in both murine and human myofibers. Fibers labeled with antibodies to human β-spectrin containing nuclei labeled with human lamin A/C were deemed to be human; fibers that failed to be labeled with both antibodies were deemed to be mouse. Fibers labeled with anti-human β-spectrin but with some nuclei that failed to be labeled with anti-human lamin A/C were recorded as human fibers with murine myonuclear contamination. Fibers that failed to be labeled with anti-human β-spectrin but were labeled by anti-human lamin A/C were not observed. Central nuclei were apparent at elevated levels in murine myofibers at all X-ray doses less than 25Gy (Fig. 1A), indicating that 25Gy was needed to block murine myogenesis completely in NRG mice. By contrast, human central nuclei were present in human myofibers at all doses and at considerably higher levels than murine central nuclei (Fig. 1B). Notably, central murine myonuclei in human fibers remained at minimal levels at doses of 25, 20, and 18Gy and increased to ~2% at 15Gy (Fig. 1C). Our results suggest that a dose of 25Gy is needed to minimize the presence of central murine myonuclei in mouse myofibers, whereas 18–20Gy of X-irradiation ensures minimal contamination of human myofibers with murine myonuclei but not suppression of murine myogenesis. We therefore use 25Gy X-irradiation before engraftment.
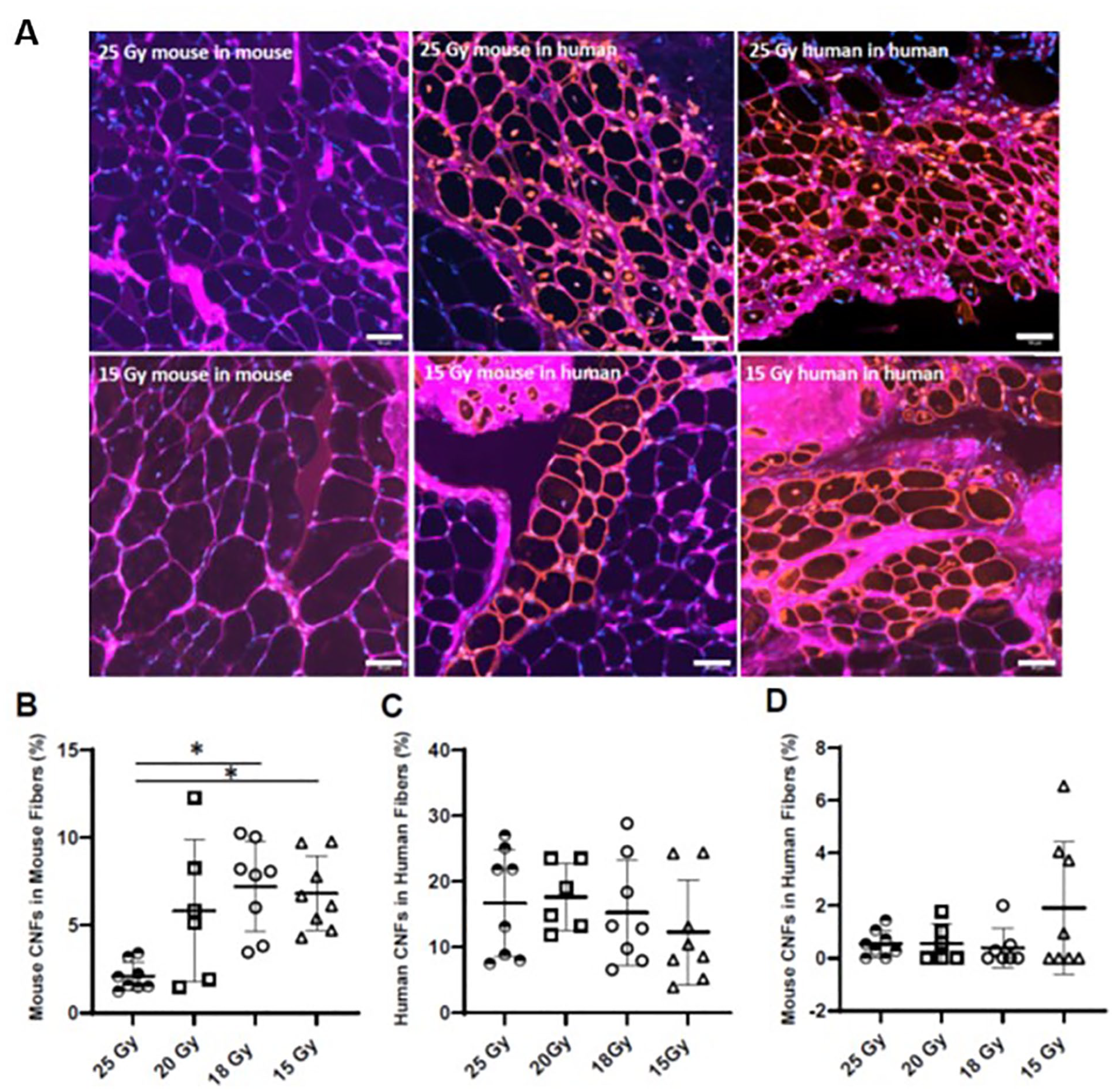
Effect of X-ray dosage on engraftment. NRG mice were subjected to X-irradiation at the doses indicated, followed by BaCl2 intoxication 2 days later and by injection of 2 × 106 15A cells into both hindlimbs 2 days later. All engrafted legs were treated for 4 weeks with iNMES. Engrafted muscles were then removed and analyzed for the number of human fibers, identified by labeling with antibodies to human β-spectrin and human lamin A/C. Although green fluorescence was expected for human-specific label of the sarcolemma, the color appears as reddish-orange, due to colabeling with antibodies to laminin or with wheat germ agglutinin. Murine myofibers, identified by the absence of human-specific labeling but the presence of labeling by antibodies to laminin or by wheat germ agglutinin are outlined in purple. Connective tissue in both human and murine regions of the engrafted muscle also appears in purple. Human myonuclei were identified as those that costain with DAPI (blue) and anti-h-lamin A/C, which appears orange in the four panels to the right. Murine myonuclei were identified as those that stain only with DAPI, which appear blue in the two panels to the left and in the murine fibers in the other panels. (B) The number of murine myofibers with central murine nuclei (CNF). (C) The number of human fibers with human central nuclei. (D) The number of human fibers with murine central nuclei. Representative images are shown in A. Each point in panels B–D represents an individual xenograft, and the entire engrafted muscle was examined. The results show that a dose of 18Gy is sufficient to minimize the contamination of human fibers with murine myonuclei, but that 25Gy is needed to suppress regeneration of murine fibers fully, as indicated by the near absence of murine fibers with murine central nuclei. *P < 0.05. Scale bars, 50 μm.
Male Mice May Be Universal Graft Hosts
As we reported earlier10,16, the sizes of the xenografts that we could generate varied widely, from only a few dozen fibers in some mice to many hundreds in others. We examined our results to learn if male or female mice were more receptive to engraftment. The data in Fig. 2 show the results of engraftment of 15A, 15B, and 15V hMPCs into the TA muscles of male and female mice that had been irradiated at 25Gy and intoxicated with N. mossambica cardiotoxin from Sigma-Aldrich (NMC-S), as described10,16.
