Abstract
Evidence for umbilical cord blood (UCB) cell therapies as a potential intervention for neurological diseases is emerging. To date, most existing trials worked with allogenic cells, as the collection of autologous UCB from high-risk patients is challenging. In obstetric emergencies the collection cannot be planned. In preterm infants, late cord clamping and anatomic conditions may reduce the availability. The aim of the present study was to assess the feasibility of UCB collection in neonates at increased risk of brain damage. Infants from four high-risk groups were included: newborns with perinatal hypoxemia, gestational age (GA) ≤30 + 0 weeks and/or birthweight <1,500 g, intrauterine growth restriction (IUGR), or monochorionic twins with twin-to-twin transfusion syndrome (TTTS). Feasibility of collection, quantity and quality of obtained UCB [total nucleated cell count (TNC), volume, sterility, and cell viability], and neonatal outcome were assessed. UCB collection was successful in 141 of 177 enrolled patients (hypoxemia n = 10; GA ≤30 + 0 weeks n = 54; IUGR n = 71; TTTS n = 6). Twenty-six cases were missed. The amount of missed cases per month declined over the time. Volume of collected UCB ranged widely (median: 24.5 ml, range: 5.0–102 ml) and contained a median of 0.77 × 108 TNC (range: 0.01–13.0 × 108). TNC and UCB volume correlated significantly with GA. A total of 10.7% (19/177) of included neonates developed brain lesions. To conclude, collection of UCB in neonates at high risk of brain damage is feasible with a multidisciplinary approach and intensive training. High prevalence of brain damage makes UCB collection worthwhile. Collected autologous UCB from mature neonates harbors a sufficient cell count for potential therapy. However, quality and quantity of obtained UCB are critical for potential therapy in preterm infants. Therefore, for extremely preterm infants alternative cell sources such as UCB tissue should be investigated for autologous treatment options because of the low yield of UCB.
Keywords
Introduction
Impressive advances in perinatal and neonatal care have led to substantial improvements in survival rates for preterm infants and neonates with severe complications. However, survival comes at a cost. Neonates with brain damage often suffer from long-term neurological motor and cognitive deficits resulting in enormous physical, psychological, and economic costs 1 –4 . Cerebral palsy (CP) is one of the most costly neurologic disabilities due to its frequency (2/1,000 births) and persistence over life 5 . Currently available treatments for patients with CP are supportive, but not curative. Apart from term infants with perinatal hypoxemia, preterm infants, monochorionic twins with twin-to-twin transfusion syndrome (TTTS), and infants with intrauterine growth restriction (IUGR) are also frequently affected by cerebral damage. Up to 10% of very low birthweight infants (<1,500 g) suffer from CP and 16% of very preterm infants (22 to 28 weeks) are complicated with severe intraventricular hemorrhage (IVH) 1,4 . TTTS is a rare but severe complication that can result in death of a co-twin and leaves up to 30% of survivors with an abnormal neurodevelopment 6 .
A new approach for neonates with brain damage is the treatment with umbilical cord blood (UCB) derived cells. UCB contains a large amount of highly proliferative progenitor cells and stem cells 7,8 . Bystander effects have been postulated to be the main mechanisms for functional recovery after cord blood transplantation 9 . Transplanted UCB cells may migrate to the affected area and deliver trophic factors that provide anti-inflammatory and antiapoptotic effects and increase the plasticity of the injured brain by enhancing neovascularization, myelination, and endogenous neurogenesis 10 –15 . In several animal models, neurological and survival benefits have been demonstrated for the application of UCB cells in the setting of stroke, ischemia, intracranial hemorrhage, and spinal cord injury 14 –23 . Current pilot studies suggested benefits of autologous cell treatments in term infants with hypoxic-ischemic (HIE) and CP 24,25 . Autologous approaches harbor a minimal risk of immunogenic reaction and infections compared to allogenic approaches.
Nevertheless, current clinical trials mostly work with allogenic UCB cells. This is mostly due to the logistic advantage of allogenic approaches. Allogenic cell products can be manufactured and be used “off the shelf” in the clinical setting 26 . The collection of autologous cells is more complicated and the quality of the resulting cell products is directly depending on the quality of the individual collection sample making it difficult to compare. In order to design valuable clinical trials, the preparation of cells from individual cell sources has to become standardized. Trials directly comparing autologous and allogenic treatment options in neonates are lacking. Those working with autologous UCB cells mostly involve mature neonates or preterm infants with a minimum of 28 + 1 to 37 + 0 weeks of gestation 24,25,27,28 .
It is unclear whether autologous UCB collection is feasible for the aforementioned high-risk populations. In very preterm infants (<30 + 0 weeks of gestations), late cord clamping and anatomic conditions limit the available amount of UCB. In emergency situations or unexpected deliveries, the collection cannot be planned. Thus, collection of UCB requires a good collaboration especially in large perinatal centers with constantly changing teams of clinicians and midwifes. The aim of the present study is to investigate the feasibility of autologous UCB collection in high-risk neonatological populations and to assess the quantity and quality of the samples collected. The successful collection of UCB in challenging anatomical conditions and emergency situations is essential prerequisite to allow for autologous treatment options.
