Abstract
C-X-C chemokine receptor type 4 (CXCR4), initially recognized as a co-receptor for HIV, contributes to several disorders, including the WHIM (Warts, Hypogammaglobulinemia, Infections, and Myelokathexis) syndrome. CXCR4 binds to its ligand SDF-1 to make an axis involved in the homing property of stem cells. This study aimed to employ WHIM syndrome pathogenesis as an inspirational approach to reinforce cell therapies. Wild type and WHIM-type variants of the CXCR4 gene were chemically synthesized and cloned in the pCDH-513B-1 lentiviral vector. Molecular cloning of the synthetic genes was confirmed by DNA sequencing, and expression of both types of CXCR4 at the protein level was confirmed by western blotting in HEK293T cells. Human adipose-derived mesenchymal stem cells (Ad-MSCs) were isolated, characterized, and subjected to lentiviral transduction with Wild type and WHIM-type variants of CXCR4. The presence of copGFP-positive MSCs confirmed the high efficiency of transduction. The migration ability of both groups of transduced cells was then assessed by transwell migration assay in the presence or absence of a CXCR4-blocking agent. Our qRT-PCR results showed overexpression of CXCR4 at mRNA level in both groups of transduced MSCs, and expression of WHIM-type CXCR4 was significantly higher than Wild type CXCR4 (P<0.05). Our results indicated that the migration of genetically modified MSCs expressing WHIM-type CXCR4 had significantly enhanced towards SDF1 in comparison with Wild type CXCR4 (P<0.05), while it was reduced after treatment with CXCR4 antagonist. These data suggest that overexpression of WHIM-type CXCR4 could lead to enhanced and sustained expression of CXCR4 on human MSCs, which would increase their homing capability; hence it might be an appropriate strategy to improve the efficiency of cell-based therapies.
Introduction
Mesenchymal stem/stromal cells (MSCs) are heterogeneous multipotent cells with evident therapeutic potential for treating intractable disorders 1 –7 . In addition to their therapeutic properties, MSCs can act as vehicles for cell-based gene therapies 8,9 . Ease of isolation, plasticity across lineages, paracrine effects, hypo-immunogenicity, immunomodulatory properties, and migratory behavior to the site of injury have attracted full attention towards MSCs 10 –12 . However, their suboptimal performance, mainly as a consequence of insufficient engraftment of applied cells, seems to question their ultimate applications 8,13,14 . Estimations showed that only a low percentage of the systemically administered cells survived to their target sites 15 . Extensive pieces of evidence suggest that migration of MSCs to inflamed tissues, the mechanism of selecting the target tissue, and crossing the endothelium have not been fully elucidated yet 16 . Despite the highly efficient migration of primary donor-derived MSCs, cultured cells rapidly lose their homing ability in vivo. It is well-known that higher levels of homing are required to achieve the optimal therapeutic effects in most cell therapy applications 17,18 .
Chemokine receptors belong to the large protein superfamily of G protein-coupled receptors (GPCRs) that mediate the trafficking of cells towards chemokines’ gradients 19 –21 . CXCR4 is a phylogenetically highly conserved receptor with 352 amino acids, which binds to stromal cell-derived factor 1 (SDF-1). CXCR4 has a seven-span transmembrane structure and, when triggered, transduces various downstream signals. CXCR4 also plays a pivotal role in some diseases, including WHIM syndrome 22 –25 .
The interaction of SDF-1 and CXCR4 plays an integral role after tissue injuries 26 –28 . It orchestrates the rapid revascularization of ischemic tissues, which is crucial to restoring organ function. SDF-1 is up-regulated at injury sites and plays the part of an efficacious chemoattractant to recruit circulating or residing cells that express the CXCR4 receptor 29,30 . After binding to SDF-1, CXCR4 is rapidly phosphorylated on its cytoplasmic tail and internalized. By internalization, CXCR4 can be either recycled back to the cell surface or degraded by lysosomes 20 . Research shows that impairment of CXCR4 internalization lengthens its outward expression and enhances the affinity of the receptor 31 . SDF-1, as a potent chemoattractant, is secreted within ischemic or inflammatory conditions. Cells expressing CXCR4 on their surface follow the gradient of SDF-1 to reach the damaged site 32 . MSCs express CXCR4 to a slight extent; however, after several passages in vitro, they abundantly lose this receptor, causing further reduction in their homing property 33 . Several studies suggested the overexpression of CXCR4 by genetic engineering as an effective strategy for improving cell therapy applications, but the short time of the receptor surface expression is still an obstacle 33 .
WHIM syndrome is a rare primary immunodeficiency disorder caused by gain-of-function mutations of CXCR4. WHIM stands for its four major clinical manifestations: Warts, Hypogammaglobulinemia, Infections, and Myelokathexis 34,35 . Myelocachexia refers to aggregation and elevation of the ratio of myeloid cells in the bone marrow, which is characterized by severe neutropenia in the peripheral blood. Previous studies highlighted that almost all known cases of WHIM syndrome are due to autosomal dominant mutations located in the cytoplasmic carboxy-terminus of CXCR4 35 –38 . Since this region plays a pivotal role in ligand-induced internalization and desensitization of the receptor, truncating mutations act to diminish the receptor’s internalization and enhance the activity of the SDF-1/CXCR4 axis 39,40 . Nine mutations have already been detected underlying the disorder. All these mutations are located in the carboxy-terminus of the receptor. Every known mutation but one truncates the carboxy-terminus of CXCR4 leading to removal of an inhibitory region. In this region, serine and threonine residues are normally phosphorylated by G protein-coupled receptor kinase (GRK) proteins upon activation by SDF-1 41,42 . The most common and well-studied WHIM variant of CXCR4 is the 1000C→T nonsense mutation, which removes 19 amino acids, creating a truncated receptor named CXCR4R334X. It accounts for most of the defined genotyped cases in the literature 43 –45 . Kawai et al. produced a retroviral vector coding R334X mutation and transduced K562 cells. The results confirmed the declined internalization of the mutant receptor and enhanced cellular migration in response to SDF-1 31 .
In the current study, we overexpressed the mutant CXCR4 gene in adipose-derived mesenchymal stem/stromal cells to evaluate the efficient superficial expression of the CXCR4 receptor. Inspired by WHIM syndrome’s pathogenesis, we hypothesized that impaired internalization of CXCR4R334X could facilitate the migration of adipose-derived mesenchymal stem cells through intensifying the SDF-1/CXCR4 axis.