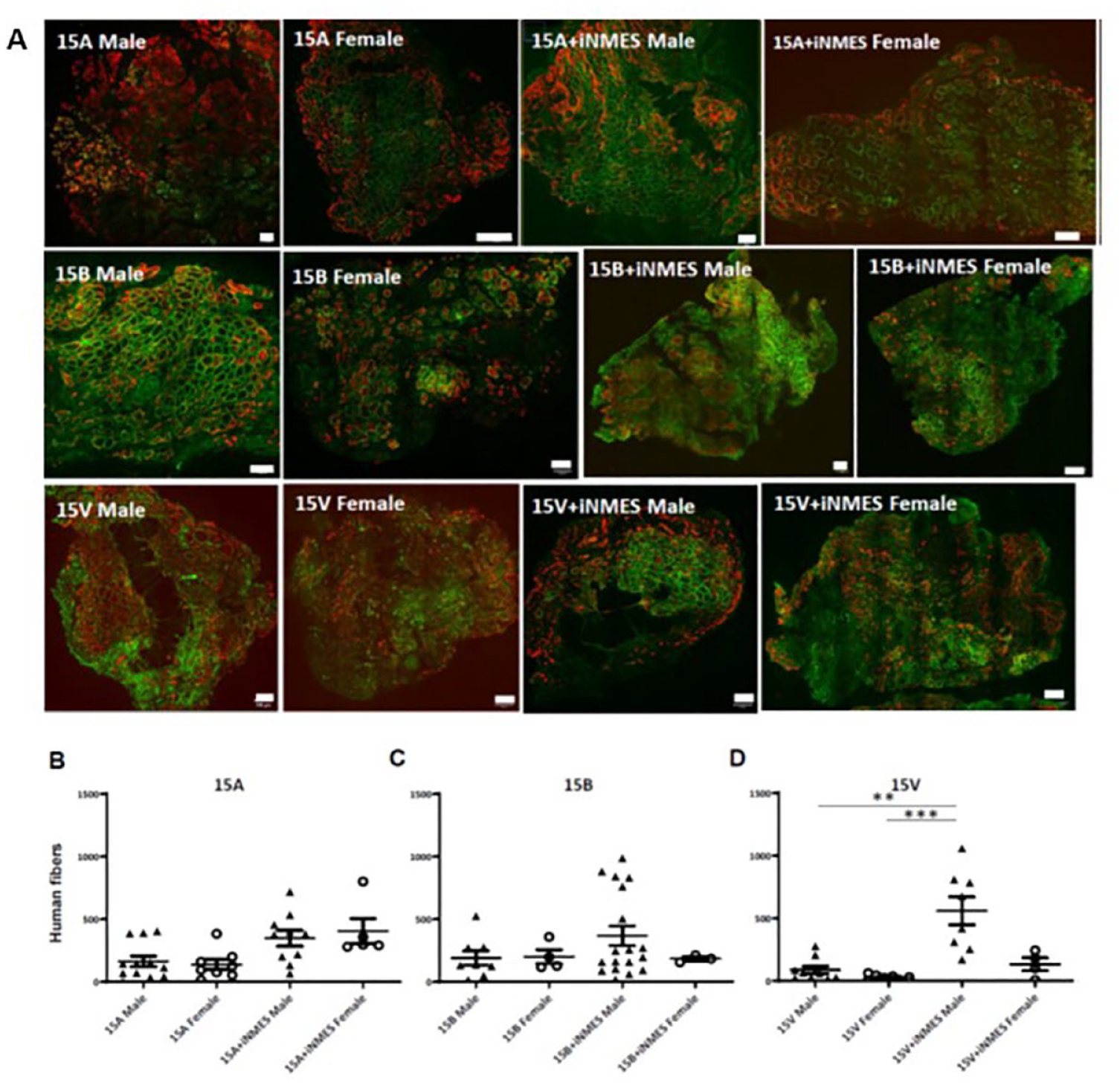
Effect of sex of the host mice on engraftment. Male and female NRG mice were dosed with 25Gy X-irradiation, intoxicated with NMC-S, and injected with either 15A (B), 15B (C), or 15V (D) cells in one hindlimb only. Some mice were treated over the following 4 weeks with iNMES; others were not, but otherwise samples were prepared and processed as in Fig. 1. Muscles were then collected, and the number of human myofibers in the grafts was quantified. Representative images are shown in A. Each point in panels B–D represents an individual xenograft, and the entire engrafted muscle was examined. The results show that 15A cells engraft equally well in male and female mice, whereas 15V cells do better in males than in females treated with iNMES. **P < 0.01; ***P < 0.001. Scale bars, 100 μm.
Although the 15A cells did not show significant differences, the 15V cells engrafted better in male mice, especially when the grafts were subjected to iNMES. Grafts with 15B cells showed no significant difference when engrafted into male vs female mice. (NB: 15A and 15B cells were from male donors; 15V was from a female donor). These results suggest that male mice are more likely than females to be universal graft acceptors. We therefore use male mice as the graft recipients in most of our studies.
BaCl2, But Not Other Myotoxins, Is as Effective as Cardiotoxin NMC-S
During the course of our work, Sigma-Aldrich ceased supplying their preparation of cardiotoxin. We invested a significant effort in identifying a replacement by testing the ability of several different commercial myotoxins to induce the loss of murine muscle fibers and then to support engraftment. We used 15V cells for these studies, treated both hindlimbs after 25Gy irradiation, injected the myotoxins into the hindlimbs of male NRG mice irradiated at 25Gy, and after injecting the cells, subjected the hindlimbs to iNMES. We compared different commercial snake vena and venom products, including Naja pallida cardiotoxin from Millipore (NPC-M) and Latoxan (NPC-L) and Notexin (Accurate Chemical, NY, USA; see Table 1), to the NMC-S (Fig. 3A). We also tested BaCl2 (Fig. 3B), which has been used by other groups for muscle xenografting (eg, Charville et al 25 ). Although we have routinely injected 18-µg NMC-S into both TA muscles, comparable concentrations of NPC-M and NPC-L were lethal to NRG mice within 24 h (data not shown). BaCl2 administered at concentrations >2%, at 50 µL/TA, was also lethal (not shown). We therefore tested each toxin over a range of lower concentrations.
Myotoxins Assayed.
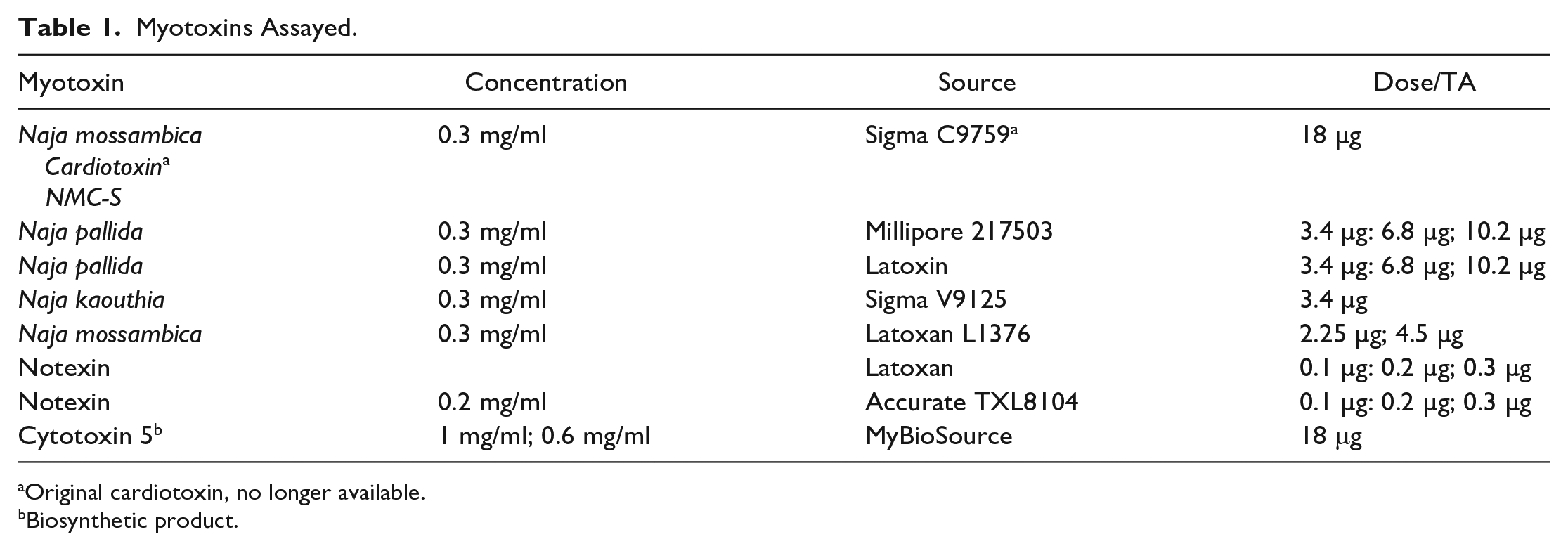
Original cardiotoxin, no longer available.
