Abstract
Cardiovascular disease is one of the most common causes of death worldwide. Mesenchymal stem cells (MSCs) are one of the most common sources in cell-based therapies in heart regeneration. There are several methods to differentiate MSCs into cardiac-like cells, such as gene induction. Moreover, using a three-dimensional (3D) culture, such as hydrogels increases efficiency of differentiation. In the current study, mouse adipose-derived MSCs were co-transduced with lentiviruses containing microRNA-1 (
Introduction
Cardiovascular disease (CVD) is main reason of death globally 1 . In the recent decades, there have been significant advances in surgical techniques and pharmacological therapies. Nevertheless, CVDs have remained a leading cause of heart failure worldwide 2 . For this reason, it seems necessary to adopt new methods, such as cardiac regenerative approaches and cell-based therapies 3 . Among cellular sources, mesenchymal stem cells (MSCs) are widely used due to their unique properties including paracrine effects, strong immunomodulation, low immunogenicity 4 , and differentiation ability into several cell lines like cardiomyocyte 5 . MSCs are found in various sources, such as bone marrow, placenta, cord blood, and adipose tissue 6 . Among these, adipose-derived MSCs have several advantages such as their accessibility, easy harvesting, low morbidity, and high expansion capacity 7 . Recently, combined strategies, such as genetic modification 8 and using biomaterials 9 have been developed to increase therapeutic efficacy of MSCs.
In several studies, using genetic modification, MSCs have been differentiated into induced cardiomyocyte-like cells (iCMs), for which both different genes and microRNAs (miRNAs) are used
10
–12
. The miRNAs bind to the 3′ un-translated region (3′ UTR) of target genes and down-regulate the expression of them
13
. Various studies implicate that miRNAs are involved in cell proliferation, differentiation, and development
14
. Moreover, different miRNAs are involved in cardiogenesis, which are known as cardiac miRNAs, such as
In muscle differentiation, serum response factor (SRF) acts as an activator of muscle genes including
Despite many advances in genetic modification of MSCs and their differentiation into iCMs, these cells are not sufficiently effective in cell-based therapy because MSC-derived cardiomyocytes in a two-dimensional (2D) cell culture are often immature and have a phenotype similar to that of fetal cardiomyocyte instead of adult cardiomyocyte 19 . Therefore, many studies have investigated various approaches of 3D cell culture, such as using polymer scaffold, decellularized extracellular matrix, and different hydrogels for further differentiation and maturation of MSCs 20 –22 . Furthermore, in myocardial infarction (MI) models, most of the cells injected straightly in infarction zone would undergo apoptosis 23 . Therefore, for increasing retention of the injected cells as well as protecting them from apoptosis in the infarction site, appropriate scaffolds can be utilized to improve healing process 23 .
Hydrogels as the first water-swollen biomaterials used in tissue engineering can create a 3D structure 24 . Chitosan is one of the widely used materials in hydrogels because of its properties such as biodegradability, biocompatibility, hydrophilicity, non-toxicity, and compressive strength due to its positive charge 25 . In cardiac regeneration, chitosan has been used in various studies as a cell carrier, increasing cell retention and viability, improving heart function by reducing infarct size and neovascularization in MI model 26 –28 .
One of the strategies in tissue engineering is the use of composite hydrogels. Chitosan-collagen (CS/CO) hydrogel is one of the most widely used composite hydrogels to improve cardiac cell differentiation and regeneration
29
. Collagen is one of the main components of extracellular matrix promoting tensile strength in heart wall. Collagen promotes survival, proliferation, and cell attachment
30
. Nevertheless, it has high biodegradability rate with weak mechanical strength
31
. But due to mechanical stability and positive charge of chitosan, combination of chitosan and collagen improves mechanical strength and reduces degradation rate of collagen
32
. Thus, in the current study, the effect of simultaneous induction of
Materials and Methods
Fabrication of Composite Hydrogel
Composite hydrogel consisting of chitosan (CS), human collagen type I (CO), and β-glycerophosphate (β-GP), as a cross-linker, was fabricated according to the previous studies 33 . Briefly, CS and CO (Sigma Aldrich, UK) were dissolved in 0.1 N and 0.02 N acetic acid and were stirred for 6 h to make 2.0% w/w and 4 mg/ml of stock solution, respectively. Then, CS and CO solutions were mixed in a ratio of 3:1 and β-GP (7.5%) was added dropwise as a cross-linker on ice bath. Next, the solution was pipetted into a 48-well plate and gel formation was performed by addition of sterile saturated NaHCO3 at 37°C for 15 min. In order to investigate the porous structure, the hydrogel was freeze-dried at -80°C for 48 h. Subsequently, the freeze-dried samples were covered with gold particles and were studied by a scanning electron microscope (SEM) (Hitachi, SU3500) with 15 kV of accelerating voltage.
