Abstract
Angiogenesis is strongly associated with ovarian hyperstimulation syndrome (OHSS) progression. Early growth response protein 1 (EGR1) plays an important role in angiogenesis. This study aimed to investigate the function and mechanism of EGR1 involved in OHSS progression. RNA-sequencing was used to identify differentially expressed genes. In vitro OHSS cell model was induced by treating KGN cells with human chorionic gonadotropin (hCG). In vivo OHSS model was established in mice. The expression levels of EGR1, SOX1, and VEGF were determined by Quantitative Real-Time polymerase chain reaction (qRT-PCR), Western blot, immunofluorescence staining, and immunochemistry assay. The content of VEGF in the culture medium of human granulosa-like tumor cell line (KGN) cells was accessed by the ELISA assay. The regulatory effect of EGR1 on SRY-box transcription factor 9 (SOX9) was addressed by luciferase reporter assay and chromatin immunoprecipitation. The ERG1 and SOX9 levels were significantly upregulated in granulosa cells from OHSS patients and there was a positive association between EGR1 and SOX9 expression. In the ovarian tissues of OHSS mice, the levels of EGR1 and SOX9 were also remarkedly increased. Treatment with hCG elevated the levels of vascular endothelial growth factor (VEGF), EGR1, and SOX9 in KGN cells. Silencing of EGR1 reversed the promoting effect of hCG on VEGF and SOX9 expression in KGN cells. EGR1 transcriptionally regulated SOX9 expression through binding to its promoter. In addition, administration of dopamine decreased hCG-induced VEGF in KGN cells and ameliorated the progression of OHSS in mice, which were companied with decreased EGR1 and SOX9 expression. EGR1 has a promoting effect on OHSS progression and dopamine protects against OHSS through suppression of EGR1/SOX9 cascade. Our findings may provide new targets for the treatment of OHSS.
Introduction
Ovarian hyperstimulation syndrome (OHSS) is a serious iatrogenic complication after ovulation induction or controlled ovarian stimulation, which can be life-threatening in severe case 1 . OHSS occurs in 20%~30% patients who undergo assisted reproductive technology 2 and the incidence of severe OHSS ranges from 2% to 9%3,4. The clinical manifestations of OHSS include ascites, hydrothorax, enlarged ovaries, respiratory distress syndrome, acute renal insufficiency, embolism, and even death 5 . A number of strategies have been developed to prevent OHSS, such as controlled ovarian stimulation, altering human chorionic gonadotropin (hCG) dose, canceling the cycle of in vitro fertilization, and intravenous albumin infusion during oocyte pickup. However, the efficacy of these approaches is unsatisfied6 –8 and novel strategies are still required.
Understanding the mechanisms driving OHSS progression will promote the development of new therapeutic approaches. It has been demonstrated that increased vascular permeability, which results in a leaking of fluid from the blood vessels to other parts of the body, is one of the major reasons responsible for the progression of OHSS9,10. Therefore, factors controlling vascular permeability/angiogenesis are reported to be closely associated with the pathogenesis of OHSS 11 . Early growth response protein 1 (EGR1) is a transcription regulator that exerts its function through binding to the promoter of target genes to regulate their transcription activity 12 . EGR1 has been reported to control angiogenesis 13 and plays an important role in the progression of OHSS11,14. However, the detailed function and mechanism of EGR1 in OHSS remains unclarified.
SRY-box transcription factor 9 (SOX9) is a member of the family of SOX proteins, which plays important roles in cell-type specification and cell differentiation 15 . Dysfunction of SOX9 has been linked to campomelic dysplasia and tumorigenesis of multiple cancers, such as lung cancer, liver cancer, breast cancer, and colorectal cancer16 –20. SOX9 plays a vital role in the regulation of angiogenesis21 –24 and SOX9 has been declared to be a downstream factor of EGR1 25 . However, whether SOX9 contributes to EGR1-mediated OHSS progression remains unknown.
Dopamine is a neuromodulator and neurotransmitter involved in a number of physiological processes 26 . Numerous researches have been performed to investigate the efficacy of dopamine agonists in preventing OHSS6,27 –30, but the exact mechanisms of dopamine against OHSS are not fully understood. In this study, we investigated the function and mechanism of EGR1 in OHSS and explored the possible contribution of EGR1/SOX9 cascade in dopamine-mediated suppression of OHSS.
