Abstract
Telocytes (TCs), a distinct type of interstitial (stromal) cells, have been discovered in many organs of human and mammal animals. TCs, which have unique morphological characteristics and abundant paracrine substance, construct a three-dimensional (3D) interstitial network within the stromal compartment by homocellular and heterocellular communications which are important for tissue homeostasis and normal development. Fibrosis-related diseases remain a common but challenging problem in the field of medicine with unclear pathogenesis and limited therapeutic options. Recently, increasing evidences suggest that where TCs are morphologically or numerically destructed, many diseases continuously develop, finally lead to irreversible interstitial fibrosis. It is not difficult to find that TCs are associated with chronic inflammation and fibrosis. This review mainly discusses relationship between TCs and the occurrence of fibrosis in various diseases. We analyzed in detail the potential roles and speculated mechanisms of TCs in onset and progression of systemic fibrosis diseases, as well as providing the most up-to-date research on the current therapeutic roles of TCs and involved related pathways. Only through continuous research and exploration in the future can we uncover its magic veil and provide strategies for treatment of fibrosis-related disease.
Introduction
Cellular Morphology
Popescu and colleges found a novel type of stromal cell called interstitial cajal-like cells (ICLC) by chance in 2005, then it was designated formally as telocytes (TCs) in 20101,2. TCs are cells containing telopodes (Tps), which is the most notable and significant traits that distinguishes TCs from other populations of interstitial cells2,3. Transmission electron microscopy (TEM) is used as the gold standard to observe typical ultrastructural characteristics of TCs4,5. TCs are ultrastructurally characterized by a small piriform, spindle or triangular cell body (containing a small amount of cytoplasm and nucleus) and extremely long and thin cellular prolongations with uneven caliber, named Tps (extending about 10–100 microns), which demonstrates a moniliform structure with thin segments (podomers) alternating with dilated regions (podoms) 3 . Mitochondria, endoplasmic reticulum, and caveolaec were observed within podoms, these are crucial for cellular metabolism and mediating the movement of TCs: calcium signaling, lipid homeostasis, mitochondrial dynamics, transport, and apoptotic signaling3,6. In the interstitial compartment, TCs are usually organized into a three-dimensional (3D) labyrinth-like network within interstitial compartment, which facilitates direct establishment of either homocellular or heterocellular contacts between Tps and other adjacent cells, such as fibroblasts, mast cell, stem cells (SCs), immunocytes, vascular endothelial cells, pericytes, muscle cells, and nerve fibers7–11 (Fig. 1). In addition, TCs can establish contacts with the connective extracellular matrix (ECM)12–14. With the aid of focused ion beam–scanning electron microscope (FIB-SEM) tomography, Tps present narrow and flattened (ribbon-like) structures and form 3D network by adherent homocellular junctions between TCs in heart and skin tissues; thus, strengthening the understanding of TCs’ spatial morphology5,15. All of these intercellular contacts were believed to be structural basis to directly regulate information communication and influence cellular behavior within interstitial compartment.

Schematic drawing depicting telocytes (TCs) interstitial system. Based on transmission electron microscope, TCs construct three-dimensional structure to integrate information by either direct homocellular/heterocellular contacts between telopodes (Tps) and adjacent cells, or indirect paracrine extracellular vesicles (EV) to influence adjacent cells, including fibroblasts (Fb), stem cells (SCs), immunocytes (IC), mast cells (MC), endothelial cells (EC), pericytes (PC), smooth muscle cells (SMC), and nerves (N). In addition, TCs can establish contacts with the connective extracellular matrix (ECM) which aims to regulate and control cellular connection and information communication. Solid and dotted arrows represent dilated podoms and thin podomers, respectively.
Under TEM, many different types of extracellular vesicles (EVs) were observed around Tps, such as exosomes, ectosomes, apoptotic bodies, and multivesicular cargos, such indirect paracrine activity were supposed to enhance functional regulation of adjacent cells16–18 (Fig. 1). In addition to special morphological features, TCs have distinct proteomic, gene profiles and miRNA imprints which are extremely different from those of fibroblasts, mesenchymal cells, or endothelial cells19–23. Given the current evidence, TCs is a unique and typical stromal cell population, with accumulation evidence and advancement on its morphology and function in the last decade.