Physiochemical Properties of CS/CO Hydrogel
Compressive strength of CS and CS/CO hydrogels was measured on a mechanical test machine (Model: INSTRON 5566). All the samples were compressed to 50% of their initial height and compressive strength of each sample was calculated with respect to compressive strain ratio. For testing swelling ratio, the freeze-dried samples were allowed to hydrate in phosphate-buffered saline (PBS) at room temperature (RT). The swollen samples were weighed at 50 minute-intervals, after removal of excess PBS by gentle blotting. Swelling ratio was calculated as follows: Q = (Ms-Md) / Md; where Q is swelling ratio, Ms is mass at the swollen state, and Md is mass at the dried state. Finally, Fourier-transform infrared (FTIR) spectroscopy was performed for evaluation of chemical structure of CS and CS/CO. FTIR spectrum was documented at the range of 400 - 4000 cm-1, using KBr pellet by FTIR spectrophotometer (FT-IR 8400 S, Shimadzu, Japan).
Isolation and Characterization of MSCs
Isolation and characterization of MSCs was mentioned in supplementary file.
Lentiviral Production and Transduction of MSCs
For lentiviral generation of
MSCs at passage 3 were used for transduction. The cells were seeded at a concentration of 4 × 105 in 6-cm petri dishes and the next day were transduced in three groups; first a group of cells that were transduced with pLenti-GFP-blank lentiviruses containing green fluorescent protein (GFP) (MSCnull), another group of cells that were infected with
Viability Assay of Transduced Cells
For evaluating their viability, transduced cells were pipetted in equal volumes at a concentration of 1×104 in 96-well plate and were kept at 37°C for 7, 14, and 21 days. Cell viability rate was determined by 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2 H -tetrazolium bromide (MTT; Sigma Aldrich) assay. At given time points, 100 μl/well of MTT was added into the cells, then was kept at 37°C for 4 h, followed by adding 100 μl/well of dimethyl sulfoxide (DMSO). The samples were monitored at 580 nm of wavelength using enzyme-linked immunosorbent assay (ELISA) reader. The non-transduced MSCs were used as a control.
Evaluation of Differentiation of Transduced Cells
Measuring expression levels of miR-1 and Myocd , and target genes in transduced cells by qRT-PCR
For verifying transduction efficiency, expression levels of
Sequences of Primers Used in qRT-PCR.

Immunocytochemistry (ICC) analysis
Twenty-one day after transduction in the 48-well plate, fixation of MSCsmiR-1 and MSCsmiR-1/Myocd was performed by 4% paraformaldehyde (PFA) for 20 min at RT and permeabilization was performed with 0.2% Triton X-100 (Sigma, USA) for 5 min at RT. Then, cells were incubated overnight with primary antibodies, at 4 °C. cTnT (ab8295, Abcam, MA, UK), NKX2-5 (ab272914) and GATA4 (ab227512) were used as primary antibodies. Subsequently, cells were incubated with fluorescence-labeled secondary antibodies for 1 h at RT. The cells nuclei were stained with 0.1μg/ml of blue-fluorescent 4’, 6-Diamidino-2-phenylindole (DAPI) DNA stain (Sigma, USA) at RT for 5 min. Finally, cells were observed by a fluorescence microscope (Olympus, Tokyo, Japan). ICC analysis for MSCs group was mentioned in supplementary file.
3D Cell Culture
Cell viability and proliferation assay for encapsulated cells into CS/CO hydrogel was performed and non-transduced MSCs were used because of eliminate transduction interference on viability test results. For this regard, MSCs were encapsulated to CS/CO hydrogel and were homogenized by stirrer for 1 min. After transferring the mixture to the culture plate, it was neutralized using sterile saturated NaHCO3 solution to pH level of 7, and then it was incubated at 37°C for 15 min. Finally, cell culture medium was added to the encapsulated cells.