Materials and Methods
Preparing Constructs and Confirmation of Subcloning
Wild type and mutant variants of CXCR4 were synthesized by GenScript (Piscataway, NJ, USA) and provided in pUC57. CXCR4 fragments were amplified with TaKaRa Ex Taq DNA polymerase (Takara Bio Inc., Kusatsu, Japan) using a conventional forward (F) and different reverse primers (R); EcoRI-CXCR4wild/mutant F (5′-ACAAATTATA
Western blot analysis
To extract the protein samples, we added RIPA lysis buffer (Sigma-Aldrich, St. Louis, MO, USA) to CXCR4-overexpressing HEK293 T cells as well as those transfected with control vector. Following centrifugation at 14,000 g for 10 min, the protein concentration was measured using a BCA assay kit (Thermo Fisher Scientific, Waltham, MA, USA).
SDS-poly acrylamide gel electrophoresis (SDS-PAGE) consisting of 5% stacking and 10% resolving gels was applied to run the protein samples. After the transfer process of the proteins onto the PVDF membrane, immunoblotting with the primary antibodies against CXCR4 (orb227928, Biorbyt Ltd, Cambridge, UK) and GAPDH (ab181602, Abcam, Cambridge, UK) was performed to detect the specific proteins of interest.
An HRP-conjugated goat anti-rabbit IgG (a6154, Sigma-Aldrich, St. Louis, MO, USA) as a secondary antibody was used to visualize the transferred proteins, and subsequently, a chemiluminescent detection technique was applied. The emitted light was detected by X-ray film and scanned for documentation.
Lentivirus Production
HEK293 T (CRL-3216) cells were purchased from the Cell Bank of Iranian Pasteur Institute and cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and incubated at 37°C in a humidified atmosphere containing 5% CO2 46 .
HEK293 T cells were transfected with the pCDH-CXCR4 lentiviral vectors (21 µg) (Wild type or Mutant) and two packaging vectors, psPAX2 (21 µg, gifted by Tronolab) and pMD2 (10 µg) by calcium phosphate method 46 for the lentivirus production process. Culture media were replaced by fresh media containing 10% FBS after 16 h, and then viral particles were harvested at 48, 72, and 96 h post-transfection. Supernatants were filtered through 0.45 µm filters (Orange Scientific, Braine-l’Alleud, Belgium), pelleted by ultracentrifugation (Beckman-Coulter ultracentrifuge XL-100 K, Brea, CA, USA) (at 28,000 rpm, 4°C for 1 h) and re-suspended in serum-free media, to increase viral concentration 46 .
Isolation and Culture of MSCs from Adipose Tissue
Lipoaspirates were collected from anonymous adults admitted to a private plastic surgery day clinic (Mashhad, Iran) to undertake an elective cosmetic procedure. The ACECR-Khorasan Razavi biomedical research ethics committee approved the project (IR.ACECR.JDMREC.1398.008), and informed consents were taken from all donors. The collected adipose samples were transferred to the laboratory under sterilized conditions and within the shortest possible time (< 8 h).
Samples were washed with phosphate-buffered saline (PBS) containing penicillin/streptomycin antibiotics. The container holding the adipose tissue was placed fixed for 15 min as stationary so that the liquid and lipid phases separated slowly. After that, 25 ml of the lower phase was extracted slowly. Next, the adipose tissue washing stage was replicated 3-4 times, followed by 0.1% collagenase type I (Thermo Fisher Scientific, Waltham, MA, USA) treatment at 37°C for 1 h. Samples were then diluted three times with PBS and then centrifuged at 800 g for 10 min. Finally, the mononuclear cells were transferred to T75 flasks containing DMEM supplemented with 10% FBS, 100 U/ml penicillin, and 100 µg/ml streptomycin at a density of 10000 cells per cm2 47 .
Characterization of Human Ad-MSCs
To ensure the purity and characterize the cells, their differentiation potential to osteocytes and adipocytes was assessed. Furthermore, we examined the expression of some specific cell surface markers as defined by the International Society for Cell and Gene Therapy (ISCT) 48 using BD Accuri™ C6 flow cytometry (BD Biosciences, San Jose, CA, USA).
Osteogenic differentiation of Ad-MSCs
Cells at passage 3 were cultured in DMEM-low glucose (Biowest, Nuaillé, France) supplemented with 10% FBS, ascorbate-2-phosphate (10 mM), β-glycerophosphate (10 mM), and dexamethasone (1 mM) (all from Sigma-Aldrich, St. Louis, MO, USA) for 14–17 days. Every 3 days, the culture medium was replaced. The differentiation ability was verified by visualizing extracellular matrix calcium deposits and the alkaline phosphatase activity of the cells. After the required time, in order to determine osteogenic differentiation, cells were fixed in 10% formalin and stained with 2% alizarin red S (Sigma-Aldrich, St. Louis, MO, USA) for 20 minutes. The alkaline phosphatase activity was determined using its substrate BCIP/NBT (Sigma-Aldrich, St. Louis, MO, USA) 49 .
Adipogenic differentiation of Ad-MSCs
To induce differentiation, we cultured the cells for 17–21 days in DMEM supplemented with 10% FBS, β-glycerophosphate (10 mM), dexamethasone (1 mM), and indomethacin (200 mM) (all from Sigma-Aldrich, St. Louis, MO, USA). This medium was replaced every 2 days, and after that, cells were fixed in 10% formalin solution and stained with 0.3% oil red O (Sigma-Aldrich, St. Louis, MO, USA) and hematoxylin-eosin to visualize the nuclei 47 .
Flow cytometric analysis of Ad-MSC surface markers
We evaluated the expression of some specific markers of MSCs to characterize the cells. Expression of superficial markers of mesenchymal stem cells, including CD90, CD44, CD73, and CD105, as well as the markers of hematopoietic cells, including CD11b, CD45, and CD34, was evaluated using flow cytometry. Firstly, 1 × 106 cells at passages 3-4 were suspended in 100 µl cold PBS containing 5% FBS. After that, cells were incubated for one h with the antibodies (all from Cytognos, Salamanca, Spain) of interest or isotype-matched controls. The expression of surface antigens was assessed using BD Accuri™ C6 flow cytometry (BD Biosciences, San Jose, CA, USA), and the data were analyzed by FlowJo 7.6 software 47,50 .
Transduction Experiments
Ad-MSCs were seeded in 6-well plates, and after reaching 60% confluency, were transduced with pseudoviruses containing pCDH-513B-1 as a control vector and Wild type or Mutant pCDH-CXCR4 vectors. Puromycin (Life technologies, Carlsbad, CA, USA) selection was performed with 2 μg/ml concentration for 2 days, and then cells were recovered in fresh media without puromycin.
