Abstract
In normal physiological conditions, restoration of a functional epidermal barrier is highly efficient; nevertheless, when it fails, one of the main consequences is a chronic ulcerative skin defect, one of the most frequently recognized complications of diabetes. Most of these chronic venous ulcers do not heal with conventional treatment, leading to the appearance of infections and complications in the patient. Treatments based on the use of autologous mesenchymal stem cells (MSC) have been successful; however, its implementation entails complications. The umbilical cord offers an unlimited source of adult MSC (ucMSC) from the Wharton’s jelly tissue with the same relevant features for clinical applicability and avoiding difficulties. It has recently been characterized by one specific subpopulation derived from ucMSC, the differentiated mesenchymal cells (DMCs). This subpopulation expresses the human leukocyte antigen-G (HLA-G) molecule, a strong immunosuppressive checkpoint, and vascular endothelial growth factor (VEGF), the most potent angiogenic factor. Considering the importance of developing a more effective therapy for wound treatment, especially ulcerative skin lesions, we analyzed DMC safety, efficacy, and therapeutic potential. By immunohistochemistry, umbilical cords HLA-G and VEGF positive were selected. Flow cytometry revealed that 90% of the DMC subpopulation are HLA-G+, CD44+, CD73+, CD29+, CD105+, CD90+, and HLA-DR−. Reverse transcription-polymerase chain reaction revealed the expression of HLA-G in all of DMC subpopulations. Upon co-culture with the DMC, peripheral blood mononuclear cell proliferation was inhibited by 50%. In a xenograft transplantation assay, DMC improved wound healing with no signs of rejection of the transplanted cells in immunocompetent mice. This study confirms that HLA-G allows allogeneic cell transplantation, and VEGF is fundamental for the restoration of the failure in blood supply. DMC population has positive effects on wound healing by promoting local angiogenesis in skin lesions. DMC could play a very important role in regenerative medicine and could be a novel allogeneic cell-therapeutic tool for wound healing.
Introduction
Wound healing after skin injury involves a complex process that affects different cellular constituents. In normal physiological conditions, restoration of a functional epidermal barrier is highly efficient; nevertheless, when it fails, one of the main consequences is an ulcerative skin defect 1 . Notably, foot ulceration is the most frequently recognized complications of diabetes 2 . Most of these chronic venous ulcers (CVUs) do not heal with conventional treatment, leading to the appearance of infections and complications in the patient 2,3 . A lot of treatments have been developed, such as elastic bandages, Unna boot, skin grafts, hydrocolloid dressings, and varicose vein surgery 4 –8 . So far none of these have been highly effective.
New treatments should therefore be developed to improve wound healing for a great number of patients. In this context, several scientific papers proposed the use of the mesenchymal stem cells (MSCs), including bone marrow and adipose tissue-derived MSC and even fibroblast lineages 9 –13 . All of these treatments with autologous MSC have been successful in wound healing; however, its implementation entails some complications such as invasive procedures that involve bone marrow puncture or liposuction surgery. Nevertheless, the umbilical cord offers an unlimited source of adult MSC from the Wharton’s jelly tissue 14 and it is discarded at delivery, avoiding any practice that causes pain or some traumatic consequences 15 . The umbilical cord MSCs (ucMSC) offer the same relevant features for clinical applicability, including are not tumorigenic at lower passages 16 and have no ethical objections.
A high proportion of ucMSC express the human leukocyte antigen-G (HLA-G) molecule 17,18 . The HLA-G is a nonclassical class I HLA molecule, which plays an essential role in fetomaternal tolerance and allogeneic tissue graft tolerance 19 –21 . HLA-G has a strong immunosuppressive activity able to inhibit the local immune response at different levels 22 –24 . This feature makes the ucMSC excellent mediators of immunosuppression, also it has the ability to induce T regulatory cells, both in vitro and in vivo 25 .
The main mechanisms involved in stem cell therapy include the provision of functional parenchymal cells, the promotion of angiogenesis, and the paracrine local response. Thus, angiogenesis plays a key role in wound healing and tissue regeneration, restoring blood perfusion, and the supply of oxygen and nutrients to injured sites 26,27 . The vascular endothelial growth factor (VEGF), the principal angiogenic factor, is the most potent mitogen on endothelial cells of arterial, venous, and lymphatic vessels. So, local VEGF expression is fundamental since the main problem for wound healing in ulcerative skin defects is the failure in blood supply.
Considering all of the above, it has recently been characterized a specific subpopulation derived from ucMSC, called differentiated mesenchymal cells (DMCs). This specific subpopulation has been characterized and always shows HLA-G and VEGF expression, allowing them to be used via allogeneic transplantation for chronic and resistant dermal wound treatments, especially CVU.
Considering the importance of developing a new therapy for wound treatment in chronic ulcerative skin defects, we analyzed DMC safety, efficacy, and therapeutic potential. We explored the specific characteristic of this DMC population in several in vitro and in vivo experiments.
Materials and Methods
This study was approved by the Comité de Bioética y Ética de la Investigación de la Facultad de Ciencias Médicas de la Universidad Nacional de La Plata (COBIMED), and the Institutional Committee for the Care and Use of Laboratory Animals (CICUAL), Facultad de Ciencias Médicas, Universidad Nacional de La Plata, Argentina.
Human Umbilical Cord Obtainment
Written informed consent was obtained from each mother before normal cesarean birth. The human umbilical cords were obtained from discarded placentas. Two samples of each umbilical cord were obtained, one for immunohistochemistry analysis and one for isolation and expansion in cell culture.
