Abstract
In animal models, cell therapies for different diseases or injuries have been very successful. Preclinical studies with cells aiming at a stroke, heart attack, and other emergency situations were promising but sometimes failed translation in clinical situations. We, therefore, investigated if human placenta-derived mesenchymal stromal cells can be injected in pigs without provoking rejection to serve as a xenogenic transplantation model to bridge preclinical animal studies to more promising future preclinical studies. Male human placenta-derived mesenchymal stromal cells were isolated, expanded, and characterized by flow cytometry, in vitro differentiation, and quantitative reverse-transcription polymerase chain reaction to prove their nature. Such cells were injected into the sphincter muscle of the urethrae of female pigs under visual control by cystoscopy employing a Williams needle. The animals were observed over 7 days of follow-up. Reactions of the host to the xenogeneic cells were explored by monitoring body temperature, and inflammatory markers including IL-1ß, CRP, and haptoglobin in blood. After sacrifice on day 7, infiltration of inflammatory cells in the tissue targeted was investigated by histology and immunofluorescence. DNA of injected human cells was detected by PCR. Upon injection in vascularized porcine tissue, human placenta-derived mesenchymal stromal cells were tolerated, and systemic inflammatory parameters were not elevated. DNA of injected cells was detected in situ 7 days after injection, and moderate local infiltration of inflammatory cells was observed. The therapeutic potential of human placenta-derived mesenchymal stromal cells can be explored in porcine large animal models of injury or disease. This seems a promising strategy to explore technologies for cell injections in infarcted hearts or small organs and tissues in therapeutically relevant amounts requiring large animal models to yield meaningful outcomes.
Keywords
Introduction
Many different preclinical animal models provided promising evidence that cell therapies may ameliorate or even cure dysfunctional tissues or replace diseased cells 1 . Among them, cell therapies for tissue regeneration after ischemic stroke, heart attack, and other emergency situations were explored2,3. In this context, ground-breaking work has been performed in rodents 4 . In such models, defects were established in a relevant number of animals, the regenerative cells were isolated in seemingly unlimited numbers from inbred mates, and a so-to-say “autologous” cell therapy was performed facilitating a statistically relevant number of cases in an individual study 4 . Large animal models complemented the studies of cellular therapy performed in rodents or other small animals, but challenges remain 5 . One challenge is the translation of animal studies into clinical success. In clinical studies, the success of cell therapies was often limited and results were sometimes inconsistent5–7. Another decision relates to the choice of optimal regenerative cells8–10. For safety considerations, the application of autologous cells is considered the best option for patients, as the transmission of infection or rejection of the cells applied is avoided when compared to applications of homologous, non-human leukocyte antigen (HLA)-matched cells 11 . The HLA group of cell surface markers, also referred to as major histocompatibility complex (MHC) or transplantation antigens, plays a central role in the regulation of the immune system and in the rejection of non-self-components, cells, or tissues upon injection, transfusion, or transplantation 12 . HLA class I molecules present internal peptides to CD8pos T lymphocytes, while class II molecules present extracellular antigens CD4pos T lymphocytes 13 . Using autologous or HLA-matched cells grants immune tolerance by the host. However, autologous cells come with some disadvantages as well. In an emergency such as a heart attack or stroke, autologous cells are not at hand for therapy in most cases in amounts and quality needed. In contrast, animal studies can be easily planned, prepared, and conducted. In clinical situations, therapy schedules for instance for cell therapy of cartilage, dermal defects, or other tissues or organs can be planned, prepared, and conducted accordingly. But in case of an emergency such a comfortable situation is not granted. Therefore, most of the aforementioned animal models may serve as proof-of-principle studies, but the clinical situation in emergencies requires other strategies.
The phenotype and nature of mesenchymal stromal cells (MSCs) from different sources have been discussed. It was suggested that MSCs express several mesenchymal antigens including CD73, CD90, CD105, CD146, CD166, and others, but lack expression of hematopoietic antigens such as CD11b, CD14, CD19, CD34, or CD45 and express little or no immune regulatory HLA class II antigens14–16. Application of MSCs has been shown to yield a therapeutic benefit in many preclinical and clinical studies17–19. Current understanding suggests that these cells act mainly by local release of regenerative factors such as cytokines and exosomes, and by limiting the inflammatory processes20–22. They thereby also limit long-term pathology associated with increased fibrosis after increased inflammation23,24. Human placenta-derived MSCs (pMSCs) are interesting candidates for cell-based therapies. They inherit several technical and biological advantages 25 . On one hand, they can be isolated from a comparably young, healthy, and well-vascularized human tissue without any harm to the donor, which legally in most countries worldwide is the mother, and without considerable ethical concerns. In addition, from the same tissue, even two types of pMSCs can be isolated, maternal pMSCs from the endometrial part of the placenta and fetal pMSCs, from Wharton’s jelly or from the tissue underneath the amniotic membrane. Recent studies provided evidence that these two distinct subsets of pMSCs are significantly different cells—albeit closely related in most of their features26,27. For instance, fetal pMSCs have significantly longer telomeres than maternal pMSCs thus facilitating even extended expansion without premature replicative senescence 27 . For autologous cell therapy, MSCs are isolated from other sources such as adipose tissues (ADSC) or bone marrow (bmMSCs). In most cases, the elderly require regenerative therapies. Thus, autologous cells inherit the “age” of the donor. In the elderly, cells prepared for therapy may have been exposed to adverse stimuli, medications with hostile side effects, and aging processes in general. Autologous MSCs from elderly donors very likely have shorter telomeres than pMSCs, thus limiting cell expansion even more. In contrast, most preclinical animal studies were performed in rather young animals, which may yield a positive bias to the outcome of such experiments. This thought is in line with the results of cell therapy in older animals reporting impaired tissue regeneration 28 .