Total RNA Extraction and Reverse Transcription
Total RNA extraction was performed using the TriPure kit (Roche Diagnostics, Mannheim, Germany), following the manufacturer’s protocol. cDNA was synthesized using Oligo-dT primer as per manufacturer’s instructions (Thermo Fisher Scientific, Waltham, MA, USA) from the DNase I treated RNA samples (Thermo Fisher Scientific, Waltham, MA, USA).
Quantitative RT-PCR
Real-time PCR reactions were performed using gene-specific primers (Table S1) and SYBR Green Master Mix (Ampliqon, Odense, Denmark) in a Bio-Rad CFX-96 system (Bio-Rad, Hercules, CA, USA) to quantify the expression of the chemokine receptor gene in transduced MSCs. To analyze the relative variations in gene expression, the 2-ΔΔ CT method was applied, and the expression level of CXCR4 was normalized to β-Actin as the reference gene.
Migration Assay
Migration assay was performed using 24-well plate transwell inserts based on the Boyden chamber principle using a polycarbonate membrane (8 µm pore size, Costar, Corning, Cambridge, MA, USA). 2.5 × 104 re-suspended MSCs in serum-free DMEM containing 0.5% bovine serum albumin (BSA) were seeded into each upper compartment. 100 ng/ml SDF-1α (BioLegend, San Diego, CA, USA), the attractive agent, was added to the bottom chamber in 600 µl volume. After 18 h incubation at 37°C and 5% CO2, the non-migrated cells were completely wiped off from the top surface of the membranes, and migrated cells on the other sides of the membranes were stained with 4′,6-diamidino-2-phenylindole (DAPI). As a control, the activity of SDF-1α-induced migration was inhibited by AMD3100 (Sigma-Aldrich, St. Louis, MO, USA), a CXCR4-blocking agent, used at a concentration of 10 µg/ml for 30 min at room temperature before chemotaxis test. We randomly selected and counted five fields from two experiments in triplicate wells, and the average numbers of cells per field were considered for statistical analysis 51 .
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 8. All data were expressed as mean ± SD. Furthermore, data were analyzed by the one-way ANOVA Dunnett’s test. P values <0.05 were considered as statistically significant.
Results
Construction and Confirming the Expression of Wild Type and WHIM-type CXCR4
CXCR4 was cloned downstream of the CMV promoter. copGFP and puromycin-resistant proteins, which could be concurrently expressed based on T2A self-cleaving peptide, were employed as selection markers (Fig. 1A.). SDS-PAGE and western blotting were used to evaluate the expression of both Wild and WHIM-types of CXCR4 in our subcloned constructs in HEK293 T cells. The results confirmed that cloning of both CXCR4 genes downstream of the CMV promoter led to the production of functional mRNAs that are translated to CXCR4 proteins with the desired molecular weight (Fig. 1B).
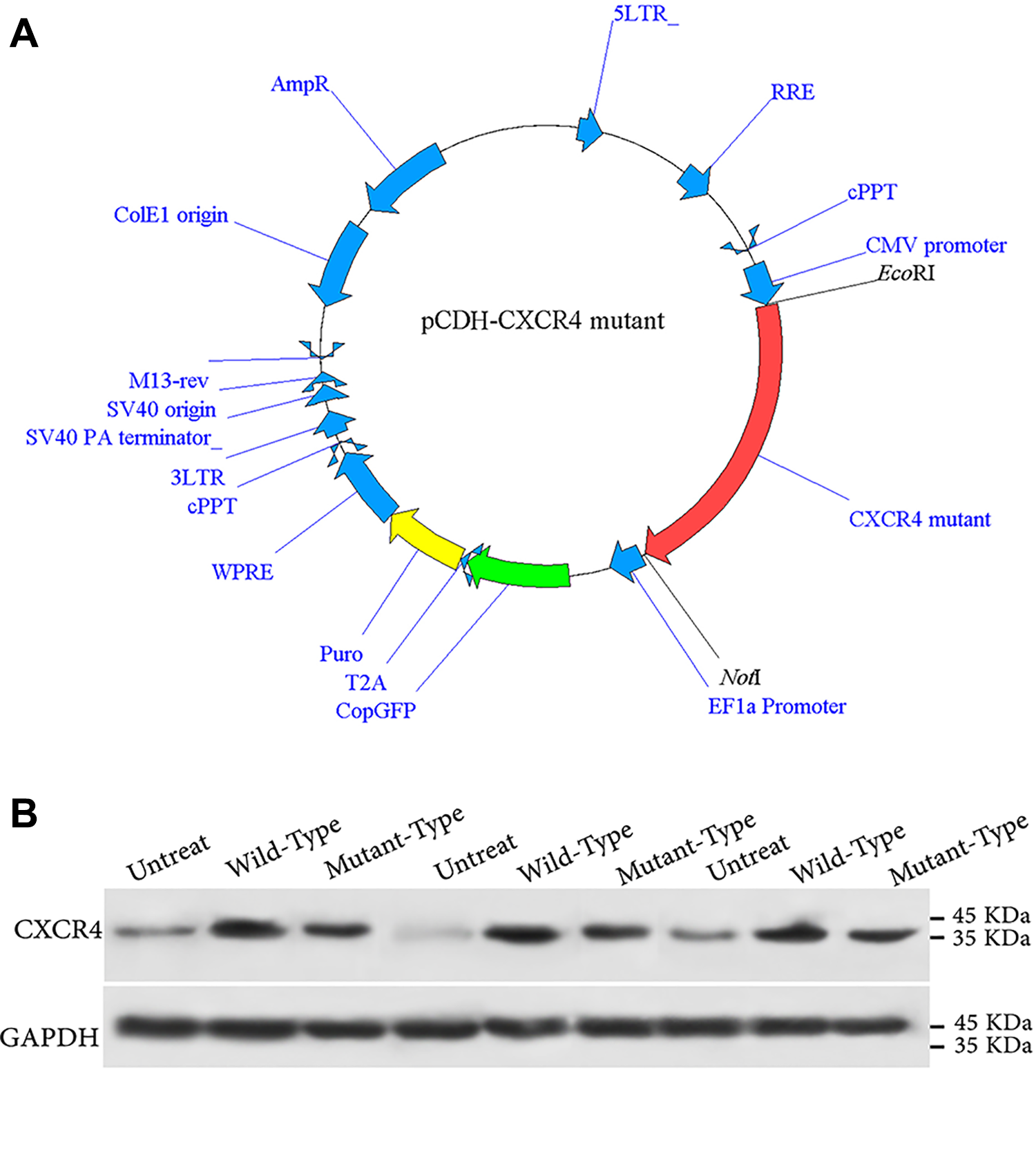
(A) The genetic map of the pCDH-513B-1 lentiviral vector containing Wild type or Mutant CXCR4 between EcoRI and NotI sites. (B) Evaluating the expression of Wild and WHIM-types of CXCR4 in the subcloned constructs by western blotting.
