Abstract
Recurrent laryngeal nerve (RLN) damage is a significant and prevalent complication of thyroid surgery. Based on the beneficial role of a human amnion/chorion membrane (HACM) allograft in wound management and nerve regeneration, we investigated whether placement of a commercially available HACM allograft on dissected RLN could reduce the occurrence and/or duration of RLN injury during thyroidectomy. Among 67 patients undergoing thyroidectomy, 100 at-risk nerves (exposure of at least 3 cm of RLN) received intraoperative placement of HACM; 205 at-risk RLNs without HACM in 134 matched patients served as controls. Patient-reported vocal analysis, physician-assessed vocal analysis, and laryngoscopic assessment of vocal-fold dysfunction were performed before and after surgery. At 24 h after surgery, 17 patients in the control group (12.5%) had documented voice changes; these changes persisted for at least 3 weeks in seven patients (5%). Only one patient (1.5%) in the HACM group had vocal changes at 24 h after surgery, which resolved within 1 week (P < 0.01). Intraoperative placement of the HACM allograft over at-risk RLNs during thyroidectomy may reduce the incidence, severity, and/or duration of intraoperative RLN injury, which could address a significant complication of head and neck surgery. A larger prospectively designed clinical study is warranted to further investigate a possible benefit of the HACM allograft in thyroid surgery and to begin to understand the mechanisms through which a clinical benefit might be mediated.
Introduction
The recurrent laryngeal nerves (RLNs) innervate intrinsic muscles of the larynx that act to open, close, and adjust tension of the vocal cords. The RLNs also carry sensory information from the mucous membranes of the larynx below the lower surface of the vocal fold, as well as sensory, secretory, and motor fibers of the trachea and esophagus. These nerves often are injured during surgical procedures on the thyroid and parathyroid glands but also are at risk during other neck surgical procedures. To reduce the risk of injury to these nerves during surgery, it is conventional to first identify and dissect them so that they can be protected during subsequent surgical activities. Despite experience, diligence, and the most careful attention of the surgeon, transient or permanent injury to the RLNs occurs in 0.5% to 30% of all thyroid and parathyroid operations1–4. More extended resections, including bilateral neck dissections, carry an even greater risk of nerve injury 5 .
Inadvertent iatrogenic damage to the RLNs induces a temporary or permanent paresis or palsy of the ipsilateral vocal cord and can lead to dyspnea and problems with deglutition. Simply touching or rubbing the nerve during dissection can result in paralysis of several months and occasionally permanently. Patients who experience an intraoperative RLN injury typically experience dysphonia for a minimum of 10 to 12 weeks, with approximately 8% to 10% having a permanent injury that does not improve with even extended time 4 . Being unable to protect the trachea, these patients frequently aspirate liquids. Attempts to decrease the risk of intraoperative nerve injury have been limited to conversion to less extensive surgery or use of nerve monitoring endotracheal tubes despite the lack of peer-reviewed effectiveness data6–10. A commercially available human amnion/chorion membrane (HACM) allograft has been shown to serve as a protective barrier during wound healing. It is also known to provide multiple extracellular matrix proteins, growth factors, cytokines, and other specialty proteins that inhibit scar formation and dramatically improve injured nerve function in animal studies, and to promote healing of skin, tendons, nerves, and corneal wounds and significantly impact soft tissue repair in human clinical trials11–18. In the clinical study described herein, we began to investigate the hypothesis that intraoperative placement of an HACM allograft directly onto dissected nerves might reduce the incidence, severity, and/or duration of intraoperative RLN injury. This first study was not designed to investigate the mechanism(s) through which any protective effect might be mediated, such as a protective barrier function provided by the allograft and/or any biologic activities inherent in the allograft material, but these questions would become a significant priority if HACM placement suggested a protective effect on nerves dissected during surgery.
Materials and Methods
Study Oversight
This study was conducted at a single site (Tampa General Hospital) and carried out in accordance with the Declaration of Helsinki and the principles of Good Clinical Practice. The study protocol was approved by the WCG IRB Connexus (1-12119531-1). All patients provided written informed consent for the collection, analysis, and publication of their anonymized outcome data.
