Abstract
Amyotrophic lateral sclerosis (ALS) is characterized by progressive loss of motor neurons. Multilineage-differentiating stress-enduring (Muse) cells are unique endogenous stem cells that show therapeutic effects on motor function in ALS mouse models. We conducted a single-center open phase II clinical trial to evaluate the safety and clinical effects of repeated intravenous injections of an allogenic Muse cell-based product, CL2020, in patients with ALS. Five patients with ALS received CL2020 intravenously once a month for a total of six doses. The primary endpoints were safety and tolerability, and the secondary endpoint was the rate of change in the Revised Amyotrophic Lateral Sclerosis Functional Rating Scale (ALSFRS-R) score. In addition, serum tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), sphingosine-1-phosphate (S1P), cerebrospinal fluid chitotriosidase-1 (CHIT-1), and neurofilament light chain (NfL) levels were evaluated. The CL2020 treatment was highly tolerated without serious side effects. The ALSFRS-R score change trended upward at 12 months post-CL2020 treatment compared with that at 3 months pre-administration, but the difference was not statistically significant. Among five patients diagnosed with ALS, three exhibited a decrease in the rate of ALSFRS-R score change, one demonstrated an increase, and another showed no change. In addition, the patients’ serum IL-6 and TNF-α levels and cerebrospinal fluid CHIT-1 and NfL levels increased for up to 6 months post-treatment; however, their serum S1P levels continuously decreased over 12 months. These findings indicate a favorable safety profile of CL2020 therapy. In the near future, a double-blind study of a larger number of ALS patients should be conducted to confirm the efficacy of ALS treatment with CL2020.
Keywords
Introduction
Amyotrophic lateral sclerosis (ALS) is a fatal neurodegenerative disease characterized by progressive loss of motor neurons. It typically occurs during middle age or later, and the selective degeneration of motor neurons results in weakness and atrophy of the limb muscles, dysarthria, and dysphagia, eventually leading to death within 5 years of onset due to paralysis of the respiratory muscles 1 . ALS can be classified into two main types: sporadic ALS, which accounts for approximately 90% of cases, and familial ALS, which comprises approximately 10% of cases and is associated with genetic inheritance. Familial ALS is linked to mutations in genes, such as Cu/Zn superoxide dismutase (SOD1)2,3, TAR DNA binding protein 43 (TDP-43)4,5, and a hexanucleotide repeat expansion of the C9orf72 gene6,7. In addition to the oral drug riluzole and free radical scavenger edaravone8,9, sodium phenylbutyrate/taurursodiol 10 has recently been approved as a new anti-ALS drug in the United States. However, its therapeutic benefits remain limited; thus, a novel and curable therapeutic strategy for ALS is still urgently required.
In 2010, multilineage-differentiating stress-enduring (Muse) cells were identified as pluripotent stem cells from bone marrow-derived mesenchymal stem cells (BM-MSCs) 11 . Muse cells can be isolated as double-positive cells for SSEA-3 (a human pluripotent stem cell marker) and CD105 (a mesenchymal stem cell marker) and can differentiate into cells representative of all three germ layers. In addition, Muse cells can be engrafted intravenously into damaged tissues and differentiate into tissue-specific cells, resulting in tissue repair and functional recovery in models of hepatitis, muscle degeneration, and myocardial infarction12–14. To harness the therapeutic potential of Muse cells, CL2020 was developed as a Muse cell-based product enriched with SSEA-3-positive cells from BM-MSCs.
In recent studies, researchers evaluated motor function parameters in an ALS mouse model, specifically in SOD1 transgenic mice. Intravenous injection of human Muse cells revealed successful homing to the lumbar spinal cords, mainly at the pia mater and underneath the white matter. After repeated intravenous administration of Muse cells, a significant improvement in hindlimb muscle weakness was observed15,16. Furthermore, a single intravenous administration of CL2020 in TDP-43 transgenic mice significantly improved limb muscle weakness (unpublished data). In addition, Muse cells have been reported to inhibit the activation of inflammatory cells and regulate immune functions 17 . Based on these results, CL2020 could be used as a novel regenerative medicine for the treatment of ALS.
As the safety of CL2020 was confirmed in clinical trials including patients with myocardial infarction and epidermolysis bullosa based on the establishment of a frozen cell manufacturing system under the Good Manufacturing Practice (GMP) system18,19, we conducted a clinical trial to evaluate the safety and therapeutic effect of CL2020 in patients with ALS.