Isolation and Characterization of Human Ad-MSCs
The isolated MSCs grew with a spindle-shaped appearance throughout the consecutive culture passages. Differentiation of the MSCs to osteocytes was confirmed after their transfer into the differentiation induction medium for about 2 weeks and staining with alizarin red S as well as verifying alkaline phosphatase activity (Fig. S1). Furthermore, differentiation of the cells to adipocytes was confirmed by oil red O staining with the emergence of lipid vacuoles with a bright red color (Fig. S1). The cells extracted from adipose tissue at passage 3 were positive for MSC superficial markers of CD90, CD44, CD73, and CD105 and mostly negative for CD11b, CD45, and CD34 (Fig. S2).
CXCR4 Ectopic Expression in Ad-MSCs
pCDH lentiviral vectors, carrying the Wild type and Mutant variants of CXCR4, were transduced into MSCs. Besides, evaluating the expression of copGFP reporter in the MSCs, 72 h after infection was indicative of the successful transduction of the cells (Fig. 2A). Flow cytometry data analysis showed ∼50% GFP positive cells 72 h after transduction of MSCs (data not shown).
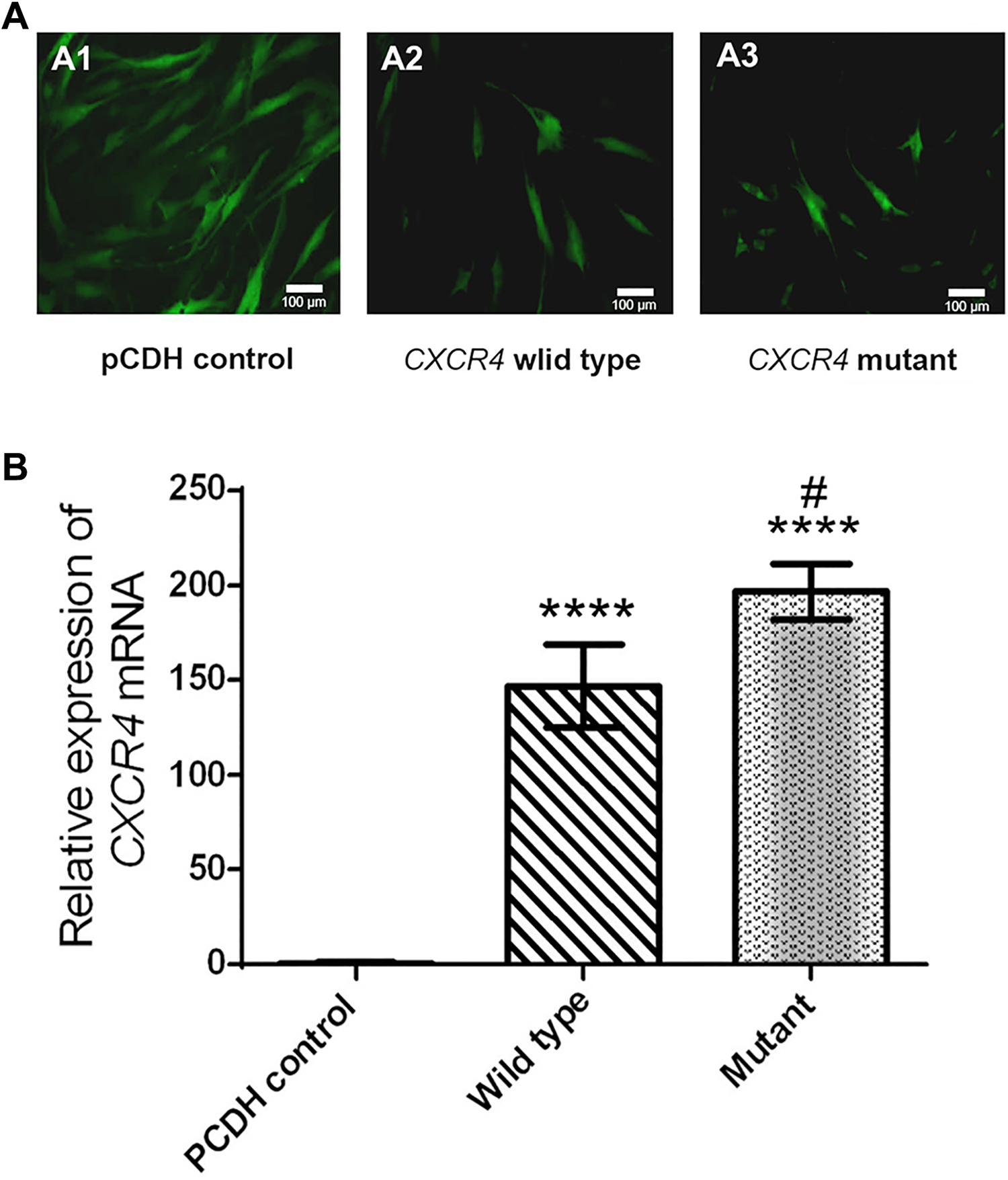
(A) Lentiviral transduced mesenchymal stem cells derived from adipose tissue 72 h after the transduction. (B) The results of CXCR4 overexpression in human mesenchymal stem cells. The results obtained from investigating the expression of the CXCR4 gene in Ad-MSCs transduced with Wild type and Mutated CXCR4 compared to MSCs containing pCDH-513B-1 vector. **** indicates significant difference compared to pCDH-control with P<0.0001, and # compared to the Wild type with P<0.05.
Following the overexpression of CXCR4 variants in human Ad-MSCs, changes in the expression of CXCR4 chemokine receptors were evaluated by qRT-PCR. Results indicated that CXCR4 expression was significantly increased in both MSC groups transduced with Wild type and Mutated CXCR4-expressing lentiviral vectors compared with cells transduced with pCDH-513B-1 vector (Fig. 2B).