Methods
Study Design
We initiated this pilot study in December 2017. This study was approved by the Institutional Ethics Committee of the Charité – Universitätsmedizin Berlin (Berlin, Germany) (EA2/206/17). The use of human UCB and all procedures in this study were conducted in accordance with the Institutional Ethics Committee of the Charité – Universitätsmedizin Berlin (Berlin, Germany) (EA2/206/17) approved protocols. Written informed consent was obtained from the patients for their anonymized information to be published in this article. The project was designed in collaboration with Vita 34 AG (Leipzig, Germany), a European cord blood bank, in order to use the established infrastructure and guarantee high standards for the banking of the acquired cord blood. To enhance dedication and compliance with the project, team briefings for all obstetricians, midwifes, and anesthetists were held discussing the current state of literature on UCB therapy in neonates and the aim of the study. All midwifes and obstetricians were trained for collection of UCB. Candidates for UCB collection were infants from four high-risk groups: perinatal symptomatic hypoxemia, extremely preterm infants (gestational age 23 + 0 to 30 + 0 weeks and/or estimated birthweight less than 1,500 g), estimated IUGR <3rd percentile for birthweight independent of gestational age, and monochorionic twins with TTTS. After emergency C-sections or other obstetrical emergency situations, UCB was always collected. Neonates were included in the study, if criteria for any of the four groups were met or if criteria for therapeutic hypothermia were met (defined as metabolic or mixed acidosis with an arterial cord pH ≤7.0 or a base deficit of >16 or an Apgar score at 10 min of ≤5 or need for positive pressure ventilation for ≥10 min). If the neonate did not meet the inclusion criteria, the UCB was discarded. Neonates with mothers positive for hepatitis B (HBsAg and/or HBeAg) or C virus (anti-HCV), syphilis, HIV (anti-HIV-1 and -2) cytomegalovirus (IgM antibodies), rubella, toxoplasma, or herpes simplex virus were excluded as were mothers under the age of 18. UCB collection was performed aseptically via in utero or ex utero techniques into cord blood collection bags (Vita 34 AG) containing 35 ml of citrate phosphate dextrose anticoagulant. Mothers who were at high risk for preterm delivery, expecting an IUGR child, or multiples with TTTS were informed about the study and prior written informed consent for collection was sought if admitted to the hospital (see Fig. 1). For deliveries in which prior collection consent had not been obtained, the local Ethics committee gave permission for obstetric staff to collect UCB and seek the mothers consent after delivery. If UCB was successfully collected and the infant met the inclusion criteria, mothers were asked to provide written informed consent for study participation. If she did not consent the blood was discarded.
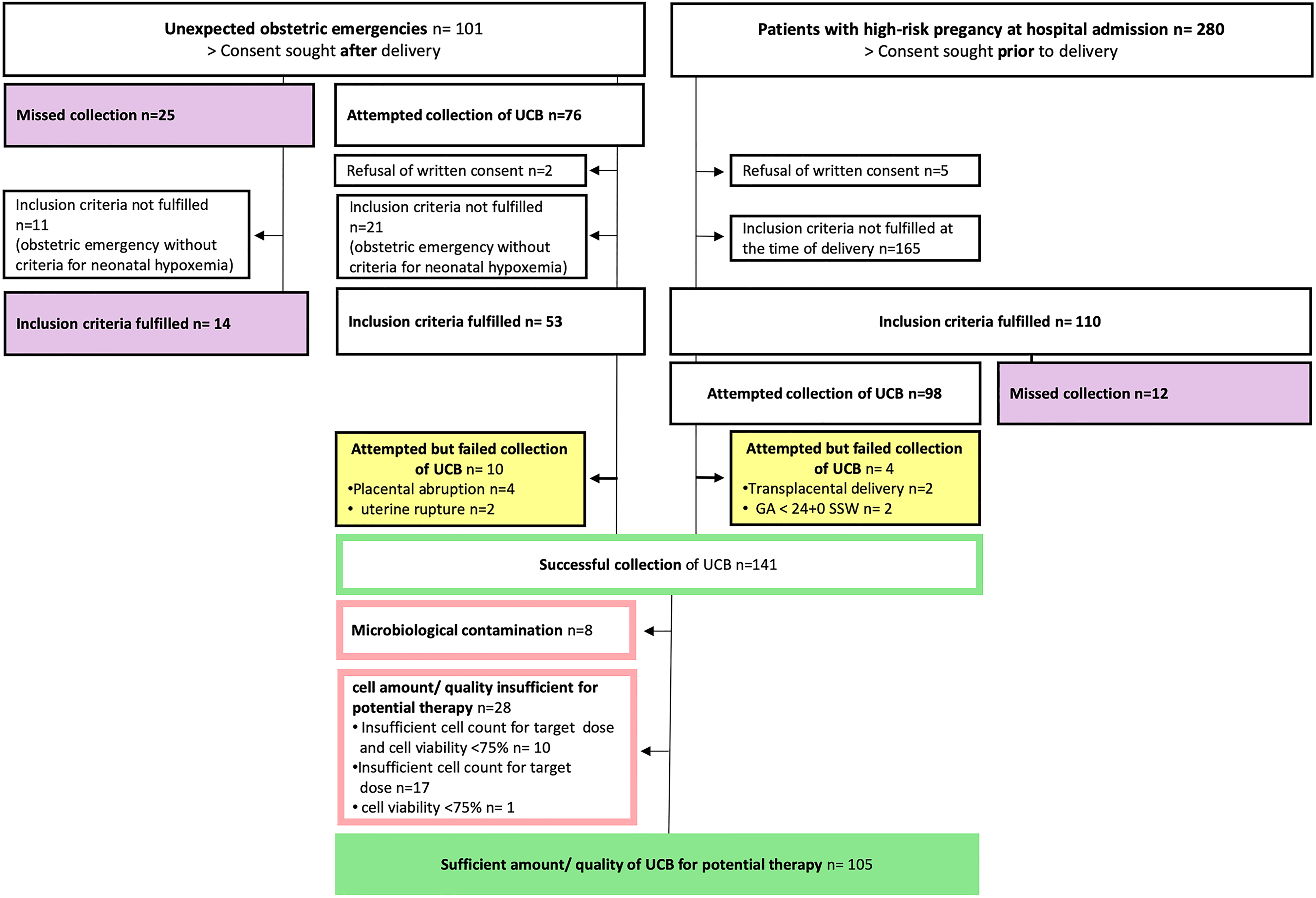
Distribution of cases. The flow chart is describing the process of case inclusion and assessment of feasibility and UCB quality/quantity. UCB: umbilical cord blood.