Immunohistochemistry
For immunohistochemistry (IHC), umbilical cord samples were transported in Formalin 10%, and the HLA-G and VEGF stain was done. Briefly, samples were fixed in 10% buffered formalin for 24 h and embedded in paraffin. Sections (5 µm) were placed on silanized slides, dried overnight, deparaffinized in xylene, rehydrated through graded alcohols, and washed in Tris-buffered saline (TBS) at pH 7.4. Endogenous peroxidase was blocked with 3% hydrogen peroxide (H2O2) for 10 min. Antigen retrieval was achieved by washing the slides in TBS and irradiating them in citrate buffer, pH 6.0 at 750 W for 2 cycles of 5 min in a microwave oven. After microwaving, the slides were washed in TBS and incubated with primary antibodies: 4H84, an IgG1 recognizing alpha1 domain common to all HLA-G isoforms (Santa Cruz Biotech., Santa Cruz, CA, USA) and VEGF-A, clone C-1 (Santa Cruz Biotech, CA, USA), both for 1 h at room temperature. Envision (DAKO Corporation, Hamburg, Germany) was used as a detection system with 3′3-diaminobenzidine (DAB, Sigma-Aldrich, St. Louis, MO, USA) as the chromogen. The sections were lightly counterstained with Mayer’s hematoxylin. Positive and negative controls were done to ensure appropriate results for each procedure. Only the MSCs from umbilical cords HLA-G and VEGF positives by IHC were selected to follow in culture, and others were discarded.
Isolation and Expansion of ucMSC and DMC
For cell isolation, umbilical cords were transported in Dulbecco’s Modified Eagle Medium (DMEM, cat. n° 52100, Gibco, NY, USA). Each cord was chopped in 5 mm length, a sagittal cut was performed to expose Wharton’s jelly and, umbilical blood vessels were carefully removed with clamps. Fragments were washed 2 or 3 times with Dulbecco’s phosphate-buffered saline (DPBS; cat. n° P3813, Sigma-Aldrich, San Louis, USA) to remove the remaining blood. Then, the face with the exposed jelly was placed against the bottom of the culture plate and minimum essential medium (α-MEM, cat. n° A1049001, Gibco, USA), and 10% of platelet lysate was added 28 . Plates were incubated at 37 ºC in a humidity atmosphere and 5% of carbon dioxide. Cell culture medium was changed every 2 to 3 days. ucMSC expansion was observed by 10 to 14 days after explantation, and those cells were amplified until passage 2/3.
Flow Cytometry Analysis
The specific surface DMC molecules were characterized by flow cytometry analysis. Cells were dissociated by incubation with trypsin and were conjugated with the followed specific cell surface markers: CD44+, CD73+, CD29+, CD105+, CD90+, HLA-G+, and HLA-DR− were used as marker (CD44-APC: Molecular probes cat. n° A14749, Eugene, OR, USA; CD73-PE: cat. n° 55027, BD Biosciences Franklin Lakes, NJ, USA; CD29-PE: cat. n° 561795, BD Biosciences Franklin Lakes, USA; CD105-PerCP-Cy 5.5: cat. n°560819, BD Biosciences Franklin Lakes, USA; CD90-FITC: cat. n° 555595, BD Biosciences Franklin Lakes, USA; HLA-G-FITC: cat. n° MA1-19591, Invitrogen CA, USA; HLA-DR-FITC: cat. n° 555560, BD Biosciences Franklin Lakes, USA). The incubation was carried out in PBS + 0.5% bovine serum albumin for 30 min at room temperature, with the primary antibodies (dilution 1:50). Flow cytometry analysis was performed in a BD Accuri cytometer. Data were analyzed with FlowJo software.
RNA Extraction, cDNA Synthesis, and RT-PCR Analysis
To confirm the HLA-G presence in DMC subpopulation, reverse transcription-polymerase chain reaction (RT-PCR) using specific primers to detect all the known isoforms were done, published previously 29 . RNA extraction was performed with TRIzol Reagent (cat. n° 15596026, Invitrogen, USA). The RNA purity and concentration were assessed using a Nanodrop spectrophotometer. For cDNA synthesis, 500 to 1000 ng of the total RNA was retro-transcribed with MMLV reverse transcriptase (cat. n° M170A, Promega, Madison, WI, USA), according to the manufacturer’s instructions. PCR was performed using Easy Taq DNA Polymerase (cat. n° AP111, Transgen Biotech, Beijing, China). Oligonucleotides used were: 257 F: 5´-GGAAGAGGAGACACGGAACA-3´ and 526 R: 5´-CCTTTGTTCAGCCACATTGG-3´.
DMC Immunomodulation Capacity Test
DMC immunomodulation activity was demonstrated by inhibition of ex vivo lymphocyte proliferation. Peripheral blood mononuclear cells (PBMCs) were freshly isolated from buffy coat leukocyte concentrates obtained from anonymous healthy human donors (they provided informed consent). Peripheral blood was diluted 1:2 with DPBS. PBMC isolation was carried on by density gradient centrifugation using Ficoll-Paque PLUS medium (cat. n° 17-1440-03, Fischer Scientific, Roskilde, Denmark). Then, PBMC were stained with CellTrace CFSE Cell Proliferation Kit (cat. n° C34554, Invitrogen, USA), a vital dye that loses fluorescence each cellular division. After this, PBMC were incubated with 5 μg/ml concanavalin-A and 300 IU/ml interleukin-2 for its activation and then co-cultured for 4 days with DMC using Roswell Park Memorial Institute 1640 medium (cat. n° 61870036, Gibco, NY, USA) + 10% fetal bovine serum. After the incubation, the lymphocytes were analyzed by flow cytometry, and the cycles were counted.
Animals
Immunocompetent adult mice, C3H/S strain, inbred in the Biotherium of the Histology Chair of the Faculty of Medical Sciences, were used. The mice were placed in individual boxes, illuminated for 12 h and alternating with 12 h of darkness (Circadian Rhythms Room) with forced ventilation and a temperature of 22 ± 2 ºC, receiving water and food “ad libitum”.