Cell Migration Assay
The migration assay results indicated that migration of both cell groups overexpressing Wild type and Mutant CXCR4 was significantly increased compared to control Ad-MSCs with pCDH-513B-1 vector and non-transduced Ad-MSCs. Furthermore, the homing of mutated cells was significantly increased compared to the Wild type (Fig. 3), which could clearly show the functional induced CXCR4 pathway in transduced cells. Migration assay showed ∼2 folds more migration capacity in MSCs expressing Mutant CXCR4 compared to Wild type protein-expressing cells (Fig. 3). It is also noteworthy that treatment with AMD3100, as a CXCR4 antagonist, reduced cell migration in both cell types. This confirms the role of the SDF-1/CXCR4 axis in expanding the migration capacity of MSCs overexpressing CXCR4.
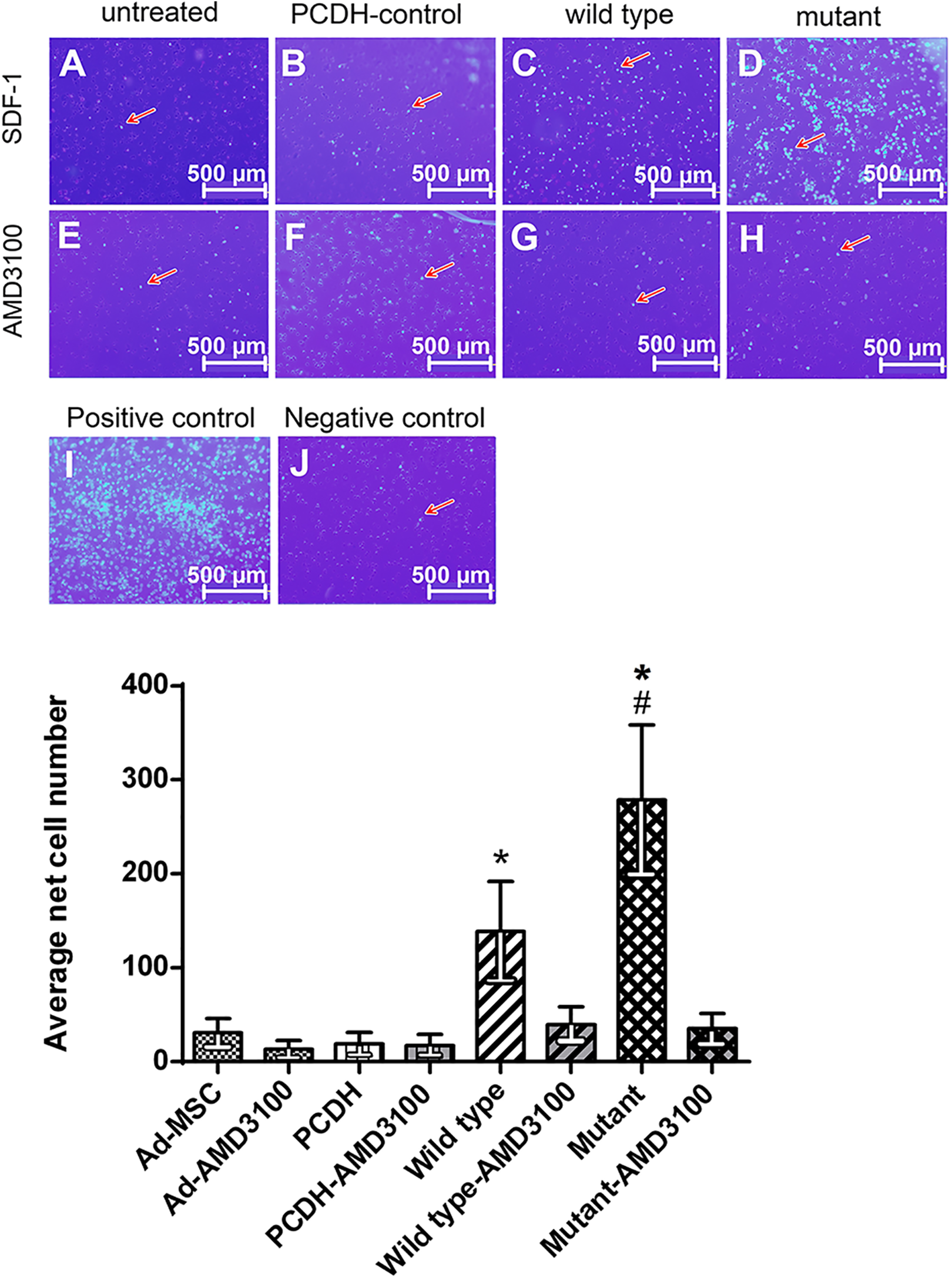
Investigating the migration of transduced cells with Mutant CXCR4 compared to the Wild type as well as Ad-MSCs containing empty vector and non-transduced Ad-MSCs. Increased migration of transduced cells (C, D) compared to the controls without treatment (A, B). To show that the migration of cells has been dependent on the SDF-1/CXCR4 axis, AMD3100 was used for inhibiting CXCR4 (E–H). Statistical investigations indicate a significant increase in migration of the transduced cells with Wild type CXCR4 compared to control groups and a substantial increase in the mutated variant compared to other groups (K). #Significant difference compared to the Wild type with P<0.05, *Significant difference compared to pCDH-control with P<0.0001. Negative control: the medium devoid of SDF-1, Positive control: the medium containing FBS.
Discussion
Advancing knowledge about the immunopathogenesis of WHIM syndrome provides a unique insight into the SDF-1/CXCR4 axis mechanism of action. We investigated the behavior of the MSCs, overexpressing CXCR4R334X, WHIM-type, in comparison with MSCs overexpressing the Wild type variant. The results showed that WHIM-type Ad-MSCs had significantly higher CXCR4 expression and in vitro migration capability than the control groups. A graphical illustration of our hypothesis, which has been confirmed functionally in this study, is depicted in Fig. 4.