The MSCs from different tissues express low levels of major histocompatibility complex (MHC) class I and very low levels of MHC class II molecules on their surfaces16,29–31. Thus, MSCs seem less immunogenic than cells expressing these MHC antigens on their surfaces. Therefore, not only autologous but also homologous MSCs may be tolerated in patients upon application in clinical situations. However, to investigate the regenerative potential of human pMSCs in allogenic clinical situations, functional disease models are required. This motivated xenobiotic preclinical studies with human pMSCs in immune-competent rodents32,33,34,35. Human pMSCs were shown to modulate inflammation and facilitate tissue regeneration in mice, rats, and other animal models of disease or injury36–39. Recently the allogenic use of human pMSCs in clinical studies was approved by the European and US American health authorities (i.e., EMA, NIH) and clinical feasibility studies reported success25,34.
Large animal models of injury, inflammation, or tissue regeneration facilitate research of surgical interventions 40 . We recently developed a porcine model of urinary incontinence to investigate the potential of pig cells for regeneration of a deficient urethral sphincter muscle35,41,42. Our data suggest that the injection of porcine ADSCs in the sphincter muscle of the urethra of an incontinent pig facilitates significant muscle regeneration in less than two months of follow-up 35 . Therefore, we set out to investigate if human pMSCs will be rejected upon injection in the urethral sphincter muscle in outbred large animals such as pigs. If pMSCs were tolerated in pigs, our studies could pave the way to investigate the regenerative potential of human pMSCs in our model of incontinence and later possibly in anatomically relevant in vivo animal models of stroke, heart attack, and other emergency situations.
Here we report that human pMSCs expanded under good manufacturing procedure (GMP)-compliant conditions were not rejected after injection in the vascularized urethral sphincter complex of pigs, nor did they induce fever or elevate inflammatory markers in serum or plasma of the animals. However, a slight local infiltration of inflammatory cells to the tissue targeted was observed. We conclude that the regenerative potential of human pMSCs can very likely be studied in a surgically relevant large animal model, in pigs.
Methods
Isolation, Expansion, and In Vitro Differentiation of pMSCs
Post-partum placenta tissue from the delivery of a baby boy was obtained from the University of Tuebingen Hospital after informed, written, and approved consent (Ethics Committee file number 341/2002V). The pMSCs were isolated from the fetal part of the placenta as described26,27. The cells were expanded in Dulbecco's Modified Eagle Medium (DMEM) media complemented with 5% human plasma and 5% platelet lysate under GMP-compliant conditions without any xenogenic components 22 . The cells were investigated for their growth patterns, expression of cell surface antigens, and in vitro differentiation capacities to comply with the inclusion and exclusion criteria defined for MSCs from different tissues14–16. Differentiation toward adipogenic and osteogenic cells was induced and visualized as described26,27. Characterized bona fide pMSCs were then utilized for the injection experiments.
Determination of Cell Surface Markers by Flow Cytometry
Expression of cell surface antigens was investigated on pMSCs by flow cytometry (FC)16,22. For FC, cells were detached (Accutase; Sigma), washed, and sedimented by centrifugation, and 5 x 10^5 cells were resuspended in 50-μL staining buffer containing the primary detection antibody (Online Supplementary Table S1) and human γ-globulin to block unspecific staining 43 . After incubation (20 min, 4°C), antibodies were washed off twice. Flow cytometry was performed as recommended by the supplier (LSR II; BD Biosciences). Unstained cells, cells omitting antibodies, and compensation beads served as controls. The data were processed by FACSDiva and FlowJo software (BD Bioscience).
Quantitative Polymerase Chain Reaction to Enumerate mRNA Levels
For isolation of RNA, pMSCs were washed, detached (trypsin-EDTA), washed again, and counted. 1 x 10^6 pMSCs were sedimented to extract RNA (RNeasy kit; Qiagen). Yield and purity of the RNA were determined by UV spectroscopy (Nanodrop; Implen). 1 µg of RNA was reverse transcribed (PrimeScript; TaKaRa), and cDNAs of target transcripts were amplified by PCR (LightCycler480; Roche) using specific oligonucleotides (Table S2). The expression levels of the target genes investigated are presented as transcripts normalized to the expression of the housekeeping genes GAPDH and PPIAγ using the 2− ΔΔcT method in each run44,45. RNA extracted from human white blood cells (WBC) of a volunteer, and from human bone marrow MSCs of a patient provided after written and informed consent (Ethics Committee file number 341/2002V), served as controls.
Transurethral Needle Injection of Cells in Gilts
The pMSCs were expanded under GMP-compliant conditions to reach not more than 75% of confluence 22 , harvested, washed, counted, and prepared for injections. Two Göttingen Minipig gilts, that is, young female pigs, were sedated by pre-medication (atropine, azaperone), followed by anesthesia using midazolam and ketamin as described 42 . The position of the urethral sphincter muscle was determined by cystoscopy and standard urodynamics (Aquarius TT; Laborie) using an air-charged balloon catheter (online supplement Fig. S1A–C) 41 . Two aliquots of 250 µL containing 6 x 10^5 pMSCs plus 6 x 10^5 FITC-labeled microparticles (PS-FluoGrün-Fi364, ø 20 µm; Microparticles GmbH) each were injected in the urethral sphincter complex by a Williams needle (Cook Medical, 35 cm length, 4 mm tip) and a cystoscope under visual control (Fig. S1D) 42 . Seven days after cell injection, gilts were sedated and sacrificed (KCl i.v. lethal), bladders and urethrae were prepared, and the area of injection was determined by imaging (IVIS Spectrum; PerkinElmer) 46 . Areas of injected pMSCs and PS-FluoGrün-Fi364-labeled particles are visualized by IVIS as false-color red-to-yellow heatmaps. The injection zone was excised to prepare cryosections. The study was approved by the local Institutional Review Board (State Animal Welfare Authorities; study # CU01-16-G, file # 35/9185.81-2) and conducted in full compliance with all relevant regulations (https://arriveguidelines.org; https://www.eara.eu/animal-research-law?lang=de) and ethics 47 .