Murine ucMSC obtaining and culture
Umbilical cord from advanced pregnant murine females was extracted, to use as controls of the DMC xenogeneic application in mice experiments. The small size, friability, and transparency of the murine umbilical cords made it difficult to manipulate, making it impossible to accomplish the same task on human cords (remove the blood vessels and position the cord sections on the plates). For this reason, it was performed a longitudinal cut in the obtained umbilical cord pieces and put these into the plates with DMEM medium + 10% FBS. After 7 to 10 days, the cells were observed around the cord fragments.
Mouse skin wounds model and treatment
To evaluate the safety and efficiency of the DMC therapy in wound healing, a modified mouse model with the dermal lesion was used 30 . The surgical procedures were performed under anesthesia. Skin wounds were executed with a disposable dermatology biopsy punch. Incisions were made through the epidermis, dermis, and subcutaneous tissue, leaving the deep fascia intact. Eighteen animals were allocated to 3 experimental groups (n = 6 in each group): group i (control) with skin lesion and without therapy, group ii with skin lesion and treatment with allogeneic transplantation of murine ucMSC (dose of 250,000 murine ucMSCs/0.1 ml PBS), and group iii with skin lesion and treatment with xenogeneic transplantation of human DMC (dose of 250,000 DMCs/0.1 ml PBS). It is necessary to take into account that mice are species characterized by rapid healing of skin wounds; therefore, we need to perform a second injury over the first one, with a week of each other, so that the wound by the second intention, which has slower healing, allows us to observe differences in wound healing process between the 3 groups (Fig. 4A). Groups ii and iii were injected with the cells (murine ucMSC or human DMC, respectively) in the subcutaneous cellular tissue at the edges of the lesion, immediately after the second injury was performed.
Wound sample evolution
Daily observation of the wound was performed by 2 independent observers. The wound healing was photographed for their subsequent analysis. For histopathological evaluation of wound evolution, samples with 1.5 to 2 mm uninjured skin margins were excised at 5 post-injury days from the third experimental group. The specimens were fixed and embedded in paraffin to obtain sections. Every section was cut and stained by routine histology.
For histological observation, the sections were stained with hematoxylin and eosin (H&E). Then, the Gomori trichrome staining was used to determine the degree of connective tissue regeneration, especially collagen fibers.
For IHC analysis, sections were rehydrated, blocked, and incubated with the primary antibodies against VEGF-A (Santa Cruz Biotech).
Statistical Analysis
Statistical analysis was performed using SPSS (IBM, Armonk, NY, USA). Statistical significance was calculated using t-tests and analysis of variance for multiple comparisons. The significance was set at P < 0.05. All values were expressed as the mean and standard deviation (SD).
Results
HLA-G and VEGF Expression in Human Umbilical Cords
Examined by IHC, only ucMSC from VEGF and HLA-G positive cords were cultured to obtain the DMC subpopulation.
DMC Characterization
By flow cytometry analysis, we checked the presence of the specific surface molecules to identify the DMC subpopulation. The cells were analyzed in passages 2 and 3 and were positive for CD44, CD73, CD29, CD105, CD90, and HLA-G and negative for HLA-DR (Fig. 1). This analysis is important since it shows that its undifferentiated state of the cells remains once isolated from the umbilical cord microenvironment.
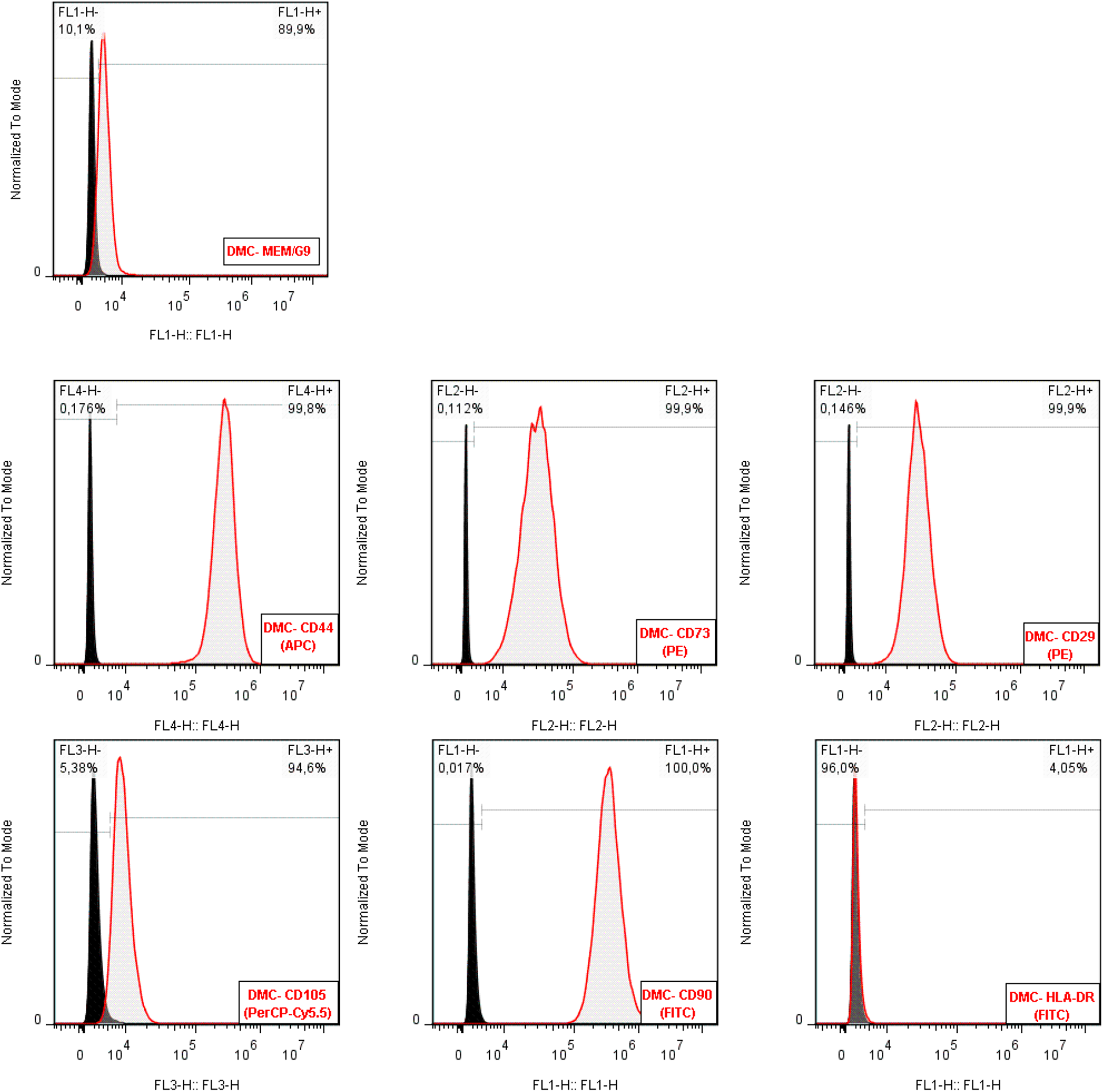
Characterization of differentiated mesenchymal cells (DMC). Flow cytometry analysis of representative DMC was performed. DMCs were negative for human leukocyte antigen (HLA)-DR and positive for CD44, CD73, CD90, CD29, CD105, and HLA-G.