Biosynthetic product.
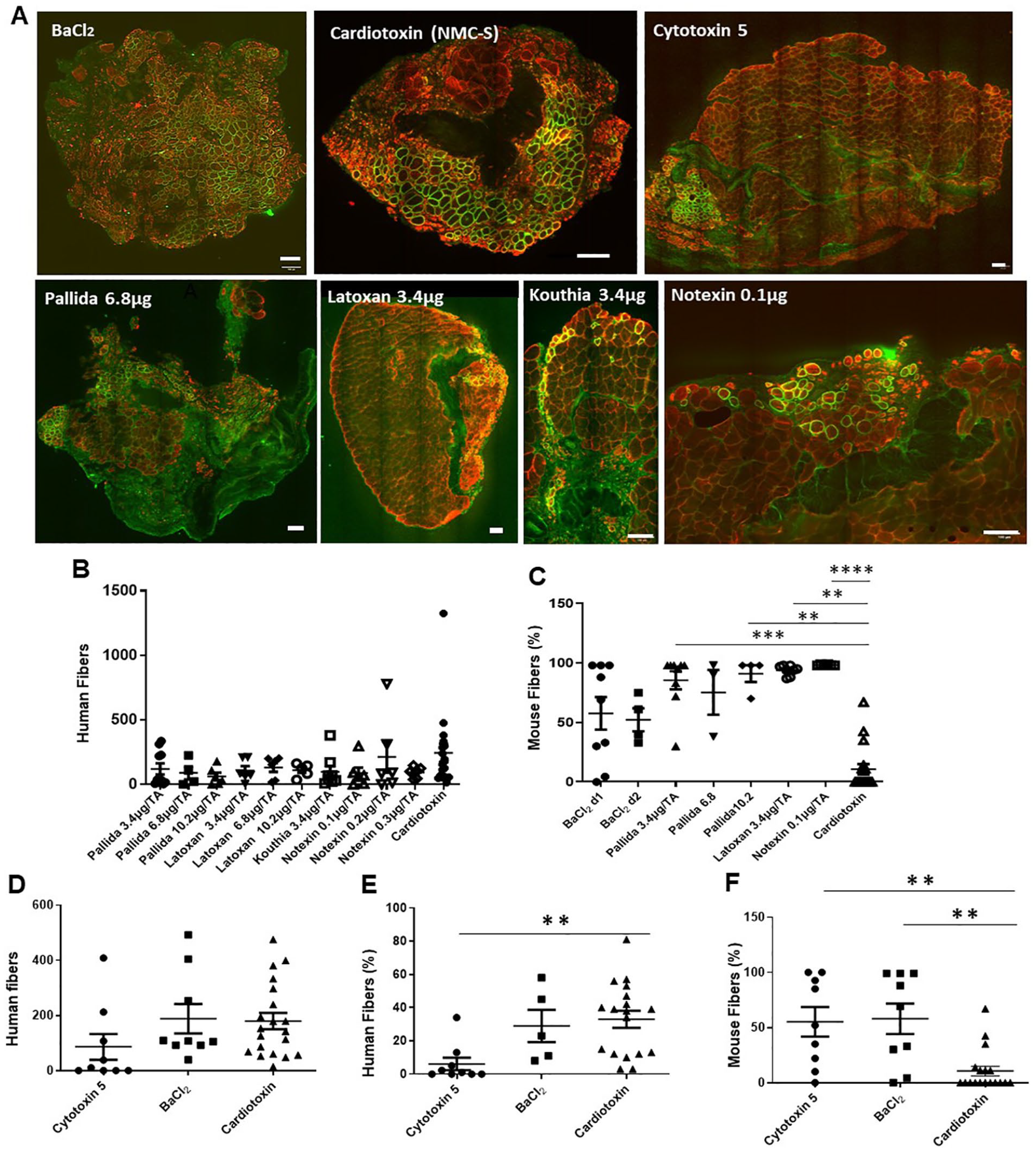
Effects of myotoxins on engraftment. Myotoxins from different sources were compared to NMC-S and BaCl2. 15V cells were used to prepare all grafts, as in Fig. 1. Panels B and C show results obtained with commercially available toxins and vena (see Table 1). Panels D–F compare results of engrafting 15V cells with a biosynthetic form of cytotoxin 5, the major component of the commercial cardiotoxin preparation, NMC-S, to BaCl2. Human fibers were counted in panels B and D, and the percentage of human fibers in the engrafted mouse muscle was determined in panel E. Panels C and F show the percentage of mouse muscle fibers remaining in the engrafted TA compartment. Representative images are shown in A. Each point in panels B–F represents an individual xenograft, and the entire engrafted muscle was examined. The results show that, although it was more variable in our hands than NMC-S and was less effective in eliminating murine myofibers, BaCl2 was statistically equivalent to NMC-S in supporting engraftment. **P < 0.01.
We found that none of the commercial myotoxins were as capable as NMC-S or BaCl2 of eliminating endogenous murine myofibers, assayed on day 2 after injection, while NMC-S may be slightly more effective in this regard than BaCl2 (P < 0.19; Fig. 3A, C). The differences between NMC-S (the Sigma-Aldrich cardiotoxin) and BaCl2 did not reach statistical significance, however. Consistent with the presence of more murine tissue and therefore less volume available for engraftment in the injected TA muscles for subsequent engraftment, the 15V grafts that developed after injection of these other vena and toxins were small and contained highly variable numbers of human fibers (Fig. 3B). In general, those myotoxins that were less effective in promoting engraftment were also less effective in eliminating the endogenous murine muscle fibers (data not shown). We conclude that, at least under these conditions, these myotoxins are not as effective as NMC-S, whereas BaCl2 is an effective replacement.
We next performed mass spectrometric analysis of NMC-S to identify its chief components. We found that, of the 4 intact polypeptides and the many peptide fragments that comprised the commercial toxin preparation, cyto-5 was by far the most abundant (Table 2), with relatively little degradation or enrichment in particular peptide fragments (Table 2; Supplemental Table 1). We tested two different commercial preparations of pure cyto-5 to learn if the pure, biosynthetic product could replace the commercial NMC-S. Our results showed that the synthetic toxin preparations did not eliminate NRG muscle any better than BaCl2 (Fig. 3F). Moreover, when we tested the commercial cyto-5 preparations to learn if they were as effective as BaCl2 in supporting the formation of human myofibers following engraftment of 15V cells), the cytotoxin was much less able to support engraftment, measured either as the absolute number of human fibers in the graft or the percent of human fibers in the engrafted hindlimb (Fig. 3D, E). Thus, cyto-5 from a commercial source is also less effective than either BaCl2 or NMC-S.
Cytotoxins in Commercial Cardiotoxin.