Materials and Methods
Study Design and Treatment Protocol
This was a single-center open phase II clinical trial to evaluate the safety and clinical effects of repeated intravenous injections of the Muse cell-based product CL2020 in patients with ALS (Japan Registry of Clinical Trials, Trial ID: jRCT2063200047). Due to the progressive nature of ALS, regular and multiple doses were considered appropriate. The selected dose of Muse cells, which demonstrated efficacy in ALS animal models 16 , was consistent with the dose used in a myocardial infarction animal model13,20. Therefore, the dose for patients with ALS in this clinical trial was set at 15 × 106 cells per dose, which was the same as the dose selected in a clinical trial for patients with cerebral infarction. Furthermore, as no safety concerns have been reported in clinical trials of patients with myocardial infarction, epidermolysis bullosa, or neonatal hypoxic-ischemic encephalopathy18,19, the same dosage was considered appropriate. CL2020 (15 × 106 cells) was administered intravenously once every 1 month for a total of six doses under open-label conditions. The primary endpoints of the study were safety and tolerability for up to 12 months after the first administration. The key secondary endpoint was the rate of change in the total Revised Amyotrophic Lateral Sclerosis Functional Rating Scale (ALSFRS-R) score over a period of 12 months. In addition, several other secondary endpoints were assessed, including changes in the ALSFRS-R scores for bulbar, limb, and respiratory functions, percent forced vital capacity (%FVC), changes in Manual Muscle Testing (MMT) scores, grip strength scores, pinch strength scores, Modified Norris Scale scores, 40-item ALS Assessment Questionnaire-40 (ALSAQ-40) total scores, and EuroQol 5 Dimension 5 Level (EQ-5D-5L) scores. These measures were used to evaluate both the safety and efficacy of the treatment for 12 months. In addition, the levels of serum tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and sphingosine-1-phosphate (S1P) were measured at different time points as exploratory endpoints, including at the beginning of the screening, at the end of the screening, and 1, 2, 3, 4, 5, 6, 8, 10, and 12 months after the first administration. These measurements aimed to evaluate standard safety biochemical and blood parameters. Cerebrospinal fluid (CSF) samples were also obtained at the beginning of screening and 2, 4, and 6 months after the first administration. Moreover, chitotriosidase-1 (CHIT-1) and neurofilament light chain (NfL) levels were also evaluated to confirm the treatment’s prognosis and therapeutic efficacy in ALS.
Participants
Prior clinical trials with CL2020 were conducted in patients with acute myocardial infarction, stroke, epidermolysis bullosa, spinal cord injury, and neonatal cerebral palsy, in which CL2020 was administered only once. In the present study, CL2020 was administered 6 times; however, because of the need to confirm the safety of multiple doses of CL2020, only five ALS patients were included.
Eligible participants were those aged 20–80 years with laboratory-supported probable, probable, or definite ALS based on the updated Awaji criteria 21 , grade 1 or 2 in the Japan ALS severity classification, symptom onset ≤24 months before screening, and %FVC ≥80% during screening. Participants were permitted to receive a stable dose of riluzole. Individuals were excluded if they had inflammatory disorders, a recent history of active neoplastic processes within the last 5 years, or active infections as determined through positive tests for hepatitis B antigens, hepatitis C virus antibodies, HIV antibodies, antibodies against human T-cell lymphotropic virus type 1, and syphilis. In addition, prior bone marrow transplantation or stem cell therapy was not permitted. Following the first 3 months of the pre-treatment period, a final inclusion criterion required a decline of −1 to −4 in the ALSFRS-R score, which was consistent with previous clinical trials22,23 (see Fig. 1: Flowchart of the trial).
