Abstract
Fireflies produce light through luciferase-catalyzed reactions involving luciferin, oxygen, and adenosine triphosphate, distinct from other luminescent organisms. This unique feature has revolutionized molecular biology and physiology, serving as a valuable tool for cellular research. Luciferase-based bioluminescent imaging enabled the creation of transgenic animals, such as Firefly Rats. Firefly Rats, created in 2006, ubiquitously express luciferase and have become a critical asset in scientific investigations. These rats have significantly contributed to transplantation and tissue engineering studies. Their low immunogenicity reduces graft rejection risk, making them ideal for long-term tracking of organ/tissue/cellular engraftments. Importantly, in the islet transplantation setting, the ubiquitous luciferase expression in these rats does not alter islet morphology or function, ensuring accurate assessments of engrafted islets. Firefly Rats have illuminated the path of transplantation research worldwide for over a decade and continue accelerating scientific advancements in many fields.
Keywords
As summer nights emerge, fireflies gracefully take flight, casting a gentle radiance that illuminates their surroundings. Unlike the luminescence observed in jellyfish and other organisms, the ethereal light emitted by fireflies stems from their synthesis of endogenous luminescent substances, luciferin, coupled with oxygen and adenosine triphosphate within their bodies. Luciferase serves as the key catalytic enzyme facilitating this luminous phenomenon in fireflies. Presently, the luciferase-based bioluminescent reaction is widely used in a multitude of scientific endeavors as a cellular beacon, contributing to the advancement of molecular biology and physiology.
In the 20th century, the scientific community witnessed a pivotal breakthrough in luciferase-based bioluminescent imaging from the cloning of the luciferase gene to the generation of transgenic animals as well as the advancement of the charge-coupled device camera technology for detecting extremely low-intensity light levels while reducing background read noise. In 1988, genetic manipulation led to cloning of the luciferase gene downstream of a small DNA fragment of the chicken alpha-skeletal actin promoter; this luciferase gene product then was microinjected into mouse embryos to generate transgenic mice expressing the luciferase gene product under the alpha-skeletal actin promoter 1 . Their pioneering work led to the generation of Firefly Rats in 2006; Hakamata and his colleagues 2 introduced the luciferase gene under a systemically expressing ROSA26 promoter in Lewis rats to establish the Firefly Rats, ubiquitously expressing the luciferase gene to emit the bioluminescent light in the presence of luciferin. Serendipitously coinciding with the elucidation of the rat whole genome sequence in 2004 3 , the Firefly Rats were deposited in a public repository, the National BioResource Project for rats 4 , which openly distributes the rat strains globally. As a result, Firefly Rats have disseminated to research institutes worldwide including Japan, United States, Canada, Germany, and China5–14.
Since all cells and organs derived from the Firefly Rats can potentially emit bioluminescent light due to the ubiquitous expression of the luciferase gene, various cells/organs harvested from the Firefly Rats have been used for tracking. Notably, the bioluminescent light can be quantified, making this animal model indispensable. With such coveted qualities, Firefly Rats have contributed to advancing transplantation studies and tissue engineering. Examples of tissue engineering include the fabrication of three-dimensional tissues by the unique cell sheet technology using Firefly Rats’ heart cells for in vitro tracking 8 and the fabrication of small intestinalized colons as a potential treatment for short bowel syndrome where the Firefly Rats were used for the organoid source for noninvasive imaging after the transplantations 10 . Foremost, the tissue from the Firefly Rats demonstrated low immunogenicity with minimal graft rejection risk. In contrast, some reporter proteins elicit immune responses, rendering engrafted tissues un-trackable. We demonstrated that the luciferase is far less immunogenic than green fluorescent protein (GFP) when they were examined in the skin graft transplantation model into the wild-type rats; the luciferase-expressing skin graft survival was over 100 days, while GFP-expressing skin graft lost the bioluminescence ~10 days 2 .
With such unique characteristics of the Firefly Rats, our team has been using them as donors for cell transplantations. We used isolated islets and hepatocytes from the Firefly Rats to transplant them into the wild-type rats for bioluminescent imaging in the long term for noninvasive tracking. We confirmed that luciferase-transgenic islets on the synthetic scaffolds transplanted in the subcutaneous site of wild-type recipients consistently emit bioluminescence over 20 weeks post-transplant (Fig. 1A) 9 , demonstrating the potential of this minimally invasive procedure of pancreatic islet transplantation under the skin. Bioluminescence of the Firefly Rat–derived hepatocytes transplanted into the spleen of the wild-type rats was detected for 22 weeks (Fig. 1B); the spleen has been recognized as the potential transplantation site for isolated hepatocytes and islets15,16. We also used luciferase-transgenic islets to demonstrate the efficacy of new treatment in the beta-cell replacement therapy for diabetes. For example, we used bioluminescent imaging to assess improvement in luciferase-transgenic islet engraftment in the subcutaneous site; we introduced clinically applicable strategy of improving the post-transplant oxygen inhalation to prevent hypoxia-induced graft loss 6 . A potential issue, specifically in the islet transplantation studies, is the effect of luciferase expression on the function and morphological phenotype of islets. However, we demonstrated that the ubiquitous luciferase expression in Firefly Rats does not alter the morphology, metabolism, and function in the isolated islets in the report recently published in Cell Transplantation 17 . This finding ensures that investigators can use isolated islets from Firefly Rats for the dual assessment of islet graft function and bioluminescence imaging.