TCs Heterogeneity: Distribution and Immunophenotype
At present, increasing evidence indicates that TCs are located in the stroma of several organs and tissues in mammal animals, such as the heart 24 , the scalp 25 , mammary gland 1 , male reproductive system26–30, female reproductive system31–33, urinary system 34 , the gastrointestinal tract 35 , spleen 36 , skin 37 , joints 12 , kidney 38 , liver and so on 39 (Table 1). Although TCs are lack of special immune markers, combined immunohistochemistry can be used to complement for the identification of TCs72,73. Basically, co-expression of CD34, vimentin, and PDGFRα is currently considered as the most noteworthy marker of TCs 7 . Multiple evidence showed that TCs express different immunophenotypes in different or even the same tissues or organs under the influence of the signaling received3,74 (Table 1). For instance, CD34, PDGFRα, vimentin, and c-kit are the most typical immune labels of cardiac TCs40,75,76. In female reproductive system, TCs are positive for CD34, c-kit, vimentin, PDGFRα, estrogen receptors (ER), progesterone receptors (PR), and T-type Ca2+ channels53,54,77,78. In terms of gastrointestinal tract, TCs are found to be double positive for CD34 and PDGFRα, and negative for c-kit35,79. Double-immunolabeling can act as a useful means to distinguish TCs from other mesenchymal cells, such as fibroblasts being vimentin-positive and CD34-negative, whereas TCs being double positive for CD34 and vimentin 76 . Giving different immune phenotypes, the researchers believe that there are different subpopulations of TCs even with the same ultrastructure and that such differences might be the basis of TCs region-specific heterogeneous functions61,80.
Immunophenotype of Telocytes in Various Organs.
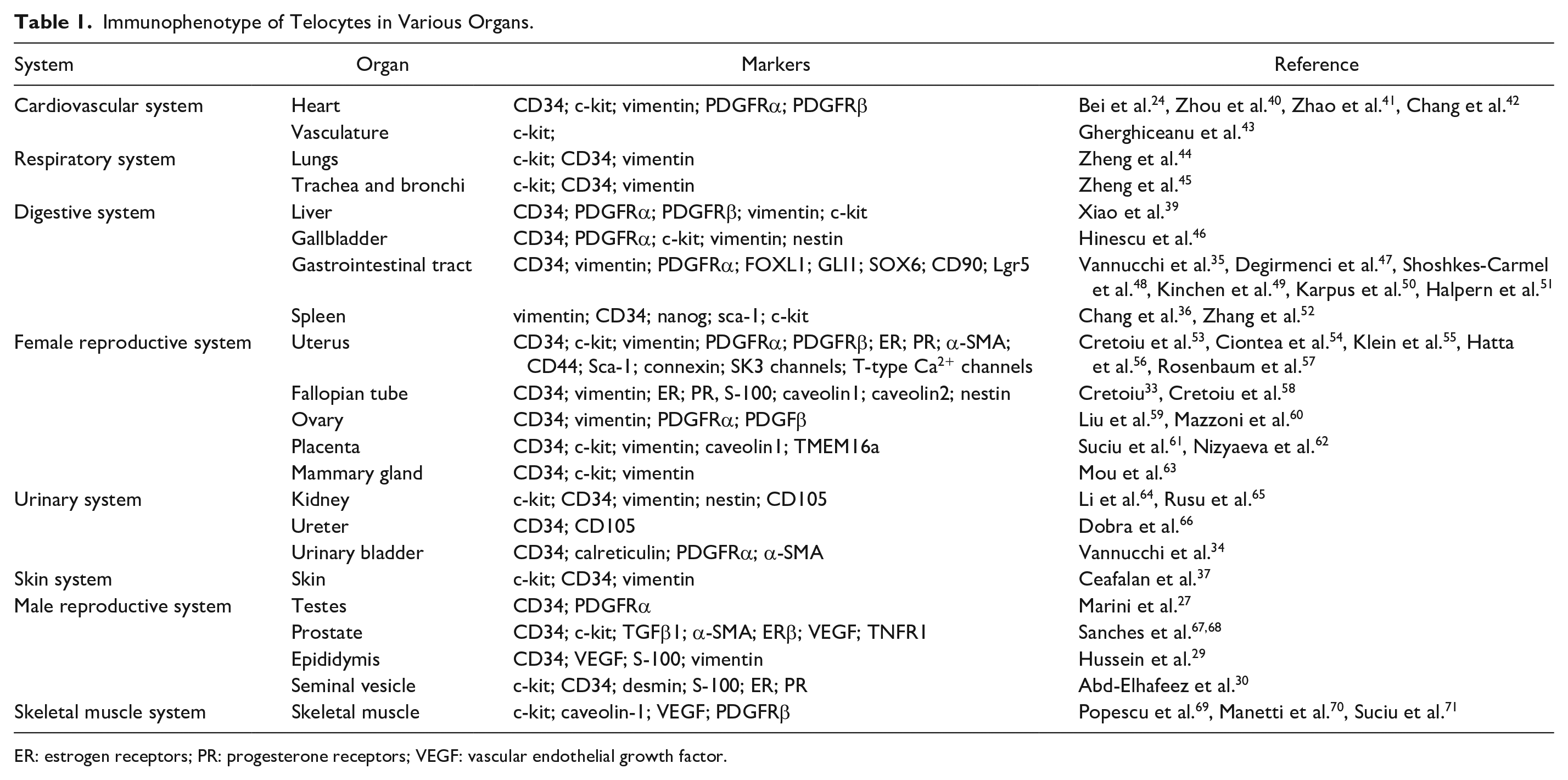
ER: estrogen receptors; PR: progesterone receptors; VEGF: vascular endothelial growth factor.