Study Design and Patients
This single arm study with a historic control group was designed to begin to investigate the idea that intraoperative placement of an HACM allograft on dissected RLNs might safely lessen or prevent intraoperative nerve damage during thyroidectomy. As a hypothesis-generating study, the sample size of 67 patients with 100 at-risk nerves was based on surgical volume over a 2-month period at a single surgical center. To establish a homogeneous background for evaluation of the HACM allograft, the patient population was those undergoing planned nerve-sparing thyroidectomy and who consented to intraoperative placement of the HACM allograft. An at-risk nerve was defined as dissection and exposure of at least 3 cm of RLN.
A control group was established from patients who had undergone nerve-sparing thyroidectomy and also had one or both RLNs at risk in the months immediately preceding conduct of the HACM arm. The intention was to match the HACM group at a 2:1 ratio (134 patients with at least 200 at-risk nerves), with matching on the basis of gender, age, thyroid pathology, central-compartment gross lymph node involvement, and extent of RLN dissection in thyroidectomy.
Normal preoperative vocal cord function as assessed by laryngoscopy was required in both the control and HACM groups. Anatomic variations of the nerve did not impact patient eligibility.
To minimize variability attributable to surgical experience and technique, all surgical procedures and follow-up care, including those in both the active and historic control arms, were conducted in a standardized fashion among two high-volume surgeons (G.L.C., R.R.) at a single surgical center over a brief 6-month period. Patient care in the surgical center is provided exclusively by specialized thyroid surgical teams, minimizing variability in surgical procedures and postprocedure care.
Surgical Procedures
Because RLN injury can be inversely correlated with the experience of the surgeon, only two surgeons whose practice is limited to thyroid surgery participated in this trial. The caseload in the control and HACM groups was equally shared between both surgeons. Furthermore, both surgeons take care to perform essentially identical operations and typically operate together in the vast majority of patients, thereby assuring that their surgical procedures and approaches to recurrent and superior laryngeal nerve identification, dissection, and preservation are standardized between them.
Patients were intubated with a #7 nerve monitoring endotracheal tube (Medtronic, Minneapolis, MN). A non-narcotic approach to anesthesia is standard, with scopolamine patch and gabapentin pretreatment prior to anesthesia, which is induced with intravenous lidocaine, followed by propofol then succinylcholine (unless a history or family history of malignant hyperthermia is identified). Intubation is always visualized with a GlideScope monitor. Patients undergoing comprehensive lateral neck dissection received further paralytic agents. All paralytic agents were reversed prior to thyroid and central-compartment surgery. All patients in both groups had normal nerve stimulation at 1 milliamp (mA) setting at the completion of the operation.
During surgery, the external branch of the superior laryngeal nerve (EBSLN) and all branches of the recurrent (or nonrecurrent) nerves were identified for sparing; patients with gross malignant invasion that prevented nerve sparing were not enrolled. External branches were identified during the initial mobilization of the thyroid upper poles, and vascular ligation did not occur until the external branch was identified. Upper poles were mobilized and Joll’s triangle opened prior to recurrent or nonrecurrent nerve identification. A standardized inferior approach was taken to identify the nerve on the right and within the immediate subfascial plane from a superior approach on the left. No incidences of nonrecurrent nerve were identified in this study.
An inferior approach was taken to identify the right RLN and the more rare nonrecurrent nerve; if not identified within 60 s, the more approachable inferior-most or most cephalad approach was pursued as an alternative. On the left, the RLN was identified in the immediate subfascial plane from a superior approach unless a mass preventing this identification, in which case a more inferior approach was pursued.
Once the laryngeal nerves were identified, nerve dissections were performed in the avascular plane of the fascia overlying the perineurium. All nervous branches were spared with the exception of minimal sensory branches, which may occur medial to the laryngeal nerves and were visualized entering the trachea subperichondral plane. Electrocautery is never used within 1 cm of any identified RLN. Bipolar electrocautery on the 8 to 10 milliamp setting and sharp dissection is only utilized in nerve microdissections. Nerves were not retracted but could be mobilized by stepwise sharp microscissor management of fascia and microvasculature of the nerves, followed by soft cottonoid “rolling” of the nerve. All nerves were inspected and tested for response to 1 milliamp nerve stimulation prior to wound closure. Meticulous hemostasis was obtained and observed. If a drain was utilized, it was placed just prior to closure and oriented as far from the nerve as possible.