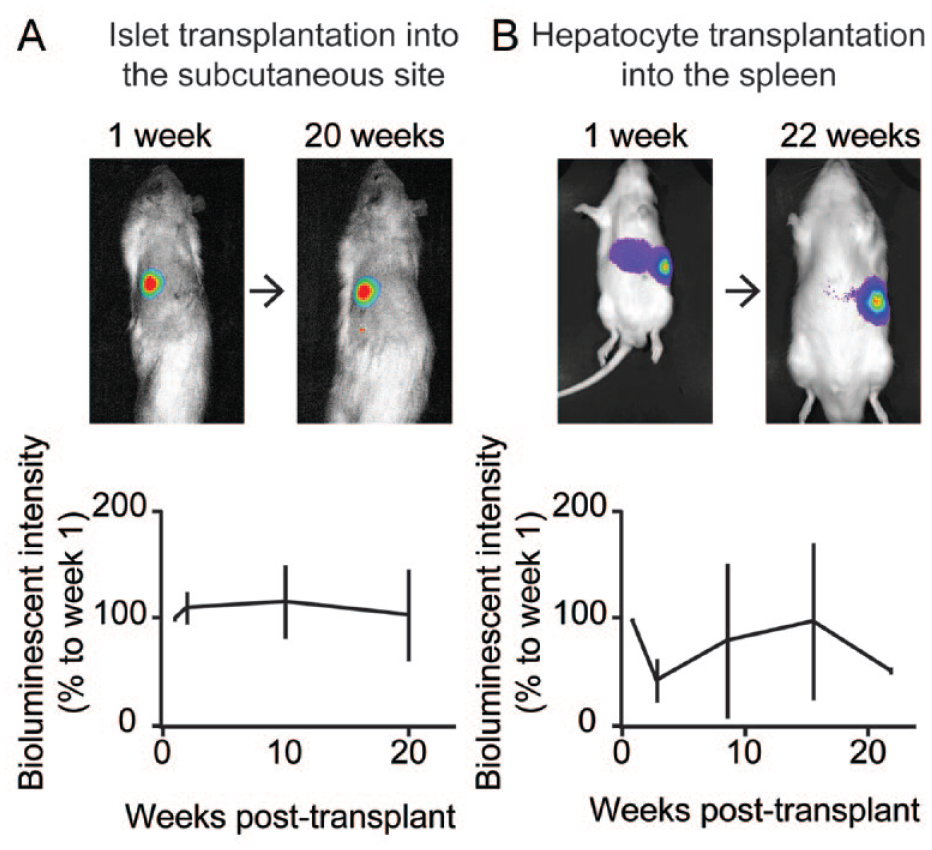
Long-term maintenance of the bioluminescence of the transplanted cells from Firefly Rats. (A) Six hundred isolated islets from the Firefly Rats were loaded on the synthetic Parylene-HT mesh scaffold and transplanted into the subcutaneous site of wild-type diabetic Lewis rats. Bioluminescent images were acquired using the Lago X platform on multiple timepoints over 20 weeks by intravenously injecting luciferin (15 mg/kg of body weight) via the tail vein under general anesthesia. (B) Six million parenchymal hepatocytes from Firefly Rats were injected into the spleen of wild-type Lewis rats. Bioluminescent images were acquired using the Lago X platform on multiple timepoints over 20 weeks by intraperitoneally injecting luciferin (30 mg/kg of body weight) via tail vein under general anesthesia. Top: Representative bioluminescent images over the observation period in the same recipients. Bottom: The bioluminescent intensity changes. Data represent the bioluminescent intensity (% to 1 week post-transplant) in each recipient. N = 3 for Figure 1A and N = 2 for Figure 1B. Error intervals demonstrate the standard deviation of the mean.
These Firefly Rats tend to experience reduced reproducibility in homozygous states. In our experience, hemizygote breeders yielded fewer homozygotes than expected (less than 25%), and these homozygotes failed to produce any offspring (unpublished). The adverse impact of luciferase expression on embryonic development could be a potential cause of the reduced reproducibility, while disruptions in embryonic development due to ROSA26-driven modifications have been scarcely reported. Alternatively, it could be a result of lethal insertion mutations commonly observed in homozygotes in any transgenic lineages. Accordingly, wild-type female rats are used for mating with male Firefly Rats to produce hemizygous luciferase-transgenic rats, ensuring the continuity of this transgenic rat strain. Remarkably, Firefly Rat strain has been maintained using this mating strategy for more than a decade in several affiliated institutions, including City of Hope Medical Center and Shanghai Jiao Tong University, for their transplantation research5,7,11,12. Firefly Rats are a critical and vitally important resource for scientific progress in many fields and, fortunately, are available to all researchers worldwide.
Footnotes
Acknowledgements
We thank Sung Hee Kil, PhD, for critical reading and editing of the manuscript. We also thank Hiroyuki Kato for processing the bioluminescent images.
Author Contributions
Conceptualization: EK, HK
Data curation and investigation and analysis: EK, YH, SE, KMS
Funding acquisition: HK
Resources: EK, YH, HK
Software: N/A
Supervision: EK, HK
Validation: N/A
Visualization: HK
Writing—Original draft: EK, HK
Writing—review & editing: YH, SE, KMS
Data Availability Statement
All data required to evaluate the conclusions in the study are present in the article. Additional data related to this article may be available to researchers upon request.
Ethical Approval
This study was approved by our institutional review board for animal studies.
Statement of Human and Animal Rights
The use of animals and animal procedures in this study were approved by the City of Hope/Beckman Research Institute and Jichi Medical University Institutional Animal Care and Use Committee.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Nora Eccles Treadwell Foundation to HK.