Methods and Materials
Cell Culture
Human granulosa-like tumor cell line (KGN) cells were purchased from the Chinese Academy of Sciences (Shanghai, China) and cultured in DMEM/F12 media (Gibco) containing 10% FBS and 1% penicillin and streptomycin. Cells were incubated in a humidified atmosphere with 5% CO2 at 37°C. Cells were passaged every 4~6 days. To induce in vitro OHSS cell model, KGN cells were treated with 10 IU/ml hCG (Solarbio, Beijing, China) for 24 h. For dopamine treatment, KGN cells were treated with 10 IU/ml hCG together with 10 μM dopamine (Macklin, Shanghai, China) for 24 h, and then cells were collected for further experiments.
Isolation of Ovarian Granulosa Cells
Infertile patients who underwent oocyte retrieval at the Third Affiliated Hospital of Guangzhou Medical University between September 2019 and June 2020 were enrolled in the study. All participating patients gave their written informed consent. Follicular fluid samples were collected from seven OHSS patients and seven age-matched non-OHSS patients. The diagnosis of OHSS was based on the 2016 published clinical practice guidelines 31 . Granulosa cells were isolated from follicular fluid samples by density gradient centrifugation. Granulosa cells were maintained in DMEM/F12 (Gibco, Shanghai, China) containing 10% fetal bovine serum (FBS). After being cultured overnight, the cells were collected for RNA isolation. The RNA sequencing was performed by Novogene (Beijing, China).
siRNA and DNA Transfection
siRNA sequences targeting EGR1 and control siRNA were purchased from GenePharma (Shanghai, China). pcDNA3.1-EGR1 overexpression plasmid was purchased from Fenghui (Hunan, China). siRNA (5 nmol) and DNA (2 μg) transfection were performed using Lipofectamine 2000 (Thermo Fisher Scientific, USA).
Quantitative Real-Time Polymerase Chain Reaction
Total RNA was extracted from KGN cells using TRIzol reagent (Invitrogen, Shanghai, China). Total RNA was reverse-transcribed into cDNA using the PrimeScript RT reagent Kit (seyotin, China) according to the manufacturer’s introductions. PCR was performed using the SYBR Green PCR Master Mix (seyotin, China). The primers used were as follows: GAPDH-F, 5’-GAGTCAACGGATTTGGTCGT-3’ and GAPDH-R, 5’-GACAAGCTTCCCGTTCTCAG-3’; VEGF-F, 5’-GGGCCTCCGAAACCATGAAC-3’ and VEGF-R, 5’-TTCTGCCCTCCTCCTTCTGC-3’; EGR1-F, 5’-GTTACCCCAGCCAAACCACT-3’ and EGR1-R, 5’-GTGGGTTGGTCATGCTCACT-3’; and SOX9-F, 5’-CAGTACCCGCACTTGCACA-3’ and SOX9-R, 5’-CCCGTTCTTCACCGACTTCC-3.’ The experiment was performed in triplicate. The relative gene expression was calculated using the 2–ΔΔCt formula and the expression of GAPDH was used as an internal control.
Western Blot
KGN cells were harvested and lyzed in RIPA buffer (Seyotin, Guangzhou, China). After incubation on ice for 30 min, cell lysate was centrifuged for 20 min at 4°C. The supernatant was collected and protein concentration was measured using the BCA method (Seyotin, Guangzhou, China). Equal amounts of protein (20 μg) were separated by SDS-PAGE and transferred onto PVDF membranes (Millipore, USA). The membrane was blocked with 5% nonfat milk and incubated with primary antibody overnight at 4°C. After three washes with TBS-tween 20, the membrane was incubated with the HRP-conjugated secondary antibody. Protein bands were detected using the Immobilon Western Kit (Seyotin, Guangzhou, China). Antibodies against VEGF (ab46154), EGR1 (ab133695), Sox9 (ab185966), and GAPDH (ab8245) were obtained from Abcam (USA).
ELISA Assay
The level of VEGF in culture medium was measured using the ELISA assay with a VEGF enzyme-linked immunosorbent assay kit (R&D Systems) according to the manufacturer’s instructions.
Chromatin Immunoprecipitation (CHIP)
The binding of EGR1 and SOX9 promoter was determined by the CHIP assay with the SimpleChIP Enzymatic Chromatin IP Kit (Cell Signaling Technology, USA) following the manufacturer’s protocol. The purified DNA was quantified by PCR using the primers, as follows: forward, 5’-CAGACTCCAGGCGCAGAAG-3’ and reverse, 5’-GACTTCGCTGGCGTTTACAG-3.’ The PCR products were analyzed by agarose gel electrophoresis.
