Abstract
The inflammatory response is an obstacle to success in both allogeneic and autologous islet transplantation. In autologous islet transplantation (AIT), however, the recipient is also the donor, permitting pretreatment of donor/recipient for a controlled duration prior to transplantation. We sought to exploit this feature of (AIT) by pretreating donor/recipients with chronic pancreatitis undergoing total pancreatectomy and autologous islet transplantation (TPAIT) to test the hypothesis that peri-transplant treatment with the FDA-approved anti-inflammatory hydroxychloroquine (HCQ) improves graft function. In this randomized placebo-controlled pilot clinical study, patients (n = 6) were treated with oral HCQ for 30 days prior to and 90 days after TPAIT. In vivo islet function was assessed via Mixed Meal Tolerance Testing before HCQ treatment, 6- and 12-months after surgery. In vitro islet bioenergetics were assessed at the time of transplantation via extracellular flux analysis of islet preparation samples from the clinical trial cohort and six additional patients (n = 12). Our study shows that HCQ did not alter clinical endpoints, but HCQ-treated patients showed greater spare respiratory capacity (SRC) compared to samples from control patients (P=0.028). Glycolytic metabolism of islet preparations directly correlated with stimulated C-peptide secretion both before and after TPAIT (P=0.01, R 2=0.489 and P=0.03, R 2=0.674, respectively), and predicted in vivo islet function better than mitochondrial metabolism of islet preps or islet equivalents infused. Overnight culture of islet preparations altered bioenergetic function, significantly decreasing SRC and maximal respiration (P<0.001). In conclusion, while HCQ did not alter clinical outcomes, it was associated with significantly increased SRC in islet preparations. Bioenergetic analyses of islet preparations suggests that culture should be avoided and that glycolysis may be a more sensitive indicator of in vivo islet function than current metrics, including islet oxygen consumption and islet equivalents infused.
Introduction
Total pancreatectomy (TP) is used for pain management in patients with chronic pancreatitis (CP) intractable to medical therapy and lacking other surgical options. Ideally, the procedure is coupled with auto-transplantation of islets of Langerhans from the resected pancreas in order to prevent or mitigate the effects of surgically-induced diabetes 1 . Although autologous islet transplants (AIT) are not subject to recurrence of autoimmunity or initiation of alloimmunity that are common to allogeneic islet transplantation used to treat type 1 diabetes 2 , both procedures lead to nonspecific inflammation-mediated damage to the islet graft. This nonspecific inflammation presents a major obstacle to success in both autologous and allogeneic clinical islet transplantation 3,4 , with estimates suggesting that up to 60% of transplanted islets are destroyed or fail to engraft 5,6 . Drugs that reduce clotting or block the action of pro-inflammatory mediators like CXCL10, MCP-1, CXCR1/2, and TNFα have been associated with improved graft function 7 –10 and TNFα blockers are commonly utilized as a peri-transplant anti-inflammatory in allogeneic islet transplantation 11 . Thus, peri-transplant anti-inflammatory treatment may protect islets from the nonspecific inflammatory challenges that occur during transplantation, resulting in preservation of β-cell function and sustained long-term insulin independence 12,13 .
Hydroxychloroquine (HCQ) is an FDA-approved anti-inflammatory commonly used to treat autoimmune diseases. Treatment with HCQ leads to cell type-dependent immunomodulatory effects including inhibition of pro-inflammatory cytokine production 14 , impaired antigen presentation 15 , decreased T cell activation 16 , and interruption of toll-like receptor signaling 17 , in addition to reducing platelet aggregation 18 . Furthermore, unlike many immunosuppressive drugs, HCQ is associated with enhanced glycemic control. Patients treated with HCQ for rheumatic diseases are less likely to develop type 2 diabetes 19 .
Although nonspecific inflammation is a major obstacle in both allogeneic and autologous islet transplantation, it is difficult to study the effect of peri-transplant anti-inflammatories on recipients of allogeneic islet transplants due to the short and unpredictable timelines involved with organ donations. It is currently not possible to study the effect of anti-inflammatories on allogeneic islet donors as islets are typically obtained from the pancreas of a deceased organ donor. In AIT, however, the recipient is also the donor and surgeries can be scheduled well in advance, making it possible to pre-treat both donor and recipient in order to minimize inflammation. We took advantage of the ability to pretreat donor/recipient for a controlled duration prior to transplantation to test the hypothesis that peri-transplant treatment with HCQ improves metabolic outcomes in patients undergoing TPAIT. In a randomized placebo-controlled pilot clinical study, TPAIT patients (n = 6) were pre-treated with HCQ for 30 days prior to and 90 days after surgery. We also measured bioenergetics in samples of the islet preparation taken at the time of transplantation from the study cohort and six additional patients. We chose to explore bioenergetics of islet preparations because, although TPAIT outcome depends on the volume of β cells transplanted, with 5 x 103 islet equivalents (IEQ)/kg generally accepted as the cut-off for consistent autologous transplant success, IEQ transplanted incompletely explains the variance associated with transplant outcome 20,21 .
Materials and Methods
Study Design
This 12-month, phase I/II randomized, double-blinded, placebo-controlled pilot trial of individuals undergoing TPAIT was approved by the Cleveland Clinic Institutional Review Board and registered with ClinicalTrials.gov (NCT03283566). Anticipated sample size (n = 10) was determined a priori using G*Power 3.1.9.7 22 for one-sided t-tests with α=0.05 and equal variance based on our group’s unpublished historical TPAIT data. We calculated that a total sample size of 10 subjects would allow us to detect a large effect size (d = 1.7) with 70% power. The intervention and control arms consisted of HCQ and placebo treatment, respectively, with equal randomization (1:1 allocation ratio). In the HCQ arm, subjects received 200 mg oral HCQ once daily for 30 days prior to scheduled TPAIT and for 90 days post-surgery. In the placebo arm, patients received placebo treatment following the same schedule as the HCQ arm. The study was double-blind, and randomization was carried out by the Cleveland Clinic Pharmacy using an alphanumeric identifier. The PI, surgeon, patients, caregivers, and researchers were all blinded to the study arms and only the research coordinator and pharmacy were un-blinded. The primary clinical endpoint was the quotient of C-peptide/glucose 90 minutes after Mixed Meal Tolerance Testing (MMTT) and secondary endpoints were stimulated C-peptide area under the curve (AUC) and the ratio of C-peptide: glucose AUC in response to MMTT, all normalized to IEQ/kg infused, at 12 months post-TPAIT.
