Abstract
Transplants comprised of encapsulated islets have shown promise in treating insulin-dependent diabetes. A question raised in the scientific and clinical communities is whether the insulin released from an implanted encapsulation device damaged in an accident could cause a serious hypoglycemic event. In this commentary, we consider the different types of damage that a device can sustain, including the encapsulation membrane and the islets within, and the amount of insulin released in each case. We conclude that the probability that device damage would cause an adverse hypoglycemic event is indeed very low.
Several published studies as well as ongoing research demonstrate the utility of transplants comprised of encapsulated islets, isolated from donated pancreata or derived from stem cells, in treating insulin-dependent diabetes. Typically, the cells are of non-autologous, generally allogeneic, nature; therefore, the encapsulation barrier may be necessary for protection of the transplant from the immune system of the host. Various capsule configurations have been proposed, including microcapsules implanted intraperitoneally or under the kidney capsule and macrocapsules of tubular or planar geometry that can be implanted in different anatomic locations, such as intraperitoneally or subcutaneously. An underlying question raised in the scientific and clinical communities is if a macroencapsulation device is damaged in an accident would a bolus of insulin sufficient to cause a severe, possibly life-threatening, hypoglycemic episode be released? Such an event, which would exacerbate the medical challenges directly caused by the injuries themselves, could occur if the encapsulation device lies beneath the skin, as is the case with a subcutaneous membrane pouch implant. In this commentary, we examine the likelihood that damage of such a device implant could cause a severe hypoglycemic event.
Rupture of the encapsulation membrane in an accident is likely if a sharp object or projectile punctures the skin and the underlying device. If no further damage takes place, the insulin that would be immediately released would be that present extracellularly within the device. Because the encapsulation membranes are readily permeable to insulin, this amount is anticipated to be relatively small; it can be calculated using mathematical models of diffusion of nutrients into the construct and their consumption by the cells and of production of metabolites by the cells and their diffusion out of the construct. Models for spherical encapsulation devices have been described 1 . Fig. 1 shows typical concentration profiles for dissolved oxygen, glucose and insulin calculated for a planar encapsulation device containing a single layer of islets that are 150 µm in diameter. The thickness of the cell compartment between the two immunoprotective membranes is, therefore, also 150 µm. Each membrane is 40 µm thick. From the concentration profile, the average extracellular insulin concentration within the device is calculated to be 1.78 × 103 ng/ml or 51.3 mU/ml (1 mU insulin corresponds to 34.7 ng). Most clinical protocols define the minimum therapeutic dose of islets for a human to be 5,000 IEQs per kg body weight, although other studies suggest that 10,000 IEQs per kg body weight are necessary for insulin independence 2 . The volume of the device that accommodates 375,000 IEQs implanted in a 75-kg individual is calculated to be 1.27 mL, whereas for 750,000 IEQs it is 2.54 mL. The amount of extracellular insulin in the device then is 65–130 mU, well below the typical bolus of 4–6 units of insulin and therefore unlikely to cause a hypoglycemic effect. Furthermore, the above number of IEQs would likely be distributed in 2–3 subcutaneous devices, so the amount of insulin acutely released from each ruptured device would be a fraction of the above amount depending on the number of implanted devices that are damaged.
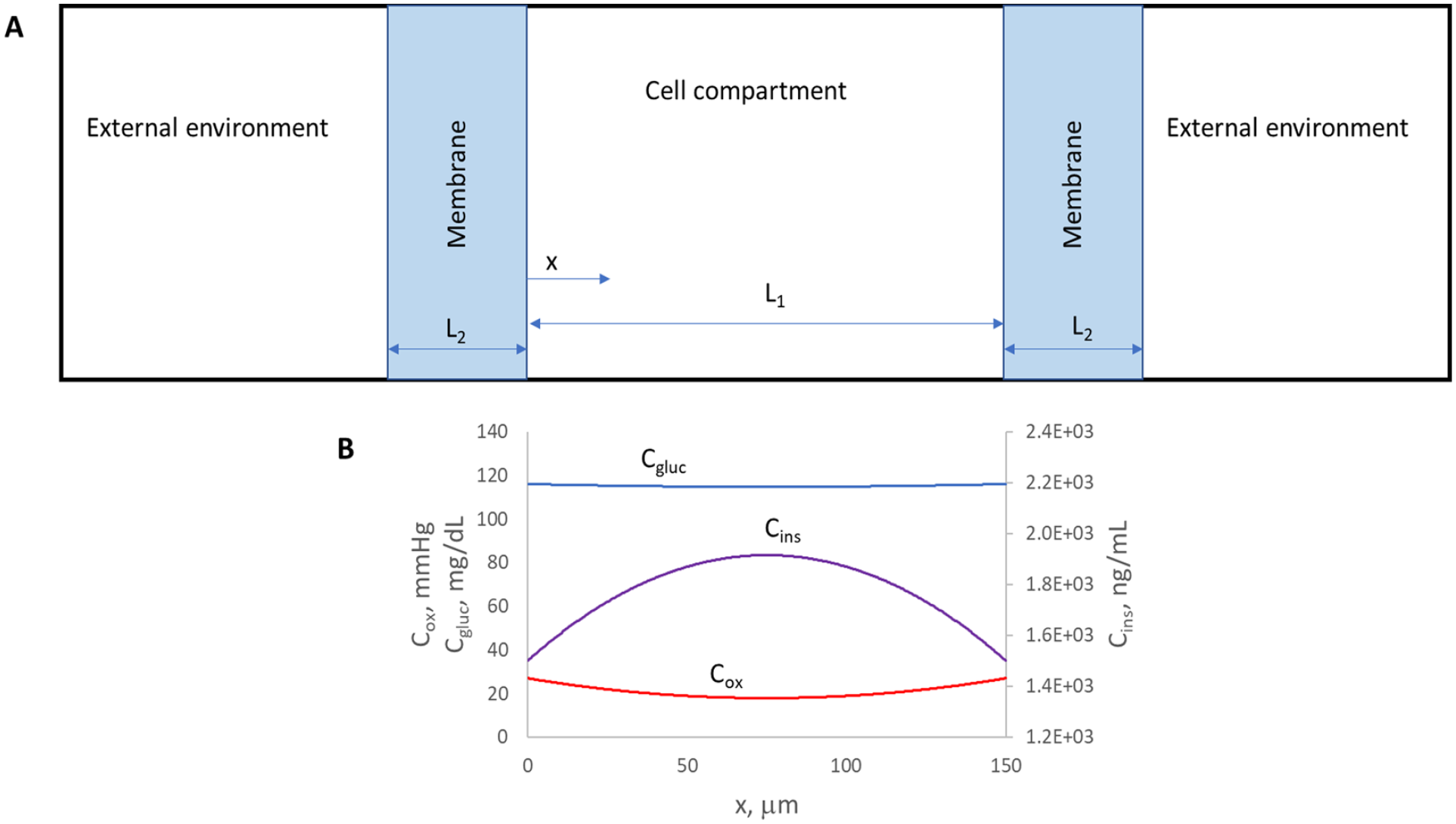
(A) Schematic of a planar islet encapsulation device containing a monolayer of islet equivalents (IEQs, cell compartment) between two immunoprotective membranes (membrane). For clarity, the device is not drawn to scale. (B) Concentration profiles of dissolved oxygen (Cox), glucose (Cgluc), and insulin (Cins) in the cell compartment of a planar construct containing a monolayer of IEQs between two immunoprotective membranes. The IEQ diameter, and therefore the thickness of the cell compartment L1, is 150 µm. This corresponds to 4.44 × 103 IEQs per cm2 of the planar device, or 2.96 × 105 IEQs/mL. The thickness of each membrane, L2, is 40 µm. The concentrations in the external environment are as follows: oxygen 60 mmHg, glucose 120 mg/dl and insulin 0 ng/ml. In the model, the oxygen and glucose consumption rates are assumed to follow Monod expressions and the insulin secretion rate the expression of Pillarella and Zydney1,3. The model parameters used were based on experimental measurements, literature values and engineering correlations. From the concentration profile, the average insulin concentration in the device is calculated to be 1.78 × 103 ng/ml.