The EBSLN was routinely identified, preserved, and tested for stimulation in every operation. Although EBSLN dysfunction can produce vocal changes, amniotic membrane allograft placement on these nerves was not a focus of this study.
HACM Allograft and Placement
The HACM allograft (AmnioFix; MiMedx Corp, Marietta, GA, USA) is a commercially available tissue matrix allograft composed of dehydrated HACM that acts as a physical, biologically active barrier and provides a protective environment to help promote healing 11 . AmnioFix HACM is provided in 3 cm × 3 cm sheets and is on formulary at the surgical center where the study was performed. The HACM allograft was cut to a shape to cover the dissected nerve, which has a gentle curve in its upper aspect and branches in its most superior aspect. The nerve was allowed to lay in its normal, curved anatomic position even if it had been displaced by the thyroid gland or central-compartment lymph nodes. The thyroid cartilage was digitally pulled cephalad and to the contralateral side to expose the greatest surface area of the nerve. The membrane allograft was held with two vascular forceps and the HACM allograft applied onto the entire length of the dissected nerve (Fig. 1, left panel).
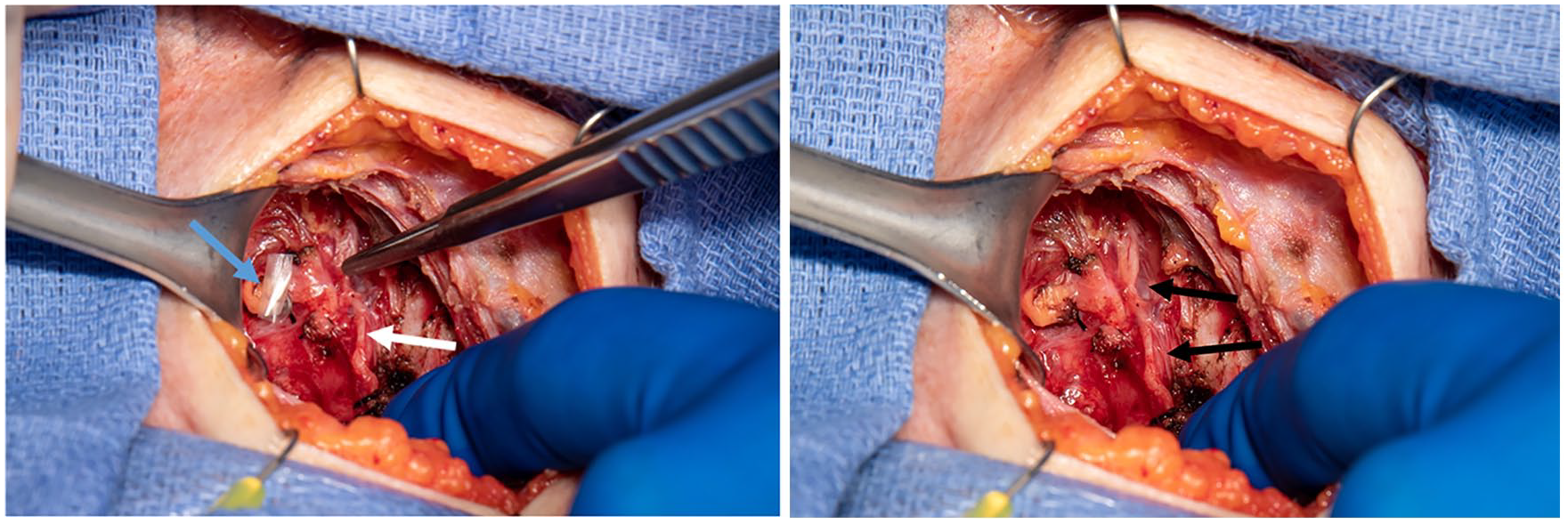
Left panel: Preparation for placement of the HACM allograft during thyroid surgery. The white arrow indicates the recurrent laryngeal nerve that has been dissected prior to thyroidectomy. The forceps on the right holds the top portion of the (nearly transparent) HACM allograft, which is being laid upon the nerve and has been pre-cut to 1 cm × 3 cm to accommodate this particular dissection. The blue arrow points to an additional smaller section of HACM, which will be draped over the preserved parathyroid gland. Right panel: Placement of the HACM allograft. Black arrows indicate the HACM allograft, which appears as a transparent blanket draped over the dissected recurrent laryngeal nerve. HACM: human amnion/chorion membrane.