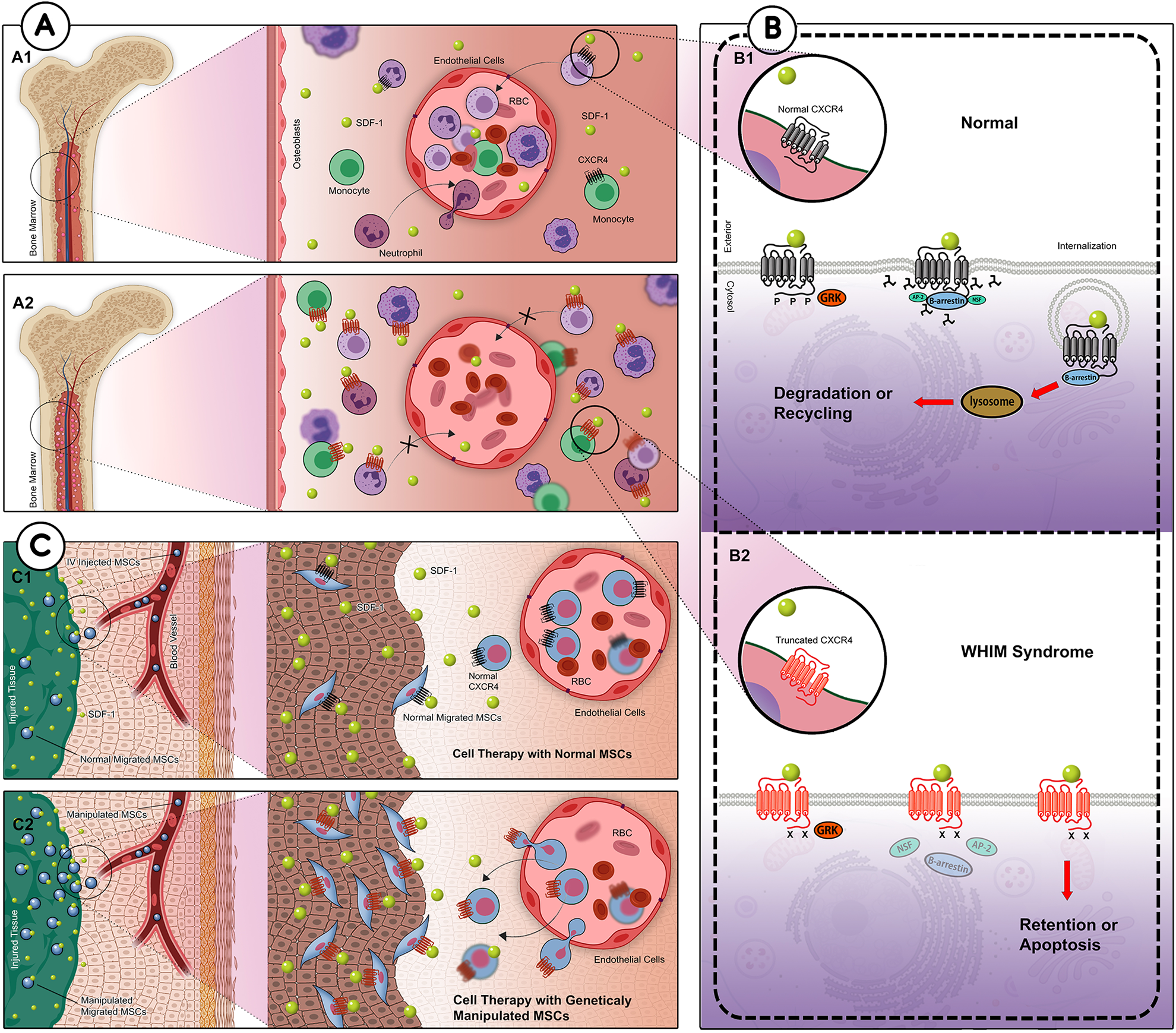
From the pathogenesis of WHIM syndrome to establishing a model to augment the SDF-1/CXCR4 axis: (A) Normal neutrophils in the bone marrow express CXCR4 and communicate with SDF1-expressing cells. Activated CXCR4 blocks passaging out from the marrow while neutrophils are un-matured. Maturation of neutrophils leads to internalization of CXCR4, which allows them to migrate to peripheral blood (A1). In WHIM syndrome, the mutant CXCR4 is not internalized, which causes retention of neutrophils in the bone marrow (A2). (B) The internalization of CXCR4 leads to transient surface expression of the receptor (B1). In this project, we genetically modified MSCs to overexpress WHIM-type CXCR4 (CXCR4R334X) by lentiviral transduction. The impairment of CXCR4R334X internalization enhanced its response to a chemotactic gradient of its specific ligand, SDF-1 (B2). (C) We hypothesized that stable ectopic expression of CXCR4R334X in MSCs could augment the SDF-1/CXCR4 axis. That being the case, systemic administration of genetically modified MSCs overexpressing WHIM-type CXCR4 may enhance the homing of transplanted cells to the injured area (C2); hence it could be an attractive strategy for efficient MSC-based therapeutics.
Genetic modification of MSCs to overexpress Wild-type CXCR4 has been an attractive approach to enhance their homing to ischemic sites both in vitro and in preclinical studies. Many researchers have employed various viral transduction techniques or transient methods to overexpress CXCR4 in stem cells (Table 1). In most cases, they inspected a significant enhancement in the migration of transduced cells towards an SDF-1 gradient in vitro. Some reported functional improvements in different preclinical conditions, include transplantation 52 –54 , myocardial infarction 55,56 , osteoporosis 57 , cerebral ischemia 58 , graft versus host disease (GVHD) 59 , acute lung injury 60 , diabetic retinopathy 61 , and colitis-associated tumorigenesis 62 models. In addition to stable overexpression of CXCR4, Hervás-Salcedo et al. showed that mRNA-transfected MSCs also possess enhanced transient CXCR4 expression and increased migration towards SDF-1 in an LPS-induced inflamed mouse model 63 . Our results regarding the comparison of Wild-type CXCR4 overexpressing MSCs with unmanipulated MSCs are in line with their results.
Overexpression of CXCR4 and its Consequences in Different Models.