Study Subjects
All subjects undergoing TPAIT for CP at the Cleveland Clinic were candidates for the study, and written informed consent was obtained. Inclusion criteria included clinically confirmed diagnosis of CP, recurrent episodes of acute pancreatitis resulting in intractable abdominal pain, chronic narcotic use, a history of failed operation(s) for CP, and a hemoglobin A1c (HbA1c) less than 8.0% pre-TPAIT. Exclusion criteria were pre-TPAIT insulin dependence, evidence of pancreatic mass or carcinoma, cirrhosis, portal hypertension and current alcohol abuse. Recruitment for this pilot clinical trial was ended in February 2020 due to the onset of the Sars-CoV-2 pandemic. Surgeries and follow-up visits were conducted at the Cleveland Clinic from December 2017-May 2018 and December 2018-May 2019, respectively, although attempts to follow up with patients continued through June 2020.
A total of nine patients were enrolled into the trial. Four were randomized into the HCQ arm with one lost to follow-up and five were randomized into the placebo arm with two lost to follow-up and one not receiving placebo treatment due to unanticipated surgery scheduling. Our final clinical analyses include a total of six subjects (n = 3 for each arm). Our in vitro studies contain data from the clinical trial subjects and six additional patients who either underwent randomization and clinical treatment but failed to complete follow-up clinical analysis or did not consent to be randomized into the trial, but consented for all other research, for a total n = 12 (4 HCQ vs 8 placebo or untreated).
Total Pancreatectomy, Islet Isolation and Transplantation
TPAIT was performed at the Cleveland Clinic (Cleveland, OH, USA). Pylorus-preserving TP with duodenectomy was performed with splenectomy to preserve perfusion to body and tail. The pancreas and duodenum were then removed intact and flushed/drained with UW solution via the splenic artery and vein, respectively. The organ was transported to the Islet Isolation Laboratory of the Institute of Cellular Therapeutics at Allegheny General Hospital (Pittsburgh, PA, USA) where islet isolation was performed as previously described. 23 Islet volume was calculated as IEQ and the preparation was transported back to the Cleveland Clinic where it was transplanted via the portal vein as previously described 23 .
Clinical Assays
Clinical assays were conducted at the Cleveland Clinic. Twelve subjects underwent MMTT to assess islet cell function before treatment with HCQ or before transplant, if untreated. Trial subjects and two additional patients also underwent MMTT at 6 and 12 months following TPAIT. After overnight fast, subjects consumed a nutritionally balanced Complete Ensure Plus® (350Kcal: 13 g protein, 11 g fat, 51 g carbohydrate). Blood was drawn at baseline and 30, 60, 90, 120, 180, and 240 min post-consumption. Plasma glucose was measured using the enzymatic reference method with hexokinase (Roche/Hitachi Cobas c 311/501 analyzers, Roche Diagnostics, Indianapolis, IN); intra-assay coefficients of variance (CV) ranged from 0.7-1.0%. Plasma insulin and C-peptide were measured using sandwich chemiluminescent immunoassays (ADVIA Centur, Siemens AG, Erlangen, Germany), with intra-assay CVs of 5.3-9.9% for insulin and 1.0-3.3% for C-peptide. HbA1c was measured using turbidimetric inhibition immunoassays (Cobas Integra 400/400 plus 800 analyzers, Roche Diagnostics), with intra-assay CVs of 0.8-0.9%. BETA-2 score was calculated from fasting blood samples according to Forbes et al 24 .
Extracellular Flux
In vitro assays were conducted at the Cleveland Clinic on islet preparation samples from 12 patients. In vitro samples were transported from Pittsburgh, PA to Cleveland, OH with the islets destined for clinical transplantation and arrived between 10:00 pm and midnight on the surgery date. Samples were suspended in 50 mL CMRL-1066 transplant media (Mediatech Inc., Manassas, VA) supplemented with antibiotics, 2 mM L-glutamine, and 0.5% human serum albumin. They were centrifuged at 220 g for 3 min (4°C), washed once with 10 mL islet culture media (Corning CMRL-1066 media containing 5.5 mM glucose, supplemented with 10% FBS, 10 mM HEPES, 20 μg/mL ciprofloxacin, and 7 mM L-glutamine), split between two non-tissue culture treated petri dishes, and cultured overnight (12-18 hr) at 37°C and 5% CO2 in 20 mL islet culture media.
Extracellular Flux data were generated on a Seahorse XFe24 metabolic analyzer (Agilent Technologies, Santa Clara, CA). Islet preparation samples were washed twice in Seahorse islet media (Seahorse XF Assay Medium containing Glutamax, Agilent #102365-100, pH 7.4, 0.22 µm-filtered, supplemented with 3 mM glucose and 1% FBS), centrifuged for 3 min at 220 g (4°C), and resuspended in 30-60 mL Seahorse islet media, depending on the pellet size. 500 µL sample suspension was added to each well of a Seahorse XF24 islet capture microplate (Agilent) to obtain approximately 1,000 islet preparation tissue aggregates (including islets and acinar tissue) per well. Aggregates were collected in the central depression within each well and trapped using the islet capture microplate screens according to the manufacturer protocol. The cell plate was then placed in a non-CO2 incubator at 37°C for up to an hour before assay. To measure islet preparation bioenergetics, the cell plate was equilibrated in the XF24e and 3 basal measurements were followed by sequential injections of glucose (20 mM), oligomycin (5 µM), carbonyl cyanide 4-(trifluoromethoxy)phenylhydrazone (FCCP; 1 µM), and a combination of rotenone (5 µM) and antimycin A (5 µM), all from MilliporeSigma (St. Louis, MO, USA). Oxygen consumption rate (OCR) and extracellular acidification rate (ECAR) were recorded following each injection until the rate stabilized. Both OCR and ECAR were normalized to the final basal measurement at 3 mM glucose before the injection of 20 mM glucose to account for differences in sample viability and/or quantity of aggregates both within and between samples. Standard metrics of mitochondrial bioenergetics including respiration, ATP-linked respiration, maximal respiration, spare respiratory capacity, proton leak, and non-mitochondrial respiration were calculated according to Kramer et al 25 . as illustrated in Fig. 1. Since we did not include an inhibitor of glycolysis such as 2-deoxyglucose (2-DG) in all assays, we assessed glycolytic metabolism using the ratio of glucose-stimulated ECAR to glucose-stimulated OCR to control for non-glycolytic acidification from mitochondrial metabolism.