Assessment of UCB Quantity and Quality
The obtained UCB was transported at room temperature in a validated transport box to Vita 34 AG cord blood bank and processed, cryopreserved, and banked according to the manufacturing authorization for cord blood transplants of Vita 34. The whole blood was cryopreserved in a rate-controlled freezer with the cryoprotectant dimethylsulfoxide. The UCB was stored in the vapor phase above liquid nitrogen at temperature between −150°C and −185°C. The UCB was banked at Vita 34 for the affected families free of charge in order to be available for future therapeutic options. Total nucleated cell count (TNC) pre- and post-processing, sterility, and viability were assessed by microscopic counting, by anaerobic and aerobic BACTEC blood culture system (Becton Dickinson, Franklin Lakes, NJ, USA) and by acridine orange staining, respectively. In cases of missed UCB collection circumstances leading to failure were assessed. Missed cases were discussed on daily ward rounds.
Assessment of Neonatal Outcome
Neonatological outcome was assessed through medical charts and radiology reports. All neonates received treatment and diagnostic tests according to standard operating procedures of the hospital, including cranial ultrasound, magnetic resonance imaging, and amplitude-integrated electroencephalograms, as applicable. HIE was defined as perinatal asphyxia accompanied by pathological amplitude-integrated electroencephalogram, seizures, or other neurological symptoms and signs. CP was diagnosed based on clinical assessment and radiological findings 29 . For the assessment of IVH, cranial ultrasound scans were graded according to the Papile scoring system 30 . Periventricular leukomalacia (PVL) was defined as cystic PVL and diagnosed by neuroimaging using cranial ultrasound 31 .
Statistical Analyses
We used descriptive statistics to characterize subject demographics and feasibility measures. Continuous variables are reported as median and range. Categorical variables are summarized as frequency counts and percentages. The correlations between gestational age and the quantity and quality measures of the UCB specimens were investigated using Spearman’s rank correlation test, as appropriate. A significance level of 0.05 (P value) was determined for all calculations. All statistical analyses were performed using IBM SPSS, v22 for Windows (IBM Inc., Armonk, NY, USA).
Results
Distribution of Cases
The distribution of cases is further summarized in Fig. 1. Between 12/2017 and 12/2018, UCB was collected from two groups: first, unexpected obstetric emergencies (n = 101) and second from patients with expected high-risk deliveries (n = 110), that had given their consent prior to inclusion. Five patients refused written consent prior to inclusion and were excluded from the study. In the first group of obstetric emergencies, UCB collection was attempted in most cases (75.2%, 76/101). UCB collected from 21 neonates in initially critical clinical condition (shoulder dystocia n = 2, placental abruption n = 3, uterine rupture n = 2, prolapse of the arm n = 2, and terminal bradycardia n = 12) and was later discarded for not meeting the inclusion criteria of symptomatic hypoxemia. Two patients refused written consent after successful collection of UCB and the collected UCB was also discarded. In the second group of expected high-risk deliveries, an even higher percentage of collections was attempted (89.1%, 98/110).
In 17.5% (37/211) of all cases the collection of UCB was missed due to organizational difficulties (obstetric emergencies n = 25, expected high-risk delivery n = 12). Of those cases 13.5% (5/37) were spontaneous deliveries, 16.2% (6/37) vacuum extractions, 18.9% (7/37) elective C-sections, and 51.4% (19/37) crash/emergency C-sections. However, only 14.6% (26/177) of the cases with fulfilled inclusion criteria were missed, including 7 cases of perinatal hypoxemia, 10 with a gestational age ≤ 30 + 0 weeks, and 9 children with IUGR.
In a small number of cases (5.6%, 10/177) the collection was attempted but failed in a clinical setting of placental abruptions, uterine ruptures, transplacental deliveries, and neonates with a gestational age of less than 24 + 0 weeks. UCB collection was successful in 79.7% (141/177, hypoxemia n = 10; gestational age ≤ 30 weeks n = 54; IUGR n = 71; TTTS n = 6). The median blood loss in cases with attempted but failed collection of UCB was 1,400 ml (range: 500–1,800 ml). The median blood loss in cases with successful collection of UCB was 500 ml (range: 250–2,500 ml).