Luciferase Reporter Assay
The SOX9 promoter region containing potential EGR1-binding sites 32 was cloned into the pGL4.10 vector. KGN cells were co-transfected with the luciferase reporter plasmids and pcDNA3.1-EGR1 or empty vector. After transfection for 48 h, luciferase activity was determined with the Dual Luciferase Reporter Gene Assay Kit (Yeasen Biotechnology, Shanghai, China) and normalized to Renilla activity.
Establishment of the Mouse OHSS Model
ICR mice (female, 5 weeks old) were purchased from Baishitong Laboratories (Beijing, China). The OHSS model was established as described previous33,34. Briefly, mice were randomly divided into three groups: control, OHSS, and OHSS+dopamine (n=6). The control group was intraperitoneally administered with 5 IU of PMSG (Solarbio, Beijing, China) on day 2 and 7 IU of hCG on day 5. The OHSS group was given 20 IU of PMSG for 4 consecutive days and 7 IU of hCG on day 5. Both the control and OHSS groups were intraperitoneally injected with a vehicle control (DMSO) from day 4 to day 6. The OHSS+dopamine group received 20 IU of PMSG for 4 consecutive days and 7 IU of hCG on day 5, followed by a supplement of dopamine (5 mg/kg/d) from day 4 to day 6. The mice were sacrificed and ovary tissues were collected on day 7. All animal experiments were performed according to the Guide for the Care and Use of Laboratory Animals and approved by the Laboratory Animal Ethics Committee of Guangzhou Medical University.
Immunofluorescence Staining
Ovary tissues or KGN cells were fixed with 4% paraformaldehyde for 10 min and permeabilized with 0.1% Triton X-100 for 10 min. Samples were then blocked with 2% BSA in PBS, followed by incubation with primary antibodies (VEGF, EGR1, or SOX9, dilution, 1:100) overnight at 4°C. Then, samples were incubated with an Alexa Fluor 488–labeled anti-mouse antibody and an Alexa Fluor 594–conjugated anti-rabbit antibody (Proteintech, Wuhan, China) for 1.5 h in the dark at room temperature. After washing with PBS, nuclei were stained with DAPI (Sigma, USA).
Immunohistochemistry
Ovary tissues collected from different treatment groups were formalin-fixed and paraffin-embedded at 4°C overnight. Ovary tissues were sectioned to 10 μm-thick slices. The sections were deparaffinized, dehydrated, and treated with 3% hydrogen peroxide in methanol. Then the slides were incubated with VEGF, EGR1, or SOX9 antibody at 4℃ overnight, followed by incubation and biotinylated by secondary antibodies. Then the slides were stained with haematoxylin and visualized under a light microscope.
Statistical Analysis
All data are expressed as mean ± SD from three independent experiments. Statistical analysis was performed using the GraphPad Prism 8 software. Difference between two groups as compared used the Student t test, while comparisons among multiple groups were analyzed by one-way ANOVA with Tukey’s multiple comparison test. Statistical significance was defined as P < 0.05.
Results
EGR1 and SOX9 Are Upregulated in Ovarian Granulosa Cells
To identify potential regulatory genes involved in OHSS progression, RNA sequencing was performed with granulosa cell samples from OHSS patients and normal women. The results showed that EGR1 and SOX9 were upregulated (Fig. 1A). Quantitative Real-Time polymerase chain reaction (qRT-PCR) assay further confirmed the increased mRNA levels of EGR1 and SOX9 in granulosa cells from OHSS patients compared with those in granulosa cells from normal women (Fig. 1B, C). In addition, EGR1 expression was positively associated with SOX9 expression (Fig. 1D). Together, these results indicated that EGR1 and SOX9 might be involved in OHSS progression.