Structural and Paracrine-Based Functional Evidence
The role of TCs is still not fully understood, but many relevant functions have been proposed, with some of them strongly confirmed by experimental data, while others are highly structural related and thus somewhat speculative. TCs contribute to the maintenance of normal organ structure, tissue homeostasis, information center, and mechanical sensing, by forming complex 3D network within interstitial compartment through various types of homocellular and heterocellular junctions
81
. First, structural or mechanical support was evidenced typically in intestinal muscularis, TCs build complex network framework through connections with various structures: smooth muscle cells, interstitial cells of Cajal (ICC), nerve bundles, blood vessels, and SCs niches; function to maintain normal peristaltic movements; and prevent tissue deformation in the gastrointestinal tract
82
. Similarly, mechanical support from TCs’ network also was observed in interstitial space of the urinary bladder during stretching activities
34
. Second, TCs contribute to cell-to-cell communication and signaling. TCs act on neighboring cells (especially SCs niches) either by direct contact or paracrine activity. Several highly expressed substances identified in TC secretome profile were carried by EVs, including cytokines, growth factors, mRNAs, epigenetic regulators like miRNAs, and other non-coding RNAs, which were believed to be involved in intercellular exchange with adjacent cells
83
. Meanwhile, stemness properties of TCs were also evidenced by multiple
Protection Against Fibrosis
Fibrosis and regeneration are two opposite side of repair process after tissue injury or chronic inflammation. During fibrosis, via epithelial–mesenchymal transition (EMT), various cells including resident mesenchymal cells, or epithelial cells, are crucial sources of fibroblasts, followed with fibroblast transformation to myofibroblasts, which was the main component of ECM and a crucial biological mediator of fibrosis 92 . Uncontrolled fibrosis is characterized by hyperplasia of myofibroblasts and subsequent excessive deposition of ECM, tissue remodeling, and scar formation, finally leads to organ malfunction, increased cancer risk, and end-stage organ disease 93 .
Increasing evidence proved that TCs abnormalities (damage or loss) are closely related to many fibrosis-related diseases, such as systemic sclerosis, ulcerative colitis (UC), Crohn’s diseases (CD), heart failure, liver fibrosis, endometriosis (EMs), and acute salpingitis (AS)72,78,79,94–96. As TCs develop connecting or supporting structure within the whole interstitial compartment, fibrosis process will simultaneously spread to TCs. However, TC damage or loss might precede or be at the beginning of the onset of fibrosis, rather than being merely a consequence of the fibrotic process 95 . Nevertheless, reciprocal causation might exist between the development of fibrosis and TCs’ damage in fibrosis diseases.
However, accumulating evidence confirmed that, TCs transplantation contributed to reduction of ECM deposition, enhanced recovery of organ function in model of myocardial infarction (MI) and renal fibrosis97,98. Therefore, TCs provide a promising therapeutic opportunity to regeneration repair instead of tissue fibrosis, although the underlying mechanism(s) still need further investigation. The purpose of this review is to summarize current findings of TCs in various fibrotic diseases, with aim to probe future clinical perspectives.
Cardiovascular System
Cardiac TCs have been most extensively studied than any other tissues or organs. TCs preset in full layers of the heart wall, including epicardium, myocardium, endocardium, and cardiac valves, with the highest cell density in epicardium 99 . Longitudinal and cross 3D networks were formed through heterogeneous and homogeneous intercellular junctions between cardiac TCs and mast cell, fibroblasts, pericytes, and cardiac progenitor 100 . TCs also provide structure and functional support for SCs niches in epicardium 88 , with bidirectional posttranscriptional signaling exchanged between TCs and SCs through EVs 101 . Although in disease-affected cardiac tissues, TCs lose its essential roles in maintaining the integrity of structure and function. The number of TCs and Tps were decreased and even lost as a consequence of quantitative and qualitative changes in ECM composition, with negative correlation to the amount of mature fibrillar collagens and positive correlation to degraded collagens102,103. Even the aging human heart was featured with a gradual depletion of TCs 104 .