Quantity and Quality of Obtained UCB
In 141 cases with successfully collected UCB, 105 units had sufficient quality and quantity for potential therapy. A total of 36 UCB units lacked sufficient quantity and/or quality for potential therapy. Eight units of UCB (8/141, 5.7%) were microbiologically contaminated. In 28 units the cell amount or quality was insufficient for potential cell therapy due to an insufficient cell count for at least one dose of the target dose (1–5 × 107 cells/kg) at birthweight and/or cell viability < 75% (for details, see Fig. 1). Almost all of these cases (82.1%, 23/28) were preterm infants with a gestational age of less than 30 weeks or birthweight of less than 1,500 g. Five cases (1.8%, 5/28) were neonates with IUGR. Characteristics of cases with collected UCB are summarized in Table 1. Quantity and quality of collected UCB cells appeared to correlate with gestational age: the stored UCB volume and the nuclear cell count were strongly related to gestational age (Rs = 0.66. P < 0.001 and Rs = 0.74, P < 0.001, respectively) (see Fig. 2). Cell viability was only weakly correlated with gestational age (Rs = 0.19, P = 0.04).
Characteristics of Cases with Successfully Collected UCB.
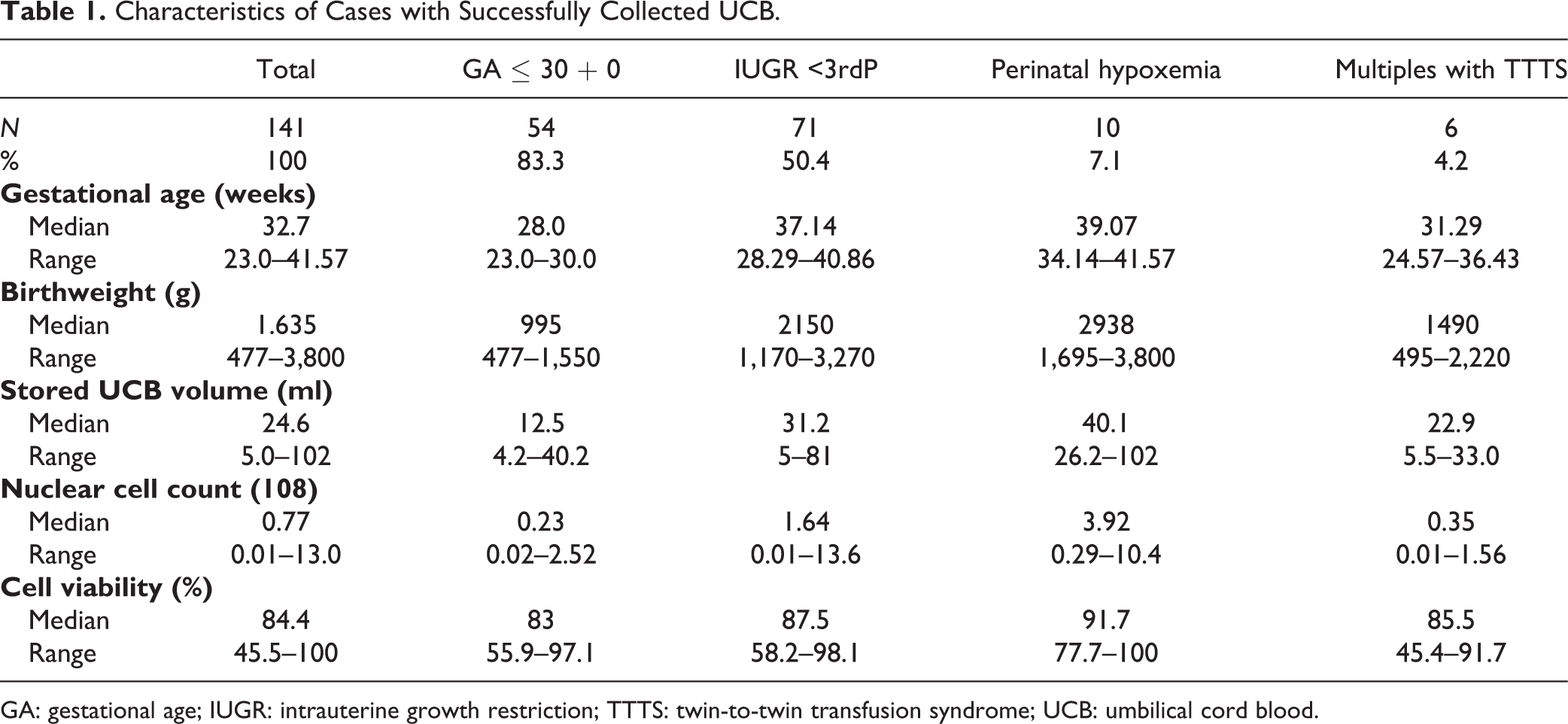
GA: gestational age; IUGR: intrauterine growth restriction; TTTS: twin-to-twin transfusion syndrome; UCB: umbilical cord blood.