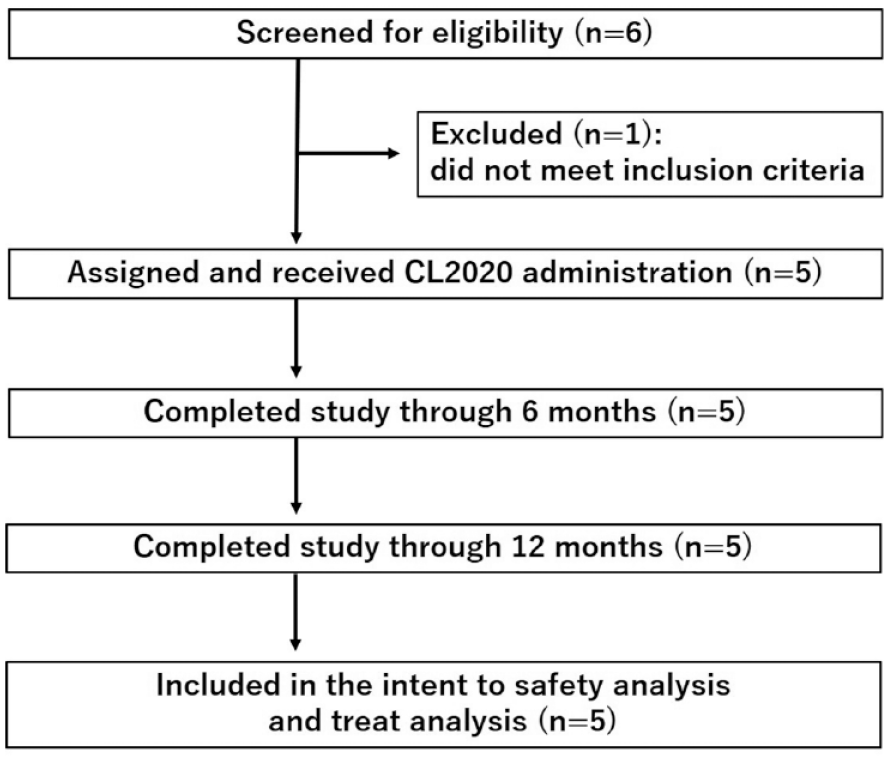
CONSORT flow diagram.
CL2020 Preparation, Administration, and Procedure Follow-Up
Clinical-grade CL2020 (15 × 10⁶ cells/15 mL) was manufactured from human (allogenic) MSCs by Life Science Institute, Inc. (Tokyo, Japan) and frozen until use. The CL2020 suspension (15 mL) was diluted with approximately 37 mL of Ringer’s acetic acid solution (Terumo, Tokyo, Japan). The diluted cell suspension (15 × 106 cells/52 mL) was administered intravenously for 10 to 15 min.
Outcome Assessments
For ALS outcome measurement, the ALSFRS-R score was used 24 . This scale consists of 12 items that assess various aspects of the disease progression and functional abilities of individuals with ALS. ALSFRS-R scores range from 0 to 48, with a lower score indicating greater impairment. These items include speaking, salivation, swallowing, writing, eating, stairs, walking, dressing, symptomatic movements, dyspnea, orthopnea, and respiratory failure.
The ALSAQ-40 was used to assess the quality of life of the participants. ALSAQ-40 scores range from 0 to 160, with a lower score indicating better quality of life. It consists of 40 questions covering eight domains: physical functioning, role limitations, emotional well-being, pain, fatigue, social functioning, sleep, and coping 25 .
The EQ-5D-5L was used to assess the individuals’ health-related quality of life. EQ-5D-5L scores range from −0.59 to 1, where 1 is the best possible health state. It consists of five dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression 26 .
Statistical Analyses
In the ALSFRS-R score assessment, changes in parameters were analyzed post hoc by calculating the slope of ALSFRS-R scores before and after the intervention. Before the intervention indicates the mean score change per month from 3 months before the intervention to the beginning of the intervention, while after the intervention indicates the mean score change per month from the beginning of the intervention to 12 months after the intervention. For hematologic tests, biochemical tests, and biomarkers, the analysis involved calculating summary statistics and two-sided 95% confidence intervals (CIs) of the mean for the change from baseline at each time point up to 12 months after the first dose. In addition, adjusted mean values were calculated, taking into account the baseline values. The adjusted means were accompanied by their two-sided 95% CIs and P-values, which were determined using a linear mixed-effects model. This statistical approach helps account for individual variations and provides more accurate estimates of the treatment effect. The model used fixed effects for time points and baseline values, considering the correlation structure between time points in the subjects. The estimability of the correlation structure was determined to ensure appropriate modeling.
The Kenward–Roger method was used to calculate degrees of freedom. Data were analyzed using SAS® (SAS Institute, Cary, NC, USA) version 9.4 or higher and expressed as mean values ± standard deviations. In all statistical analyses, statistical significance was set at P < 0.05.