Expression of HLA-G Molecule in DMC
The HLA-G expression in the DMC subpopulation was confirmed by the RT-PCR technique. HLA-G has 7 protein isoforms by alternative splicing, with isoforms 1 to 4 to be membrane-bound and isoforms 5 to 7 to be soluble/secreted. By RT-PCR analysis, all studied DMC lines expressed some HLA-G isoform since the used primers (257 Fw/ 526 Rv) can detect all the known isoforms (Fig. 2).
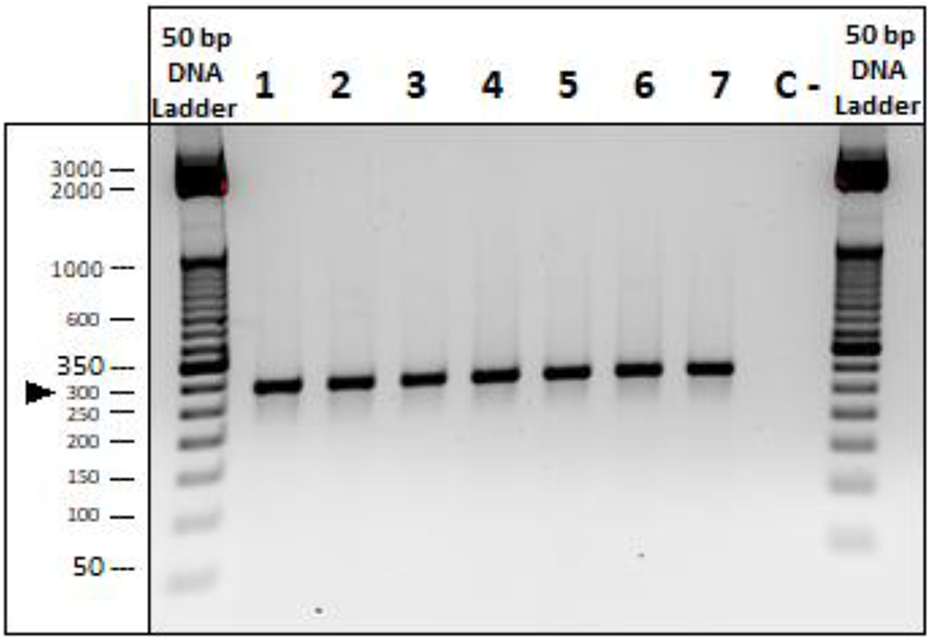
HLA-G was highly expressed on the DMC. Quantitative polymerase chain reaction revealed a steady expression of HLA-G mRNA in 7 DMC subpopulations using the primers 257 Fw/ 526 Rv, (290 bp) to detect all the known HLA-G isoforms. DMC, differentiated mesenchymal cell; HLA, human leukocyte antigen.