Viability and proliferation assays in CS/CO hydrogel
For evaluating their viability, MSCs were pipetted in equal volumes with a density of 1×105 in 96-well plate and were kept at 37°C for 1, 3,7, and 14 days. Cell viability rate was examined by MTT assay, as previously described. Cell free hydrogel and 2D culture of MSCs were used as control. Moreover, for showing cell proliferation potential in the hydrogel, the cells were labeled with 1,1’-dioctadecyl-3,3,3’,3’ –tetramethylindocarbocyanine perchlorate (DiI) (Thermofisher), a red cell membrane dye, according to manufacturer’s instructions. Finally, on days 1, 3, and 7, fluorescence microscopy was performed.
SEM analysis
Twenty-one days after transduction, the MSCs embedded in hydrogel was prepared for SEM analysis. The medium was aspirated, and glutaraldehyde (2.5%) was added to the cells and they were kept for 1 h at 4°C and after washing with PBS, dehydration was performed by ascending series of ethanol and SEM was performed as previously described 34 .
Encapsulation of Transduced Cells in CS/CO Hydrogel and Evaluation of their Differentiation into iCMs
3 days after transduction, MSCsmiR-1/Myocd were trypsinized and encapsulated in CS/CO hydrogel. Then, like the 2D culture, qRT-PCR and ICC assay for differentiation assessment of encapsulated transduced cells were performed. Total RNA was obtained from the cells on 7, 14, and 21 days after infection. Subsequently, after cDNA synthesis, qRT-PCR was performed for evaluation of
In vivo Studies
Induction of myocardial infarction (MI) in rats and patch transplantation
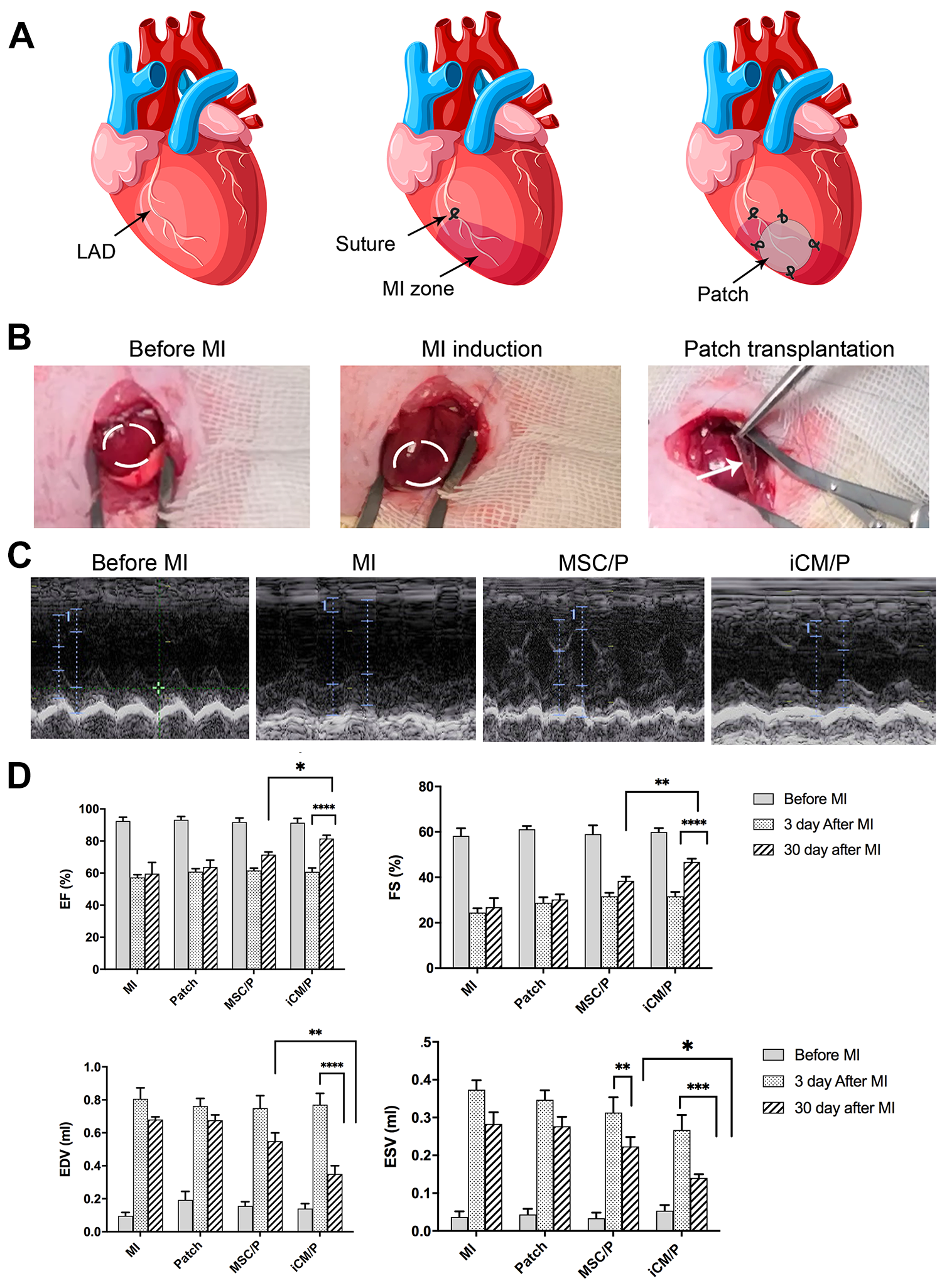
In the current study, 20 male Sprague Dawley (SD) rats (6–8 weeks old, and 180–250 g) were used. All the animal experiments were conducted according to the guidelines approved by Care and Use of Laboratory Animals and the Ethics Committee of the Tarbiat Modares University, Iran (IR.MODARES.REC.1398.125). The rats were anesthetized by intramuscular injection of 50 ml/kg of ketamine hydrochloride and 5 mg/kg of xylazine. Next, the thoracotomy was performed, the pericardium was opened and a 5-0 silk suture was narrowed close the left anterior descending (LAD) coronary artery. After 10 min, color of the area under the suture was changed and patches were transplanted on MI zone. The transplanted patches (P), comprising of CS/CO hydrogel placed on Polydimethylsiloxane (PDMS) were