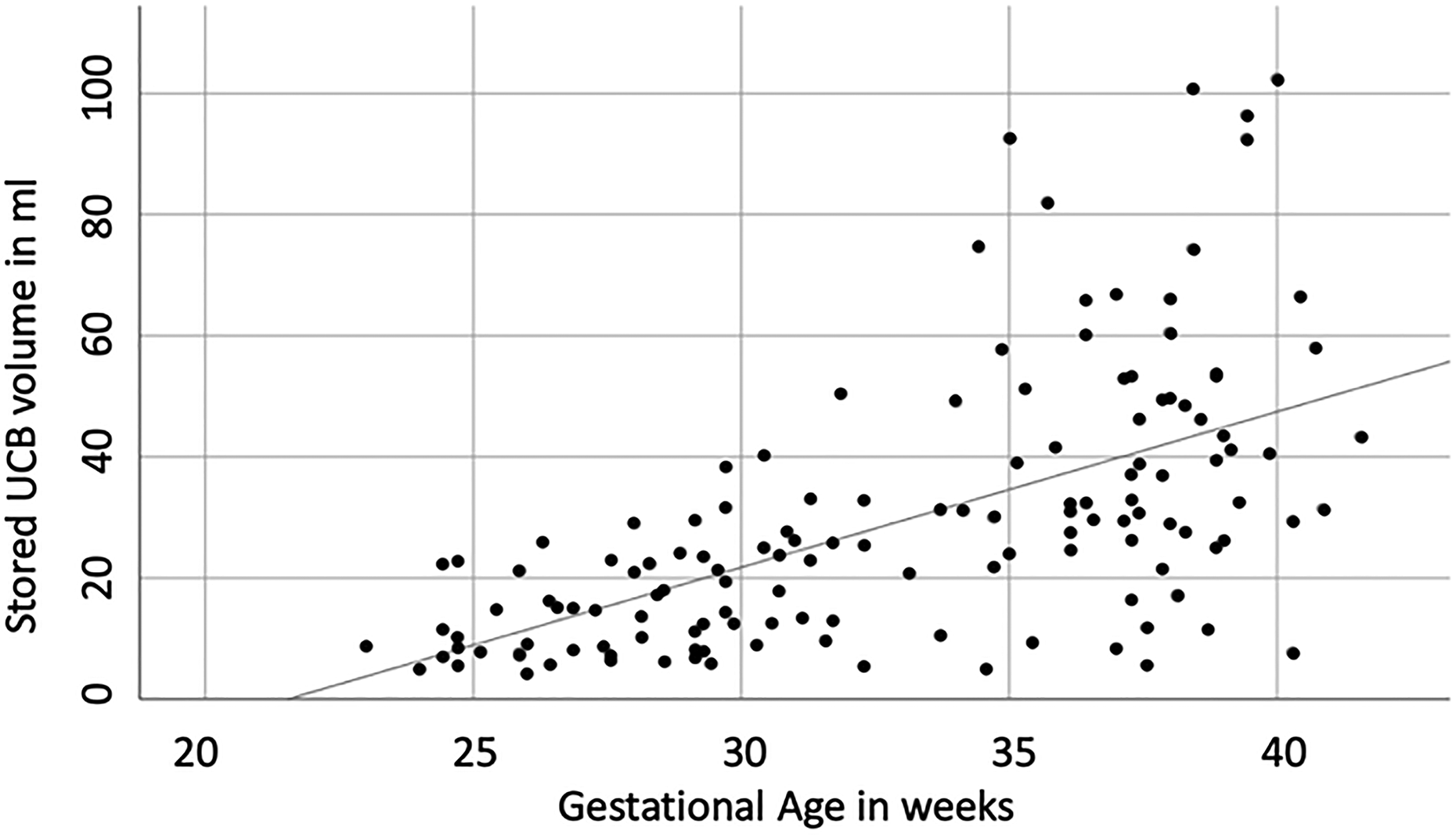
Volume of collected UCB in correlation to gestational (Spearman rank order coefficient 0.66, P < 0.001). UCB: umbilical cord blood.
Training Effect
In the first 3 months (December to February) of the project, 41 cases were enrolled. Thirty-six percent of these cases were missed (14/41). At this point only obstetricians were trained to collect the UCB and collection kits were stored in a collective place. After February 2018, a systematical training system and logistic changes were implied. Teams of all disciplines involved (obstetricians, neonatologists, midwifes, anesthetists, and surgical assistants) received trainings and were informed about the study. Trainings were repeated monthly. Missed cases and incorrectly collected samples were discussed by the research staff during daily ward rounds with midwifes and obstetricians in the delivery suite. During an established weekly perinatal conference, where important cases were discussed between obstetricians, midwives, and neonatologists, weekly updates on the progress of the study were given and outcome of patients with collected UCB were discussed. Collection kits were now placed in all delivery areas and operating suites. With these efforts, there was a learning curve within the team (see Fig. 3) and an average of only 8.8% of enrolled cases (12/136) were missed during the rest of the year (March 2018 to December 2018). The positive impact of daily supervision by the research personnel for successful UCB collection is illustrated by a period of 2 weeks in April 2018, where a peak of missed collections can be observed (three cases) while both doctors responsible for the study were not on site for a period of 2 weeks simultaneously.