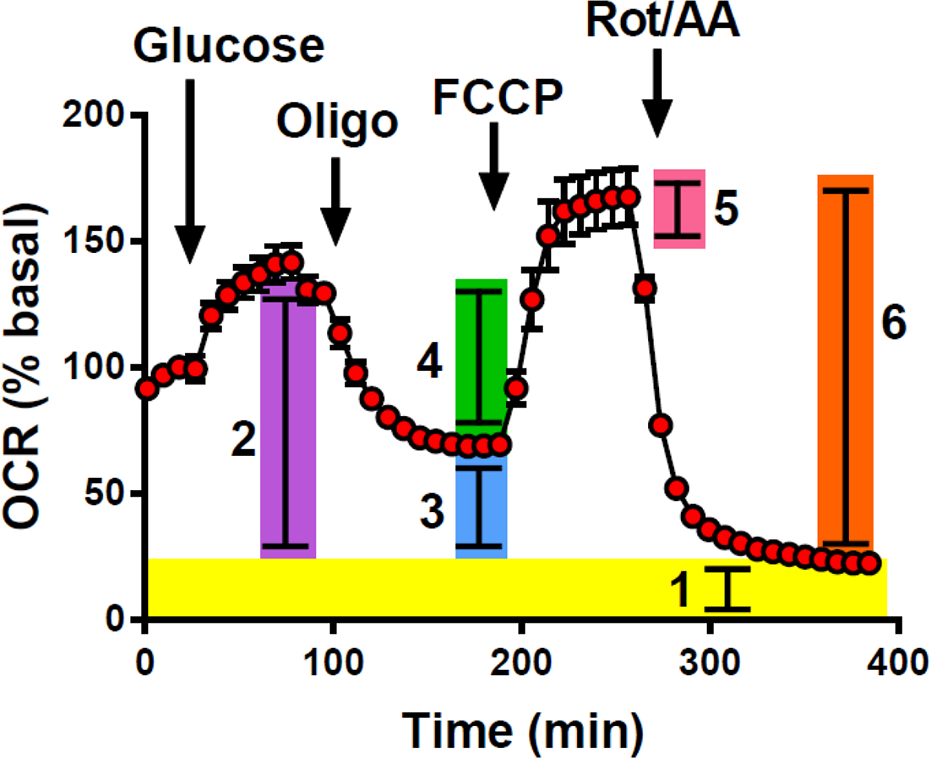
Statistical Analysis
Continuous data are presented as mean ± standard deviation (SD), mean ± standard error of the mean (SEM), or median (interquartile range), as specified. Categorical data are summarized as counts and percentages (n, %). Stimulated glucose, insulin, and C-peptide were computed by calculating the incremental area under the curve (iAUC) during MMTT using GraphPad Prism 6 (GraphPad Software Inc., California). If necessary, values below the detection limit were substituted with half the assay detection limit. Correlation analyses were conducted using Pearson or Spearman correlation tests, depending on whether or not the data distribution violated the assumption of normality tested via D’Agostino & Pearson omnibus normality test. For correlation analyses, metrics of islet preparation bioenergetics were normalized to patient weight, as the ability of a graft to maintain glycemic control depends on patient mass. For comparisons of islet bioenergetics between HCQ and placebo-treated patients, islet bioenergetics were normalized to IEQ in order to remove β-cell volume as a variable and, since the sample size for HCQ treatment was too small to test for normality, we used nonparametric Mann-Whitney tests. The clinical effect of HCQ and the effect of overnight culture on bioenergetics of islet preparations were analyzed using two-way repeated measures ANOVAs followed by Sidak’s multiple comparisons tests. All other demographic and clinical variables were compared using Fisher’s permutation test, which is distribution-free. All analyses were two-tailed with α=0.05 using GraphPad Prism 6. A priori and post-hoc power analyses were conducted using G*Power 3.1.9.7 (Universität Düsseldorf, Germany).