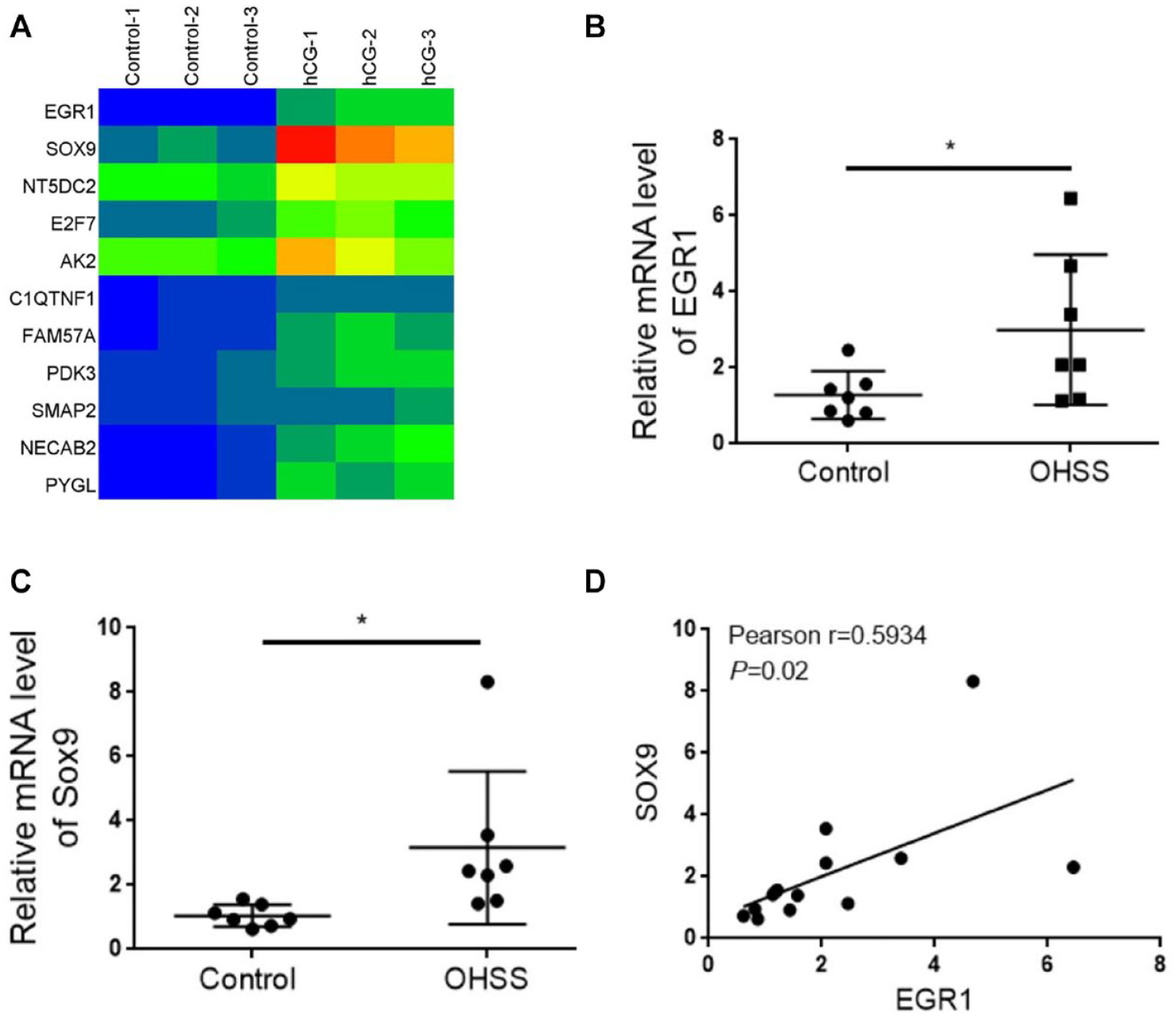
EGR1 and SOX9 are upregulated in ovarian granulosa cells from OHSS patients. (A) RNA-sequencing determining the differently expressed genes in ovarian granulosa cells from OHSS patients and normal women. (B) qRT-PCR assay examining the mRNA level of EGR1 in ovarian granulosa cells. (C) qRT-PCR assay examining the mRNA level of SOX9 in ovarian granulosa cells. (D) The association between EGR1 and SOX9. SOX9: SRY-box transcription factor 9; OHSS: ovarian hyperstimulation syndrome; qRT-PCR: Quantitative Real-Time polymerase chain reaction; EGR1: early growth response protein 1.
EGR1 and SOX9 Expressions Are Significantly Increased in Ovaries of OHSS Mice
To verify the function of EGR1 and SOX9 in OHSS, an experimentally established OHSS mouse model was utilized. HE staining showed that the size of ovaries was remarkedly increased in OHSS mice compared with control mice (Fig. 2A). VEGF is commonly used as a marker of OHSS 33 . Consistently, a significant elevation in VEGF level was observed in ovary tissues of OHSS mice in comparison with ovary tissues of control mice (Fig. 2B). These results indicated that the OHSS mouse model was constructed successfully. In addition, we observed that the expression levels of EGR1 and SOX9 were also obviously enhanced in ovaries of OHSS mice, when comparing with those in ovaries of control mice (Fig. 2C).