used in three groups; P (cell-free patch), MSC/P, and iCM/P. In MSC/P and iCM/P groups, MSCs and iCMs were encapsulated in CS/CO hydrogel and then were placed on PDMS. For tracking the transplanted cells in rat heart tissue, DiI staining was used to label cells before transplantation according to manufacturer’s structure. In MI group, the LAD was sutured but no patches were transplanted on MI zone. Finally, rats were sacrificed by an intravenous injection of sodium thiopental (100 ml/kg) and the hearts were harvested for qRT-PCR, SEM, and histological analyses.
Echocardiography
Echocardiographic images of the hearts from 20 rats were obtained using a GE Vivid 7 echocardiography machine before MI, 3 days after MI (baseline), and 30 days after MI from different groups. For measuring left ventricular (LV) function, fractional shortening (FS), ejection fraction (EF), End-systolic volume (ESV), and End-diastolic volume (EDV) were assessed by echocardiography. The percentage of fractional shortening (FS) was calculated as follows: FS (%) =[(LVEDd−LVESd)/LVEDd] × 100 where LVEDd is LV end-diastolic dimension and LVESd is LV end-systolic dimension. All the measurements were performed based on averaged three sequential cycles of cardiac and were assessed by two independent operators who were blinded to treat and control groups of the animals.
RNA isolation and qRT-PCR analysis
Total RNA isolation, cDNA synthesis, and qRT-PCR were performed for
Histological analysis and immunohistochemistry (IHC)
Samples were prepared for histological analysis according to previous study 35 . Briefly, samples underwent 10% formalin fixation for 24 h and were embedded into paraffin wax. Paraffin-embedded tissues were cut at a thickness of 3µm and were prepared for histological assay. For the routine histological assessment was performed on tissue sections by Hematoxylin and Eosin (H & E) staining 36 . Moreover, IHC analysis was performed according to previous protocols 37 and paraffin-embedded sections were immunostained with cTnT, NKX2-5, and GATA4 antibodies. The images were observed by inverted fluorescence microscope (Olympus, Japan).
Statistical analysis
All data were represented as mean ± standard deviation (SD) of at least three experiments. The differences between test and control groups were analyzed with the unpaired t-test, one-way or two-way ANOVA using GraphPad Prism v9.0 (USA).