Results
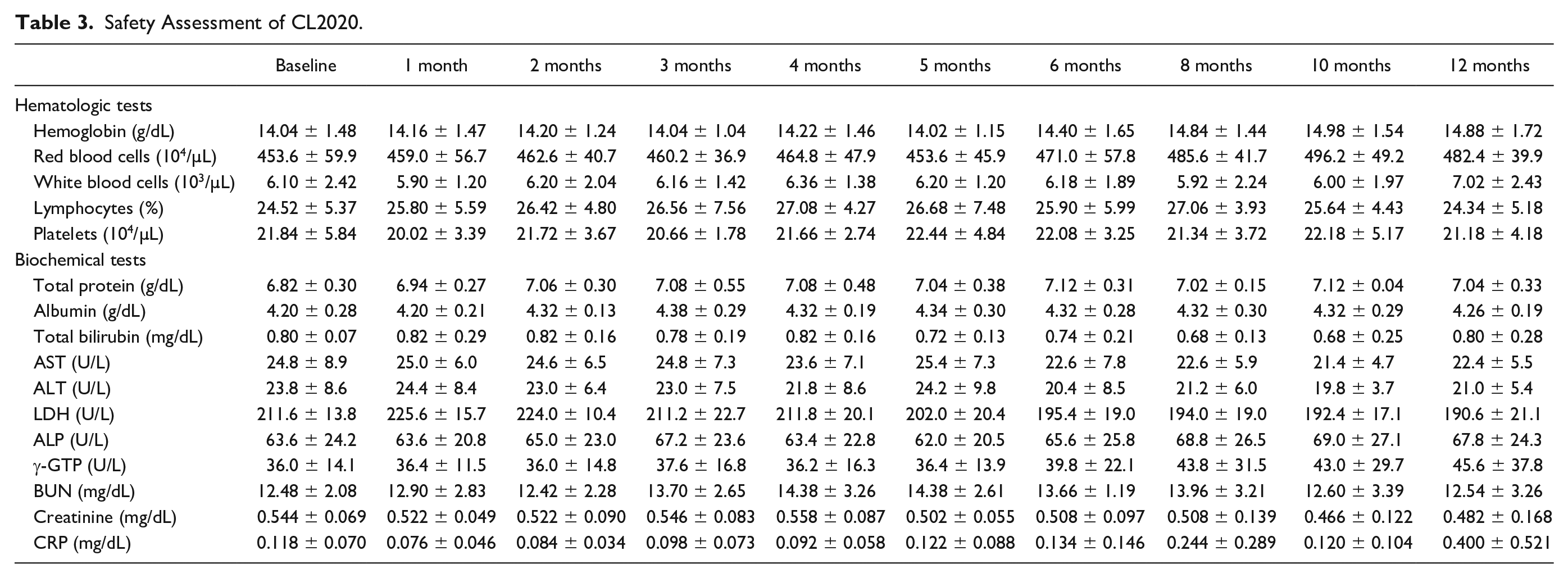
In this study, five patients with ALS received CL2020 (male-to-female ratio of 3:2), and their clinical forms were limb-onset (Table 1). We started recruiting patients in February 2021. Enrolment was completed in April 2021, and the last patient completed the trial in September 2022. A total of 28 adverse events occurred in five patients up to 12 months after the first dose (Table 2). Common adverse events were headache (four cases) and fatigue (three cases). The only serious adverse event was a bone fracture in Patient 2, which was graded as grade 3 according to the Common Terminology Criteria for Adverse Events version 5.0. There were no significant changes in the patients’ vital signs, medical examination results, electrocardiogram results, oxygen saturation values, or laboratory test results, including hematological and biochemical test results (Table 3).
Clinical Characteristics of the Patients with ALS.

ALSFRS-R, Revised Amyotrophic Lateral Sclerosis Functional Rating Scale.
Adverse Events.

MedDRA, Medical Dictionary for Regulatory Activities.
Safety Assessment of CL2020.
The change in the ALSFRS-R scores exhibited a declining trend over the course of 12 months (baseline, 40.6 ± 2.6, 95% CI = 37.4–43.8; 6 months, 39.0 ± 4.8, 95% CI = 33.0–45.0, P = 0.094; 12 months, 37.6 ± 5.2, 95% CI = 31.2−44.0, P = 0.022). However, the rate of change in the ALSFRS-R scores showed a trend toward improvement at 12 months post-treatment compared with that at 3 months pre-CL2020 administration; however, the difference was not statistically significant (Fig. 2, pre-treatment: β = −0.47 vs. post-treatment: β = −0.25, P = 0.096). Of the five patients with ALS, three (Patients 3, 4, and 6) showed a decrease in the rate of change in the ALSFRS-R score, one showed an increase, and one showed no change (Table 1).

Longitudinal follow-up of the changes in the ALSFRS-R scores.