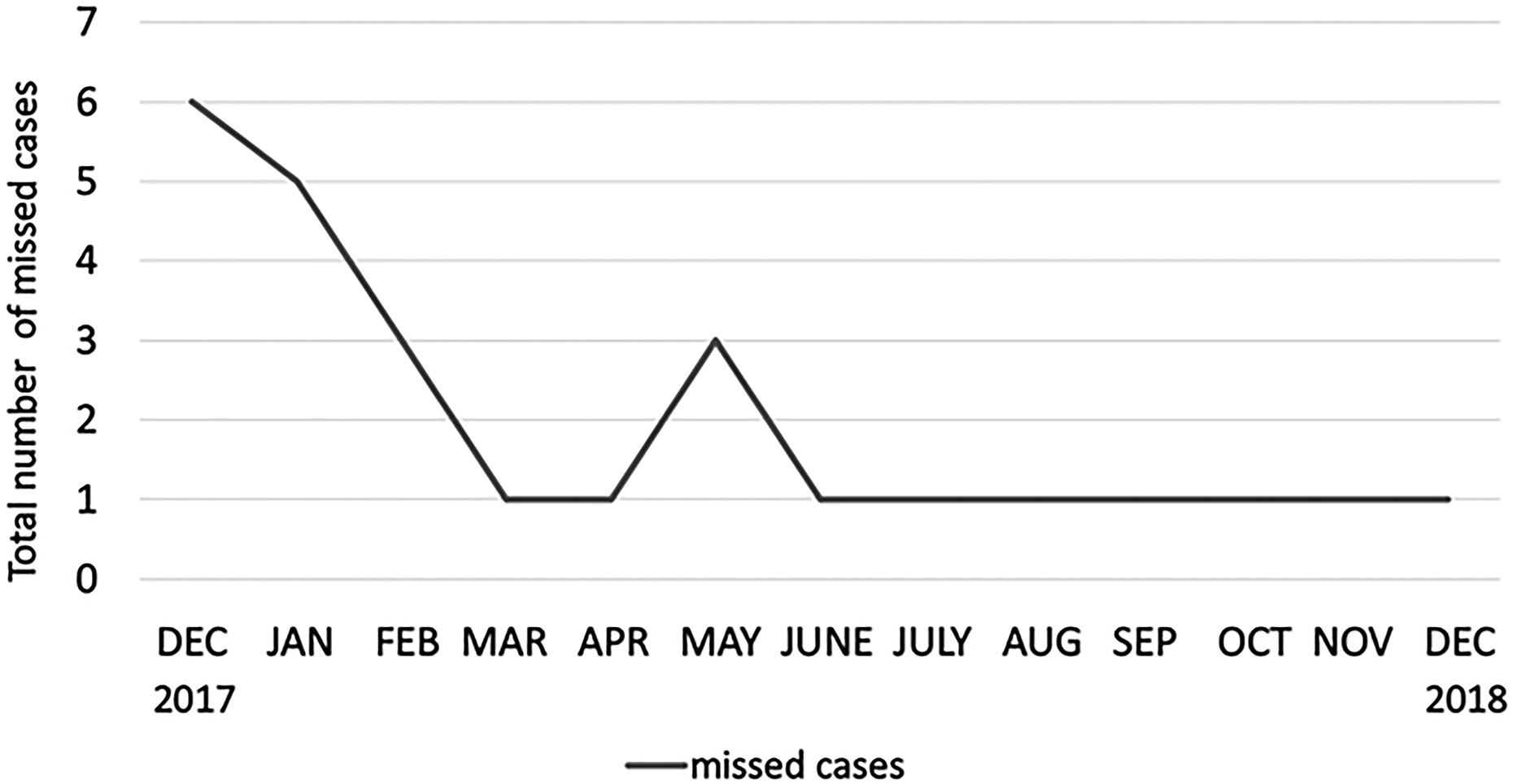
Total number of missed cases per month during the recruitment period. A learning curve within the team can be observed. Only cases with fulfilled inclusion criteria were taken into account.
Outcome of Included Neonates
Six (3.4%) of all included neonates deceased. Four of whom were extreme preterm infants with a gestational age of less than 25 weeks and deceased within days after birth, one was discovered to suffer from a complex congenital heart anomaly and one from a congenital cardiomyopathy. As illustrated in Table 2, 10.7% (19/177) of all included neonates developed brain lesions. The most common events were IVH (9/177, 5%) and HIE (7/177, 4%), followed by PVL (2/177, 1%) and CP (1/177, 0.5%). Within the different groups, neonates with TTTS had by far the highest prevalence of perinatal brain damage with 80% (4/5). 36.8% (7/19) of children with perinatal hypoxemia suffered from HIE and 11.6% (8/69) of neonates born with a gestational age of ≤30 + 0 weeks suffered from IVH (n = 7) or PVL (n = 1). Within the group of neonates with IUGR none (0/82) were affected by perinatal brain damage.
Manifestation of Perinatal Brain Damage in 177 Included Neonates of Four High-risk Groups.

CP: cerebral palsy; GA: gestational age; HIE: hypoxic ischemic encephalopathy; IUGR: intrauterine growth restriction; IVH: intraventricular hemorrhage; P: percentile; PVLM: periventricular leukomalacia; TTTS: twin-to-twin transfusion syndrome.
Discussion
Integration of UCB Collection into Routine Proceedings
The present single-center study assessed the feasibility of UCB collection in neonates at high risk of perinatal brain damage. Our data show that the collection of autologous UCB in neonates at high risk of brain damage is organizationally possible but challenging. Multidisciplinary collaboration, daily team briefings, and personal engagement were essential for the success of the study. Figure 3 illustrates the training effect in the study.