Results
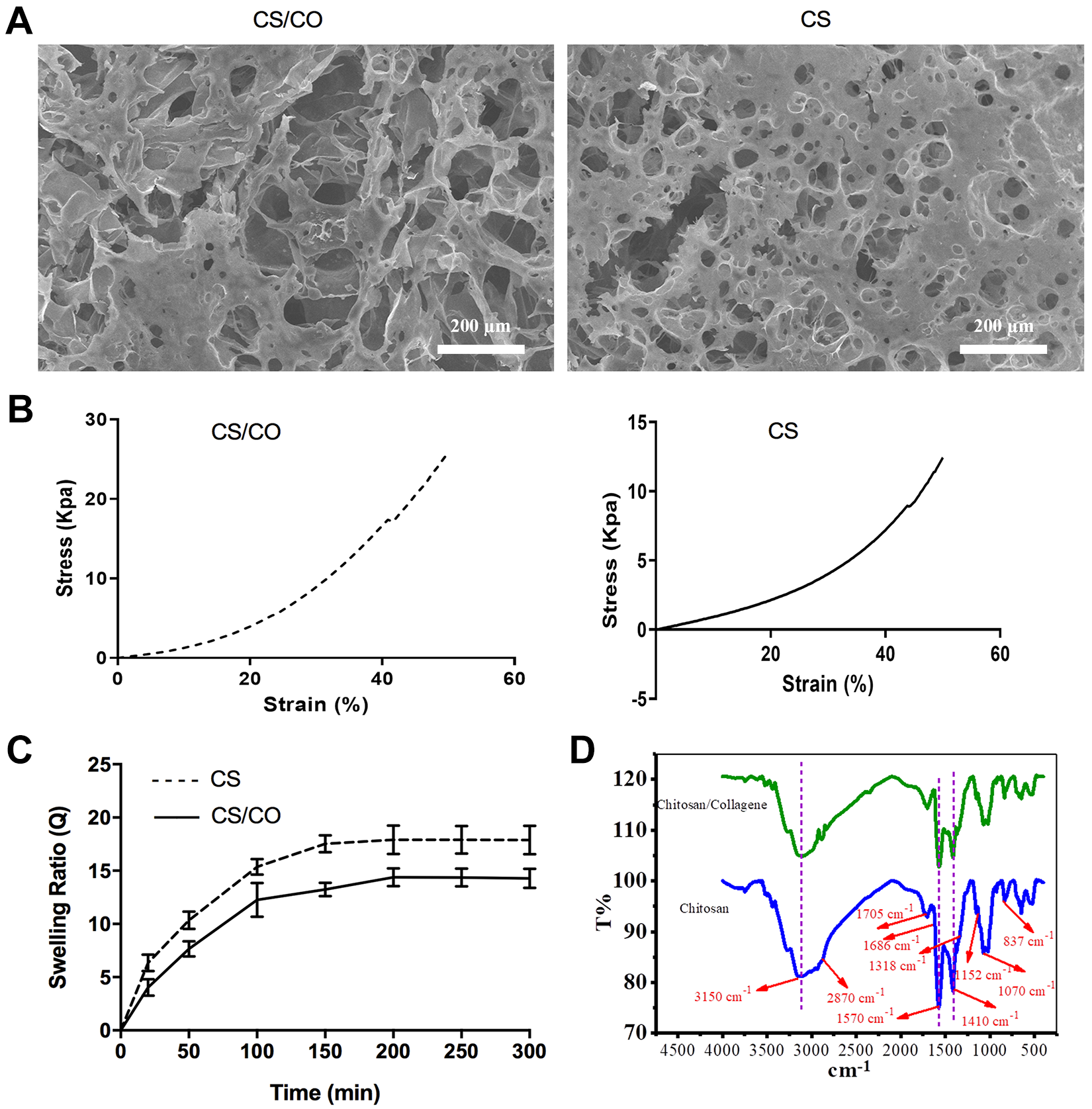
Morphology and characterization of the CS/CO hydrogel
SEM analysis was performed for determining morphological structure of CS and CS/CO hydrogels. The composite hydrogel exhibited a structure with CO fibrils dispersed within CS matrix. Moreover, interconnecting pores and creasing patterns were formed inside the hydrogels (Fig. 1A). CS and CS/CO hydrogel showed porous structure with 51.57% and 56.68% porosity, 17–89 µm and 21–109 µm pore size, respectively. This means that the pore size in both hydrogels allows the exchange of nutrients and biological molecules between cells. Stress-strain curve showed that pure CS and CS/CO hydrogels indicated a gradually enhancement in stiffness. Moreover, the CS/CO hydrogel showed a significant increase (**