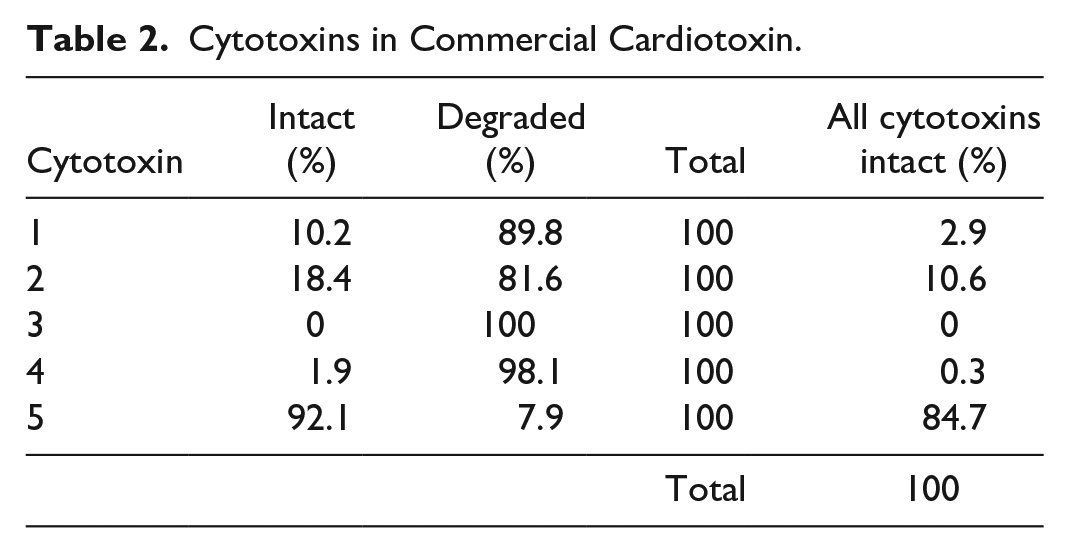
Finally, we compared different regimens for injecting cells with BaCl2: injecting from 1 to 3 days, injecting the BaCl2 twice, on day 1 and again on day 3 after intoxication, and increasing the BaCl2 dose slightly, from 2% to 2.5%. (We did not go higher, as such doses can be lethal). Only changing the day of BaCl2 injection to day 3 showed a statistically significant decrease in engraftment of 15A cells followed by iNMES (Fig. 4). 15V cells with and without iNMES sowed no significant differences when the days after BaCl2 were compared. Thus, the time frame in which we inject cells following BaCl2 may need to be adapted for each cell line and how it is treated subsequently.
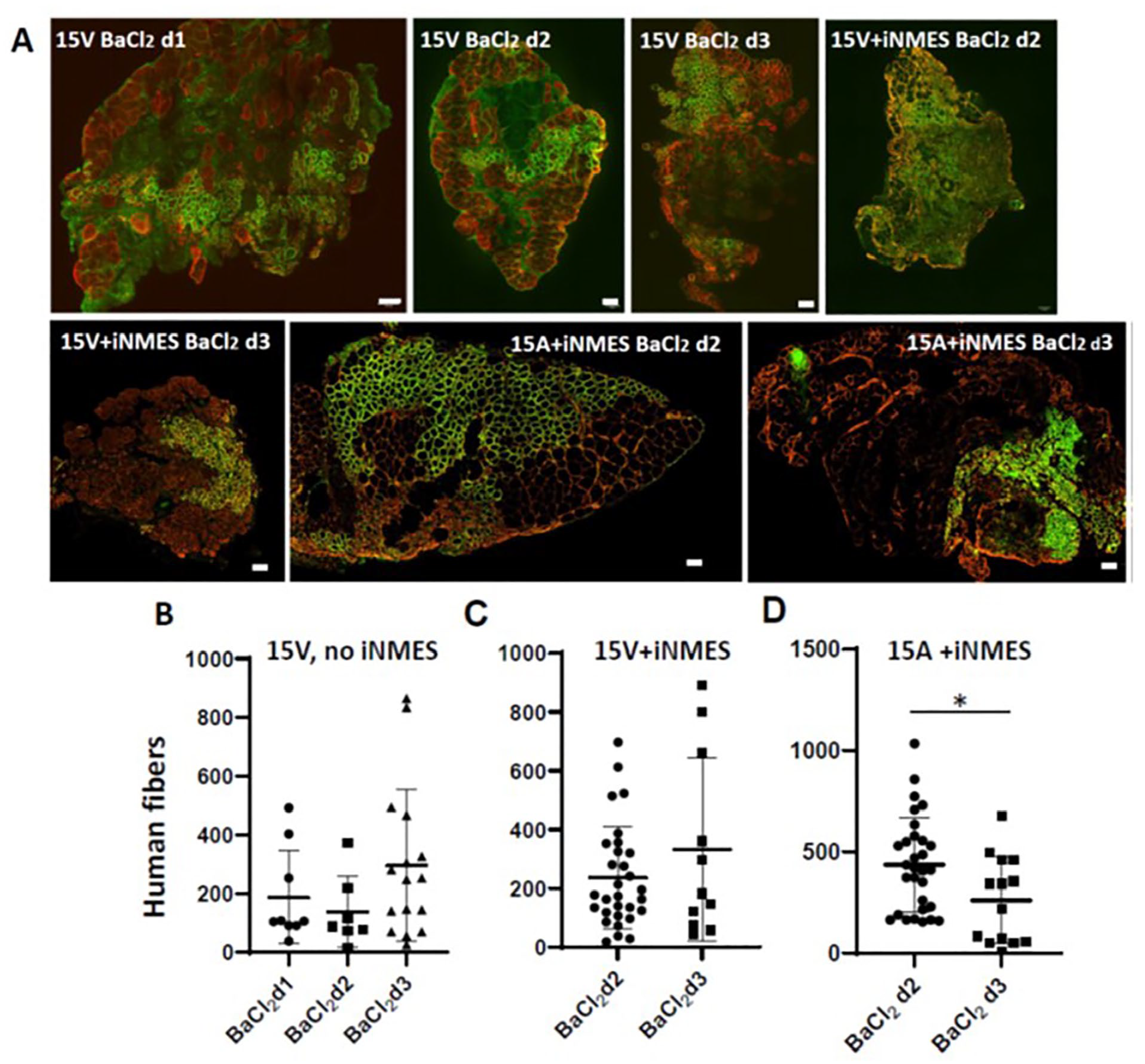
Effect of initiating engraftment at different times after BaCl2 treatment. Male NRG mice were dosed with 25Gy X-irradiation and 2 days later injected with BaCl2. At different times thereafter, the TA compartments were injected with either 2 × 106 15V (B, C) or 15A (D) cells. iNMES was applied as indicated. Representative images are shown in A. Please refer to Fig. 2 for images of a 15A graft initiated 2 days after injection of BaCl2. Each point in panels B–D represents an individual xenograft, and the entire engrafted muscle was examined. The results show no significant differences between d1 and d2. Scale bars, 100 μm.
Other Cell Lines and Different Culture Conditions Yield Grafts of Different Sizes
We previously reported on the engraftment of LHCN cells and of two cell lines obtained from members of a family with FSHD, 15V (healthy control donor) and 15A (FSHD donor)10,16. Here, we compared 15A and 15V further and also examined four other hMPC lines, 16A, A4, C6, and C12 (see Materials and Methods), to test their ability to engraft after intoxication with BaCl2, with and without iNMES. We injected TA muscles with 2 × 106 cells of each line in volumes of 60 µl. The results (Fig. 5A) show that the number of fibers of human origin in the grafts was greatest with the 15A, 15B, and 15V grafts. Fewer human fibers formed with 16A, C6, or C12 cells. These results indicate that different cell lines engraft to different extents and that the four cell lines that were less able to engraft were also unresponsive to iNMES.