EGR1 and SOX9 expressions are elevated in ovaries from OHSS mice. (A) HE staining of ovary tissues from OHSS mice and control mice. (B) Immunofluorescence staining detecting the levels of EGR1 and SOX9 in ovaries from OHSS mice and control mice. (C) Immunochemistry analysis determining the expression of VEGF in ovary tissues from OHSS mice and control mice. EGR1: early growth response protein 1; SOX9: SRY-box transcription factor 9; OHSS: ovarian hyperstimulation syndrome; HE: Hematoxylin-eosin; VEGF: vascular endothelial growth factor; DAPI: 4,’6-diamidino-2-phenylindole.
HCG Induces EGR1 and SOX9 Expression in KGN Cells
KGN is a cell line derived from human ovarian granulosa cell tumors, which is often used to study the function of normal granulosa cells 35 . To investigate the implication of EGR1 and SOX9 in OHSS at the cell level, an hCG-induced OHSS cell model was used. After treating KGN cells with hCG, the mRNA level of VEGF was significantly increased (Fig. 3A). Consistently, the concentration of VEGF in the culture medium of hCG-treated KGN cells was remarkably elevated compared with control cells (Fig. 3B). Western blot analysis also displayed that the protein level of VEGF was much higher in hCG-treated KGN cells than that in control cells (Fig. 3C). The aforementioned results indicated that the hCG-induced OHSS cell model was established successfully. Then, we determined the abundance of EGR1 and SOX9 in hCG-treated KGN cells. The mRNA levels of EGR1 and SOX9 were significantly increased (Fig. 3D, F) and immunofluorescence staining showed that protein levels of EGR1 and SOX9 were obviously enhanced in hCG-induced KGN cells compared with control cells (Fig. 3E, G).

VEGF, EGR1, and SOX9 levels are increased in hCG-induced KGN cells. (A) qRT-PCR analysis of the mRNA level of VEGF in hCG-treated KGN cells. (B) ELISA determining the content of VEGF in the culture medium of hCG-treated KGN cells. (C) Western blot analysis of the protein expression of VEGF in KGN cells treated with hCG. (D) qRT-PCR analysis of the mRNA level of EGR1 in control and hCG-treated KGN cells. (E) Immunofluorescence staining examining the levels of EGR1 and SOX9 in hCG-treated KGN cells. (F) qRT-PCR analysis of the mRNA level of SOX9 in hCG-treated KGN cells. (G) Immunofluorescence staining determining the levels of EGR1 and SOX9 in hCG-treated KGN cells. VEGF: vascular endothelial growth factor; EGR1: early growth response protein 1; SOX9: SRY-box transcription factor 9; hCG: human chorionic gonadotropin; KGN: human granulosa-like tumor cell line; qRT-PCR: Quantitative Real-Time polymerase chain reaction; OHSS: ovarian hyperstimulation syndrome; DAPI: 4,’6-diamidino-2-phenylindole; ELISA: enzyme-linked immunosorbent assay; GAPDH: Glyceraldehyde-3-phosphate dehydrogenase. *P < 0.05; **P < 0.01.
EGR1 Promotes OHSS Progression Through Transcriptional Regulation of SOX9
To verify the activity of ERG1 in OHSS development, we knocked down EGR1 in KGN cells with three siRNA sequences. si-EGR1-3 was most efficient and was chosen for further experiments (Fig. 4A). qRT-PCR and immunofluorescence analysis revealed that silencing EGR1 resulted in a significant downregulation in hCG-induced VEGF expression at both mRNA and protein levels (Fig. 4B, C). hCG treatment elevated the mRNA expression of SOX9 in KGN cells and this effect was reversed by depletion of EGR1 (Fig. 4D). In agreement with this result, silencing of EGR1 decreased the fluorescence intensity of SOX9 that was increased by hCG (Fig. 4E). Given that EGR1 is a transcription regulator, we hypothesized that EGR1 promoted SOX9 expression through transcription. As expected, luciferase reporter assay displayed that overexpression of EGR1 markedly enhanced the luciferase activity of SOX9 promoter (Fig. 5A). The direct interaction between EGR1 and SOX9 promoter is required for EGR1 to regulate SOX9 transcription. Consistently, the CHIP assay showed that EGR1 immunoprecipitated the promoter of SOX9 (Fig. 5B). Collectively, these data indicated that EGR1 contributes to OHSS progression through modulation of SOX9 transcription.