The allograft was placed as soon as possible following nerve dissection and verification of effective hemostasis. A cottonoid was used to contour the graft over the exposed surface of the nerve. Placement of the allograft takes only seconds, and it is left to cover the nerve as a protective blanket as the remainder of the operation is completed in typical fashion. Once placed (Fig. 1, right panel), efforts were made to manipulate the allograft as little as possible. The allograft is semi-transparent so the nerve can still be seen and thus avoided as usual. The overlying membrane prevents desiccation of the nerve and also prevents direct contact of the nerve by surgical instruments, sponges, and suction as the operation continues. The allograft is left in place as an intimate covering over the nerve at the time of wound closure, providing a protective layer to prevent the overlying muscle from rubbing or otherwise causing irritation to an exposed nerve in the postoperative period and possibly preventing scar tissue from forming directly on the nerve. It was also envisioned that the allograft would provide the potential active biological benefits associated with HACM, including release of multiple extracellular matrix proteins, growth factors, interleukins and cytokines, as well as tissue inhibitors of metalloproteinases (TIMPs); the release of these factors from the HACM has been previously observed and reported 11 but was not directly investigated in this hypothesis-generating study.
Assessment of Clinical Benefit and Safety
All patients in the control and HACM groups were evaluated by laryngoscopy preoperatively to establish normal nerve function as a condition of inclusion and to serve as a baseline assessment. During surgery, nerve function was intermittently monitored via the Medtronic NIM™ nerve monitoring system. All patients were stimulated at 0.9 mA to verify stimulation integrity. When the patients have completely emerged from anesthesia and are prepared for discharge from the postanesthetic care unit, they provide a self-assessment of vocal changes. Any patient reporting any subjective change in voice underwent laryngoscopy at that time. Otherwise, all patients had documentation of vocal cord function via endoscopy within 7 days of surgery (typically within 48 h). Fiberoptic laryngoscopy was performed to assess function of the superior laryngeal nerve and RLN. In addition, anatomic and muscular function was directly analyzed. Throughout the surgical follow-up period and at recovery, laryngoscopy was performed by a single physician examiner (G.L.C.) to minimize variability.
A validated instrument of vocal quality (Voice Handicap Index) was utilized to allow patients to self-report their quality of voice presurgery and postsurgery. This assessment was obtained from every patient prior to surgery, the evening of their operation, the following day, at 1- and 3-week timepoints after surgery, and until resolution of any subjective or objective voice changes.
Safety of intraoperative placement and ongoing residence of the allograft on the dissected laryngeal nerves was further assessed by monitoring signs and symptoms of reactions during the postoperative recovery period. Wound reaction was graded based on an inflammatory wound scale and was assessed by physical inspection and photographic documentation at day 1 and at 1 and 3 weeks postoperatively.
Statistical Considerations
A sample size of 100 at-risk nerves was identified for the active arm based on the number of patients undergoing thyroidectomy by the study surgeons (G.L.C., R.R.) in a brief (2-month) period at the surgical center. The decision was taken to generate a twofold larger historic control group to provide a firm basis against which to assess safety and efficacy of the allograft; with 67 patients in the HACM allograft group, a matching control group of 134 patients was curated which included 205 at-risk nerves, helping to confirm the similarities of the surgical procedures in both groups. Because there was no prior use of the HACM allograft in this setting, the sample size was not statistically derived for this hypothesis-generating study but simply reflected the number of patients who underwent planned thyroidectomy during a 2-month period and met the prespecified criteria of at least 3 cm of RLN dissected from surrounding tissues. A two-tailed Fisher’s exact test was used to analyze the percentage of patients with RLN injury, assessed as subjective or objective voice changes, at each observation timepoint. Observations in this first study may serve as a baseline for statistical planning for a subsequent larger study.
Results
Demographics
The study group was comprised of 100 consecutive at-risk nerves within 67 patients (Table 1) who underwent thyroidectomy in June or July 2019 and met the criteria consistent with a comprehensive nerve dissection, defined as at least 3 cm of nerve dissected from surrounding tissues. Approximately 50% of patients (n = 34) had two at-risk nerves and 50% had only a single at-risk nerve. All 100 dissected RLNs were overlaid with the HACM allograft.