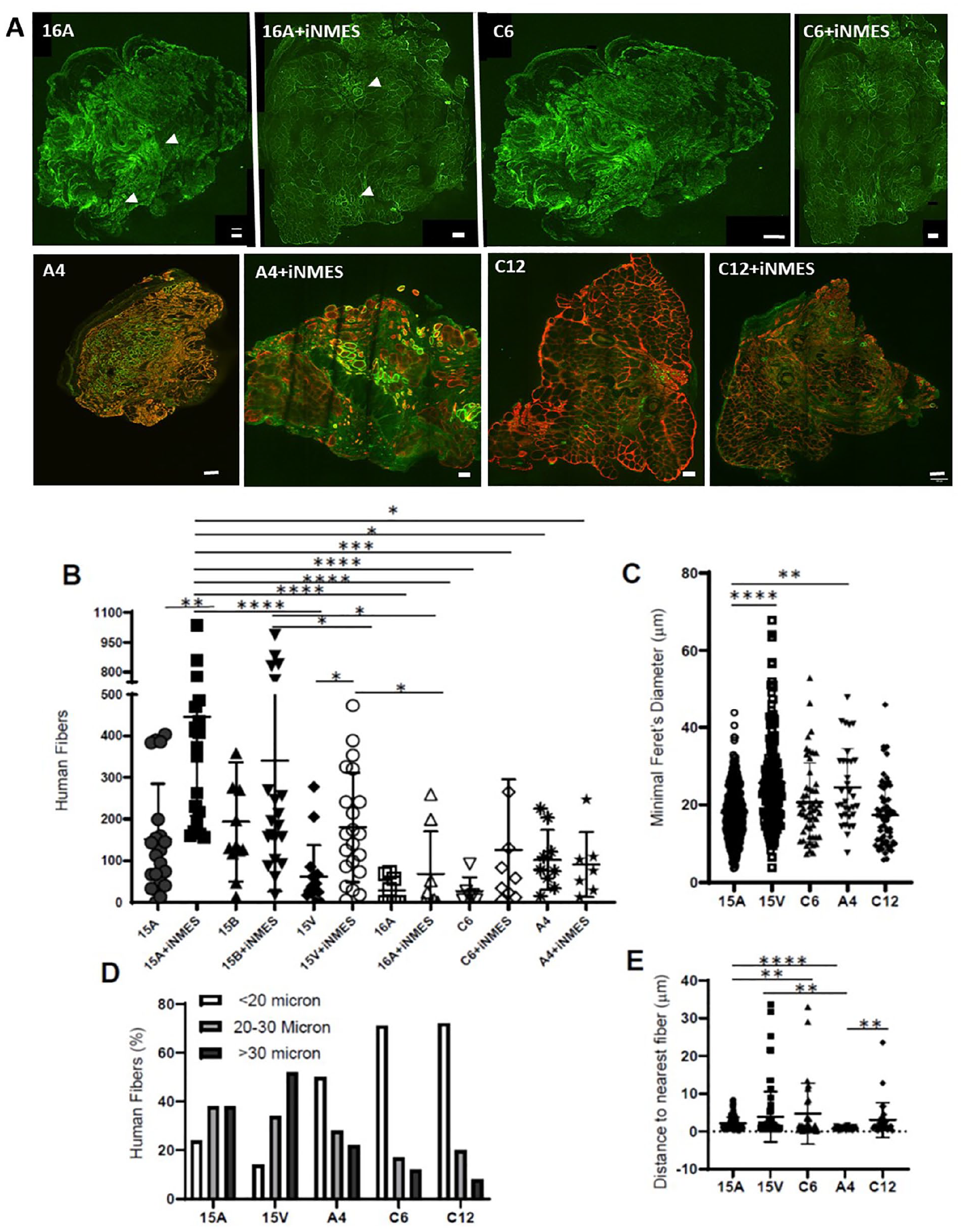
Comparison of grafts formed by different cell lines. Mice treated as in Fig. 1 with 25Gy X-irradiation were engrafted with one of several different cell lines, at 2 × 106 cells/injection. Some mice were subjected to iNMES for 4 weeks; others were not. Human fibers were then analyzed for number (B), minimal Feret’s diameter (C), distribution of diameters (D), and closest fiber-to-fiber distances (E). Representative images are shown in A. (N.B.: The image quality of the 16A grafts was poor, so only the green channel, showing outlines of the human fibers [white arrowheads], is shown for these two panels. The green fluorescence in A4+iNMES panel of A, representing labeling by human-specific antibodies, appears greenish-yellow.) Please refer to Fig. 2 for images of 15A and 15A+ iNMES. Each point in panels B and E represents an individual xenograft, and the entire engrafted muscle was examined. The results show that 15A, 15V, and 15B form the grafts with the largest number of human fibers and that iNMES promotes the formation of more fibers with some cell lines but not several others. Furthermore, the grafts that contain the largest number of human fibers also have the largest number of large-diameter fibers that are more closely packed together. *P < 0.05; **P < 0.01; ***P < 0.001; ***P < 0.0001. Scale bars, 100 μm.
Comparison of the properties of the fibers in the grafts examined in Fig. 5A shows further differences between grafts in terms of fiber diameter (Fig. 5B, C), as well as the interfiber spacing (Fig. 5D). As with fiber numbers, the 15A and 15V lines generated grafts that were composed of the largest fibers with the closest spacing and so more closely resembled mature skeletal muscle tissue than the grafts formed by the other cell lines.
Mode of Injecting hMPCs or Access to Running Wheels Has No Significant Effect
We also tested several variations of our standard injection procedures, which are to wait 2 days after intoxication of the TA of the left hindlimb and inject 2 × 106 hMPCs in a single bolus along the length of the muscle. We doubled the number of injections over a period of days and tested the potential benefits of engrafting both hindlimbs rather than only one, with single or multiple injections, using a single needle for both hindlimbs or one needle per TA, the possible benefit of using a different gauge needle for the IM injections (28 g vs the 27 g needle that we have used routinely), or maintaining the engrafted mice in cages with running wheels to increase the sizes of the human xenografts formed by 15V cells. The results (Fig. 6) show that the cells engrafted approximately equally well under all conditions. Nevertheless, to optimize our yield of grafts and minimize our use of mice, we now routinely inject both legs, although with only a single bolus of hMPCs. We found no significant differences when we varied the gauge of the needle used for injection, and so we continue to use the 27-g needle for this purpose.