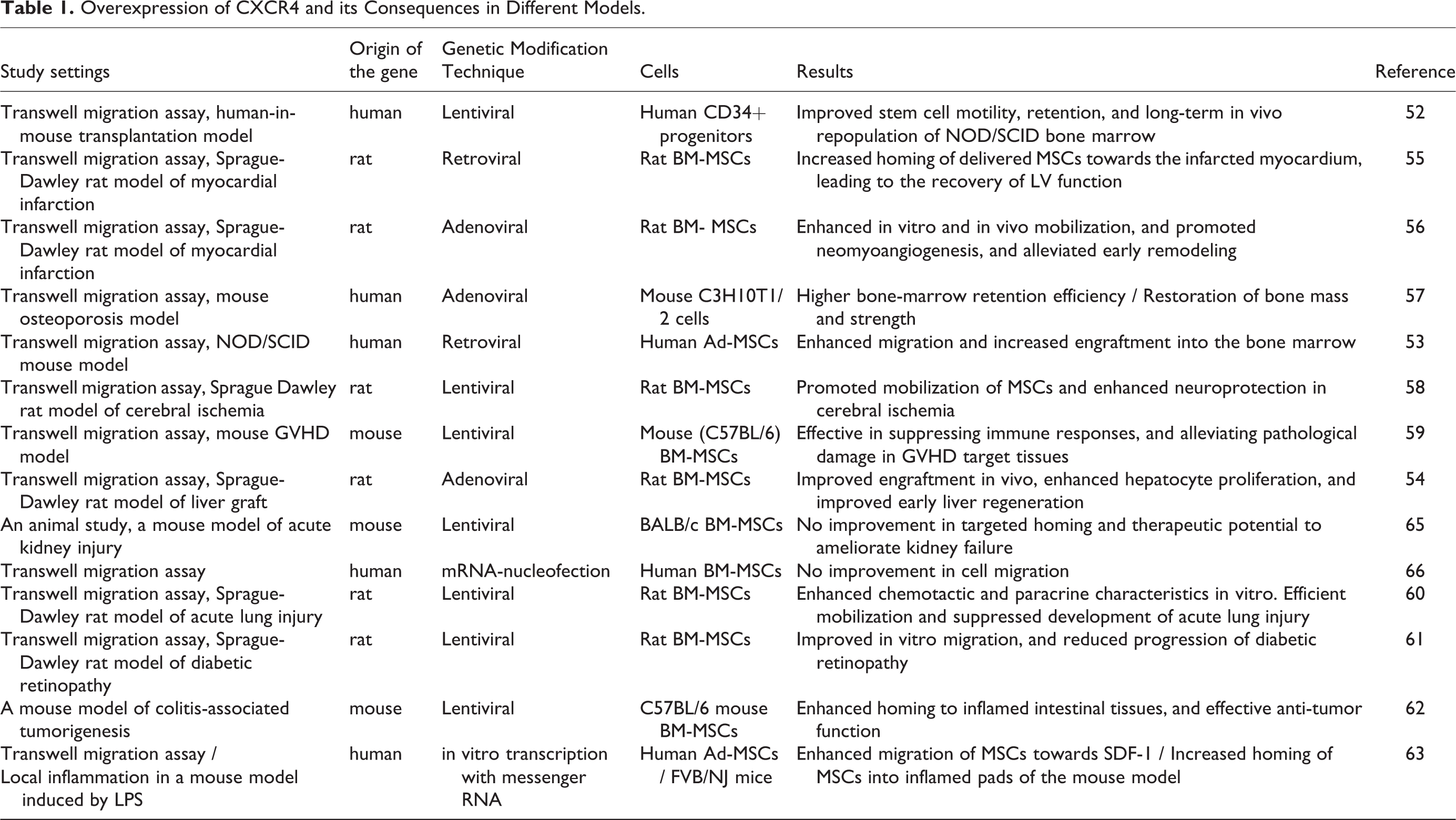
Freitas et al. addressed the application of genetically modified MSCs for non-union bone repair by their immunomodulatory and reparative potentials. They reviewed studies manipulating different candidate genes, e.g. BMP-2, BMP-4, SOX11, CXCR4, CBFA1, OSX, VEGF, and hTERT, and reported accelerated bone regeneration in MSCs overexpressing CXCR4 or genes that reinforced the SDF-1/CXCR4 axis, such as SOX11 64 .
In contrast to most studies, few researchers have claimed that genetic manipulation of stem cells does not improve their migratory property or in vivo outcome. Gheisari et al. overexpressed CXCR4 and CXCR7 in mouse BM-MSCs and evaluated their ex vivo homing property and renoprotective capacity in a murine model of acute kidney failure. They illustrated that the up-regulation of SDF-1 receptors could not enhance the engraftment rate of transduced MSCs and their therapeutic effect in renal injury. They attributed the discrepant findings to (1) the probable chromosomal abnormalities in MSCs during various ex vivo expansions, (2) compensatory mediators for the SDF-1/CXCR4 axis, and (3) the source of cells 65 . Similarly, Wiehe et al. overexpressed CXCR4 in human MSCs by mRNA-nucleofection with a GMP-grade protocol. SDF-1 stimulation of the modified cells increased cytosolic Ca2+ significantly, and different MAP kinases were activated. Although they confirmed the efficacy of CXCR4 modification, the migration capacity of the cells did not improve. They claimed that the SDF-1/CXCR4 axis appeared not to be involved in the homing of MSCs 66 .
The prolonged retention of mature myeloid cells in bone marrow followed by neutropenia, known as myelokathexis, is the primary manifestation of the WHIM syndrome. The rest of the signs and symptoms could be secondary to this condition. Some researchers have assumed this condition as approximately a pathognomonic sign of WHIM syndrome 45 . It was later explained that normal myelopoiesis results from a balance of CXCR2 release and CXCR4 retention signals. They were enhancing signals from CXCL8/CXCR2 and retarding signals from SDF-1/CXCR4 that determined the rate of neutrophil passage out from marrow stroma to peripheral blood and the distribution of neutrophils between the two compartments. The gain-of-function mutations of CXCR4 account for more than 95% of WHIM syndrome studied cases, whereas the loss-of-function mutations of CXCR2 have been reported only in two siblings 41,45 .
McDermott et al. in 2011 found that AMD3100 (Plerixafor) serves as a potent antagonist for CXCR4R334X identically to the Wild-type CXCR4. They indicated that the mutation in CXCR4 enhanced the chemokine receptor signal strength and decreased receptor down-regulation without affecting its sensitivity to AMD3100. They also investigated neutrophil chemotaxis mediated by SDF-1 in healthy donors and a WHIM patient. AMD3100 was an active antagonist to both. As the WHIM patient responded significantly to SDF-1, AMD3100 was considered a potent therapeutic agent 67 .
In the current study, we used AMD3100 to inhibit SDF-1-induced migration as controls for modified Ad-MSCs. The results showed that the migration of both transduced groups with Wild type and Mutant CXCR4 when inhibited by AMD3100 has no significant difference with control groups (Ad-MSCs with pCDH-513B-1 vector and non-transduced Ad-MSCs). It confirmed the substantial role of CXCR4 in the improved migration of modified Ad-MSCs. Additionally, as sustained binding of SDF-1 and CXCR4 is usually caused by maintained expression of CXCR4 at the cell surface 20 , our results regarding effective inhibition by AMD3100 could substantiate CXCR4R334X surface expression in the mutant group.