Inhibitory Effect of DMC on PBMC Proliferation
DMC inhibited lymphocyte proliferation when it was co-cultured with activated lymphocytes, confirming their immunosuppressive effect. In Fig. 3, we can observe that the mean proliferation percentage of responder PBMCs was significantly lower than without adding the DMCs. When the activated PBMCs were cultured alone, the number of cell divisions reached 6, whereas when the PBMCs were exposed to DMC, no only the number of cell divisions diminished by 50%, also the percentage of non-dividing PBMC increases from 27 to 58 %
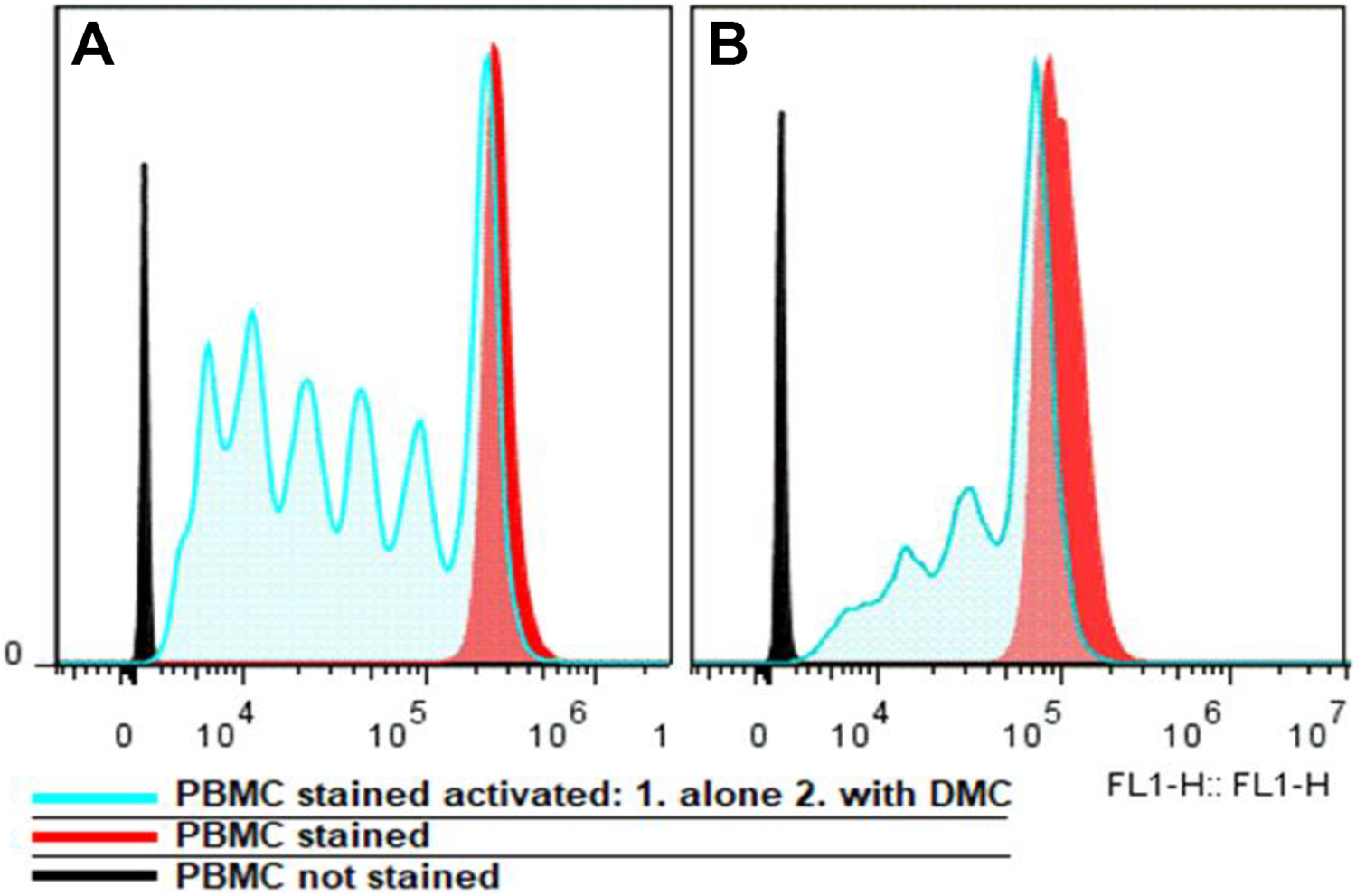
Inhibitory effect of DMC on PBMC proliferation. (A) Normal proliferation cycles of activated PBMC and (B) inhibition of proliferation cycles of PBMC with the co-culture with DMC. DMC, differentiated mesenchymal cell; PBMC, peripheral blood mononuclear cells.
Mouse Skin Wounds Model, In Vivo Assay
The use of laboratory animal tests offers an excellent option to evaluate DMC’s advantages and disadvantages. The tolerance of human DMCs was further investigated in vivo by xenotransplantation of the cells into immunocompetent C3H/s mice. Mice express an HLA-G protein equivalent, the Qa2 molecule. It has a functional and structural homology 31,32 .
Macroscopic analysis