Demographics and Characteristics of Patients.

NIFTP: noninvasive follicular thyroid neoplasm with papillary-like nuclear features.
The control group consisted of 134 patients who were matched for age, extent of operation, pathologic diagnosis, presence of central-compartment lymph node metastases, and soft tissue extension (Table 1) who underwent thyroidectomy (February–May 2019) at the same center with the same two surgeons. In this group, 205 nerves met the criteria consistent with a comprehensive dissection; 51% (n = 68) of patients had two at-risk nerves, and 49% (n = 66) of patients had only a single at-risk nerve.
The median age and age range was evenly distributed across the HACM allograft and historic control groups. In addition, the fraction of patients with thyroid malignancy, central-compartment lymph node metastases, and soft tissue extension in the central compartment as the basis for requiring surgery was not significantly different between groups, demonstrating an even distribution for the extent and complexity of RLN dissection between the groups. The HACM allograft group had a higher percentage of patients with gross disease in the central neck compartment and a lower percentage of patients with disease isolated to the thyroid gland. Although this trend was not statistically significant, it is anticipated that the more extensive malignancy in the HACM group would be associated with a higher rate of RLN injury, and therefore, further attempts to balance this criterion were not made.
Safety
There was no effect of HACM allograft placement on respiratory ability and no patient in either arm experienced wound infection or any effect on swallowing, or required any assistance with swallowing function. Safety of the HACM allograft was supported by the absence of objective or subjective evidence of adverse events other than vocal change in a single patient and normal postoperative pain, which was expected in this study. Anecdotally, patients in the HACM allograft group had fewer complaints of pain following surgery, but this was not a measured endpoint and will be assessed in a future study.
Clinical Outcome
Patients were monitored for all possible manifestations of RLN injury, including dysphonia, aphonia, difficulty breathing, and difficulty swallowing. The degree of vocal changes ranged from self-assessed vocal change to overt hoarseness. There were no patients with aphonia or respiratory compromise or aspiration in this series. All postoperative findings in both groups were limited to vocal changes which were scored based on the subjective patient-reported vocal analysis scale, physician assessment of vocal changes, and laryngoscopic assessment of objective vocal-fold dysfunction. Results of this assessment are presented in Fig. 2. In the first 24 h after surgery in the control group, 17 patients (12.5%) encompassing 17 nerves at risk reported subjective changes of voice. At the 1-week assessment, nine patients (7%) reported persistent vocal changes, and seven patients (5%) continued to report vocal changes at the 3-week assessment. All patients recovered within 3 months after surgery, which was confirmed by fiberoptic examination.

Incidence and persistence of patient and/or physician-reported vocal dysfunction after thyroidectomy with recurrent laryngeal nerve dissection.
Among the 67 patients in the HACM allograft arm, a single patient with a single dissected nerve experienced a vocal change at day 1 after surgery (P < 0.01, Fisher’s exact test two-sided). This dysfunction was fully and permanently resolved at the 1-week timepoint and documented subjectively, objectively, and by laryngoscopy.
Discussion
The prevalence of laryngeal nerve injury during head and neck surgery despite the best preventive efforts is remarkable, with permanent nerve injury documented in 0.5% to 5% of patients and transient injury in 1% to 30% of patients, with even higher rates in more extensive procedures1–5. Patients with resultant impairment in phonation report feelings of seclusion, frustration, anxiety, fear and depression, diminished social interactions, and ability to fully participate in their work environments19,20. Associated morbidities of dyspnea and deglutition can lead to malnutrition, weight loss, dehydration, choking, and aspiration pneumonia.
Well-differentiated thyroid carcinoma is the indication for 50% of thyroid surgeries performed in the United States. Whether due to improved diagnostics or an actual increase in incidence, thyroid carcinoma incidence has quadrupled in the past four decades 21 . The small decrease in incidence in the most recently reported 5-year period (2013–2017) is likely attributed to a change in biopsy guidelines for nodules <1 cm in the absence of certain high-risk characteristics22,23. However, incidence continues to increase among adolescents and young adults, with increasing trends for large and small tumors 24 . Changes in prevalence of several risk factors, including low-dose radiation exposure 25 and excess body weight 26 , may contribute to increasing incidence rates in more recent birth cohorts and it is not clear what impact the increase in incidence in children, adolescents, and young adults will have in future.