Results
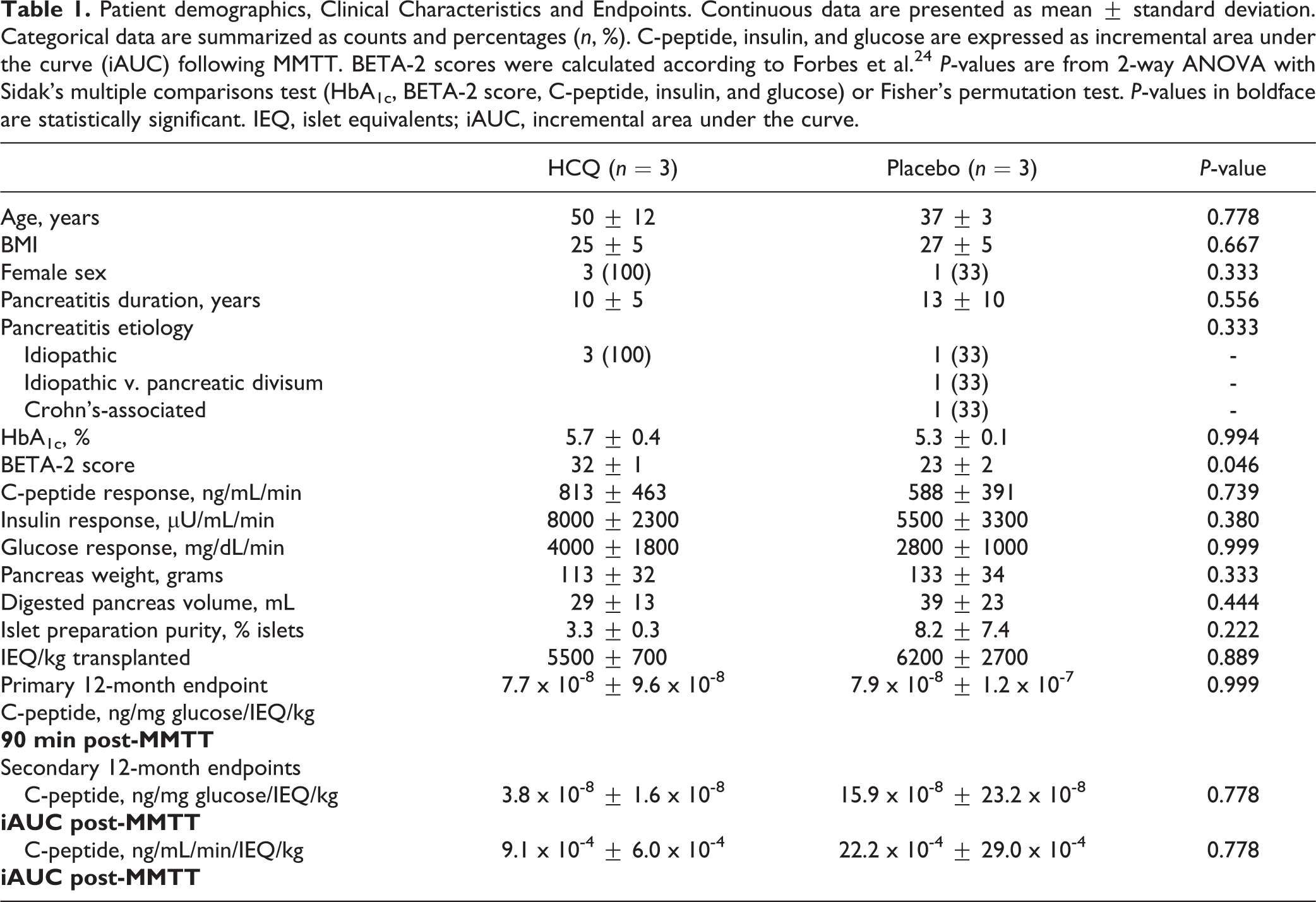
Among clinical and demographic characteristics of patients randomized into each arm, only BETA-2 score was significantly different (Table 1). Metrics of islet function were significantly poorer after TPAIT, including BETA-2 score (F2,8=207.5, P<0.0001), HbA1c (F2,8=12.7 P=0.003), and stimulated response to MMTT (iAUC for glucose, insulin, and C-peptide: F2,8=6.7, P=0.020, F2,8=28.3, P=0.0002, and F2,8=20.9, P=0.0007, respectively; Fig. 2). However, primary and secondary clinical endpoints were not significantly different between HCQ and placebo-treated patients (Table 1), nor were additional measures of clinical outcome including BETA-2 score, HbA1c, daily insulin requirement (divided as bolus and basal depending on clinical judgment), and response to MMTT at 6- and 12-months post-transplant (Tables 2 and 3, respectively). Although there was no significant effect of HCQ treatment on these clinical parameters, there was a significant interaction between HCQ treatment and time post-transplant on BETA-2 score (F2,8=10.5, P=0.006), as the higher BETA-2 score in patients randomized into the HCQ arm was not maintained post-HCQ treatment and TPAIT.
Patient demographics, Clinical Characteristics and Endpoints. Continuous data are presented as mean ± standard deviation. Categorical data are summarized as counts and percentages (n, %). C-peptide, insulin, and glucose are expressed as incremental area under the curve (iAUC) following MMTT. BETA-2 scores were calculated according to Forbes et al. 24 P-values are from 2-way ANOVA with Sidak’s multiple comparisons test (HbA1c, BETA-2 score, C-peptide, insulin, and glucose) or Fisher’s permutation test. P-values in boldface are statistically significant. IEQ, islet equivalents; iAUC, incremental area under the curve.