In the control group i, the normal wound healing process took a period of 12 ± 1.03 days, and pilosity was recovered in 25 ± 1.13 days. In group ii, murine ucMSC-treated mice, lesions healed after 9 ± 0.75 days, and pilosity was recovered in 20 ± 1.02 days. In group iii, DMC-treated mice, the skin wound healed at 5 ± 0.81 days and the area regained its hairiness on 10 ± 0.90 days. The observed time values between group i and iii (control vs DMC treated mice) are statistically significant (P < 0.01) (Fig. 4B, 5).
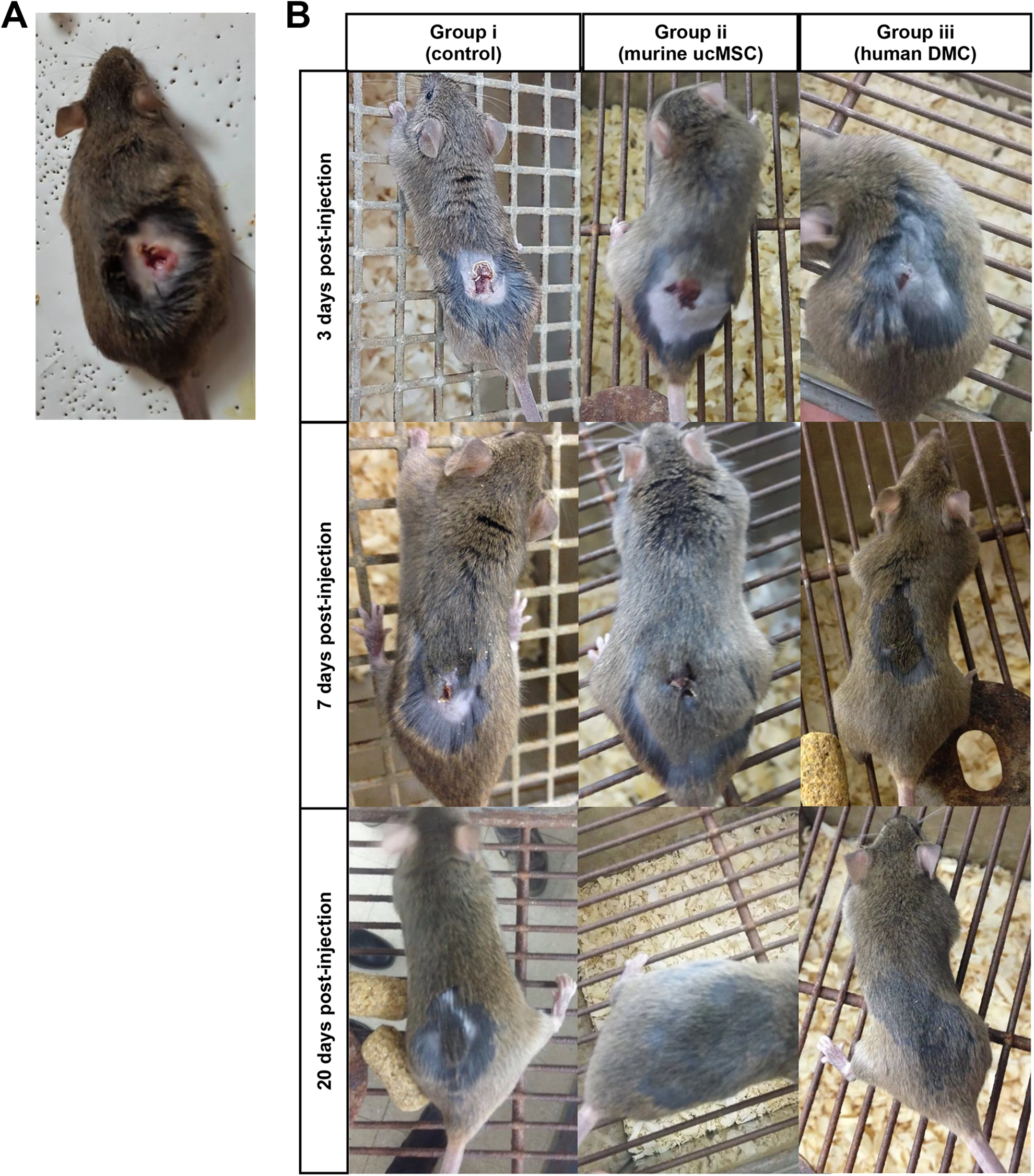
Images from the trial in mice. (A) Photography of initial dorsal lesion in C3H/s mice. (B) Photographs of wound healing and pilosity evolution at 3, 7, and 20 days post-treatment in the 3 analyzed groups.
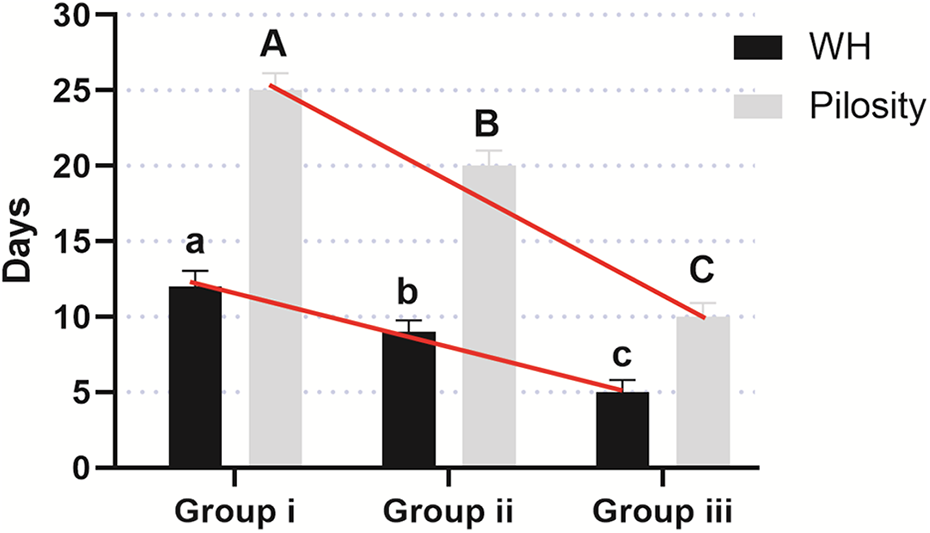
Comparisons of WH and pilosity in the 3 analyzed mice groups. The figure shows significant variations (in days) for WH and pilosity between the 3 analyzed mice groups. WH, wound healing.
Histological observations of wound healing
Histological observation provided key information in the wound healing process analysis. H&E stained sections of the injured region showed a disordered appearance concerning the normal tissue structure and the typical granulation tissue in healing wounds in the 3 analyzed experimental groups. Also, the histological analysis of the skin sections of animals from groups ii and iii at day 5 post-treatment (injected with murine ucMSC and human DMC, respectively), did not show large quantities of lymphocytes and others infiltrating immune cells in the area of the lesion (Fig. 6).