In 82.5% of all cases the collection of UCB was attempted (174/211). In 23 of these cases UCB was collected prophylactically but the inclusion criteria were not met (see Fig. 1) and the UCB was discarded. One hundred and seventy-seven cases fulfilled the inclusion criteria. In 80% of these cases (141/177) the collection was successful. Integrating the collection of UCB into routine proceedings is essential to overcome organizational difficulties of the collection, especially in emergency settings. The collection rate in cases with expected high-risk deliveries was much higher (89.1%, 98/110) than the rate of attempted collections in obstetric emergencies (75.2%, 76/101). Emergencies such as crash C-sections and vacuum extractions complicate the collection of UCB as they challenge the team and may distract from the collection of UCB. 67.6% (25/37) of all missed cases in this study were vacuum extractions and emergency C-sections. In another study of UCB collection for banking, 16.6% of cases were missed due to dystocial child birth and urgent C-section 32 . A possible future approach to overcome organizational failures may be the system of “all collect.” To guarantee the collection of UCB in tricky situations, UCB could just be routinely collected after every delivery just like arterial blood-gas analyses are routinely performed from UCB after delivery. Whether an “all collect” system is worth the effort has to be determined in the future depending on chances of actual use of autologous UCB units.
Impact of Gestational Age on Quality of UCB Cells
Collection of UCB may not only be challenging for organizational reasons but also for technical reasons: first, a greater loss of maternal blood seems to complicate the collection of UCB. In 10 cases of attempted but failed UCB collection the median maternal blood loss was 1,400 ml (range: 500−1,800 ml). This can be explained through the placenta being spared during maternal centralization of blood circulation. Situations like placental abruption and uterine rupture are therefore not only life threatening for the unborn child and the mother but may also influence the collection of UCB. Second, the gestational age of the affected child appears to be a major issue for the quantity and quality of the collected UCB. Both the stored UCB volume and nuclear cell count correlated with increasing gestational age. In 42.6% (n = 23/54) of UCB units collected from preterm neonates with a gestational age of ≤30 + 0 weeks, the sample was insufficient for transplantation either due to a too low amount of TNC for at least one dose containing the target cell number at birthweight or viability of less than 75% of cells. Conversely, almost all cases (23/28) with an insufficient quantity of UCB cells for transplantation were preterm infants.
To date, several studies have shown that the cord blood volume and TNC are positively correlated with birthweight, but none of them have investigated cord blood collection in extreme preterm infants. Clinical trials involving autologous UCB cells mostly involve mature neonates or have included preterm infants with 28 + 1 to 37 + 0 weeks of gestation 24,25,27,28,33 . Ballen et al. noted that each 500 g increase in the infants’ weight caused a 6%, 11%, 22%, and 28% increase in the volume, TNC, colony-forming units, and CD34+ cell concentration, respectively 34 . In preterm infants UCB volume is additionally lost through late cord clamping, which is a recommended standard operating procedure through several best practice committees, including the ACOG (American College of Obstetricians and Gynecologists) and the AWMF (Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften) 35 . Faster cord clamping results in the maintenance of blood flow and collection of a greater blood volume 36,37 .
Alternative Cell Sources for Extreme Preterm Infants
Studies comparing quality and quantity of preterm and term cord blood cells are lacking. An animal model of Huang et al. shows that autologous UCB term and UCB preterm cells are both effective but appear to differ in mechanisms of neuroprotection 38 . For clinical translation of cell therapies apart from effectiveness, dosing is another major issue. Recent studies have shown that for mature neonates ≥35 weeks of gestation autologous cord blood units collected through trained stuff harbor an adequate cell number for planned cellular therapy 24 . For preterm infants, on the other hand, quantity of collected autologous UCB cells is critical as shown in this study. Therefore, for extremely preterm infants with a gestational age of ≤30 weeks or a birthweight of ≤1,500 g alternative sources for perinatal cells should be considered. UCB is a rich source of primitive multipotent stem/progenitor cells 39 including mesenchymal stromal cells (MSCs). MSCs are a promising cell type with therapeutic potential for neonatal brain disorders and can be harvested not only from UCB but also from umbilical cord Wharton’s jelly or from nonperinatal sources such as bone marrow or adipose tissue 40 . Beneficial effects of MSC-based therapies have been demonstrated mainly in animal models of bronchopulmonary dysplasia but lately also in models of IVH, neonatal stroke, and HIE 41 –50 . Current phase 1 clinical trials have shown human UCB-derived MSCs to be safe in the treatment of severe IVH in preterm infants and bronchopulmonary dysplasia 51 –53 . We therefore suggest umbilical cord Wharton’s jelly as another alternative cell source for preterm infants with an insufficient quantity of UCB cells.
Role of Autologous UCB Application in Comparison to Allogenic Application
Until today banking of autologous UCB has been organized mostly through private cord blood banks. Current data show that at the time there is only a very low possibility of future use for banked autologous cord blood units collected in the absence of a known indication from a healthy infant 54,55 . A different approach is targeted collection from high-risk populations that could benefit from autologous cord blood. With 10.7% (19/177) a high proportion of the included children in this study suffered from perinatal brain damage indicating that targeted collection of UCB in these high-risk populations may be worthwhile. Because none of the children with IUGR suffered from brain lesions, we suggest that this group may be excluded for a more efficient approach in the future. Other neonatological populations that may benefit from autologous cord blood are preterm infants developing bronchopulmonary dysplasia or neonates undergoing surgical correction of congenital cardiac defects 47,56 –59 .