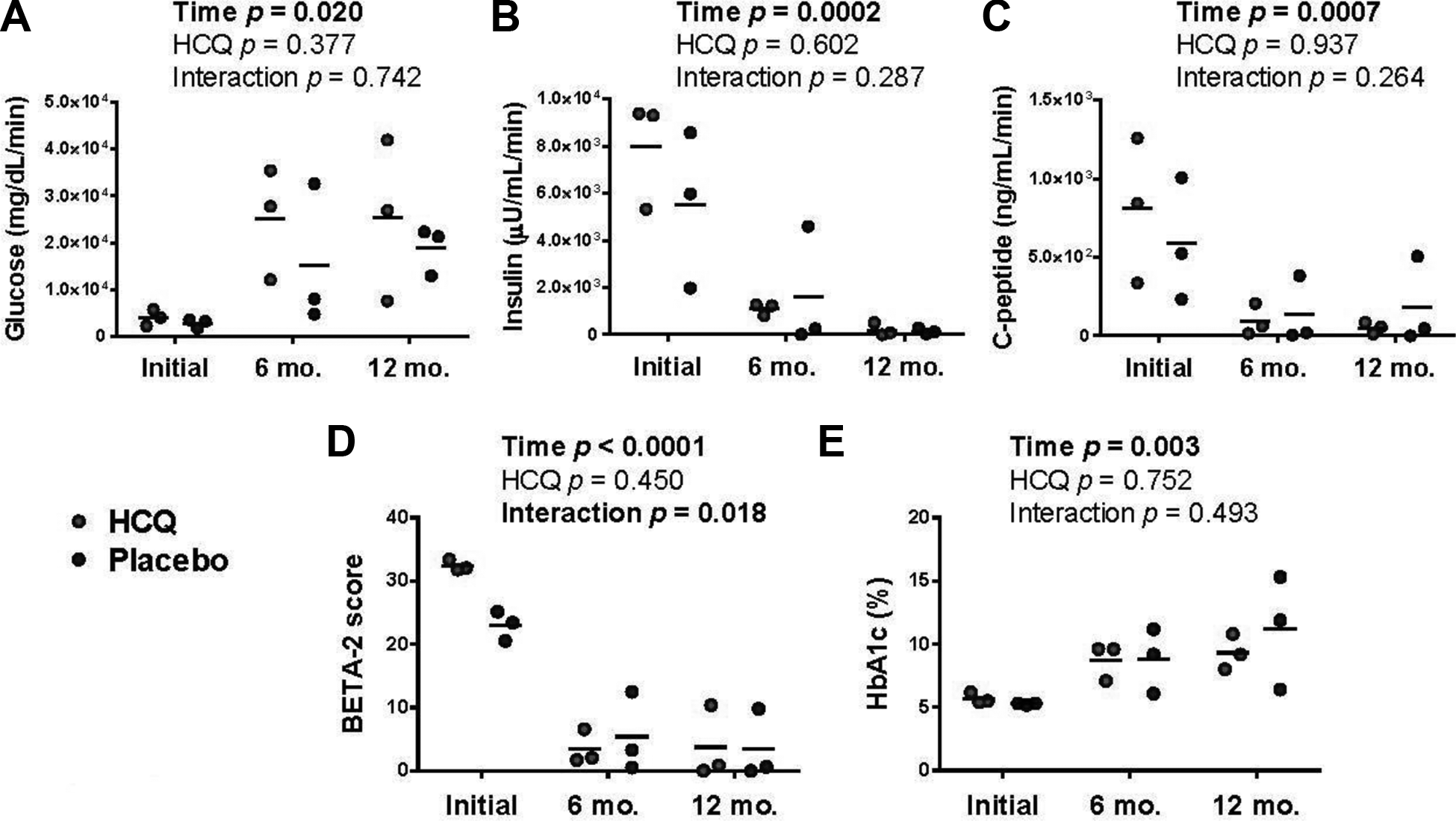
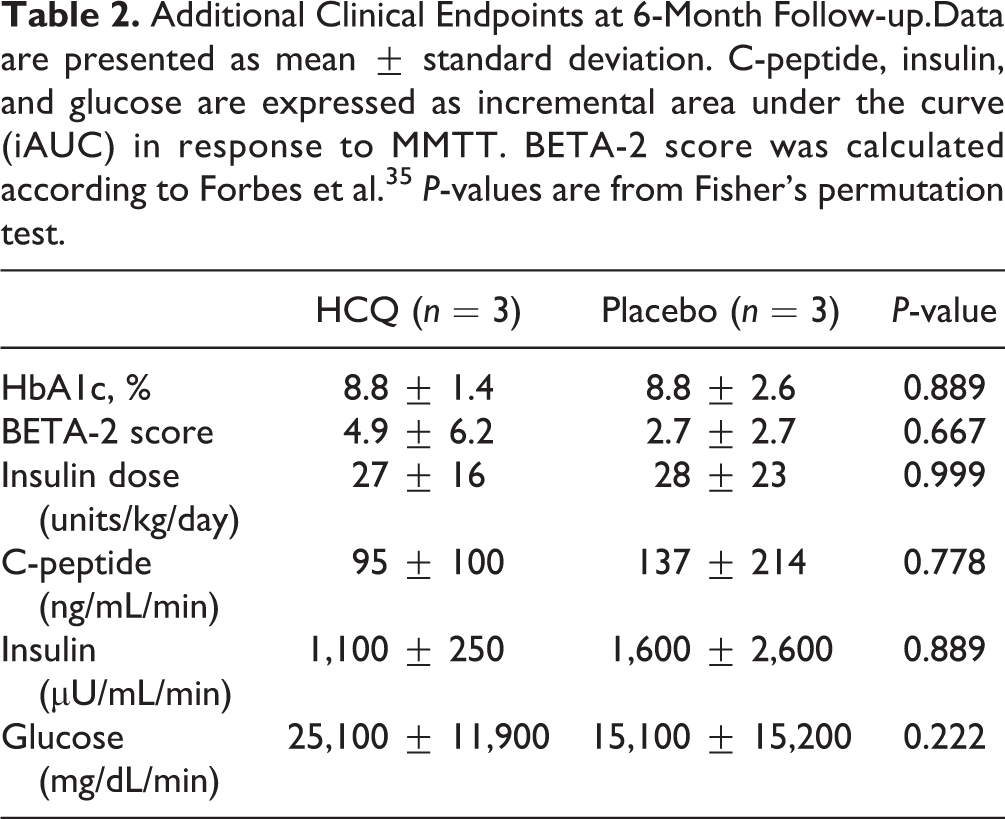
Additional Clinical Endpoints at 6-Month Follow-up.Data are presented as mean ± standard deviation. C-peptide, insulin, and glucose are expressed as incremental area under the curve (iAUC) in response to MMTT. BETA-2 score was calculated according to Forbes et al. 35 P-values are from Fisher’s permutation test.
Additional Clinical Endpoints at 12-month follow-up. Data are presented as mean ± standard deviation. C-peptide, insulin, and glucose are expressed as incremental area under the curve (iAUC) in response to MMTT. BETA-2 score was calculated according to Forbes et al.35 P-values are from Fisher’s permutation test.