The change in the %FVC showed a gradual downward trend over the 12-month period (baseline, 100.38 ± 12.77, 95% CI = 84.53–116.23; 6 months, 98.10 ± 17.55, 95% CI = 76.30–119.90, P = 0.515; 12 months, 93.68 ± 13.55, 95% CI = 76.86–110.50, P = 0.012). The change in the %FVC was maintained in two patients and showed a decreasing trend in three patients.
The change in the total MMT score showed a decreasing trend over time (baseline, 53.2 ± 4.4, 95% CI = 47.7–58.7; 6 months, 51.0 ± 4.2, 95% CI = 45.7–56.3, P = 0.004; 12 months, 47.3 ± 3.8, 95% CI = 41.2–53.3, P = 0.009). By 12 months, the total MMT score decreased in four of the five CL2020-treated patients and remained unchanged in one.
The total Modified Norris Scale score demonstrated a decrease over time up to 12 months (baseline, 88.4 ± 5.0, 95% CI = 82.2–94.6; 6 months, 83.8 ± 13.0, 95% CI = 67.6–100.0, P = 0.119; 12 months, 79.4 ± 14.0, 95% CI = 62.0–96.8, P = 0.042). For individual patients, the total Modified Norris Scale score remained almost unchanged in three patients and decreased in two throughout the 12-month period.
The ALSAQ-40 score showed a transient decrease at 1 month, followed by an increase over time up to 12 months (baseline, 75.6 ± 14.9, 95% CI = 57.1−94.1; 1 month, 67.4 ± 10.9, 95% CI = 53.8−81.0, P < 0.001; 6 months, 79.6 ± 16.0, 95% CI = 59.7−;99.5, P = 0.192; 12 months, 94.4 ± 18.5, 95% CI = 71.4−117.4, P = 0.002).
Serum IL-6 levels exhibited a significant increase of 0.2744 pg/ml (95% CI = 0.0833−0.4655, P = 0.006) at 3 months and 0.3804 pg/ml (95% CI = 0.0388−0.7220, P = 0.030) at 6 months after administration (Fig. 3A). Serum TNF-α levels also showed a significant increase of 1.988 pg/ml (95% CI = 0.390–3.586, P = 0.016) at 3 months, 1.422 pg/ml (95% CI = 0.703−2.141, P < 0.001) at 4 months, 1.946 pg/ml (95% CI = 0.134−3.758, P = 0.036) at 6 months, 2.066 pg /ml (95% CI = 0.747−3.385, P = 0.003) at 8 months, and 1.526 pg/ml (95% CI = 0.090−2.962, P = 0.038) at 10 months (Fig. 3B). Meanwhile, serum S1P levels showed a significant decrease of −30.0 pg/ml (95% CI = −57.5 to −2.5, P = 0.034) at 4 months and −59.6 pg/ml (95% CI = −86.5 to −32.7, P < 0.001) at 12 months (Fig. 3C). Cerebrospinal fluid CHIT-1 levels showed a significant increase of 541.2 pg/ml (−95% CI = 398.5 to 683.9, P < 0.001) at 4 months and 861.0 pg/ml (−95% CI = 396.8−1,325.2, P = 0.003) at 6 months (Fig. 3D). However, CSF NfL levels showed a tendency to increase but the change did not reach statistical significance (Fig. 3E).

Longitudinal changes in biomarkers. (A–E) The data of each biomarker was expressed as the mean ± SD.
Discussion
In this study, the safety and efficacy of CL2020 were investigated in five patients diagnosed with sporadic ALS after multiple intravenous administrations (CL2020 once every 1 month for a total of six doses). Despite repeated intravenous administration of CL2020 prepared from the bone marrow cells of other individuals, the treatment was highly tolerated without pulmonary embolisms, anaphylactic shock, or other serious side effects. However, there was one serious adverse event in Patient 2, which was a bone fracture occurring 12 months after the initial dose. After a thorough investigation, a causal relationship between the bone fracture and CL2020 treatment was ruled out. Unfortunately, this accidental adverse event may have contributed to a worsened clinical score, including the ALSFRS-R, in Patient 2.
Despite the enrolment of patients whose ALSFRS-R scores worsened by 1−2 points during the screening period, the ALSFRS-R score did not worsen in three out of five patients until 6 months after the administration of CL2020, indicating that multiple doses of CL2020 may prevent the deterioration of symptoms in patients with ALS. In addition, the rate of change in the ALSFRS-R scores showed a tendency to improve at 12 months after treatment when compared with that at 3 months before treatment (period pre-treatment: β= −0.47 vs. post-treatment: β= −0.25), supporting the therapeutic effect of CL2020 in ALS. A previous study reported that intrathecal transplantation of autologous mesenchymal stem cell neurotrophic factor-secreting cells (MSC-NFT) inhibited ALS progression (Phase 2a, 14 ALS patients, pre-treatment: β = −1.4 vs. post-treatment: β = −0.6). Although direct comparisons may be difficult due to different cell characteristics, the beneficial effects of MSC-NFT are similar to those observed in this study 27 .