Analysis of Inflammatory Parameters in Blood Samples
White blood cell (WBC) counts and other hematology parameters were enumerated in EDTA-blood samples of gilts prior to injection of pMSCs, (day 0), and on days 3 and 7 of follow-up by a hematology analyzer (Sysmex XP 300). Blood samples from two other gilts served as controls. C-reactive protein (CRP) and interleukin (IL)-1β (Quantikine Porcine Immunoassays; R&D Systems), serum amyloid A (SAA), and haptoglobin (Laboklin, Bad Kissingen, Germany) were determined in plasma samples by ELISA as described 48 . In addition, haptoglobin was detected in plasma samples by Western blot 49 . 100 µg of protein was used in the analyses. Unspecific antibody binding was blocked (5% milk powder in 0,1%Tween/PBS) and haptoglobin was detected by antibody (1:700 abcam ab231013, in 5% milk powder/PBS, 1 h, room temperature). Unbound primary antibodies were washed off, detection reagents added (1:2000 anti-rbt-HRP in 5% milk powder/PBS; Dako), washed off again, and haptoglobin expression was visualized using a WB-scanner (C-Digit, LiCor) as described 50 . Signal intensities were computed, and graphs were exported using the proprietary software (LiCor).
Histology of Tissue Sections and Polymerase Chain Reaction of Chromosomal DNA
Cryosections (20 µm, CM1860 UV; Leica) were generated from the area of cell injection, mounted, and stained by HE chemistry to visualize the tissue targeted41,46. Cryosections of porcine spleen and lymph node tissues served as positive controls. To detect infiltrating leukocytes, cryosections were blocked (5% milk powder in 0,1% Tween/PBS), incubated with anti-CD45 antibodies (1:200 in 1% BSA/PBS, 37°C, 90 min, black humidified box, Invitrogen), washed 3 times (0,1% Tween 20 in PBS), reacted with secondary AF555-labeled anti-ms antibodies (1:200 in 1% BSA/PBS, 37°C, 45 min, black humidified box, Invitrogen), washed again, counterstained by DAPI, and recorded by fluorescence microscopy (DMi8; Leica with LAS X software). To detect the human male pMSCs injected in the porcine urethra, tissue was scratched off from eight consecutive crysections to isolate DNA (DNeasy extraction kit, Qiagen) 46 . Male chromosomal DNA was detected in cryosections by a hot start PCR of the SRY gene (Table S2) employing 50 cycles for DNA amplification (denaturation: 94°C for 30s, annealing: 65°C for 30s, extension:72°C for 20s) followed by primer extension (72°C for 5 min.; LighrCycler, Roche) The PCR products were mixed with DNA loading buffer, separated by 2% agarose gel electrophoresis, and visualized (GelRed Nucleic acid, Biotinum). DNA extracted from human female (HeLa) and human male (RT4) cell lines, and from a boar served as controls.
Results
Characterization of Human Placenta-Derived Stromal Cells
For quality measures the expression of mesenchymal stromal cell markers was investigated in human pMSCs by FC (Fig. 1). The cells failed to express the hematopoietic markers CD14, CD34, and CD45, but expressed the mesenchymal antigens CD73, CD90, and CD105 15 , as well as adhesion molecules CD146, and CD166 (Fig. 1). Of note, for CD90 and CD146 two subsets were noted, respectively 27 (Fig. 1). Expression of SUSD2, a candidate MSC stemness marker, was very low51,52 (Fig. 1). Differentiation of pMSCs along the adipogenic and osteogenic lineage was successfully demonstrated in vitro (Fig. S2). A prominent expression of immune checkpoint ligand CD276, but very little HLA-ABC, and no HLA-DR were detected (Fig. 1). We therefore explored the transcript levels encoding some representative immune-regulatory HLA molecules by RT-qPCR (Fig. S3). Class II transcripts for HLA-DRA, HLA-DQB1, HLA-DRB1, and the class II-associated chaperone CD74 were recorded in white blood cells (WBS), but much less in pMSCs and bmMSCs. HLA-DQB1 was not expressed in pMSCs. Class I transcripts HLA-A and HLA-B were detected in WBC, and less in pMSCs and bmMSCs, respectively (Fig. S3). The data confirmed that human pMSCs expressed the expected cell surface markers. The low expression of HLA-class I and class II suggested that the human pMSCs employed in this study seem not to be prominent antigen-presenting or immunogenic, but possibly rather immune modulatory cells25,53,54.