Multiple studies revealed therapeutic value of TCs. During acute experimental MI, TCs experienced significant loss in the first 1 or 2 days, while increased around the neovascularization border zone 30 days later, with direct physical contact or very narrow intercellular cleft (80–120 nm) to endothelial cells, and released vesicles containing various angiogenic micro-RNAs, therefore indicated that TCs were involved in neo-angiogenesis during late stage of MI 105 . Cardiac TCs transplantation in rat model of MI dramatically enhance the number of c-kit/CD34 double positive cells in the infarcted area, resulted in a substantial reduction in infarct size and collagen deposition, increased microvessel density (MVD) in infarcted and marginal zones, enhanced ventricular remodeling and post-infarcted cardiac function after 14 weeks41,97. Therapeutic value of TCs was also evidenced by its support on SCs niches, TCs abnormalities (damage or loss) will inevitably influence EVs secretion and impact local microenvironment, thus affecting the proliferation and differentiation of SCs 106 . Transplantation of human-induced pluripotent stem cell (iPSC) can protect cardiac function and alleviate ventricular remodeling of MI in mice, by reconstruction of the interstitial network of TCs and angiogenesis within the infarcted myocardium. In which, authors believed that TCs may play a unique role in contributing to the observed functional recovery 107 . More recently, cardiac TCs-EVs are capable of transferring macromolecular signals such as miRNA to adjacent cells, hence altering their transcriptional activity. Transplantation of miRNA-21-5P in rat MI model, the most abundant miRNA in cardiac TCs exosome, can facilitate angiogenesis, increase MVD in infarcted and border zones, reduce myocardial infarct size and fibrosis, and improve myocardial function via targeting and silencing Cdip1 gene to inhibit the apoptosis of microvascular endothelial cells 108 (Fig. 2). In calcific aortic valve disease (CAVD), TCs-EVs injection can reduce valve calcification and valve interstitial cells apoptosis by transferring miRNA-30b, and inhibiting wnt/β-catenin/Runx2 axis 110 (Fig. 2).

Schematic network that integrates the experimentally confirmed signaling pathways and functions of telocytes (TCs) involved in disease occurrence and treatment. ➀ In rat model of renal fibrosis, TCs transplantation can indirectly increase the expression of hepatocyte growth factor (HGF)
Giving these results, transplantation of TCs may ameliorate fibrosis of cardiac via following mechanisms: (
Skin System
Skin TCs preset mainly in dermal reticular layer, with a few in dermal papilla layer. In normal skin, FIB-SEM tomography demonstrates that, TCs develop spatial 3D networks, Tps construct heterocellular contacts to surrounding cells: mast cells, fibroblasts, adipocytes, blood vessels, nerves and adnexal structures of skin. TCs maintain normal tissue structure by controlling their activity10,15,37. TCs were found surrounding SCs niches in human normal skin and acting as nurse cells 94 . Similarly, EVs were observed along with Tps, indicated rich amount of information exchange and functional regulation to adjacent cells from skin TCs 15 .
In skin diseases, such as psoriasis and systemic sclerosis (SSc), which manifest as over-production of autoantibodies and progressive fibrosis, TCs experience ischemia-induced cell degeneration: severe ultrastructural damages (swollen mitochondria, cytoplasmic vacuolization, lipofuscin bodies) or almost completely disappeared in full layer of skin as disease progressive94,117. Authors speculated that, skin TCs might be more susceptible to chronic ischemic microenvironment of SSc 94 . In consequence, damage or loss of TCs may affect the 3D structure of skin ECM. First, in normal dermis, Tps were usually collagen-embedded or lining elastic fibers, whereas Tps were surrounded with large and abnormal aggregates of elastin and collagen fibers in SSc skin to limit their spreading into the interstitium, thus impact normal construction of 3D structure 94 . Next, damage or loss of TCs might contribute to abnormal activation of fibroblasts and mast cells in SSc skin, lead to excessive deposition of ECM and fibrosis10,37,94. Last but not least, damage or loss of TCs was accompanied by disappearance of vascular wall-resident SCs niches, this will inevitably impact SCs-mediated tissue regeneration and lead to fibrosis 94 . Interestingly, loss of TCs was found to accompany fibrosis of multiple visceral organs in SSc, such as gastric wall, myocardium and lung 103 .