Increasing evidence suggests that pro-inflammatory cytokines (IL-6 and TNF-α) and S1P-related signaling may contribute to the pathogenesis of ALS15,28. Muse cell was reported to have anti-inflammatory activity that suppressed the secretion of inflammatory cytokines in basic research and clinical research, including research on acute myocardial infarction 18 . However, in the present study, serum IL-6 and TNF-α levels tended to increase up to 12 months after CL2020 administration (Fig. 3A, B). Serum IL-6 and TNF-α levels do not appear to be significantly different from those of ALS natural history patients (serum IL-6: 2.75 pg/ml, serum TNF-α: 2.97 pg/ml)29,30. Based on this finding, it is speculated that CL2020 treatment could not suppress the secretion of inflammatory cytokines in ALS patients, probably because ALS is a progressive disease that gradually exacerbates chronic inflammation. In contrast, serum S1P levels continuously decreased for up to 12 months (Fig. 3C). CL2020 expresses S1P receptor 2, which allows for a strong response to S1P 15 . Notably, a functional antagonist of S1P, fingolimod, was reported to show beneficial effects with modulation of immune response in ALS mouse model 31 . Therefore, administration of CL2020 might functionally neutralize S1P, thereby attenuating S1P-related signaling in ALS patients.
Cerebrospinal fluid CHIT-1 and NfL levels are associated with ALS progression and patient survival32,33. In the present study, even after CL2020 administration, CHIT-1 and NfL levels tended to increase for up to 6 months, but they were relatively lower than those of ALS natural history patients (CSF CHIT-1: 22,015 pg/ml, CSF NfL: 7693.8 pg/ml)34,35, indicating that ALS patients with a relatively slow rate of disease progression were included in this trial.
Considering the results of clinical scores, including the ALSFRS-R, and the biomarker findings mentioned above, it can be concluded that CL2020 treatment alone may have slowed down ALS progression but was not fully capable of halting it. In other words, several pathways may exist in parallel in the pathological progression of ALS 36 , and CL2020 may attenuate only one of them and not alter another pathway related to CHIT-1 or NfL. Therefore, combination therapy with other drugs, such as edaravone 9 or sodium phenylbutyrate/taurursodiol 10 , should be considered in the future to advance the treatment of ALS.
There are some limitations in interpreting the results of this study, including the small number of patients (n=5), the relatively short observation period (6 months after final administration), the lack of comparisons, and post hoc analysis of ALSFRS-R slope. Thus, a double-blinded study including a larger number of ALS patients with a longer observation period should be conducted to evaluate the effectiveness of the CL2020 treatment.
Taken together, these results suggest that there are no safety concerns associated with multiple doses of CL2020 in patients with ALS. The administration of multiple doses of CL2020 in patients with ALS has the potential to prevent the worsening of symptoms in these patients.
Footnotes
Acknowledgements
The authors extend their heartfelt gratitude to all the participating patients and their families.
Author Contributions
T.Y. and K.A. conceived, designed, and coordinated the research; T.Y., Y.N., R.S., K.T., Y.O., T.Y., Y.K., N.M., Y.T., C.M., and R. M. performed the research and participated in the discussion; T.Y. and K.A. analyzed the data and wrote the paper; and T.Y. and K.A. obtained funding. All the authors have read and approved the final draft of the manuscript.
Availability of Data and Materials
The data that support the findings of the present study are available from the corresponding authors upon reasonable request.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Research Ethics and Patient Consent
The study protocol was approved by the Institutional Review Board of Okayama University Hospital (approval no. R20201702) and conducted in compliance with “The Act on Securing Quality, Efficacy, and Safety of Products Including Pharmaceuticals and Medical Devices” in Japan, the “Ordinance on Good Clinical Practice of Regenerative Medical Products,” and principles of the Declaration of Helsinki. Written informed consent was obtained from all participants. All laboratory processes were clearly defined and controlled to satisfy the GMP regulations.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.Y. and K.A. have patents for the therapeutic use of CL2020 for ALS. The other authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Life Science Institute, Inc. and a Grant-in-Aid for Scientific Research (23K08543).