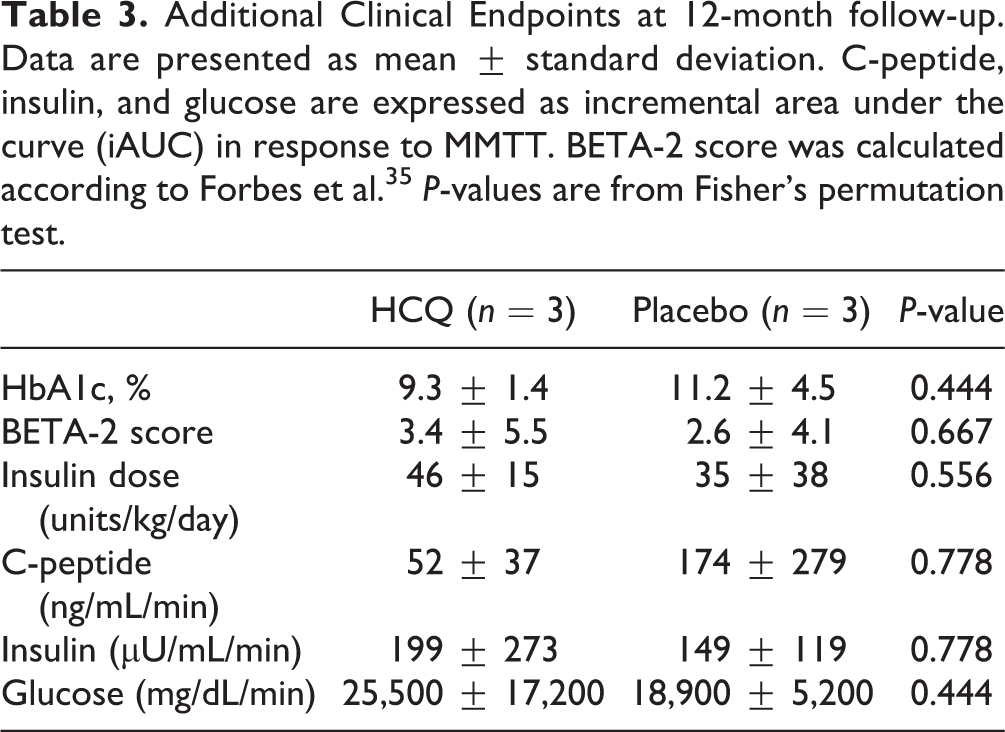
We analyzed seven bioenergetic parameters of autologous islet preparation samples stimulated with 20 mM glucose: respiration, ATP-linked respiration, maximal respiration, spare respiratory capacity (SRC), proton leak, non-mitochondrial respiration, and glycolytic metabolism (Fig. 1). CQHCQ treatment was associated with significantly higher SRC in autologous islet preparations compared to preparations from patients not treated or treated with placebo (P=0.028, Fig. 3A). The increased SRC was not a function of increased islet volume, since the comparisons were conducted after normalization to IEQ.
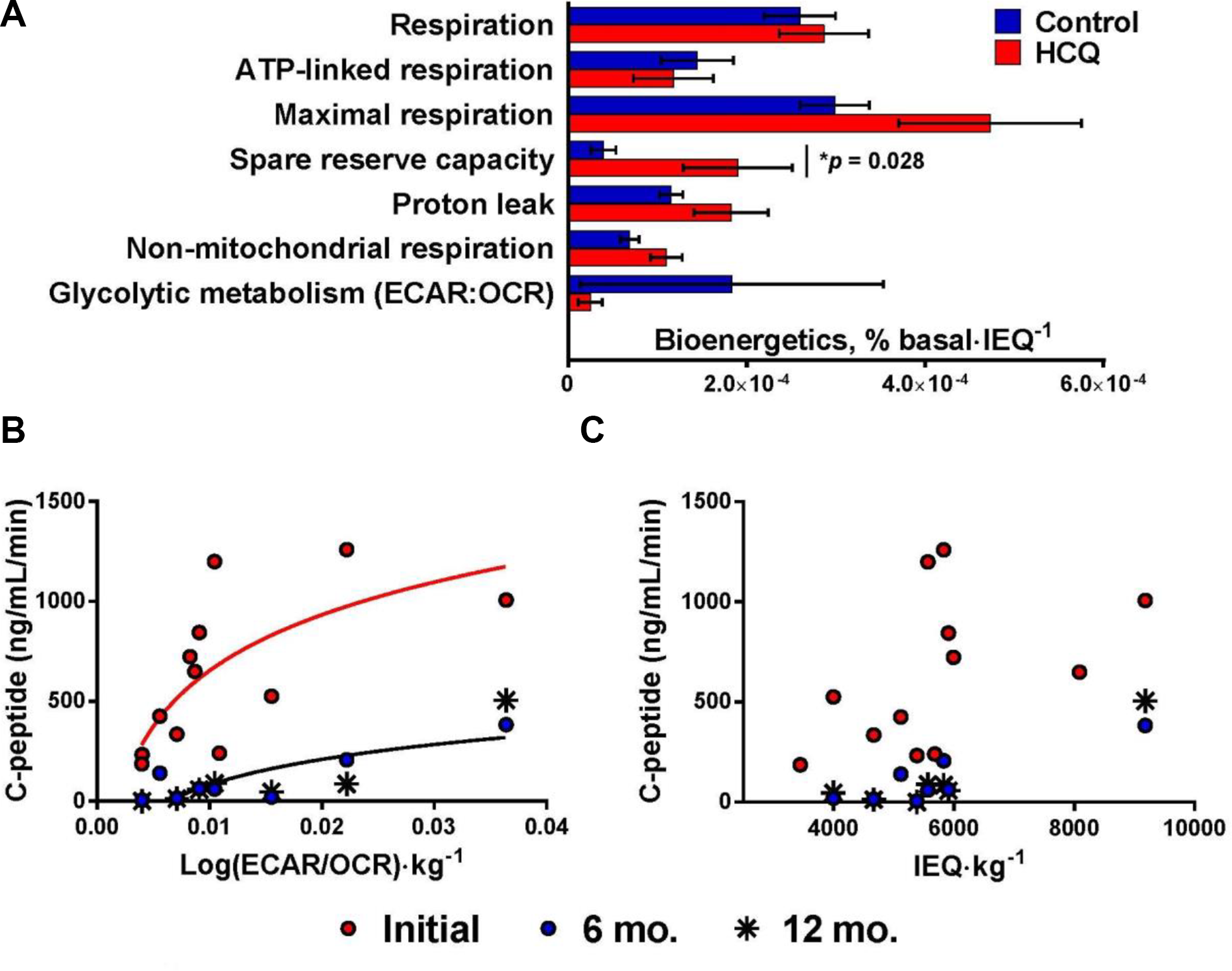
In order to determine the relationship between in vitro bioenergetic function of islet preparations and in vivo islet function after engraftment, we analyzed the correlation between bioenergetic parameters of the islet preparation (normalized to patient weight) with stimulated C-peptide in response to MMTT. We found that glycolytic metabolism (ECAR: OCR ratio/kg) of islet preparations significantly correlated with stimulated C-peptide (Fig. 3B) while metrics of mitochondrial function, such as respiration, ATP-linked respiration, maximal respiration, or SRC did not. Glycolysis of islet preparations directly correlated with stimulated C-peptide both before (P=0.01, Spearman’s r=0.699) and 12 months after TPAIT (P=0.03, Spearman’s r=0.821). The relationship between in vitro glycolytic metabolism of islet preparations and in vivo islet function was stronger than that between islet volume (IEQ/kg) and in vivo islet function (Fig. 3C). Islet volume showed a non-statistically significant trend toward correlation with stimulated C-peptide before (P=0.11, Pearson’s r=0.484) and 12 months after TPAIT (P=0.09, Spearman’s r=0.714). Glycolytic metabolism (ECAR/OCR) of islet preparations was not significantly correlated with IEQ (P=0.43, Spearman’s r=0.252), and normalizing glycolytic metabolism to IEQ did not improve the correlation between glycolytic metabolism and in vivo islet function.