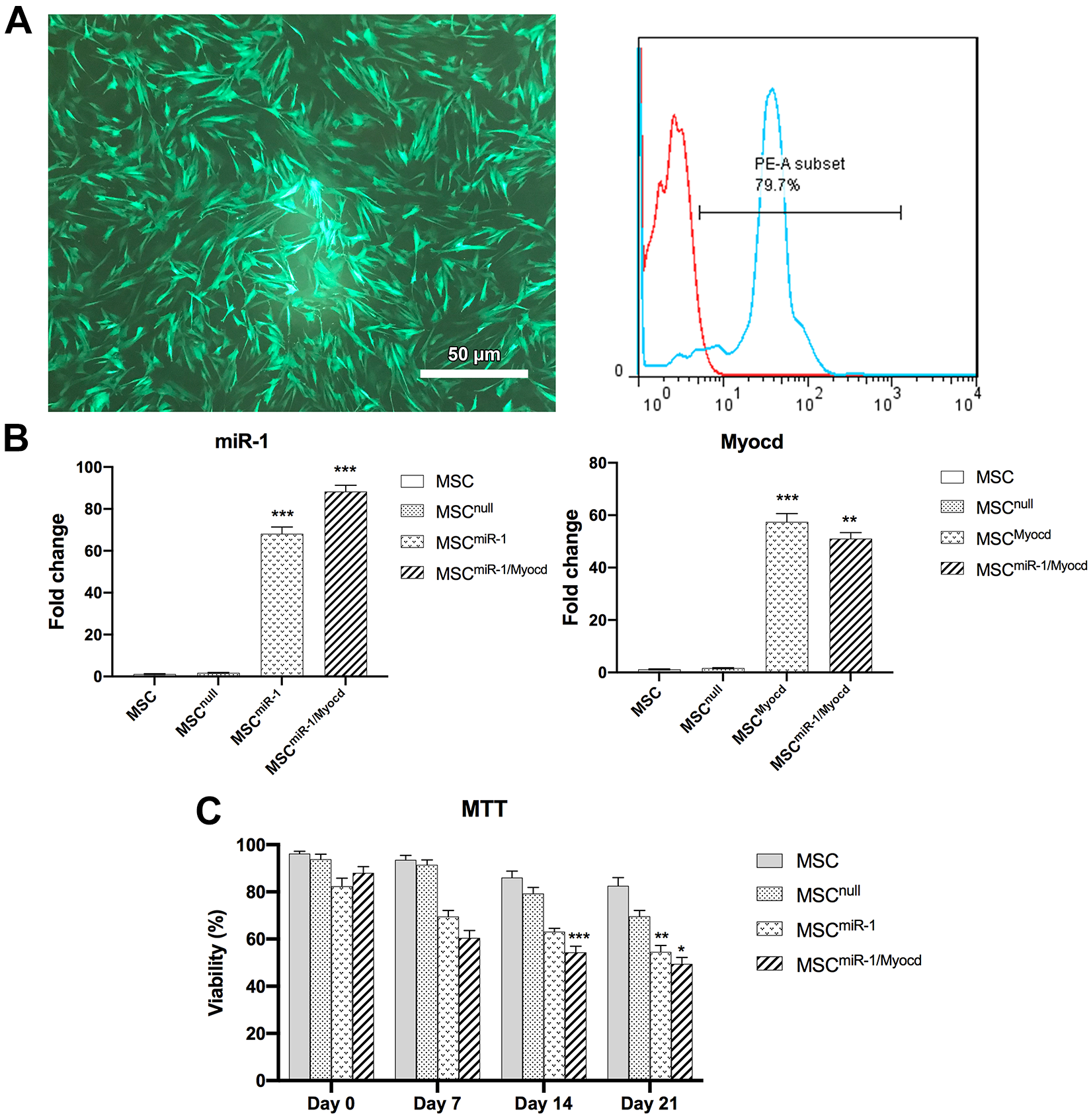
Up-Regulation of miR-1 and Myocd Expression in Transduced Cells
Transduction efficiency was performed by fluorescent microscopy and flow cytometry assessment, which about 80% of cells were transduced (Fig. 2A). Expression levels of
Differentiation of Transduced Cells into iCM
Seven days after transduction, morphological changes were observed in different groups of transduced cells. On day 7, morphology of MSCmiR-1 and MSCmiR-1/Myocd groups was found to be short spindle- or star-shaped (Fig. 3A). As expected, morphology of control groups (MSC and MSCnull) did not change (Data were not shown). Twenty-one days after transduction, further differentiation and maturation was observed. The cells became polygonal or spindle-shaped and were connected to adjacent cells (Fig. 3A).