Similar results were reported in bleomycin-induced mouse model of scleroderma, an early stage of SSc; obvious cellular degeneration in ECM of dermis, such as breaking and shorting of Tps, nuclear fragmentation, progressive reduction, and disappearance of TCs were observed with aggravation of skin fibrosis, and authors suggested that TCs injury occurred in the beginning of fibrosis rather than the result of fibrosis 118 . In addition, TCs were considered to be source of myofibroblasts during the formation of dermal fibrosis, although the progressive reduction of CD34 in TCs was not parallel to the increased α-SMA expression in myofibroblasts. This might be explained that, only part of TCs transition into myofibroblasts, with most of the rest experience degeneration and necrosis 118 . This opinion was consistent with others, in which human resident CD34+ stromal cells/TCs have progenitor capacity and are a source of α-SMA+ cells during tissue repair process119–121. In addition, giving the contact between TCs and macrophages in normal skin, and the fact that TCs enhanced classically activated macrophages (M1) differentiation 114 , authors proposed that TCs injury might lead to alternatively activated macrophages (M2), which was an important participator for promoting tissue fibrosis in many fibrotic diseases 118 .
So far, there is no report concerning the therapeutic application of TCs in skin diseases.
Liver
TCs mainly located in Disse space of liver, provide 3D structural support to hepatic stellate cells (HSCs), hepatocytes and SCs in interstitial compartment 39 . Hepatic fibrosis and liver cirrhosis are characterized by excessive activation of HSCs and deposition of ECM 122 . Close relationship between TCs and liver injury and fibrosis was documented. In human liver fibrosis, TCs, which were labeled with four different double immunofluorescence markers (CD34/PDGFRα or CD34/PDGFRβ or CD34/Vimentin or CD34/c-kit), all demonstrate severe reduction of TCs, with obvious collagen deposition, accumulation of inflammatory cells, and necrosis. However, researchers were unable to determine whether TCs damage was the cause or result of liver fibrosis 72 . Ultrastructural damage of TCs was also present in rat model of aflatoxin B1–induced liver injury 123 . Two potential mechanisms were proposed for TCs involvement in liver fibrosis. First, disruption of TCs based 3D interstitial structure will lose both paracrine loaded with exosomes and direct heterocellular contact on HSCs, subsequently contribute to abnormal activation of HSCs and fibrosis 101 , just like activation of fibroblast in skin tissue fibrosis 94 . Second, damaged TCs might lose their structural and functional support to hepatocytes and SCs regeneration, finally lead to liver fibrosis 87 .
Meanwhile, for treatment purpose, TCs showed its potential in tissue repair after liver injury. In murine model of partial hepatectomy, hepatic cell proliferation rate increased significantly at 48 and 72 hours, accompanied by a peak of TCs and hepatic SCs at 72 hours, indicate that TCs are participator closely related to hepatocytes and SCs regeneration 73 . What’s more, in Npc1 mutant mice, which manifested as enlarged spleen and altered metabolism of cholesterol and glycolipid, significantly increased splenic TCs were observed. Authors proposed that splenic TCs might act as a defender for enlarged spleen via recruiting hematopoietic SCs and macrophages to reduce progressive splenic damage and malfunction 52 . However, the involved mechanisms still need investigation.
Respiratory System
TCs were located in interstitial of lung or around terminal bronchioles, with 3D network connections between Tps and alveolar epithelial cells, nerves, blood vessels and SCs 89 . Lung TCs had specific gene and protein profiles which distinguished itself form other mesenchymal cells: SCs, fibroblasts, alveolar type II cells, airway basal cells and lymphocytes22,124–126. At gene level, Capn2, Fhl2 and Qsox1 were over-expressed in chromosome 1, which suggested TCs might be involved in regulating tissue homeostasis and maintaining structural integrity, anti-inflammation and alleviating fibrosis in lung diseases 127 . Among them, Capn2 plays a crucial role in morphogenesis and tissue homoeostasis. Fhl2 is associated with reversing inflammation and slowing fibrosis 128 . Qsox1 is involved in oxidative protein folding, cell cycle control and ECM remodeling. The most down-expression of Pde5 in chromosome 3 is associated with development of pulmonary fibrosis and interstitial lung disease 22 . Protein profiles of lung TCs showed, elevated superoxide dismutase and acid ceramidase could reduce oxidative stress and inhibit fibrosis during injury 23 .