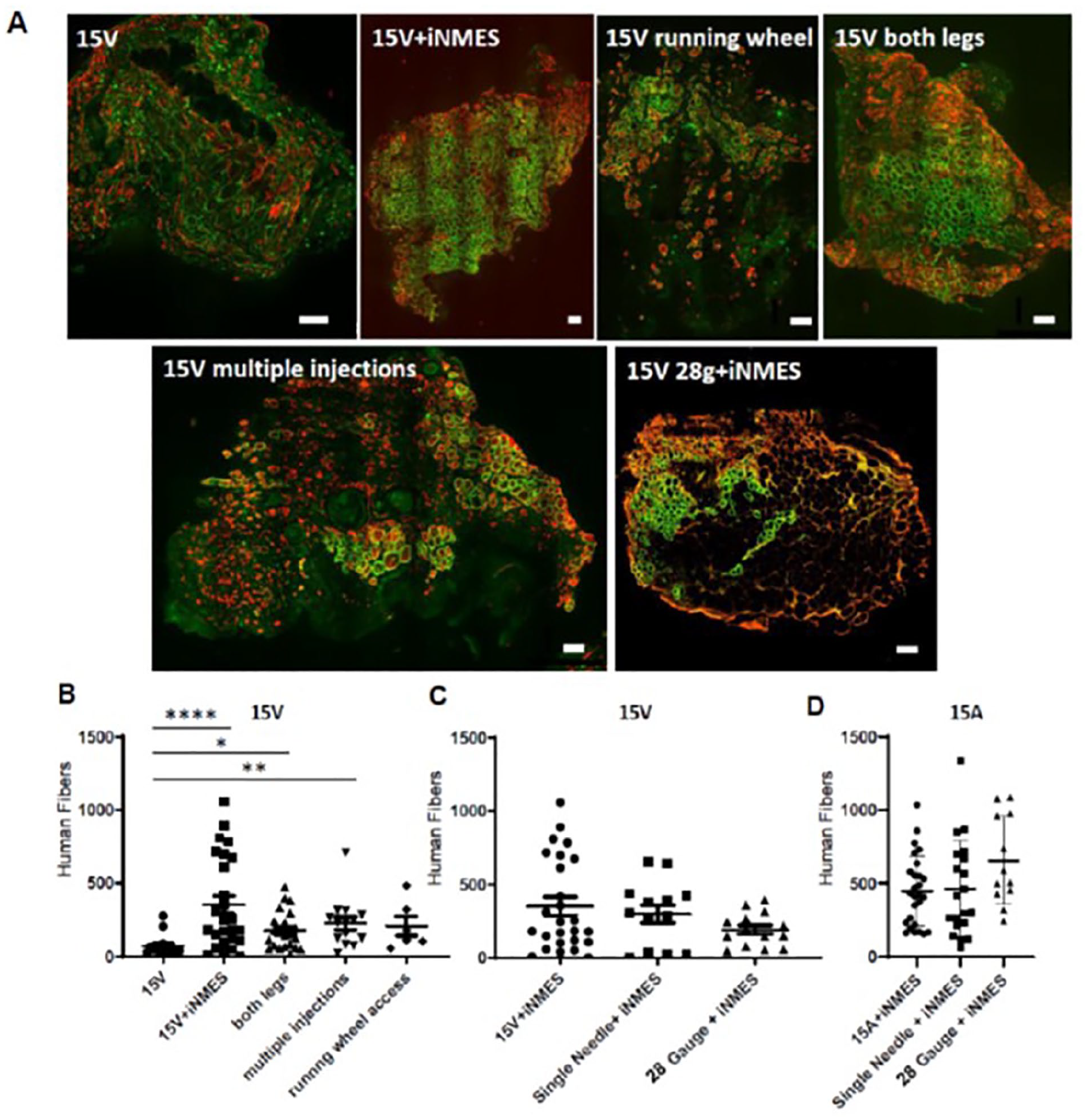
Effects of varying injection protocols and caging on engraftment. Mice treated as in Fig. 1 with 25Gy X-irradiation were engrafted with either 15V (B) or 15A (C) cells under different conditions. 15V grafts were either treated with iNMES or not treated; instead they were treated normally, injected in both hindlimbs, injected 1/week for 4 weeks into the same hindlimb, or injected into mice that were given ad libitum access to running wheels. Alternative modes of injection of both 15V and 15A cells were tested to learn if the use of a larger gauge needle (28g vs 27g) or pooling cells to ensure that a single needle was used for a single injection per TA muscle (“single needle”) yielded larger grafts. Representative images are shown in panel A. The occasional spot of yellow is due to overlap of the red and green fluorescence. Each point in panels B–D represents an individual xenograft, and the entire engrafted muscle was examined. The results show that none of the alternative methods we tested gave grafts as robust as those obtained with iNMES but that several tended to improve the size of 15V grafts when iNMES was not applied. *P < 0.05; **P < 0.01; ***P < 0.0001. Scale bars, 100 μm.
Nine to 12 iNMES Sessions Yield Grafts of Similar Sizes
Our results, explained earlier, show that iNMES increases the ability of some cells to engraft and mature in the mouse hindlimb. As iNMES is time-consuming and requires that mice be periodically anesthetized, we first tested if three sessions of iNMES weekly were needed to promote engraftment. We found that iNMES for a total of 9 or 12 sessions showed no significant difference in the xenografting of either 15A or 15V cells (Fig. 7A, B; NB: both protocols yielded grafts with significantly more 15A myofibers than grafts that were not subjected to iNMES, however).
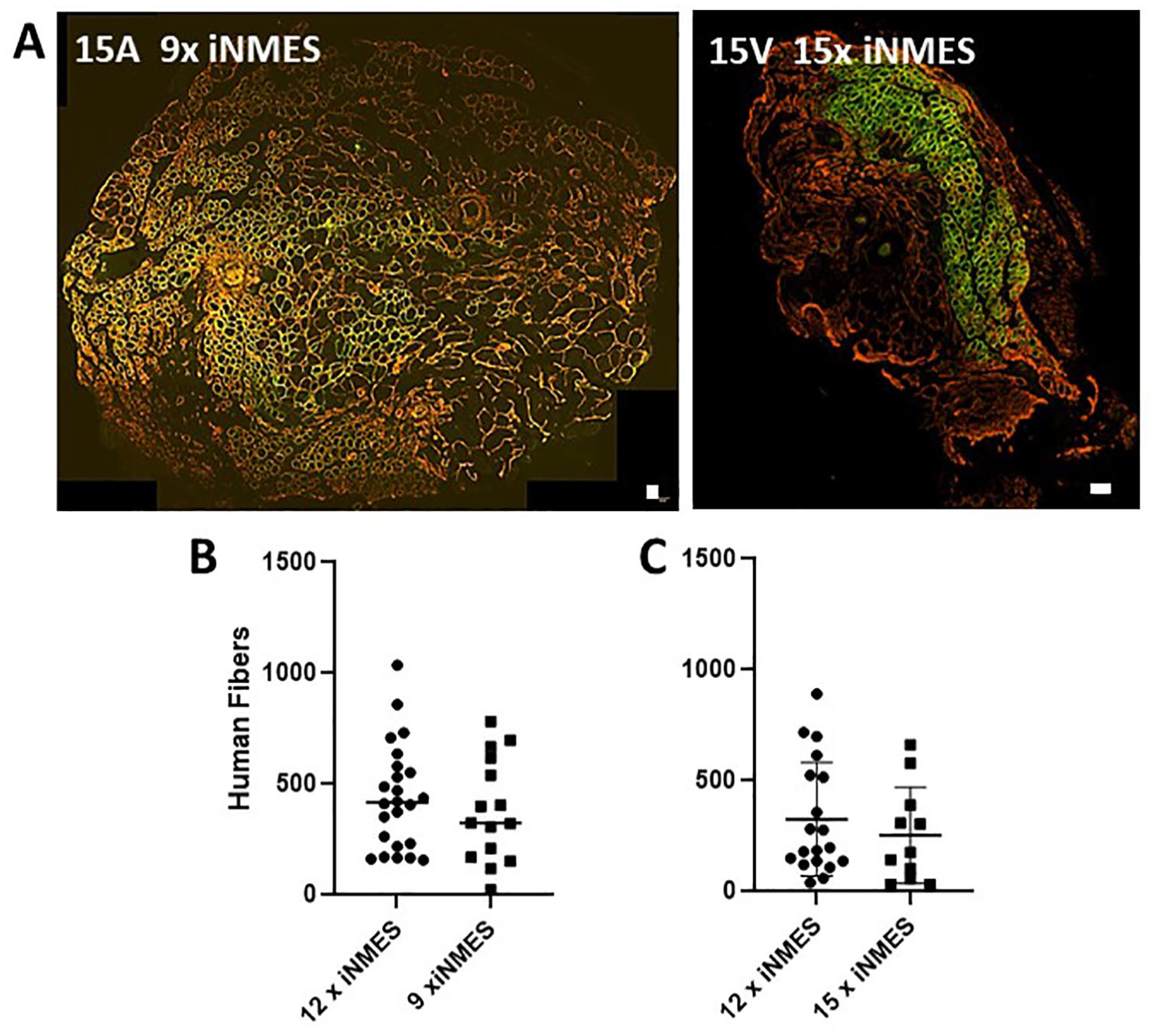
Effect of varying the number of iNMES sessions on engraftment. Mice treated as in Fig. 1 with 25Gy X-irradiation were engrafted with either 15V (B) or 15A (C) cells and then subjected to 9, 12, or 15 bouts of iNMES. Human fibers per graft were then counted. Representative images are shown in panel A. Please refer to Fig. 2 for images of 15A and 15V with 12 sessions of iNMES. (NB: The green fluorescence in the left panel of A, representing labeling by human-specific antibodies, appears greenish-yellow.) Each point in panels B and C represents an individual xenograft, and the entire engrafted muscle was examined. The results show no significant differences. Scale bars, 100 μm.