Autologous approaches harbor a favorable risk profile as they minimize the risk of immunogenic reactions and infections, such as graft versus host disease or even undiagnosed infectious agents (e.g., prions). The safety of an innovative treatment is of particular importance in critical patients such as newborn children. Nevertheless, collection of autologous cells is more complicated and the preparation and manufacturing of cells from individual cell sources has to become standardized to design valuable clinical trials for autologous treatment.
Allogenic approaches have a logistic advantage because cells can be manufactured and be used “off the shelf” in the clinical setting 26 . Furthermore, some UCB cells like MSCs lack MHC II antigens and are therefore immune privileged even in the allogenic setting 60,61 .
Reliable studies directly comparing efficiency and safety of allogenic and autologous treatment options are lacking. In a small case series with only seven patients with HIE, Bae et al. reported lower levels of proinflammatory cytokines and greater improvement in gross motor performance in intravenous application of allogenic UCB compared with that of autologous UCB 62 . To date, no larger study has been published. Early phase clinical studies suggest that properly administered autologous UCB is effective in the treatment of brain damage 24,25,63 .
Overall, autologous UCB units appear to be comparable to allogenic UCB units in quantity and quality of cells. While autologous options appear safer, allogenic options harbor logistic advantages. It has to be determined whether the additional expenditure and logistic challenges related to autologous approaches are reasonable. However, this decision should not depend on the effort but on efficiency of treatment.
Limitations of the Study
We classified cases to have an insufficient quality of cells if the contained number of cells was too low for at least one treatment with the target dose at birthweight (minimum 1–5 × 107 cells/kg). The target dose was based on the dose range presently used in allogeneic transplant after myeloablative chemotherapy. However, the effective dose in the autologous setting and for the treatment of brain lesions is not known and could be lower or higher. If UCB is transplanted at a later age, higher doses may be needed to be beneficial for a higher weight. The optimal dose and optimal timing for cell transplantation remain to be defined. Therefore, the clinical translation of the collected data is complex and the number of cases with sufficient quantity and quality of cells could vary from our current calculations.
The collected volume of UCB units varied widely. These differences can be explained through the variety of gestational ages and birthweights. However, UCB has been collected by different obstetricians and midwives, and the impact of individual sampling techniques was not monitored. In the future, standardized sets with a specified vacuum instead of using gravity could be developed and used to make UCB collection more comparable and possibly improve generated volumes.
Conclusion
Our data show that the autologous collection of UCB in neonates at high risk of brain damage is possible through extensive multidisciplinary collaboration required among neonatologists, the labor and delivery ward, and the cord blood bank. In the aforementioned high-risk population, the prevalence of brain damage is high with 10.7% (19/177) and may justify the effort made to obtain autologous cells from UCB or other perinatal sources from these children. The nuclear cell count and stored UCB volume correlated strongly with gestational age (P < 0.001), and the targeted dose was often not reached in very preterm infants. We therefore suggest that for preterm infants with a gestational age ≤30 + 0 weeks, umbilical cord MSCs should be investigated as an alternative cell source to UCB.
Footnotes
Acknowledgments
This study was supported by research grant from the Vita 34 AG, a cord blood bank, without which this work could not have been done. We are especially grateful to Dr Thomas Böse, Dr Josefin Zschaler, and Dr Susan Hetz for their advice and assistance. Moreover, we are grateful that Vita 34 AG offered banking of the obtained UCB units free of charge for 18 years for the included patients and their families. We thank all midwives, nurses, and the administrative staff for their work on behalf of this study. We are indebted to the patients and families for their commitment to participating in this trial.
Authors’ Contributions
AS: Manuscript writing, conception and design of the study, collection and assembly of data, information of patients, data analysis, and interpretation; TB: Administrative support, conception and design of the study, data analysis and interpretation, review of the manuscript, and final approval of manuscript; HSF: Administrative support, data analysis and interpretation, review of the manuscript, and final approval of manuscript; RD: Collection and assembly of data, information of patients, review of the manuscript, and final approval of manuscript; C-RW: Collection and assembly of data, information of patients, review of the manuscript, and final approval of manuscript; AS: Collection and assembly of data, information of patients, review of the manuscript, and final approval of manuscript; CJ: Administrative support, data analysis and interpretation, review of the manuscript, and final approval of manuscript; CB: Administrative support, conception and design of the study, provision of follow-up data, review of the manuscript, and final approval of manuscript; and WH: Conception and design of the study, review of the manuscript, and final approval of manuscript.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article. Raw data were generated at Charité University Hospital Berlin. Additional data supporting the findings of this study are available from the corresponding author AS on request.
Ethical Approval
This study was approved by the Institutional Ethics Committee of the Charité – Universitätsmedizin Berlin, Berlin, Germany (EA2/206/17).
Statement of Human and Animal Rights
The use of human umbilical cord blood and all procedures in this study were conducted in accordance with the Institutional Ethics Committee of the Charité – Universitätsmedizin Berlin (Berlin, Germany) (EA2/206/17) approved protocols.
Statement of Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The authors report funding from the Vita 34 AG, Leipzig, in form of financing a full-time position for a physician for 2 years. This position was filled by AS. The other authors indicated no potential conflicts of interest. The Vita 34 AG performed the transport and preparation of the obtained UCB free of charge. All collected data and results of this study were intellectual property of the authors and not the Vita 34 AG and the authors alone bear the responsibility for data analysis and interpretation. The corresponding author has the permission of the other authors to submit the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vita 34 AG, Leipzig, Germany, in form of financing a full-term position for one physician for 2 years.