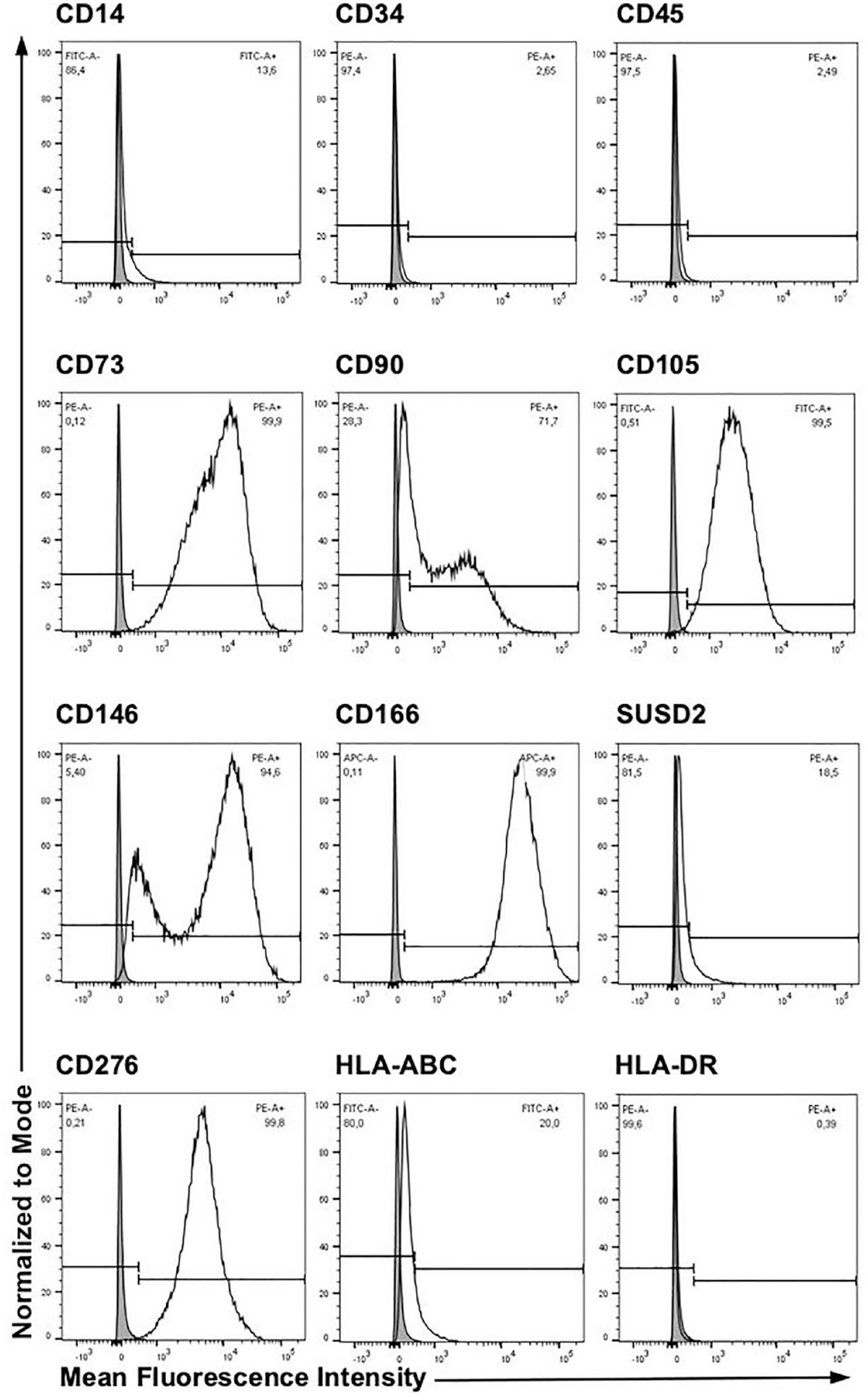
Expression of cell surface molecules on human pMSC by flow cytometry. The cells were expanded in xeno-free media under GMP-compliant conditions, harvested, and reacted with antibodies as indicated. Human pMSCs failed to express the hematopoietic markers CD14, CD34, and CD45, but expressed the mesenchymal antigens CD73 and CD105, and adhesion molecule CD166. For CD90 and CD146 two subpopulations were recorded, SUSD2 was very low. The immune checkpoint antigen CD276, alias B7H3, was high, HLA-ABC low, and HLA-DR negative. In the contour plots, x-axes indicate the log of the mean fluorescence intensities (MFI), and y-axes the numbers of events normalized to mode. Gates were set to include viable cells only.
Analysis of the Urethral Tissue Targeted
To investigate if human pMSCs are immunogenic upon xenogenic application across mammalian species in pigs, a feasibility study was performed. To this end, the sphincter complex of the porcine urethra was localized (Fig. S1A–C), and two aliquots of human pMSCs were injected in the area of maximal wall pressure of the sphincter muscle by transurethral needle injection under visual control (Fig. S1D). Seven days after cell injection, gilts were sacrificed to prepare the urethra. This tissue was scanned for fluorescence signals by an IVIS imaging device and the co-injected fluorescent microparticles facilitated the localization of the injection sites by the imaging technology in each urethra treated (Fig. 2A, 2B). The urethral tissue appeared normal in cryosections of injected areas. Signs of tissue injury by needle injections were not observed but a mild local infiltration of mononuclear cells was noted by HE-staining (Figs. 2C, 2D, S4, S5). In contrast to our previous study employing human bmMSCs, signs of necrosis or encapsulation were not observed upon injection of human pMSCs 42 . Green fluorescent particles—appearing as yellow dots due to the red immunofluorescence of the CD54pos cells—were detected in consecutive cryosections specifying the proper localization of the injection site (Fig. 2E, 2F). By immunofluorescence, a few CD45-positive lymphocytes were observed in the red fluorescence channel overlapping the area of the fluorescent particles (Fig. 2G, 2H). To prove the specificity of the anti-CD45 staining protocol, tissue samples of porcine spleen and lymph node stained with the anti-CD45 reagents, and a prominent fluorescence was observed (Fig. 2I, 2K). This confirmed that a locally confined lymphocyte infiltration was observed at sites of xenogenic pMSC injection.