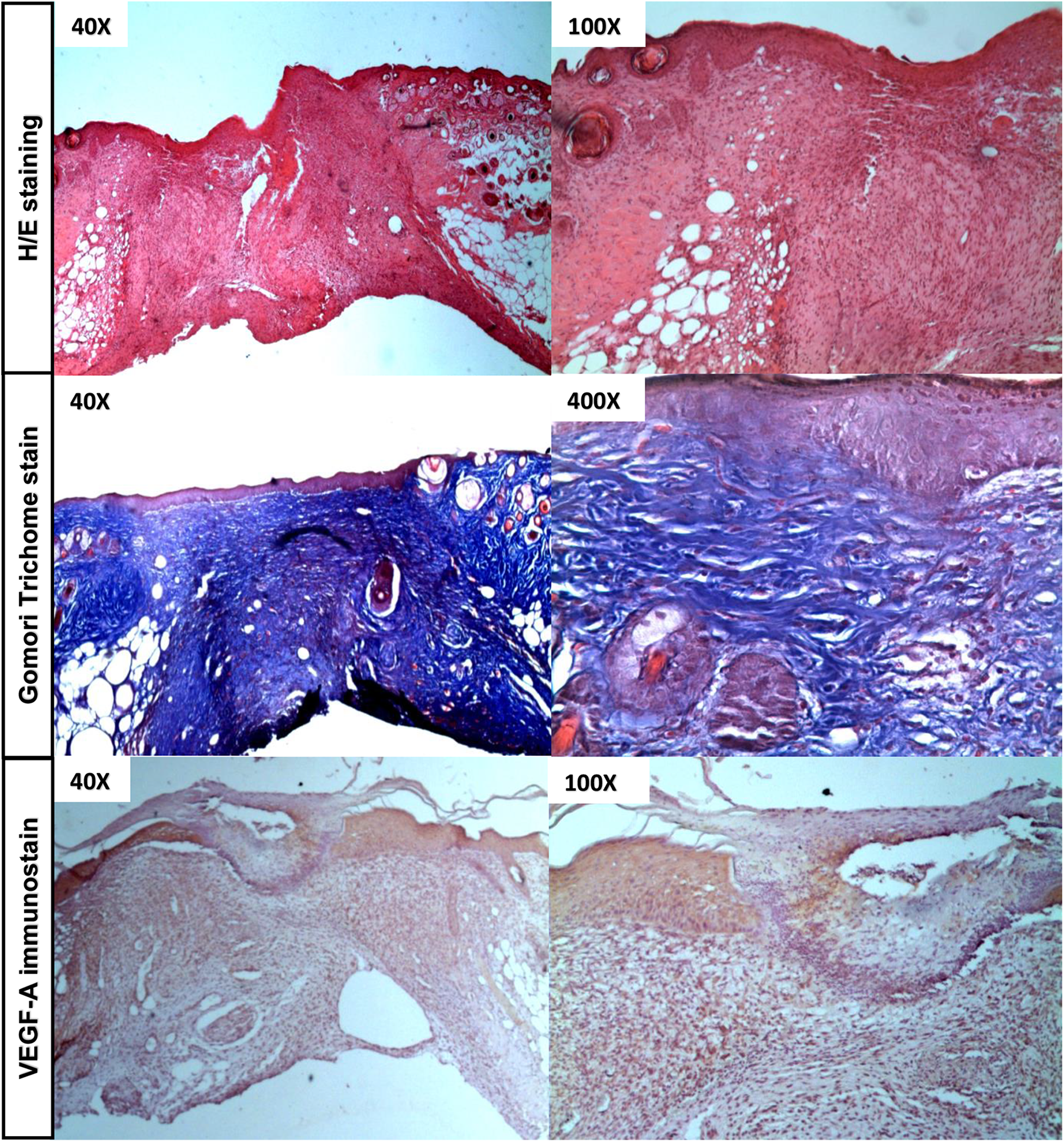
Histology analysis by H&E, Gomori trichrome, and IHC stain. Skin sections were obtained from group iii of mice, after 5 days of human DMC transplantation. Epidermis, dermis, and hypodermis were observed. By H&E staining, we could observe the typical granulation tissue. With the Gomori trichromic, we observed a good organization of collagen fibers of the connective tissue. For IHC, formalin-fixed tissue sections were immunostained using primary antibody VEGF-A, clone C-1 (Santa Cruz Biotech, CA, USA); 3′3-diaminobenzidine (Sigma, St. Louis, MO, USA) is the chromogen. We can observe a high number of immunomarked fibroblasts (intracytoplasmic) expressing VEGF-A in the cellular dermis. DMC, differentiated mesenchymal cell; H&E, hematoxylin and eosin; IHC, immunohistochemistry; VEGF, vascular endothelial growth factor.
Using the Gomori stain, we analyzed the appearance and organization of the collagen fibers in connective tissue. We initially observed in the control group a pattern of fine and isolated fibers without actually forming a thick collagen package or bundles. However, in groups ii and iii, injected with murine ucMSC and human DMC, respectively, progress was observed in the organization and packaging of collagen, compared with that described in group i, evidencing an active advance of the regeneration process (Fig. 6).
Immunohistochemistry Analysis
The immunolabeled sections with VEGF-A antibody showed intense positive labeling in connective tissue cells, especially in fibroblasts, scattered around the granulation tissue developed in the area of the lesion. In group iii, xenotransplanted with human DMC, more than 70% of the observed fibroblasts expressed VEGF-A (Fig. 6).
Discussion
The failure in the restoration of a functional epidermal barrier can lead to the establishment of an ulcerative skin defect 1 . CVU is an important skin chronic disease that compromises the life quality of a great number of patients. According to the published by the International Diabetes Federation in 2015, every year between 9.1 and 21 million diabetic people around the world develop foot ulcers, most of which do not heal, leading to the appearance of infections and complications in the patient, and may even lead to amputation of the affected limb 2 . Not just diabetes, obesity, and a sedentary lifestyle in older people are also common causes of this morbidity 33,34 .