Silencing EGR1 inhibits hCG-induced VEGF and SOX9 expression in KGN cells. (A) The knockdown efficiency of siRNAs (si-EGR1-1, si-EGR1-2, and si-EGR1-3) for EGR1 was determined by qRT-PCR. (B, C) qRT-PCR and immunofluorescence staining showing that silencing EGR1 decreased the mRNA and protein expression of VEGF in KGN cells treated with hCG. (D, E) qRT-PCR and immunofluorescence staining showing that knockdown of EGR1 reduced the mRNA and protein levels of SOX9 in KGN cells treated with hCG. EGR1: early growth response protein 1; hCG: human chorionic gonadotropin; VEGF: vascular endothelial growth factor; SOX9: SRY-box transcription factor 9; KGN: human granulosa-like tumor cell line; qRT-PCR: Quantitative Real-Time polymerase chain reaction; DAPI: 4,’6-diamidino-2-phenylindole. **P < 0.01.

EGR1 binds to the SOX9 promoter. (A) Luciferase reporter assay determining the effect of overexpression of EGR1 on the SOX9 promoter. (B) CHIP and electrophoresis analysis of the direct binding of EGR1 and the SOX9 promoter. EGR1: early growth response protein 1; SOX9: SRY-box transcription factor 9; CHIP: chromatin immunoprecipitation; OE-NC: overexpression-negative control; IgG: immunoglobulin G. **P < 0.01.
Dopamine Suppresses OHSS Progression Through EGR1/SOX9 Axis
Next, we explored the therapeutic effect of dopamine on OHSS progression in vitro. hCG treatment markedly elevated VEGF mRNA level in KGN cells and administration of dopamine suppressed this enhancement (Fig. 6A). ELISA assay showed that hCG increased VEGF content in the culture medium of KGN cells and this enhancement was abolished upon dopamine exposure (Fig. 6B). Immunofluorescence staining also showed that hCG-induced VEGF protein expression was reduced by the treatment of dopamine (Fig. 6C). These data indicated that dopamine protects against OHSS progression. In addition, treatment with dopamine reduced the mRNA levels of EGR1 and SOX9 in hCG-treated KGN cells (Fig. 7A, B). Consistently, the protein expressions of EGR1 and SOX9, which were increased by hCG, were also decreased by dopamine (Fig. 7C). Collectively, these results suggested that dopamine hampers OHSS through suppression of EGR1/SOX9 expression.
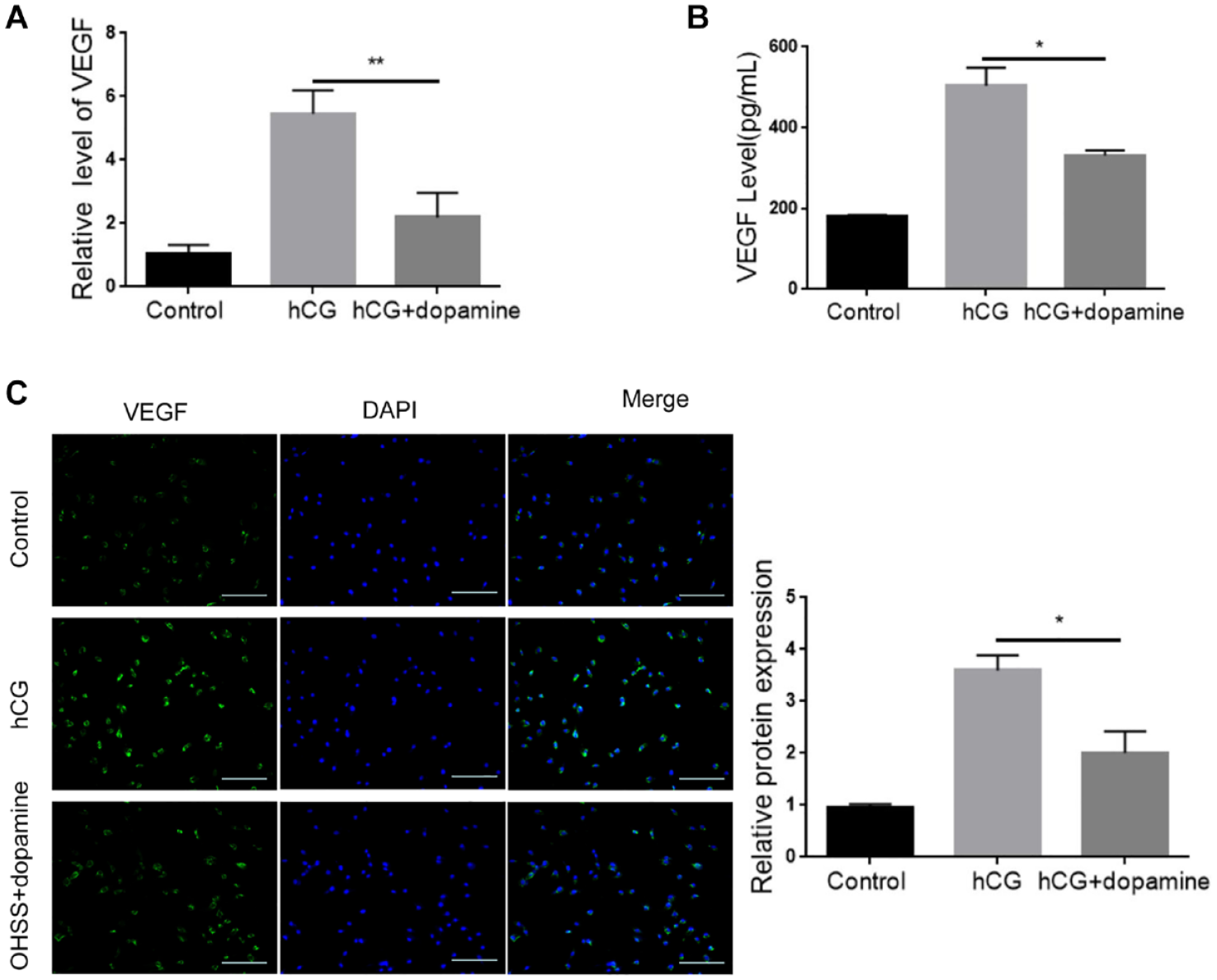