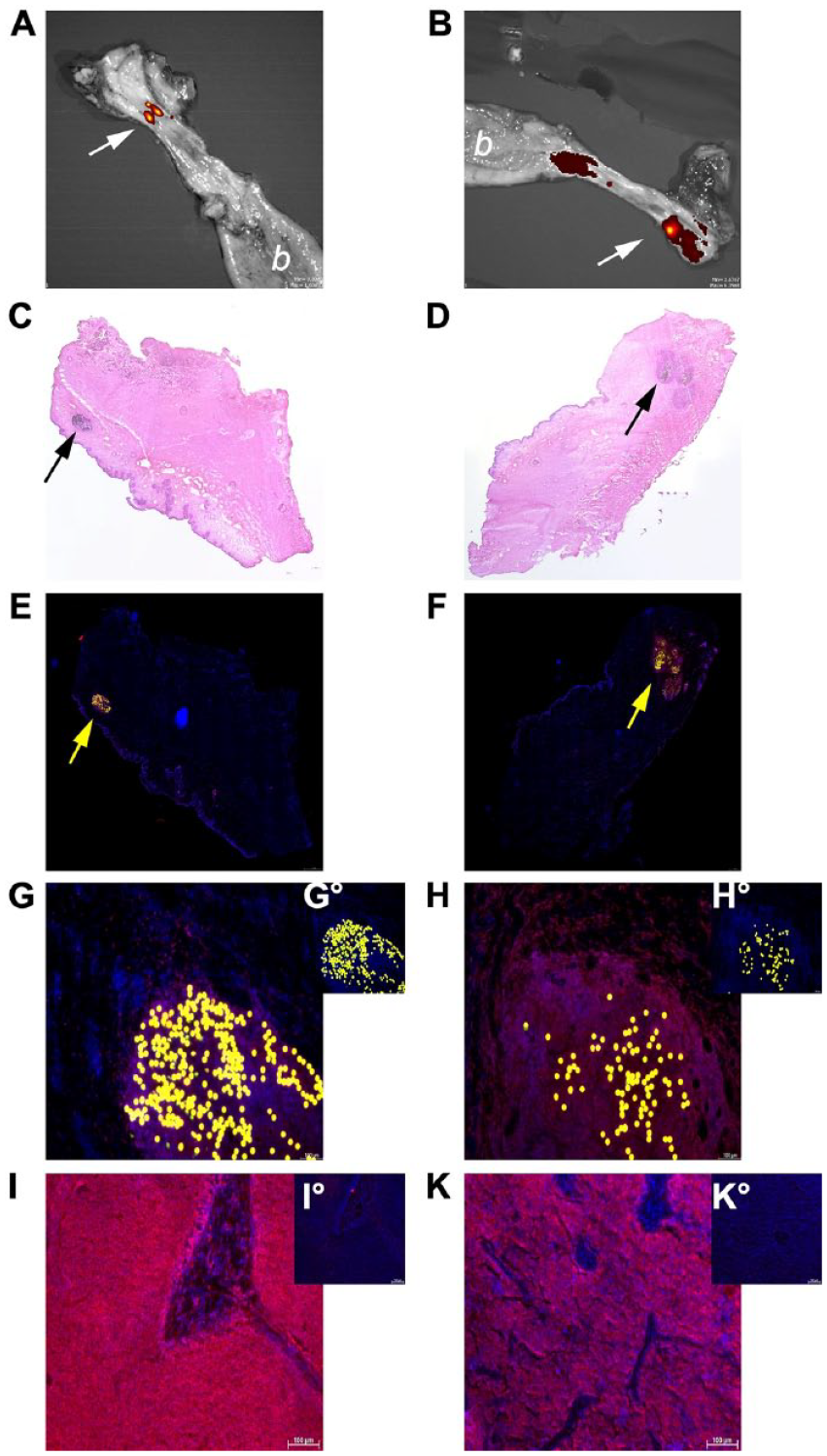
Analyses of the regions of interest in the sphincter complex in the porcine urethrae. Seven days after cell injection, the two gilts were sacrificed and the urethrae were prepared. Fluorescence indicating the spots of cell injection were visualized in the explanted urthrae by IVIS (white arrows; A, B; the bladders are labeled by a white “b,” respectively). Cryosections were generated from this region (C—H°). HE staining of cryosections showed normal tissue situations. In the samples, the urothelial layer appeared as a pleated violet line (*), and the submucosal and muscular layers of the urethra in a pink hue. Signs of necrosis were not noted, and the microparticles co-injected with the pMSCs were observed as dark dots (black arrows, C, D). These two micrographs are shown enlarged in Fig. S4, S5, respectively. In consecutive cryosections, cell nuclei were counterstained by DAPI (blue fluorescence, E—H°), and the fluorescent microparticles were recorded and visualized as yellow dots (yellow arrows, E, F). Enlarged micrographs showed a locally circumscribed minor infiltration of CD45pos cells in pig 1 (G) and a moderate locally circumscribed infiltration of CD45pos cells in pig 2 (H, red fluorescence) in the injection zone marked by microparticles. The corresponding detection antibody controls are included as small inserts (G°, H°). Staining CD45 in cryosections of porcine lymph node (I) and spleen (K) samples served as positive controls (red fluorescence). The corresponding detection antibody only controls are inserted (I°, K°). Micrographs from both samples of pigs (pig 1: left, pig 2: right) are shown. Size bars indicate 500 µm (C, D), 1 mm (E, F), and 100 µm (G—K), respectively.
Inflammatory Responses in Blood and Plasma Samples
To investigate if xenogenic injection caused a general inflammatory response, a blood count was performed. Changes in white or red blood cell counts, hemoglobin, hematocrit, mean corpuscular volume, hemoglobin per volume or per erythrocyte, and platelet count were not detectable in blood samples taken prior to pMSC injections (d0) when compared to samples prepared 3 (d3) or 7 (d7) days after the injections, respectively, nor in comparison to samples from not-treated control animals (Fig. 3A). The values determined matched published data 55 . In pigs, elevated haptoglobin is a sensitive and robust marker for necrosis and inflammation in an acute phase response 56 . An increase of haptoglobin was not observed by Western blot during follow-up in both pigs (Fig. 3B). Complementary, an increase of haptoglobin or SAA was not recorded during follow-up by ELISA. The values recorded remained in the physiological range (Table S3)57,58. The IL-1β concentrations were determined as well. In all samples collected prior to pMSC injection and during follow-up, IL-1β concentrations remained below the detection level of the ELISA (approx. 20 pg/mL; data not shown). CRP prior to cell injection was 510 µg/mL, which did not rise above this level during follow-up (not shown). Moreover, determining the body temperature of each individual gilt, and inspecting their behavior, drinking, and voiding patterns during follow-up did not indicate a fever reaction or any other maladies (data not shown). This suggested that the xenogenic pMSCs did not provoke considerable inflammatory responses in pigs.