Although so far, many therapies have been developed to treat this disease, none turned out, for different reasons, to be successful in all cases. In this context, therapy with MSC has shown to be a promising therapeutic strategy 12,13,35 . The use of autologous MSC has been successful despite, as mentioned above, significant limitations. In contrast, the ucMSCs have enormous therapeutic potential given their infrequent characteristics. The umbilical cords are obtained from the discarding of normal deliveries, thus avoiding any traumatic and potential complications for obtaining cells, and also, they are a source of a large number of cells, expandable in culture 14 . The ucMSC are multipotent cells and can differentiate into mesenchymal lineages, such as osteocytes, chondrocytes, and adipocytes. This characteristic was studied by multipotent assay and published in a previous article 28 . Thus, the presence of ucMSC in the wound could be effective not only by promoting the cell proliferation from the adjacent tissues but also by differentiating into these mesenchymal cell populations. Another relevant aspect of ucMSC is the high level of HLA-G expression, up to 90% 9,18 . All DMC, obtained for these experiments, were HLA-G positive used in lower passages. The expression of this checkpoint is the main reason for the allogeneic cell acceptance due to the effect in local immune response blocking. HLA-G exerts its action binding to 2 main cell membrane receptors: ILT2 and ILT4 22 –24 . The ILT2 receptor is expressed on the surface of many immune cells, including monocytes/macrophages, dendritic cells (DC), B-lymphocytes, a subpopulation of T-lymphocytes, and natural killer cells, while the ILT4 receptor is expressed only by neutrophils and myeloid cells. The presence of these receptors on almost all cells of the immune system explains the important immunosuppressive effect exerted by HLA-G 22 –24,36 . The observed results with the co-culture between DMC and PBMC confirm that the DMCs have an important immunosuppressive effect through the proliferation of activated lymphocytes. On the other hand, DMCs do not express the HLA-DR molecule, an HLA class II antigen. It is a cell-surface transmembrane protein that is expressed on antigen-presenting cells, also found in bone marrow MSC 37 . Since HLA-DR expression is essential for the presentation of peptides or antigens, initiating, eventually, a specific immune response 38 its absence is fundamental for the allogenic DMC transplantation. Confirming these observations, it is worth mentioning 2 published studies, which evaluate the toxicity of MSC and their exosomes administered in monkeys, and no stem cell transplantation-related toxicity was found for human ucMSC 39,40 .
Stem cell therapy activates various key cellular processes for damaged tissue regeneration. These include the contribution of enabling parenchymal cells, whose action promotes angiogenesis and induces paracrine action in neighboring cell populations, all of which contribute to the regeneration and repair of the injured tissues 9,10,26 . As we have already mentioned, angiogenesis plays a key role in wound healing, being especially important in chronic ulcerative skin lesions, by restoring blood perfusion and delivering nutrients to injured sites 26 . Thus, VEGF expression, the most potent mitogenic agent of endothelial cells for angiogenesis, repair, and capillary tube remodeling, by DMC subpopulation, is essential for the treatment of this type of pathology 41 –43 .
Considering all the above, DMCs are excellent candidates to use for wound healing treatment. First, the high HLA-G expression and a lack of HLA-DR expression make them one of the most appropriate cell populations for allogeneic transplantation, and second, these cells can release various growth factors, including VEGF, which exert their paracrine effect in the area where the cells are applied, activating the regeneration processes in normal adjacent tissues, similar to the autologous MSC effect 11,12 .
To evaluate the safety and efficiency of DMC-based therapies, in a clinically representative in vivo model, with an intact immune system we have used the C3H/s mice. Previous works have reported that mice have an HLA-G functional and structurally equivalent, called Qa2 31,32 . Their murine PIR-B receptor, equivalent to the human ILT4 receptor, is expressed in myeloid cells and B lymphocytes, activating the differentiation of regulatory and suppressing T cells 44,45 . In the present study, the immunomodulatory DMC activity was further demonstrated by their effect as xenotransplants in immunocompetent C3H/s mice without any immunosuppressive treatment. Due to this functional homology between HLA-G and Qa2, HLA-G expression from human DMC injected in mice can induce immunosuppressive effects, very similar to what happens in humans, avoiding cell rejection by the host. The in vivo assays results were highly positive since the DMC population significantly diminished the period required for wound healing. Although the experimental lesion produced in mice is not due to the common pathogenesis observe in ulcerative skin defect, the process of dermis and epidermis healing and regeneration are similar, which allows us to predict the DMC subpopulation future behavior when it is applied to human dermal ulcerative lesions. Histological analysis of the skin sections confirmed this observed improvement in wound healing and the absence of an inflammatory process added when DMCs were transplanted. The repair and remodeling process, after DMC applying, showed the same regeneration pattern as that observed in the control group but faster. Thus, we have shown that human DMC could be transplanted in a xenogeneic form in mice. Another noteworthy observation was that the mice group injected with human DMC was the one that healed the fastest. This improved ability of DMC compared with murine ucMSC could be owing to homogeneity in this human cell population concerning murine ones, in which different cell lineages possibly coexist, some of which may have been less effective in the wound healing process, decreasing its reparative capacity. It demonstrates the importance of using a specific, well known, homogenous, and characterized cell population, like DMC, to optimize its positive impact, ensuring maximum efficacy and safety in the treatments. Finally, no less important is the fact that these cells are obtained from disposable tissue so that their manipulation and therapeutic use does not bring any ethical objection.
Conclusion
DMC population has been characterized and studied both in vitro and in vivo assays demonstrating its safety and efficacy to the proposed therapy. The data reported here indicate that the DMC population allows allogeneic transplantation due to the HLA-G expression and has positive effects on wound healing by promoting local angiogenesis in dermal skin lesions. Thus, DMC could play a very important role in regenerative medicine and maybe a novel cell-therapeutic tool for dermal healing diseases.
Footnotes
Acknowledgments
The authors are particularly grateful to Javiera Marini for assistance in immunohistochemistry experiments and Dario Espinosa for technical assistance.
Ethical Approval
This study was approved by the COBIMED (Comité de Bioética y Ética de la Investigación de la Facultad de Ciencias Médicas de la Universidad Nacional de La Plata) and the Institutional Committee for the Care and Use of Laboratory Animals, Facultad de Ciencias Médicas, Universidad Nacional de La Plata, Argentina. Protocol number P01-03-2018.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the guidelines of COBIMED (Comité de Bioética y Ética de la Investigación de la Facultad de Ciencias Médicas de la Universidad Nacional de La Plata), and the animal procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals (CICUAL), Facultad de Ciencias Médicas de La Universidad Nacional de La Plata, Buenos Aires, Argentina.
Statement of Informed Consent
Written informed consent was obtained from each mother before umbilical cord donation. The donation is anonymous. The informed consent has been approved by the COBIMED (Comité de Bioética y Ética de la Investigación de la Facultad de Ciencias Médicas de la Universidad Nacional de La Plata).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Program of the MINCyT (Ministerio de Ciencia y Tecnología) of Argentina (code 11/M230).