The islets released from a ruptured device would be subjected to the immune system of the host. Some of the islets might exit the body via bleeding or first aid to the wound. The now non-immunoprotected islets remaining within the host are also unlikely to cause hypoglycemia. The islets that are alive and functional, before their rejection, would continue to secrete insulin in a glucose-responsive manner. As the islets undergo rejection, they would release intracellular insulin; however, this would be a gradual process extending over days or weeks; by then the victim would be in a medical setting where blood glucose levels would be closely monitored. Furthermore, glucagon released from alpha cells destroyed concomitantly with beta cells might partially counteract the effects of insulin and further reduce the likelihood of hypoglycemia.
The worst-case scenario is that a major injury
However, this worst-case scenario is highly unlikely as the accelerations required to cause extensive cell lysis are extreme and much higher than those observed during a lethal sudden impact such as in a car accident. Studies with fibroblasts revealed that acceleration alone does not damage the cells even when it is of a magnitude of 1,000 g 5 . Studies with brain tissue have established an acceleration greater than 150 g as the common criterion for injury 5 and the National Highway Traffic Safety Administration (NHTSA) standard for a sudden impact acceleration on a human that would cause severe injury or death is 75 g for a “50th percentile male” (https://hypertextbook.com/facts/2004/YuriyRafailov.shtml). Although cavitation bubbles can damage cells, such bubbles are unlikely to occur at the injury site; even if they do, the number of permeabilized islets would be small as cavitation events are stochastic and localized 5 . With regard to the anatomic placement of the device, if the device is placed subcutaneously in an arm or leg that is severely injured, first aid maneuvers to save the patient’s life, such as a tourniquet applied to reduce bleeding, might attenuate systemic release of insulin. If the implant is placed subcutaneously in the abdominal area 2 , it is unlikely that the patient would survive an injury that would cause extensive lysis of the islets.
Based on the above analysis, we conclude that the probability that device damage by sub-lethal injury causes a detrimental hypoglycemic event is quite low. In any event, wearing a medical bracelet with an appropriate warning would seem prudent. Obviously, a patient with an irreparably damaged transplant would require exogenous insulin therapy until a new device is implanted. Regardless of the location or extent of injury, all patients with islet transplants who sustain trauma should be carefully monitored for glucose levels as the responses of the transplanted cells to physiologic stresses are, as yet, difficult to predict.
Footnotes
Acknowledgements
The authors thank Evangelos Doukakis for providing the MATLAB code that was used for the modeling computations.
Author Contributions
Klearchos K. Papas (K.K.P.) and Athanassios Sambanis (A.S.) conceived the subject matter of the commentary and wrote the paper. A.S. generated the computational results. Charles W. Putnam (C.W.P.) and Robert C. Johnson (R.C.J.) made substantial conceptual contributions and edited the manuscript.
Availability of Data and Material
All data generated or analyzed during this study are included in the main text of the article.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Research Ethics and Patient Consent
There were no animals or human subjects involved in generating the material of this commentary.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K.K.P. and R.C.J. have a financial interest in Procyon Technologies LLC. A.S. is a paid consultant to Procyon Technologies LLC. C.W.P. has no financial interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge partial support by NIH/NIDDK grant # DP3DK106933 (K.K.P.), JDRF grant #2-SRA-2018-685-S-B (K.K.P.), and Procyon Technologies LLC.