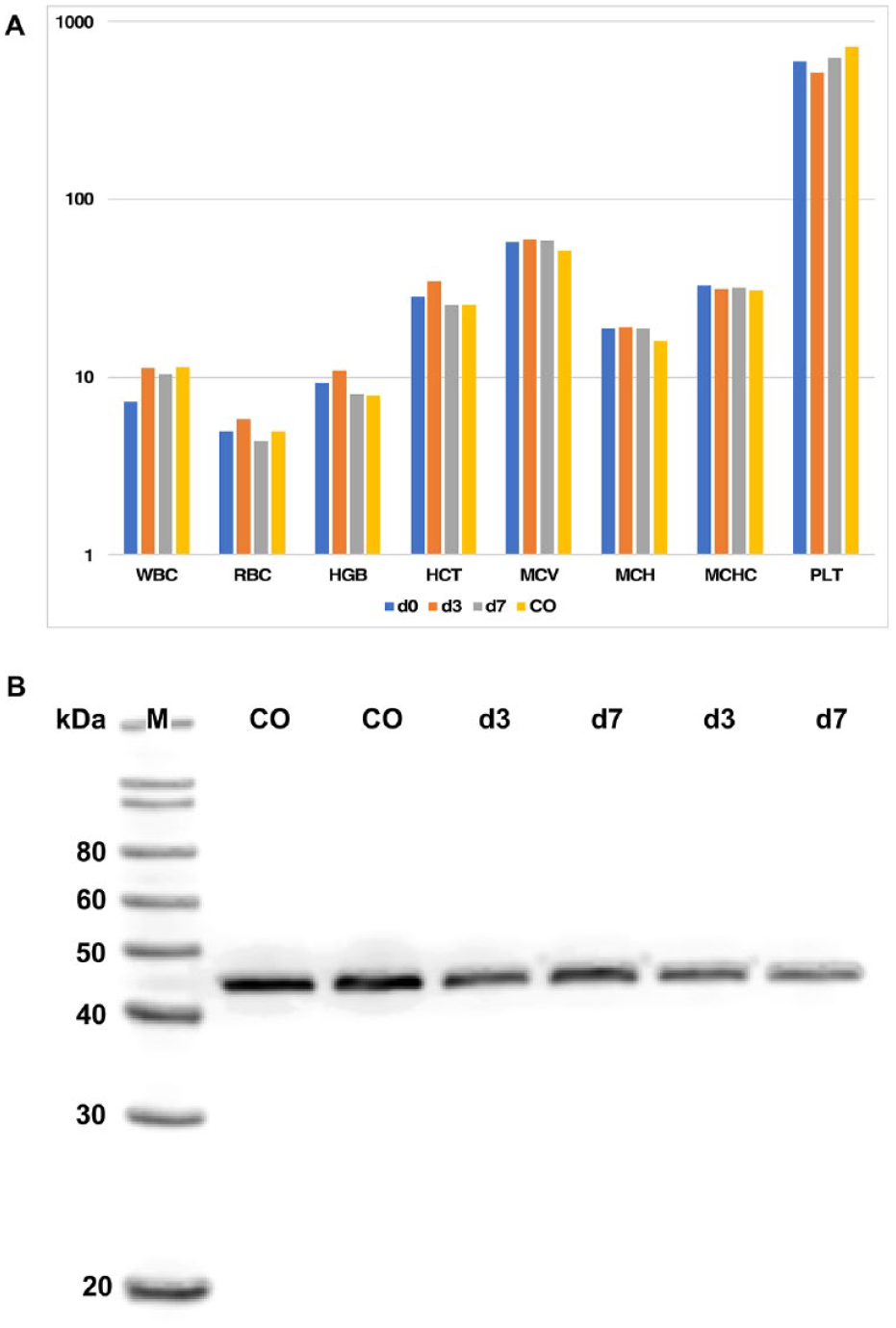
Inflammatory parameters in peripheral blood after cell injection. (A) EDTA blood was collected from the gilts prior to (d0), three (d3), and seven (d7) days after injection of xenogenic pMSCs, respectively, as indicated. EDTA-blood from untreated gilts served as controls (CO). Relevant differences over time after injection of xenogenic pMSCs in the counts of white (WBC) or red blood cells (RBC), concentrations of hemoglobin (HGB), hematocrit (HCT), mean corpuscular volume (MCV), hemoglobin per volume (MCH) or per erythrocyte (MCHC), and platelet count (PLT) or between treated animals versus not-treated controls were not recorded. The x-axis presents the individual parameters as recorded by the automated analyzer, and the y-axis the log normalized mean values in arbitrary units. (B) Levels of haptoglobin in plasma samples collected from the two control gilts (CO) and animals on day three (d3) or seven (d7) after cell injection were determined by Western blot as indicated. An elevation of haptoglobin after injection of human pMSCs was not recorded in comparison to controls. A size marker (M) confirms the expected molecular mass of approx. 44 kDa of the porcine haptoglobin beta-chain.
Detection of Human Cells in Porcine Tissue by PCR of Chromosomal DNA
Amplification of the human male SRY DNA by PCR was performed from urethral tissue samples to detect human chromosomal DNA or DNA fragments in the urethral tissue 7 days after application (Fig. 4). DNA from the human female HeLa cells, human male RT4 tumor line, and from cells of a boar served as positive and negative controls, as well as a species specificity control. By PCR bold signals for SRY amplification were detected in DNA from cryosamples prepared precisely from the site of cell injection of both gilts, while samples from adjacent cryosections more than 1 mm apart from the injection center yielded faint or no PCR products (Fig. 4). This may indicate that the human pMSCs were probably not moving over larger distances from the immediate injection area marked by fluorescent particles during follow-up of 7 days. Human male DNA generated bold SRY signals while female DNA did not (Fig. 4). Boar DNA did not yield specific PCR products (Fig. 4). This indicated that at least some of the chromosomal DNA of the injected human male pMSCs remained at least in part intact for no less than 7 days after injection of the cells in pigs. Alternatively, some pMSCs may have remained intact at the injection site. In addition, at least some of the DNA or some of these cells had not (yet) been eliminated by the host within this period of follow-up. Taken together, this feasibility study provides evidence that the regenerative potential of human pMSCs can be explored in a porcine large animal model.