The prevalence of thyroid nodules and the malignant potential of nodules increases with age, with 50% of patients over 65 years of age having nodules on ultrasound examination27–29. As improvements in health care have increased life span, we anticipate that the number of thyroid surgeries will continue to increase. Without better methods for prevention of intraoperative injury to the RLNs, we can only expect more patients to be impacted.
No procedures, techniques, or medical adjuncts developed to date have reduced the incidence or duration of intraoperative laryngeal nerve damage. Recent evidence suggests that intraoperative neural monitoring can be used to adapt surgical strategy in attempt to prevent vocal cord palsy 30 and guidelines have been published to provide guidance to the surgeon31,32, but even this is not entirely effective at preventing nerve damage that occurs during thyroid surgery. With the medical, psychological, and psychosocial impact of laryngeal nerve injury, the medical community is faced with a highly impactful surgical complication that can be highly prevalent. A number of animal studies suggest that an HACM allograft can prevent the occurrence of perineural adhesions, reduce fibrosis, accelerate nerve repair, and improve nerve function following injury22–24. Aside from the barrier function offered by the allograft, presumed improvement in the injury microenvironment, and prevention of adhesions during healing, these studies have demonstrated upregulation of multiple neurotrophic factors associated with axonal regeneration, leading to improved nerve conduction velocity and amplitude, often in dramatic fashion. Thus, we hypothesized that application of a commercially available allogeneic allograft derived from HACM might reduce the incidence, severity, and/or duration of RLN injury, assessed as vocal cord dysfunction.
The human amniotic membrane is comprised of two distinct tissues, the amnion and the chorion. Grafts derived from the amnion and chorion membrane have been used medically for over a century with the first uses in skin transplantation and with burned and ulcerated skin33,34. When used in patients with burns, the membrane provides significant pain relief owing to adhesion to the wound surface and coverage of dermal nerve endings. It also prevents wound surface drying, which accelerates wound healing, lending itself to management of wounds in the oral cavity35,36. More recent applications of amnion membrane allografts include chemical or thermal burns, ulcers, traumatic wounds, surgical incisions, and ophthalmic conditions 37 . With the emergence of new preservation techniques, they have become more widely used in the medical community17,38,39.
Physiologically, the membrane provides a barrier function but is also metabolically active, with its metabolic activities mediated through growth factors, cytokines, and chemokines that are produced by cells in the membrane and can facilitate healing38,40–42. Amniotic membrane tissue also is rich in mesenchymal stem cells and epithelial cells which promote nerve, axon, and myelin regeneration 43 . The paired amnion/chorion membrane allograft used in this study provides a more robust supply of growth factors and cytokines than single layer amnion grafts; possesses antibacterial, antiviral, and anti-inflammatory effects; and is nontumorigenic with low immunogenicity.
While the specific mechanism through which HACM may afford protection to the RLNs cannot be determined in this study, based upon the known properties of HACM we hypothesized that the membrane may lessen insult and damage to the nerves in two distinct manners—a physical barrier to trauma and direct application of active biological proteins. During the surgical procedure, the graft serves as a protective blanket keeping stray instruments and sponges away, preventing desiccation, and possibly even diffusing any electrical current away from the nerve. In the postoperative setting, the membrane continues to protect the nerve from irritation from any overlying muscle which could rub on or move across its surface as the phases of healing are taking place. It is likely that the decrease in vocal cord dysfunction seen in the initial 24-h period in patients receiving HACM is primarily due to this physical protective phenomenon rather than the actions of cytokines and other active proteins.
Although the active biological effects of HACM have been well studied for its ability to alter wound healing, preventing scar formation, and improve injured nerve regeneration, it is not clear what role this is playing in these patients. It would seem likely that the biological benefits (if any) HACM provides may begin during surgery, but the effects of these growth factors and active proteins are likely primarily active in the hours and days following the operation which may facilitate a more rapid repair of any intraoperative or postoperative injuries that occur. Possibly this explains the rapid 1-week recovery of a dysfunctional nerve in one HACM patient as has been shown previously in animal models. Additional studies will be required to glean a detailed understanding of the mechanism through which the allograft affects the occurrence and duration of intraoperative RLN injury.