Meanwhile, TCs show its therapeutic potential in repair of tissue damage result from lung inflammation, which was characterized with excessive ECM deposition and pulmonary interstitial fibrosis. Similar in other tissues, TCs form close relationship with SCs niches in lung, support and nursing through nanocontact or paracrine activity on SCs, initiate and promote SCs-based tissue repair during acute lung injury 89 . TCs can reduce oxidative stress and tissue damage via increased miRNA-146a-5p, then downregulate CREB1/DUOX2 pathway in mice model of inflamed lungs 116 (Fig. 2). In ventilation-induced mice lung injury, TCs transplantation can alleviate inflammation, promote angiogenesis via vascular endothelial growth factor (VEGF) and improve lung function 129 . TCs transplantation in mouse model of lipopolysaccharide (LPS)-induced acute respiratory distress syndrome (ARDS) can relieve acute lung injury, promote angiogenesis and tissue repair through miRNA-21a-3p-PI3K(p110α)/AKT/mTOR signal pathway 115 (Fig. 2). Co-transplantation of TCs and mesenchymal stem cells (MSC) significantly relieve alveolar inflammation and injury, mechanism including enhanced migration, proliferation of MSC and TCs’ nutritional support for MSC 130 . Intraperitoneal administration of TCs yield reduced pulmonary inflammation and edema, and facilitated proliferation and differentiation of airway epithelium by providing nutrients with TCs-derived mediators and exosomes 109 (Fig. 2).
Urinary System
In urinary system, TCs mainly distributed in renal cortex interstitium and upper lamina propria of the renal pelvis, ureter and urinary bladder 131 . In renal cortical interstitium, TCs appeared around blood vessels and renal tubules, accompanied by EVs release surrounding Tps 38 . Loss or damage of TCs is closely related to occurrence of ureteral wall fibrosis. In experimental obstructive hydronephrosis, TCs reduction and collagen deposition were observed in thickened ureteral wall 132 . In patients with ureteropelvic junction obstruction (UPJO), decrease of TCs was accompanied by increased ratio of collagen to muscle contents 133 . These experiences further confirmed TCs’ roles for maintaining normal ureter structure.
TCs also showed its potential in therapeutic purpose. Renal fibrosis is the end stage of various renal diseases, which eventually leads to the destruction of renal parenchyma and renal failure. In a renal ischemia–reperfusion injury (IRI) model,
Gastrointestinal Tract
TCs were widely distributed in full layer of gastrointestinal tract 35 , especially constituted the intestinal SCs niche within mucosal layer47,48. TCs support entire epithelium by forming a subepithelial plexus extending from stomach to colon tissues47,48. Due to multiple subtypes of TCs (expressing different immunophenotypes), there was no unified definition of such mesenchymal cells. They were generally identified or named by specific morphological characteristics.
Inflammatory bowel disease (IBD), including CD and UC, are chronic recurrent diseases and common cause of extensive intestinal wall fibrosis134,135. TC showed normal morphology and distribution likely maintaining their supposed roles in unaffected ileal segments 79 . While in IBD cases, architectural disorder and fibrosis of intestinal wall was accompanied by diseases severity related loss of TCs79,95. Loss of TCs was paralleled by decrease of c-kit positive ICC at myenteric plexus, these might explain dysmotility of gastrointestinal tract in IBD 136 . In addition, damaged TCs were embedded in ECM and lead to destruction of 3D structure due to hypoxia, further cause deformation of the gastrointestinal structure and impaired contacts with around cells, including immunocytes, fibroblasts, smooth muscle cells, ICC, and so on. Moreover, when studying human adipose tissue and intestinal wall affected by inflammation and repair (appendicitis, diverticulitis of large bowel and Crohn’s disease of the terminal ileum), the observed CD34+ TCs undergone a series of activation, proliferation and differentiation to α-SMA+ stromal cells (myofibroblasts), indicating that CD34+ stromal cells/TCs have progenitor capacity and are source of α-SMA+ stromal cells during tissue repair120,137.