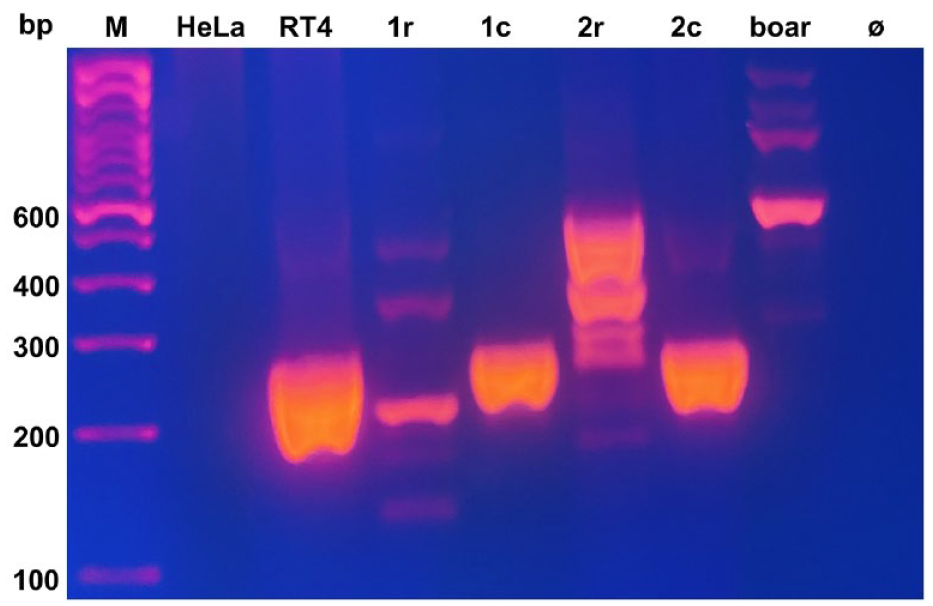
Detection of human DNA in porcine urethrae 7 days after pMSC injection. Tissue was scratched off from eight consecutive cryosections of the center of confirmed pMSC injection (lanes 1c, 2c) or from remote areas (1r, 2r) of the treated gilts 1 and 2, respectively. Chromosomal DNA was isolated and PCR was employed to amplify a specific sequence of the human SRY gene. In cryosamples with confirmed pMSC injection (compare Fig. 2C –2H), a prominent SRY-PCR product was recorded (lanes 1c, 2c), while a sample from remote sections from gilt 1 yielded a lower signal (lane 1r) and remote samples from gilt 2 yielded no specific product in the expected product range of 254 bp (2r). The PCR protocol was highly specific, as only human male DNA generated a 254 bp PCR product (lane RT4), while DNA from female cells did not produce an SRY PCR product (lane HeLa). The protocol was species-specific, as porcine male DNA failed to amplify a 254 bp PCR product (boar). A 100 bp marker was included (lane M) to indicate the product sizes in base pairs (bp).
Discussion
In a variety of clinical studies, MSCs were applied to support or modulate tissue regeneration and inflammation 59 . A metastudy indicated that human MSCs will work in different mammalian species 60 . However, only a few of the studies presented data on larger animals (i.e., sheep, pigs, and dogs). In most studies reviewed (63%), the human MSCs were given at remote sides (i.v., i.p., s.c.) 60 . However, local administration of human bmMSCs in vascularized tissue was performed in a large animal study, and the bmMSCs were injected in porcine hearts 61 . Six days after cell application bmMSCs were not detected anymore but inflammation was observed even in pigs under immune suppression by cyclosporine 61 . This indicated that the medical potential of human bmMSCs and possibly of most other human-derived MSCs can probably not be investigated in large preclinical animal models under largely unbiased conditions. By contrast, pMSCs inherit unique properties, which could be utilized in future clinical applications. For instance, the expression of MHC molecules in pMSCs is very low, and MHC class II is not upregulated in pMSCs by IFNy but in other MSCs62,63. Our data are in line with this notion as transcripts encoding HLA-DRA, HLA-DQB1, and HLA-DRB1 were lower in pMSCs when compared to bmMSC, while expression of HLA class I transcripts were determined at comparable levels. These differences in MHC expression and possibly in the MHC regulation might have contributed to the rejection, necrosis, and encapsulation of human bmMSCs observed in porcine tissues in our previous cell injection study 42 . Expression of CD74, a chaperone regulating expression and antigen presentation by HLA class II molecules for immune responses, was found lower in pMSCs compared to bmMSCs as well 64 . This corresponded to the low expression of HLA-DR on pMSCs as determined by FC. Moreover, pMSCs isolated from fetal tissues share features of young cells such as significantly longer telomeres when compared to the pMSCs isolated from endometrial tissues of the same placenta 26 . At the same time, the risk of heterotopic ossification especially in vascularized tissues is less pronounced in pMSC when compared to bmMSCs26,27,65. In addition, the regenerative potential of pMSCs, and their secretion of growth factors, cytokines, and exosomes promoting wound healing is well described 25 .
Human pMSCs served in several xenogenic models to cope with lethal radiation, improve diabetes-associated heart function, support regeneration from experimental stroke, or complement cancer therapy in small mammals and were applied in most studies distant to the diseased location using subcutaneous, intraperitoneal, intravenous, or intramuscular routes36,38,39,66. It was shown that pMSCs act at remote sites to facilitate functional tissue regeneration. A xenogenic study applied the pMSCs right in the injured muscle of mice to treat ischemia 37 . This suggested that pMSCs may support muscle regeneration locally as well. We complement these studies and provide experimental evidence—to the best of our knowledge for the first time—that human pMSCs can be injected in vascularized tissues of pigs without the need for immune suppression by, for example, tacrolimus or corticosteroids. Immune suppressive therapy could bias wound healing and other regenerative processes. Thus, the translation in clinical situations may become cumbersome.