Co-injection of Extracellular Materials Does Not Promote Engraftment
Finally, we considered the possibility that grafts were limited and variable in size due to a paucity of sites into which the hMPCs could embed. We tested this by suspending the hMPCs in media containing laminin, Geltrex (a basement membrane preparation), gelatin, or GrowDex, a hydrogel. None of these additives improved engraftment of A4 and 15A cells, compared to engraftment followed by iNMES, but Matrigel may have beneficial effects on engraftment by C6 cells (Fig. 8).
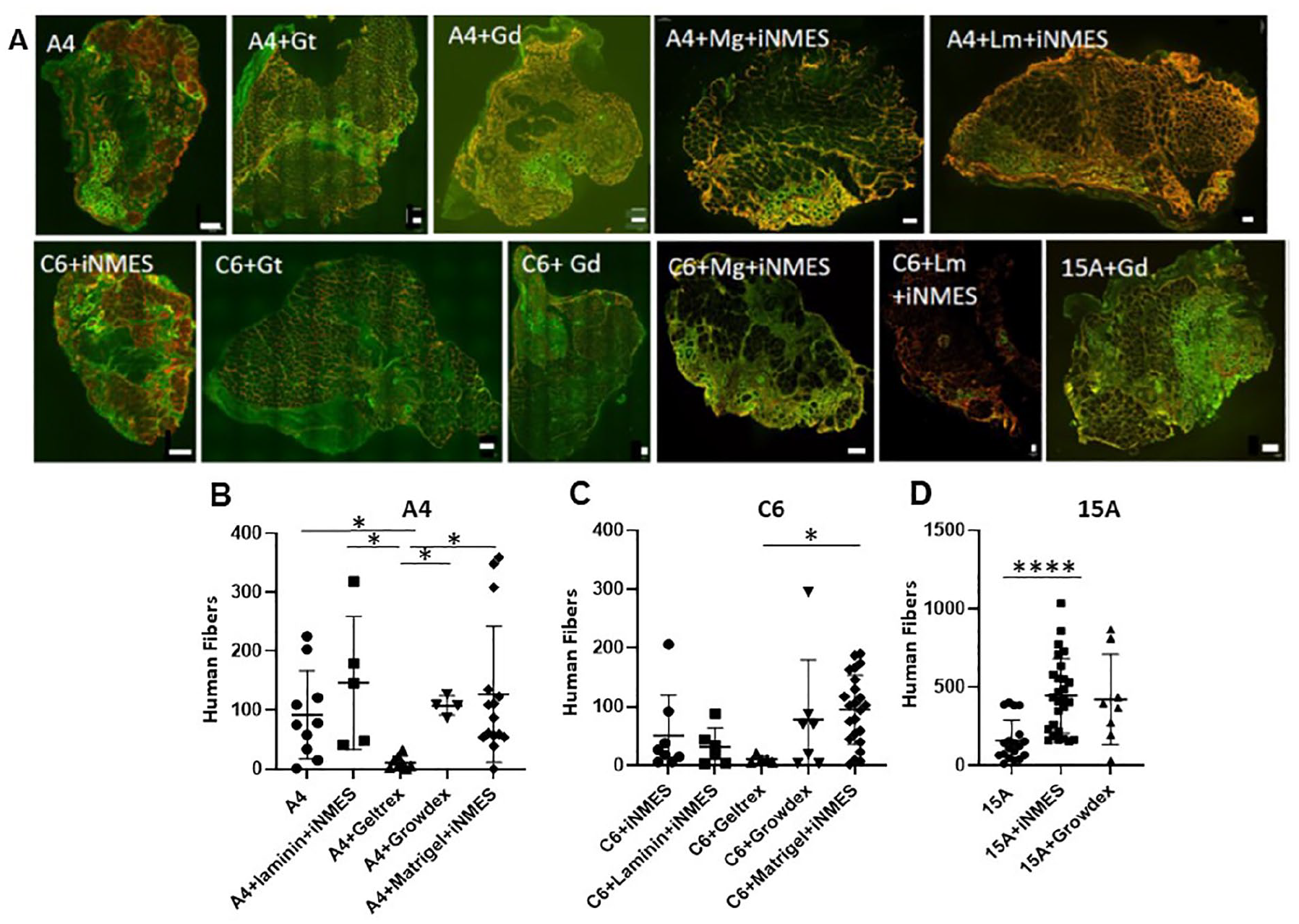
Effect of adding extracellular materials to grafts formed by A4, C6, and 15A cells. Mice treated as in Fig. 1 with 25Gy X-irradiation were engrafted with either A4 (B), C6 (C), or 15A (D) cells, supplemented with Growdex (Gd), Geltrex (Gt), laminin (Lm), or Matrigel (Mg), as described in Materials and Methods. Some grafts were subjected to iNMES, as indicated. After 4 weeks, human fibers per graft were counted. Representative images are shown in panel A. Please refer to Fig. 2 for images of 15A and 15A+ iNMES. The regions appearing yellow are due to overlap of the red and green fluorescence. Each point in panels B–D represents an individual xenograft, and the entire engrafted muscle was examined. The results show that none of the treatments significantly improve engraftment of A4, C6, or 15A cells and that Geltrex significantly inhibits engraftment of A4 and C6 cells. *P < 0.05. Scale bars, 100 μm.
Discussion
We performed these studies to optimize our methods for generating xenografts of mature human muscle tissue in mice for different hMPC lines and to minimize the considerable effort required with each mouse to succeed. We examined different cell lines, male vs female host mice, a range of doses of X-irradiation, several methods of cell culture and injection, different myotoxins, and different protocols for treating the mice after injection. Some of our experiments were necessitated by the termination of production of a cardiotoxin previously commercially available, NMC-S10,16. Our results show that xenografts generally are more robust when the hosts are male mice, their hindlimbs are X-irradiated at 25Gy, treated with BaCl2 as the myotoxin, engrafted with a single bolus of 2 × 106 hMPCs, and subjected to iNMES three times per week for 4 weeks.
Factors Affecting Engraftment
Although these parameters closely resembled those we described in our earlier papers10,16, we were surprised to discover that an important factor in determining the success of engraftment may be the sex of the host, as we found that grafts of 15V cells were much more robust in male mice than in female mice. Although these results led us to use only male mice in subsequent studies, we do not know how broad this sex-dependent difference is. Engraftment of other cell lines, like 15A, does not show the same benefits of using males over females. Although other cell lines may yield more promising results with females, the ability of the 15V cells to engraft more effectively into male mice is similar to earlier observations on xenotransplantation of some tumor cells, in which males supported more robust engraftment than females26–28, perhaps due to the expression by the engrafted cells of androgen receptors 27 . The 15V cell line was from a female donor 20 , but we do not know if it expresses androgen receptors.
Yet another factor that can affect engraftment is whether cells are injected as a free suspension or suspended in a matrix or hydrogel. Suspending cells in a commercial basement membrane preparation, GelTrex, in other extracellular materials, or in Growdex, a hydrogel, appears to reduce the engraftment of some cell lines, such as C6 and A4, that normally engraft less efficiently than 15A of 15V, whereas Matrigel may promote engraftment of C6. Our results with these materials contrast with the beneficial effects that similar materials have on engraftment of satellite cells 29 and cancer cells (reviewed in Benton et al 30 ). We speculate that they may reduce engraftment by curtailing the ability of hMPCs to spread from the sites of injection to the sites in the muscle where they must embed to grow and mature into muscle.