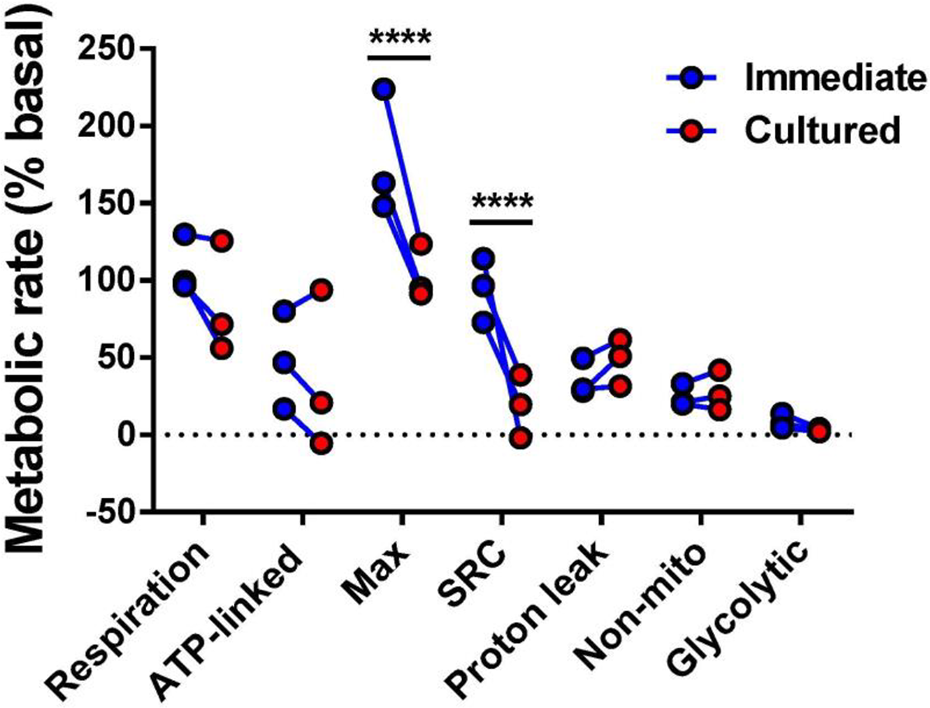
For each of three patients (1 HCQ-treated) the islet preparation sample was divided in half, with one half of the sample assayed immediately upon arrival to the Cleveland Clinic (at which time the clinical islet preparation was infused into the patient) and the other half of the sample assayed after overnight culture. A two-way repeated measures ANOVA showed that metrics of metabolic function were altered by overnight culture (F1,14=33.98, P<0.0001), but not all metrics were altered to the same direction/degree (Interaction F6,14=9.89, P=0.0002). Metrics of efficient metabolic function (ATP-linked respiration, maximal respiration, SRC and glycolytic metabolism) tended to decrease with overnight culture while proton leak, a metric of mitochondrial inefficiency, tended to increase (Fig. 4). Both SRC and maximal respiration significantly decreased after overnight culture (Sidak’s multiple comparisons test, P<0.0001).
Discussion
HCQ did not alter clinical outcomes in this pilot trial, but the statistical power to detect an effect of HCQ on clinical endpoints was limited by the clinical sample size (n = 6), which provided only 65% power to detect even an extremely large effect (e.g., d=2). In addition, the clinical outcomes themselves reported from this pilot trial are suboptimal compared with outcomes from larger studies conducted both at our center and others. In this trial, only 1 placebo-treated patient (17%) was insulin-independent at 12 months post-TPAIT, and median HbA1c was 10% with a range of 6.4-15.3 while a study of 36 patients treated between 2008 and 2014 at the Cleveland Clinic resulted in 12 patients (33%) that were insulin-independent at a median follow-up time of 28 months post-TPAIT with a median HbA1c of 6.8% and a range of 5.4 –10.3% 26 . In a study of 371 patients treated between 2009 and 2018 at the University of Minnesota, 121 patients (33%) achieved insulin-independence at 12 months post-TPAIT, and median HbA1c was 6.1% with a range of 5.7–6.9% 27 .