Dopamine inhibits hCG-induced VEGF expression in KGN cells. (A) qRT-PCR determining the mRNA level of VEGF in KGN cells treated with hCG alone or together with dopamine. (B) ELISA examining the content of VEGF in the culture medium of KGN cells treated with hCG alone or together with dopamine. (C) Immunofluorescence staining detecting the protein expression of VEGF in KGN cells treated with hCG alone or together with dopamine. hCG: human chorionic gonadotropin; VEGF: vascular endothelial growth factor; KGN: human granulosa-like tumor cell line; qRT-PCR: Quantitative Real-Time polymerase chain reaction; DAPI: 4,’6-diamidino-2-phenylindole; ELISA: enzyme-linked immunosorbent assay. *P < 0.05; **P < 0.01.

Dopamine suppresses hCG-induced EGR1 and SOX9 expression in KGN cells. (A) qRT-PCR analysis of the mRNA level of EGR1 in KGN cells treated with hCG alone or together with dopamine. (B) qRT-PCR analysis of the mRNA level of SOX9 in KGN cells treated with hCG alone or together with dopamine. (C) Immunofluorescence staining determining the protein levels of EGR1 and SOX9 in KGN cells treated with hCG alone or together with dopamine. hCG: human chorionic gonadotropin; EGR1: early growth response protein 1; SOX9: SRY-box transcription factor 9; KGN: human granulosa-like tumor cell line; qRT-PCR: Quantitative Real-Time polymerase chain reaction; OHSS: ovarian hyperstimulation syndrome; DAPI: 4,’6-diamidino-2-phenylindole. *P < 0.05.
Dopamine Attenuates the Pathogenesis of OHSS in Mice
To further investigate the effect of dopamine in OHSS progression, an OHSS mouse model was used. The results showed that the ovary size, body weight, and ovary weight were markedly increased in the OHSS group compared with the control group. However, administration of dopamine attenuated these parameters in OHSS mice (Figure 8A–C). Immunohistochemistry analysis revealed that the protein level of VEGF was elevated in the ovaries of the OHSS mice and this increase was blocked by dopamine (Fig. 8D). Moreover, administration of dopamine reduced the levels of EGR1 and SOX9 in the OHSS mice (Fig. 8E). Together, these results implied that dopamine ameliorates OHSS progression through the EGR1/SOX9 cascade.

Dopamine inhibits the pathogenesis of OHSS by downregulation of EGR1 and SOX9. (A) Representative ovaries from different treatment groups (Control, OHSS, and OHSS+dopamine). (B) Body weight of different treatment groups. (C) Ovarian weight of different treatment groups was determined. (D) Immunofluorescence staining determining the protein level of VEGF in ovaries of different groups. (E) Immunochemistry staining determining the protein levels of EGR1 and SOX9 in ovaries of different groups. *P < 0.05.