The extent to which muscle and hindlimb tissue are damaged prior to hMPC injection may also affect subsequent engraftment. Our hypothesis when we began these studies was that the more the damage the endogenous murine skeletal muscle experienced upon intoxication, the poorer the subsequent engraftment would be. We tested two damaging agents, X-irradiation and myotoxins. Remarkably, both yielded results that indicate that engraftment is in fact better when harsher, more damaging conditions are used. X-irradiation at 25Gy but not at lower doses was optimal for engraftment, as it, but not lower doses, fully suppresses the expansion of murine satellite cells and their contribution to the new muscle tissue that develops after hMPCs are introduced. X-ray doses of 25Gy but not lower can occasionally cause scabbing or sloughing of the skin, which, when serious, requires that the mouse be euthanized. It is nevertheless optimal for our purposes. Similarly, the myotoxins that cause the maximal loss of murine muscle tissue, especially NMC-S (N. mossambica cardiotoxin) and BaCl2, are the reagents that promote optimal engraftment, perhaps because they create larger niches in which hMPCs can embed and grow. Notably, the primary component of N. mossambica cardiotoxin, cyto-5, is not nearly as effective as the crude commercial mixture. As crude N. mossambica cardiotoxin preparation is no longer commercially available, we now use BaCl2 routinely.
Engraftment of a majority of the cell lines we have assayed so far is also improved by obliging the engrafted mice to use their hindlimbs, either by iNMES, on running wheels, or simply by injecting both hindlimbs so that neither can be favored as the grafts develop. These observations are consistent with the fact that exercise can promote engraftment 31 as well as muscle growth and maturation. The different effects of iNMES on the engraftment of the hMPC lines we have studied are likely due to their expression of different levels of several voltage-activated ion channels, which can have a profound effect on myogenic growth and differentiation32–36.
Finally, we believe that the rate at which the hMPCs grow in tissue culture may be related to how well they engraft. Although we have not examined this in detail, the 15A and 15V cells, which form the largest grafts in our hands, have doubling times in tissue culture of approximately 34 h, whereas the A4 and C6 cells, which form smaller grafts, have doubling times of approximately 67 h. We have seen that other hMPC lines that grow more slowly in vitro also engraft poorly. This question deserves further study.
Uniqueness of Our Approach
We have been criticized for not acknowledging the many laboratories on which our methods are based 37 . We did, in fact, acknowledge the earlier work of the pioneers in muscle xenografting in our initial paper, in which we first described our methods 10 . To be clear here, as we were in that publication: many of our methods (X-irradiation; myotoxin intoxication; the use of immunocompromised mice; injection of cells; analysis of the resulting tissues) were modified from methods that were initially developed by other laboratories.
Our approach is unique in several respects, however. (i) Our use of a dose of X-irradiation of 25Gy fully suppresses myogenesis in NRG mice without having adverse effects on engraftment. This dose also minimizes the presence of murine myonuclei in human myofibers. The minimization of murine myonuclear contamination of human myofibers may be essential for studies of FSHD, as the gene products produced by healthy murine myonuclei have the potential of suppressing the pathogenic program in FSHD. This is also likely to be the case with other xenograft-based studies of muscular dystrophies, especially when a small amount of the murine gene product can restore function compromised by the pathogenic mutation. (ii) Our use of iNMES in xenografting is also distinctive, although it has a basis in previous literature38–43. (iii) Equally important is our use of immortalized clonal cell lines of hMPCs. The ultimate success of using immortalized clones of human muscle precursor cells for xenografting was far from obvious when we began our studies, and it was only confirmed when our first paper was published 10 . This aspect of our work is significant, as clones can be manipulated to study genetic and epigenetic features in ways that primary cells, typically used by other laboratories, cannot. In addition, the use of immortalized cells allows scaling of our methods to generate hundreds of mice carrying the same disease-causing mutations, for further characterization of the disease, identification of biomarkers, and discovery and testing of therapeutics in mature human muscle tissue. Furthermore, the clones are readily available to other laboratories that may wish to replicate or expand on our findings. These advantages, too, are not applicable to primary cultures.
Future Investigations
Although we have made significant progress in understanding the contributions of different factors to successful engraftment and growth of human muscle tissue in mice, there are a several questions that remain to be addressed.
The variability in our results continues to be troublesome, as it requires that we study a large number of engrafted mice for our results to reach statistical significance (and, as our results here show, even high numbers may not yield statistically significant differences). The differences among cell lines, although not surprising for studies of different clones, is also a challenge, as it suggests that each line may require its own optimized conditions for engraftment. This is certainly the case for the use of iNMES and is likely to be true for several of the other factors we have studied as well. This will make comparisons among lines more difficult.
Finally, we have been frustrated by the fact that large grafts of >1,000 human fibers are infrequent. If these were generated routinely, we would be able to perform physiological studies on the grafts, but with lower numbers, any functional differences in the grafts may be difficult to observe against the contribution of the remaining murine muscle tissue. Methods to eliminate murine tissue more effectively and to grow engrafted tissues more robustly are therefore still needed. The fact that Sigma-Aldrich NMC-S, which in our hands was the most efficient in eliminating murine myofibers, is no longer available has made this issue challenging. To address it, we are testing additional alternatives to our routine methods that use BaCl2. The introduction of other growth factors or manipulation of the transforming growth factor beta (TGF-β) family of signaling molecules that control muscle growth (reviewed in the works of Lee 44 and Chen et al 45 ), already briefly investigated in the context of xenografting8,46, may offer additional avenues for improving the quality and size of human muscle tissue in mice, as well. With further improvements to our methods, we anticipate that xenografts will be as useful for physiological and pathophysiological studies of mature human skeletal muscle as they already are for genetic, epigenetic, and biochemical studies.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241242624 – Supplemental material for Optimization of Xenografting Methods for Generating Human Skeletal Muscle in Mice
Supplemental material, sj-docx-1-cll-10.1177_09636897241242624 for Optimization of Xenografting Methods for Generating Human Skeletal Muscle in Mice by Andrea O’Neill, Anna Llach Martinez, Amber L. Mueller, Weiliang Huang, Anthony Accorsi, Maureen A. Kane, David Eyerman and Robert J. Bloch in Cell Transplantation
Footnotes
Acknowledgements
The authors thank Ujwala Pimparkar for her technical assistance and the laboratories of Drs. Woodring E. Wright (Southwestern Medical School, Dallas TX; deceased), Silvère van der Maarel (University of Leiden, The Netherlands), and Vincent Mouly (Institut de Myologie, Paris, France) for their generous gifts of the cell lines used here (WEW: 15A, 15V, 15B, 16A; SvdM and VM: 4A, C6, C12). The authors are grateful to Steven Kazmirski, then at Fulcrum Therapeutics, for providing the cytotoxin 5 and to Fulcrum Therapeutics for providing invaluable collaborative support.
Authors’ Note
This paper is dedicated to the memory of Dr. Woodring Erik Wright, a treasured colleague whose insights and support were invaluable at the early stages of our research.
Authors’ Contributions
A. O’Neill, A. Llach Martinez and A. L. Mueller grew the cells, prepared engrafted mice, treated them as described in the text, and collected and analyzed the engrafted muscles. A. O’Neill prepared the figures. W. Huang and M. A. Kane performed the mass spectroscopic analysis of commercial cardiotoxin. A. Accorsi and D. Eyerman provided useful suggestions and coordinated with staff at Fulcrum Therapeutics to enable us to obtain different preparations of cytotoxin 5. R. J. Bloch supervised the study of the grafts, coordinated the collaboration with Fulcrum Therapeutics and with Drs. Huang and Kane, and wrote the paper.
Availability of Data and Materials
All data will be made available upon request. Materials will be shared upon request, if stocks remain.
Ethical Approval
Our studies were approved by the Institutional Animal Care and Use Committee of the University of Maryland, Baltimore.
Statement of Human and Animal Rights
This article does not contain any studies with human subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by NINDS 1R21NS086902 (RJB), Friends of FSH Research (RJB), The FSH Society (ALM and RJB), NIAMS 5F31AR070621 (ALM), and NIGMS T32GM08181 (fellowship support for ALM, M. Trudeau, and A. Meredith, PIs). This research was also supported in part by the University of Maryland, Baltimore, School of Pharmacy Mass Spectrometry Center (SOP1841-IQB2014).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