Although there is no current application of cell transplantation for treatment of gastrointestinal diseases, TCs demonstrate its promising therapeutic value. TCs provide structural and functional support for SCs niches within gastrointestinal epithelium by providing non-epithelial sources of Wnts ligands and R-Spondins 3 (RSPO3), both were important regulator for WNT signaling pathway47,48,111. As we know, Wnt/β-catenin pathway is pivotal and indispensable for sustaining the self-renewal and proliferation of intestinal SCs 138 . Knockout of Porcn or Wntless gene required for Wnts secretion and RSPO3 in Foxl1- and Gli1-expressing intestinal stromal cells, resulted in reduction of SCs population, defect epithelial proliferation and crypt collapse47,48,111 (Fig. 2). These indicates that TCs act as the central coordinator of intestinal renewal, responsible for SCs mediated tissue repair and maintaining stability of local intestinal environment. Interestingly, single-cell RNA sequencing revealed a mesenchymal cell population, expressing SOX6, F3, CD142 and Wnt genes, was identified near the colonic crypt niche. Breakdown of intestinal epithelial structure in UC was accompanied by considerable reduction of these cells. Abundant Wnt signal for SCs proliferation further proves the classification of observed interstitial cells as almost similar to TCs under studying 49 .
Gallbladder TCs is a new player in gallstone disease. Current studies mainly focus on decreased density of TCs in patients with cholelithiasis, indicated close regulation of gallbladder and extrahepatic bile duct movement, bile component alterations, and chronic inflammatory process91,139,140. However, there is no solid evidence reporting the involvement of TCs in ECM remodeling or fibrosis in gallbladder tissues.
Female Reproduction System
TCs existed in various parts of female reproductive system, including vagina, cervix, uterus, uterine tubes, and ovary. Site-specific TCs subpopulations was observed with different immunophenotypes and may be related to specific functions, such as immunomodulation and immunosurveillance, muscular layer contractility, pregnancy maintenance, and tissue regeneration32,55,141. In female reproductive system, TCs specifically express ER and PR, suggested that its functions and activities were periodically controlled by hormones58,142. In normal oviduct, TCs distributed in lamina propria and muscular layer, involved in maintenance of normal structure and function, by creating 3D network with smooth muscle, blood vessels, nerve fibers, and so on 143 . TCs and Tps wrap around SCs to form SCs niches 96 .
Gynecological conditions are often accompanied by tissue fibrosis, such as premature ovarian failure (POF), EMs, intrauterine adhesions, AS, uterine leiomyoma, and ectopic pregnancy59,144–147. In mouse model of cyclophosphamide-induced POF, ovarian parenchymal cell injury is coupled with TCs reduction in fibrotic stroma which may be linked to reduced estrogen levels, resulting in damage of ovarian microenvironment and function 59 . In EMs- or AS-affected rat oviduct, decrease or loss of TCs was accompanied by extensive ultrastructural damage, including collapse of interstitial 3D network and disruption of TC-SCs niches, finally lead to oviduct fibrosis and tubal factor infertility78,96. Similar results were reported in clinical specimen from EMs and tubal ectopic pregnancy, in which, damage and loss of oviduct TCs was observed accompanied with fibrosis and reduced tubal motility 148 . On the contrary, in patients with uterine myoma 147 and ectopic pregnancy 146 , TCs appear compensatory increase in fallopian tube, then will decrease the cilia and muscular movement of oviduct, then lead to female fertility disorders146,147. In obstetric conditions, with the aid of TEM and immunophenotype studying, loss and impairment of TCs in human preeclampsia placenta (under the influence of hypoxia and malnutrition) was observed and differentiated into fibrocytes in fibrotic villi stroma, indicated that TCs potentially have functions related to immunomodulation, angiogenesis, and fibrosis 62 .
Uterine leiomyoma is characterized by ECM overproduction in myometrium, TC was found disappeared in leiomyoma and lose its control on tissue homeostasis, with excessive ECM deposition
149
. TCs are crucial components in pathogenesis of leiomyoma formation, supposed mechanisms including (
TCs show emerging therapeutic role in gynecologic conditions, as evidenced by its proactive impact on peritoneal macrophage and endometrial stromal cells (ESCs) in our group.
Mammary Gland
In human mammary gland, TCs mainly distributed around capillaries and breast ducts, forming spatial 3D network structure with immunocytes, capillaries, lymphocytes, macrophages, and mast cells 1 . Rat mammary TCs was crucial for stromal structure and breast development, and TCs in different physiological stages demonstrate different immunohistochemical and ultrastructural characteristics 152 . Mammary resident CD34+ Stromal Cells/TCs was proposed as the origins for cancer-associated fibroblasts (CAFs) in invasive lobular carcinoma of breast, which may act as a guide for neoplastic cells, assist in ECM deposition and remodeling, tumor growth, invasion, metastasis, and angiogenesis, as well as modulating tumor immunity121,153.