Discussion
Severe OHSS is a life-threatening problem. In this study, using both mouse and cell models, we found that EGR1 and SOX9 are involved in OHSS progression. Moreover, we confirmed the preventive activity of dopamine in OHSS and demonstrated that dopamine ameliorates OHSS by inactivation of the EGR1/SOX9 signaling.
Angiogenesis-related proteins are closely associated with the pathophysiology of OHSS36,37. EGR1 is a key regulator of angiogenesis 38 . It has been reported that EGR1 expression can be upregulated by hCG in granulosa cells, which is related to follicle maturation and ovulation 39 . Treatment with GnRH antagonist cetrorelix decreases EGR1 and VEGF expression and thus meliorates moderate and severe OHSS 11 . In rats, EGR1 binds to the promoter of Tie1 and enhances its transcription, thus triggering the progression of OHSS 40 . In agreement with these findings, we found that ERG1 expression was upregulated in ovarian granulosa cells of OHSS patients and ovaries of OHSS-modeled mice, as well as in hCG-treated KGN cells. VEGF is a key mediator involved in OHSS, which can be induced by hCG and elevates vascular hyperpermeability 33 . Agents or factor commonly regulate OHSS progression through modulation of VEGF expression41,42. Yang et al. 43 have reported that EGR1 inhibits the migration and angiogenesis of HTR-8/SVneo cells through downregulation of VEGF. Consistently, our results showed that silencing EGR1 reduced the level of VEGF that was elevated by hCG and thus contributed to the development of OHSS.
SOX9 is a transcription regulator that plays a central role in many cellular processes including angiogenesis. It has been reported that SOX9 expression is associated with angiogenesis in patients with gastric cancer 44 . Exosome circCMTM3 enhances angiogenesis in liver cancer through regulation of miR-3619-5p/SOX9 cascade 24 . Overexpression of miR-206 inhibits angiogenesis and invasion of TNBC cells through downregulation of SOX9 45 . However, the function of SOX9 in OHSS has not been reported. In this study, we found that SOX9 expression was increased in ovarian granulosa cells of OHSS patients and ovarian tissues of OHSS mice. It has been reported that depletion of SOX influences the expression of VEGF and alters the proliferation and migration of mesenchymal stem cells 46 . Here, we found that hCG increased VEGF expression, which was companied with elevated ERG1 and SOX9 expression. Previous studies demonstrated that SOX9 is a downstream effector of ERG1 32 . Consistently, we found that EGR1 directly bound to the promoter of SOX9 and modulated SOX9 expression. Taken together, our results implied that EGR1 modulates VEGF expression through regulation of SOX9.
Dopamine is a neurotransmitter that plays a critical role in many biological processes 47 . A number of studies have been conducted to test the usage of dopamine agonists in preventing OHSS27 –29,48. Dopamine has been reported to decrease VEGF expression and VEGFR2 phosphorylation to inhibit pulmonary edema 49 . Our results verified that dopamine could significantly reduce VEGF expression in hCG-treated KGN cells and ameliorate OHSS in mice, confirming the preventive potential of dopamine in OHSS. Moreover, we found that the expression levels of EGR1 and SOX9 were downregulated upon dopamine in both ovaries of OHSS mice and hCG-treated KGN cells. Therefore, our data indicated that dopamine alleviates OHSS through inactivation of the EGR1/SOX9 axis.
In this study, we found that EGR1 and SOX9 are increased in granulosa cells from patients with OHSS. Our further experiments verified that EGR1 promotes the progression of OHSS through transcriptionally regulated SOX9. Moreover, our data revealed that dopamine protects against OHSS through the EGR1/SOX9 cascade. Our findings indicate that EGR1 may serve as a potential target for the treatment of OHSS.
Footnotes
Acknowledgements
The authors thank all the members of the laboratory.
Author Contributions
H.W. and W.C. contributed to conception and design, Z.L. provided administrative support, Y.H. and Y.S. provided study materials or patients, H.W. and W.C. contributed to collection and assembly of data, and Y.L. and Y.Z. contributed to data analysis and interpretation. All authors contributed to manuscript writing, and accorded final approval of the manuscript.
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author (email:
Ethical Approval
All animal experiments were approved by the Ethics Committee of Guangzhou Medical University (GZXK2020-06).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (No. 81302079).