Among 67 patients representing 100 dissected laryngeal nerves on which the HACM allograft was applied, only a single patient (1.5%) with a single at-risk nerve (1%) reported voice disturbance 24 h after thyroidectomy, which was fully recovered within 1 week. In contrast, in a historic control group of 134 patients with 205 dissected laryngeal nerves not exposed to the allograft membrane, 17 patients (13%) with a total of 17 dissected laryngeal nerves suffered a voice disturbance 24 h after surgery; in nine patients (7%), the voice disturbance persisted for 1 week and seven patients (5%) experienced continued dysphonia at 3 weeks after surgery. The reduction in occurrence of dysphonia at 24 h after surgery was statistically significant (P < 0.01, Fisher’s exact test, two-tailed) but less significant at the 2- (P < 0.05) and 3-week (P = 0.099) timepoints due to the small sample size. The persistence of dysphonia is not likely attributed to surgical experience, technique, or postsurgical care as the same high-volume surgeons and surgical care team performed all procedures at the same institution in a very brief time frame for both cohorts of patients.
The present study was limited to patients undergoing thyroidectomy with at least 3 cm of dissected RLN. This requirement was based on the study authors’ consensus that 3 cm of dissected nerve was sufficient to define an “at-risk” nerve. It is not envisioned that the beneficial effect of HACM allograft placement will be limited to such extensively dissected nerves, and subsequent clinical studies will evaluate the impact of allograft placement when dissection of less than 3 cm of nerve is undertaken.
Although no patients in this study suffered permanent RLN injury, it would be inappropriate to diminish the impact of temporary recurrent nerve paralysis which seriously affects both patients and care providers. It well known by surgeons operating in the neck that even transient RLN injury has a tremendous impact on patient’s physiological and physical well-being. Inability to return to work, social isolation, fear, aspiration, and vocal rehabilitation are just a few things that happen commonly in these patients. Even short-term vocal dysfunction can have profound psychosocial impact on all involved. The ability to significantly impact temporary vocal cord dysfunction—and not just permanent paralysis—following thyroid surgery can have profound benefits for both patients and surgeons alike. When considering that transient or permanent injury to the RLNs occurs in up to 30% of all thyroid and parathyroid operations and approximately 8% to 10% of patients have a permanent injury that does not improve with even extended time1–4, it will be important to evaluate the HACM allograft in a setting that involves more investigators with variable experience and differences in surgical technique.
For patients who experience nerve damage during head and neck surgery, a significant reduction in the occurrence and duration of intraoperative RLN dysfunction may be a highly impactful advance in thyroid and other forms of head and neck surgery. To validate the results observed in this first study, a randomized multicenter study to further characterize the efficacy and safety of the HACM allograft in this setting is planned.
Conclusion
Transient or permanent injury to the RLNs occurs in up to 30% of all thyroid and parathyroid operations, and approximately 8% to 10% of patients have a permanent injury that does not improve over time. This type of injury, including even short-term vocal dysfunction, has a tremendous impact on patients’ physical and psychosocial well-being; this highly important and prevalent complication of thyroid surgery presents a significant unmet medical need. Data from this small study suggest that placement of a commercially available HACM allograft on RLNs dissected during thyroid surgery may lessen the incidence and/or duration of intraoperative nerve injury. Further investigations will be required to confirm this observation and investigate the mechanism by which any benefit of the HACM allograft is mediated.
Footnotes
Author Contributions
Dr. Gary L. Clayman served as principal investigator and primary author. Dr. Rashmi Roy served as co-investigator and author. Dr. James Norman served as co-investigator and author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Thyroid Cancer and Parathyroid Foundation, Tampa, Florida, USA.
Ethical Approval
Ethical approval to conduct and report this study was obtained from WCG IRB Connexus (approval number/ID #1-1219531-1).
Statement of Human Rights
All procedures in this study were conducted in accordance with the WCG IRB Connexus approved protocol (approval number/ID #1-1219531-1).
Statement of Informed Consent
Written informed consent was obtained from the patient(s) for their participation in the study and for their anonymized information to be reported.