The involution of post-lactational mammary gland is featured as alveoli collapse and ECM remodeling. TCs involved actively in such involution process in Mongolian gerbil, by surrounding collapsed alveoli and breast ducts with synthesis of Matrix Metallopeptidase 9 (MMP9) and VEGF, which was essential for ECM digestion and remodeling, and angiogenesis respectively 13 . Therefore, TCs have therapeutic potential in maintaining the dynamic balance of CAFs or ECM either in carcinoma of breast or fibrotic diseases.
Male Reproduction System
In male reproduction system, TCs distributed in inner genitalia, including prostate, testes, epididymis, seminal vesicle26–30. Prostatic TCs existed in interacinar region around the acinar smooth muscle, where they created Tps network around acini to support alveoli and smooth muscle differentiation, as well as the maintenance of interstitial compartment homeostasis at different phases 67 . The prostatic TCs undergone dynamic alterations under castration, such as phenotypic transitions, Tps loss, or folding, which can be reversed by testosterone, indicated androgen-dependent TCs play a key role in prostate tissue organization and reversing prostate involution by connecting with alveoli and smooth muscle 154 . Furthermore, TCs contributed to age-related structural alterations in the prostate by synthesizing VEGF and expressing TNFR1, which promoted angiogenesis and established a pro-inflammatory microenvironment respectively 14 .
Testicular TCs distributed and formed complex reticular structures around blood vessels and seminiferous tubules, with close contact between its long Tps and adjacent cells, such as peritubular myoid cells, mononuclear cells, intertubular steroidogenic Leydig cells 27 . TCs also existed in testes of different species60,155,156, with their unique structural distribution and potential roles including spermatogenesis control, structure maintenance, lipid metabolism, and cell signal transmission. In male fish, TCs produce MMP-2 and MMP-9, indicated TCs can undergo de-differentiation and contribute to ECM reorganization and tissue remodeling 60 . TCs in seminoma almost totally disappeared, accompanied by severe degeneration of testicular architecture and interstitial fibrosis. TCs damage/loss activate α-SMA+ myoid cells which contribute to tumor invasion and metastasis 157 . This is similar in breast cancer, TCs were considered the source of CAFs and facilitate tumor invasion and metastasis 153 . Therefore, modulation of TCs activity might provide a therapeutic approach in cancer biology. However, the relationship between the loss of TCs and disease development was still controversial. Whether abnormal seminoma microenvironment leads to damage or loss of TCs, or disruption of TCs-based signal network further trigger pathological changes of testicular tissue, will be an interesting topic and worth of in-depth investigation.
Skeletal Muscle
TCs located in endomysium and perimysium of skeleton muscle, form special 3D interstitial network with all type of cells within muscular tissue, support paracrine signaling trophic substance (such as VEGF) to both satellite and non-satellite, suggesting a key role in integrating signals for skeletal muscle fibers, regeneration and repair after trauma 69 . Moreover, by cell culture obtained from explant, TCs support and establish heterogeneous junctions with muscle SCs niche and exhibited specific capacities, including high proliferation capacity (CD105+, Ki67+), pluripotent capacity (Oct4+), and angiogenesis (VEGF+) 158 . TCs’ nursing role in neighboring satellite cell (SC)-mediated skeletal muscle regeneration was also evidenced by TC-SC morpho-functional interaction following damaged skeletal muscle condition 70 . In human fetal skeletal muscle, series changes of CD34+ TCs was observed during early stages of myogenesis, with peak number, immunopositivity and richest reticular network with blood vessels and myotubes at 10 to 11.5 weeks, suggesting their potential involvement in the early steps of myogenesis 159 .
Although lack of reports on TCs in fibrosis treatment, current studies lay the ground and provide new attractive target in the field of regenerative medicine in skeleton muscles repair.
Conclusion and Future Perspectives
In conclusion, this review summarized current findings that TCs, as a novel stromal cell population, are involved in fibrosis diseases in several organs or tissues, with confirmed therapeutic evidence in heart, liver, gastrointestinal tract, lung, kidney, female reproduction system. Due to intricate junctions and specific paracrine traits, TCs have been a spotlight to in-depth understanding its underlying molecular mechanisms. Giving that TCs are responsible for the process of fibrotic conditions, a pressing need for future research works to better discover and expand its relevant prophylactic or therapeutic roles in fibrosis-related diseases.
Footnotes
Acknowledgements
The authors thank the reviewers for their insightful comments.
Author Contributions
XJW took part in the conception of this review, drafted the manuscript, and prepared the figures and table. TQC and XJY revised and gave the final approval of submission.
Ethical Approval
This study was approved by authors’ institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant numbers 81971335 and 81571415).