In spite of the small clinical sample size and less-than-optimal clinical outcomes, our study comprehensively characterized the functional metabolic rates of autologous islet preparations as a predictor of graft quality and produced some valuable insights into both the analysis of islet preparations pre-transplant and their use as a predictor of graft quality post-transplant. First, these in vitro analyses yielded some support for the hypothesis that peri-transplant HCQ treatment is beneficial in TPAIT: samples of islet preparations taken from HCQ-treated patients showed greater mitochondrial SRC (Fig. 3). SRC represents the potential respiratory capacity that can be drawn on in the event of stress or increased energetic demand, and is associated with cell viability, longevity, and resistance to stress 28 . It is possible that the effect of HCQ on clinical outcomes was masked by post-transplant variability in this small cohort and that a beneficial effect of HCQ, as detected in islet preparations, may indeed be detectable in clinical outcomes in a larger study.
Our data also support the hypothesis that in vitro bioenergetics of islet preparations can provide insight into in vivo graft function, previously identified by Papas et al 29 . who showed that OCR of autologous islet samples correlated with clinical transplant success better than IEQ infused. The utility of in vitro bioenergetics in predicting clinical outcomes is likely due to the ability of bioenergetic parameters like OCR to reflect both islet quantity and quality, while IEQ is a metric only of quantity. Strong predictors of in vivo islet function can inform clinicians about the probability that an islet preparation will yield a functional graft. This knowledge is especially valuable in the context of allogeneic transplantation, in which case the recipient suffers not only from nonspecific inflammation but alloimmunization and a recurrence of autoimmunity. Since the recipient of an allogeneic islet transplant has the option of waiting for another donor, bioenergetic indicators of a poor islet preparation could prevent patients from risking immunization with a low quality graft.
Our study builds on Papas et al.’s discovery that mitochondrial metabolism of autologous islet samples correlated with clinical transplant success better than IEQ infused by using an analytical platform that allows for simultaneous functional characterization of mitochondrial and glycolytic metabolism via OCR and ECAR, respectively. To our knowledge, our study represents the first attempt to simultaneously measure mitochondrial and glycolytic metabolism of autologous islet preparations and to characterize the relationship between the glycolytic rate of autologous islet preparations and clinical outcome. In doing so, our study identified glycolytic metabolism (glucose-stimulated ECAR: OCR ratio) as the strongest predictor of in vivo islet function, both before and after TPAIT (Fig. 3B, C)—outperforming both OCR and IEQ infused. Furthermore, in vitro ECAR: OCR of islet preparations was not significantly correlated with IEQ, suggesting that glycolytic metabolism was not simply a functional measure of islet quantity but contributed additional information regarding islet quality.
A strong correlation between glycolysis and islet function is to be expected according to the metabolic hypothesis of glucose-stimulated insulin secretion. First-phase insulin secretion depends on ATP produced via glucose metabolism, which triggers ATP-dependent K+ channels to open and induce Ca2+ influx 30 . Glycolysis then, as the first pathway in glucose metabolism, should correlate with glucose-stimulated islet function. Since O2-dependent mitochondrial metabolism of glucose produces the majority of ATP per glucose molecule, it is also reasonable that islet OCR would correlate with TPAIT outcome, as previously shown 29 . Our data, however, indicate that glycolysis of islet preparations may be a much more sensitive predictor of autologous islet graft function than either IEQ or mitochondrial metabolism.
Finally, by running the same bioenergetic assay both immediately upon the arrival of islet preparations to the Cleveland Clinic (at which time it was transplanted into the patient) and after overnight culture (n = 3 patient samples), we show that even short culture (12-18 hr) altered the bioenergetics of islet preparations (Fig. 4). Metrics of metabolic efficiency (glucose-stimulated, ATP-linked, and maximal respiration, SRC, and glycolytic metabolism) tended to decrease after overnight culture while proton leak—a metric of mitochondrial inefficiency—tended to increase. Maximal respiration and SRC, both of which are associated with cell viability and longevity, significantly decreased after overnight culture. Although in vitro islet culture is more common during allotransplantation, in which surgeries are scheduled only after deceased donors are secured, our data suggest that culture negatively affects the function of islet preparations, which may have negative consequences on in vivo graft function in both autologous and allogeneic transplantation. Furthermore, these data suggest that bioenergetics should be measured at the time of transplant, without additional culture, if they are to accurately reflect the function of the islet preparation infused.
Our in vitro bioenergetic analyses were limited by the lack of a pharmacological glycolysis inhibitor. We controlled for mitochondrial contributions to acidification a posteriori by analyzing the glucose-stimulated ECAR: OCR ratio rather than ECAR alone, but it is possible that another metabolic pathway contributed to extracellular acidification. Future studies should incorporate a glycolysis inhibitor to resolve this possibility.
Despite the study’s limitations, we took advantage of the ability to pretreat donor/recipients for a controlled duration prior to scheduled TPAIT in order to tackle nonspecific inflammation, an obstacle that is common to both autologous and allogeneic islet transplantation. We included a 30-day anti-inflammatory pre-treatment; a duration that is not possible even for the recipient in most cases of allogeneic islet transplantation. Although this pilot trial did not detect any differences in clinical outcome associated with peri-transplant HCQ treatment, it reveals new insights into the bioenergetic analysis of islet preparations before transplantation. In vivo HCQ treatment was associated with increased SRC in autologous islet preparations, which may serve to protect grafts from bioenergetic peri-transplant stress. In addition, we showed a significant negative impact of overnight culture on bioenergetic function of islet preparations and a correlation between in vitro glycolytic metabolism of islet preparations and in vivo graft function. Since glycolytic metabolism performed better than OCR or IEQ infused in predicting clinical outcome, our data suggest that glycolysis may be the most sensitive bioenergetic indicator of islet function.
Footnotes
Acknowledgements
This study was funded by the Cleveland Clinic Foundation. BH and KFA designed the clinical trial and REM and JPK designed the non-clinical portion of the study. REM conducted in vitro analyses, analyzed and interpreted all data, conducted statistical analyses, and wrote the manuscript. KFA, VTSM, SL, MW, TS, WW, and BH collected and/or interpreted clinical data. RB performed islet isolation. All authors revised the manuscript and approved the final version and agree to be held accountable for all aspects.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no subject in this article and informed consent is not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This reserach is funded by Cleveland Clinic Foundation.