Kawai et al. developed a WHIM rat model with CXCR4R334X with a truncated cytoplasmic tail. They indicated that in addition to the CXCR4 variant, the type of cell utilized could also affect the receptor’s performance. As in the present study, the CXCR4R334X variant was expressed in mesenchymal stem/stromal cells for the first time; the functional differences and the divergent impact of this receptor on its downstream pathways are expected 68 . The authors assume that studying the mesenchymal stromal cells of a diagnosed WHIM patient (preferably caused by the 1000C→T nonsense mutation) could shed light on the mechanisms behind this investigation). Further, Hickstein and West examined the migration of neutrophils in an in vivo model of WHIM by developing a zebrafish model 69 . In both cases, the mutated CXCR4 model of WHIM was not able to be internalized following attachment to SDF-1. Even the development of inflammation could not cause the migration of neutrophils from bone marrow to peripheral blood 68,69 .
Future Directions
The method implied in the current investigation relied on the lentiviral-based approach with the benefit of being highly efficient and providing better conditions to examine our hypothesis 70 . However, close consideration of safety aspects is pivotal to minimizing unforeseen detrimental consequences in any genetic modification approach. By confirming our hypothesis, it would be crucial to conduct a preclinical study 71 . Upon successful animal model studies, recent gene-editing methods could also be employed to accelerate the translational process, including AAV-mediated gene therapy, zinc finger nucleases (ZFNs), transcription activator-like effector nucleases (TALENs), and clustered interspaced short palindromic repeats (CRISPR) in combination with CRISPR-associated protein (Cas) 70 . Another considerable strategy to facilitate the clinical application could be transiently expressing Mutant CXCR4, using other safer approaches such as in vitro transcription 63 . As a prominent cell-free method, extracellular vesicles (EVs) could also attenuate the potential risk of genetic engineering and pave the way towards the clinic. Ciullo et al. showed that systematic infusion of EVs derived from CXCR4-overexpressing cells significantly enhanced cardioprotection compared to non-engineered EVs in a rat model. By detecting CXCR4 at the surface of exosomes derived from genetically modified cells, they introduced the notion of “migration” for cell derivatives based on the SDF-1/CXCR4 axis 72 .
Conclusion
In this study, by modeling WHIM syndrome’s pathogenesis, a mutated variant of the CXCR4 gene was proposed for expression in mesenchymal stem cells derived from adipose tissue. CXCR4R334X variant in these cells was overexpressed using viral transduction. In summary, using MSCs with ectopic expression of the mutant CXCR4 resulted in increased targeted migration of the cells. CXCR4R334X variant overexpression could be proposed to enhance the homing efficiency of stem cell implantation. Despite the lack of molecular modeling for CXCR4R334X to show the clear mechanistic function of the mutant protein at the molecular level, our study may pave the way for recruiting cells obtained from genetic disorders with known gain-of-function or loss-of-function mutations as ideal models for regenerative strategies.
Supplemental Material
Supplemental Material, sj-docx-1-cll-10.1177_09636897211054498 - Mesenchymal Stem/Stromal Cells Overexpressing CXCR4R334X Revealed Enhanced Migration: A Lesson Learned from the Pathogenesis of WHIM Syndrome
Supplemental Material, sj-docx-1-cll-10.1177_09636897211054498 for Mesenchymal Stem/Stromal Cells Overexpressing CXCR4R334X Revealed Enhanced Migration: A Lesson Learned from the Pathogenesis of WHIM Syndrome by Hamid Reza Bidkhori, Ahmad Reza Bahrami, Moein Farshchian, Asieh Heirani-tabasi, Mahdi Mirahmadi, Halimeh Hasanzadeh, Naghmeh Ahmadiankia, Reza Faridhosseini, Mahtab Dastpak, Arezoo Gowhari Shabgah and Maryam M. Matin in Cell Transplantation
Supplemental Material
Supplemental Material, sj-jpg-1-cll-10.1177_09636897211054498 - Mesenchymal Stem/Stromal Cells Overexpressing CXCR4R334X Revealed Enhanced Migration: A Lesson Learned from the Pathogenesis of WHIM Syndrome
Supplemental Material, sj-jpg-1-cll-10.1177_09636897211054498 for Mesenchymal Stem/Stromal Cells Overexpressing CXCR4R334X Revealed Enhanced Migration: A Lesson Learned from the Pathogenesis of WHIM Syndrome by Hamid Reza Bidkhori, Ahmad Reza Bahrami, Moein Farshchian, Asieh Heirani-tabasi, Mahdi Mirahmadi, Halimeh Hasanzadeh, Naghmeh Ahmadiankia, Reza Faridhosseini, Mahtab Dastpak, Arezoo Gowhari Shabgah and Maryam M. Matin in Cell Transplantation
Supplemental Material
Supplemental Material, sj-jpg-2-cll-10.1177_09636897211054498 - Mesenchymal Stem/Stromal Cells Overexpressing CXCR4R334X Revealed Enhanced Migration: A Lesson Learned from the Pathogenesis of WHIM Syndrome
Supplemental Material, sj-jpg-2-cll-10.1177_09636897211054498 for Mesenchymal Stem/Stromal Cells Overexpressing CXCR4R334X Revealed Enhanced Migration: A Lesson Learned from the Pathogenesis of WHIM Syndrome by Hamid Reza Bidkhori, Ahmad Reza Bahrami, Moein Farshchian, Asieh Heirani-tabasi, Mahdi Mirahmadi, Halimeh Hasanzadeh, Naghmeh Ahmadiankia, Reza Faridhosseini, Mahtab Dastpak, Arezoo Gowhari Shabgah and Maryam M. Matin in Cell Transplantation
Footnotes
Acknowledgments
The authors gratefully thank Dr. Nasser Sanjar-Moussavi for providing us with liposuction samples.
Author contributions
Conception and design of the study: ARB, RF, NA, and MMM. Acquisition of data: HRB, MF, AHT, HH, and MM. Analysis and interpretation of data: MF, HH, and MMM. Drafting and revising the manuscript: ARB, HRB, MF, AGS, and MMM. All authors have approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to conduct and report this study was obtained from the ACECR-Khorasan Razavi biomedical research ethics committee (IR.ACECR.JDMREC.1398.008).
Statement of Human and Animal Rights
Procedures in this study were entirely conducted in accordance with the ACECR biomedical research ethics committee approved protocols.
Statement of Informed Consent
Written informed consents were obtained from the patients for their anonymized information to be published in this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Ferdowsi University of Mashhad (Grant No: 2908/1388) and Academic Center for Education, Culture, and Research (ACECR) (Grant No: 2107/1394).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