However, our feasibility study must leave several aspects of future applications of pMSCs in clinical situations open. The pMSCs were produced under GMP-compliant conditions. Thus, the injection media contained human serum and platelet lysate 22 . This could have provoked a minor local reaction in the pigs as observed by local infiltration of CD45pos cells. Moreover, the cells were injected in a healthy environment. Thus, the regenerative potential of the pMSCs was not explored. Based on our current data we cannot discriminate between a regenerative and tissue repair or wound-healing infiltration of CD45pos cells or a process tilting toward extended inflammation and tissue destruction. But the absence of HLA-DQB1 and low expression HLA-DRA and DRB1 transcripts in pMSCs would favor a non-inflammatory process. This is supported by little HLA-ABC molecules on pMSCs and no HLA-DR as determined by FC. In contrast, the immune checkpoint antigen CD276 was expressed on the pMSCs with a rather high signal intensity. Thus, ameliorating at least in part T-cell mediated responses toward regenerative processes 54 . This still needs to be investigated in an appropriate injury model in more detail. Future studies should include sensitization of the host by multiple injections of pMSCs and dose escalations to explore the immune tolerance against the human pMSCs in pigs. Moreover, the cohort size in the treated as well as in the control cohort is two gilts only. Therefore, statistical analyses could not be performed and the outcome must be interpreted with care. However, these results may pave the way for future larger animal studies to confirm the preliminary data reported here.
Besides immune modulation, clinical applications of pMSCs inherit other advantages as well. Medical grade pMSCs are approved by EMA and FDA for allogenic clinical use 34 . The cells employed for this study were produced and characterized under comparable and GMP-compliant conditions. Batches of human pMSCs may be produced for allogenic therapies and stored in hospitals for immediate application to treat patients suffering for instance from a stroke, heart attack, or other emergency situations. Immediate availability of regeneration-competent cells is key for the clinical success of such cell therapies as isolation, expansion, quality management, and preparation of autologous cells for a “subito”- application takes too much time and effort. Moreover, in pigs, even batches of functional subsets of human pMSCs could be selected using relevant disease models and testing individual preparations or lots of pMSCs 25 . Due to an abundant number of pMSCs available from one placenta, minimal expansion under GMP-compliant conditions for in-depth characterization and in vivo testing should not be a limiting factor. This would facilitate the pre-screening of optimal batches for distinct clinical situations by using aliquots of a given batch in porcine disease models and the application of the remaining cells for therapies.
Production of MSCs from term placenta for homologous applications seems ethically less problematic and less invasive for the donor when compared to the isolation of MSCs from bone marrow or other sources. Manufacturing autologous induced pluripotent stem cells (iPSCs) and their in vitro differentiation to generate functional regeneration-competent cells takes time and effort as well. For the treatment of stress urinary incontinence, autologous skeletal tissue was harvested, minced during surgery, and directly injected into the urethral sphincter muscle 67 . This strategy may be applicable to the regeneration of other striated muscles. However, it is currently unknown if this strategy could be applied to the treatment of stroke or heart attack as well.
Conclusion
This feasibility study provides evidence that human pMSCs can be applied and thus functionally explored in pigs as a large animal model of injury or disorders. Studies in pigs facilitate surgical interventions in living animals of clinically relevant size. Moreover, the metabolism of omnivore pigs seems suitable for combinations of surgical as well as pharmaceutical interventions. However, due to the small cohort size of two animals only, the results of this proof of principle study must be interpreted with some care. Nonetheless, this report might serve as a basis for future preclinical confirmatory studies of xenogenic cell therapy employing human placenta-derived cells.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241226737 – Supplemental material for Xenogenic Application of Human Placenta-Derived Mesenchymal Stromal Cells in a Porcine Large Animal Model
Supplemental material, sj-docx-1-cll-10.1177_09636897241226737 for Xenogenic Application of Human Placenta-Derived Mesenchymal Stromal Cells in a Porcine Large Animal Model by Niklas Harland, Jasmin Knoll, Bastian Amend, Tanja Abruzzese, Harald Abele, Peter Jakubowski, Arnulf Stenzl and Wilhelm K. Aicher in Cell Transplantation
Footnotes
Acknowledgements
The authors thank the midwives, nurses, and physicians of the obstetrics unit at the University Hospital for providing placenta tissue samples.
Author Contributions
BA, AS, and WKA: Conceptualization and methodology, funding.
NH, JK, TA, HA, P.J.: Investigation, experiments, data processing.
All authors: Writing. All authors have read and approved the final of the manuscript.
Data Availability Statement
The data are available to colleagues from academia upon justified request.
Ethical Approval
The study was conducted in full accordance with the Declaration of Helsinki and all other relevant regulations. It was approved by the Institutional Review Board, the Ethics Committee of Eberhard-Karls-Universität Tübingen and the University Hospital, under file number 341/2002V.
Statement of Human and Animal Rights
The animal study was approved by the Institutional Review Board, the Animal Welfare Authorities of the State of Baden-Württemberg, Administration at RP in Tübingen, under project number CU01/20G and file # 35/9185.81-2. The animal study was conducted according to ARRIVE 2.2 (![]() ) and in full compliance with all other relevant regulations.
) and in full compliance with all other relevant regulations.
Statement of Informed Consent
Written and informed consent was obtained from the donor of placenta tissue for the isolation of pMSCs.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We express our great gratitude to the Adolf-Leuze-Foundation (Owen/Teck, Germany) for an unrestricted educational grant that enabled us to acquire the GloMax system and ELISA reader needed for precise determination of cytokines in porcine plasma and serum levels in due time and at short notice. We thank the veterinarians, surgery assistants, and animal wardens of the animal housing and surgery unit at the University of Tübingen for their technical support. We thank Mrs. Conny Bock for expert technical assistance in cell cultures, and Chaim Goziga for the preparation of the artwork. This project was supported in part by DFG grants Ai 16/27-1 PoTuS (#429049495), Ai 16/29-1 MyoRepair (#468616715), and in part by institutional funds.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