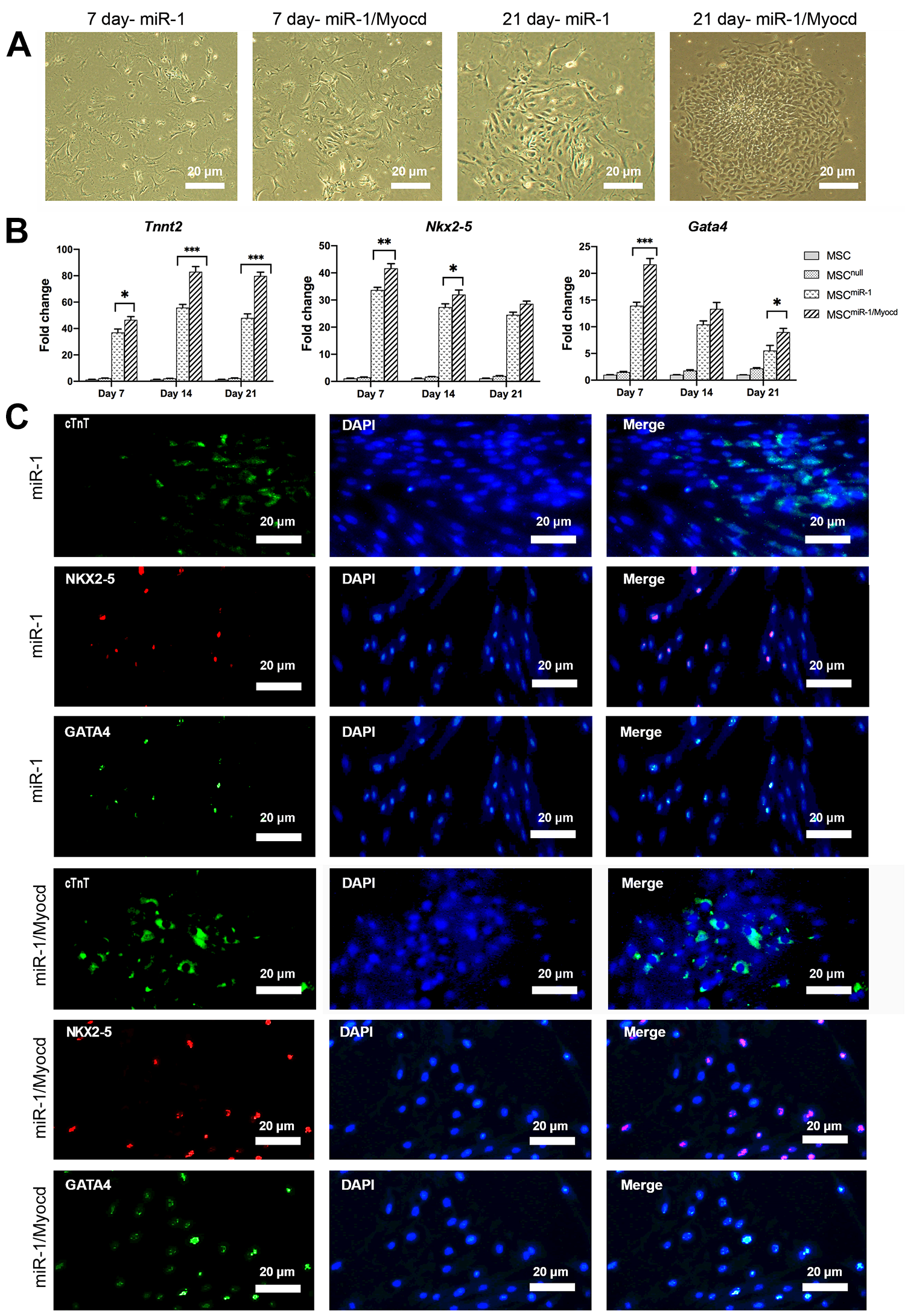
As shown in Fig. 3B, mRNA expression of the genes involved in cardiogenesis, such as
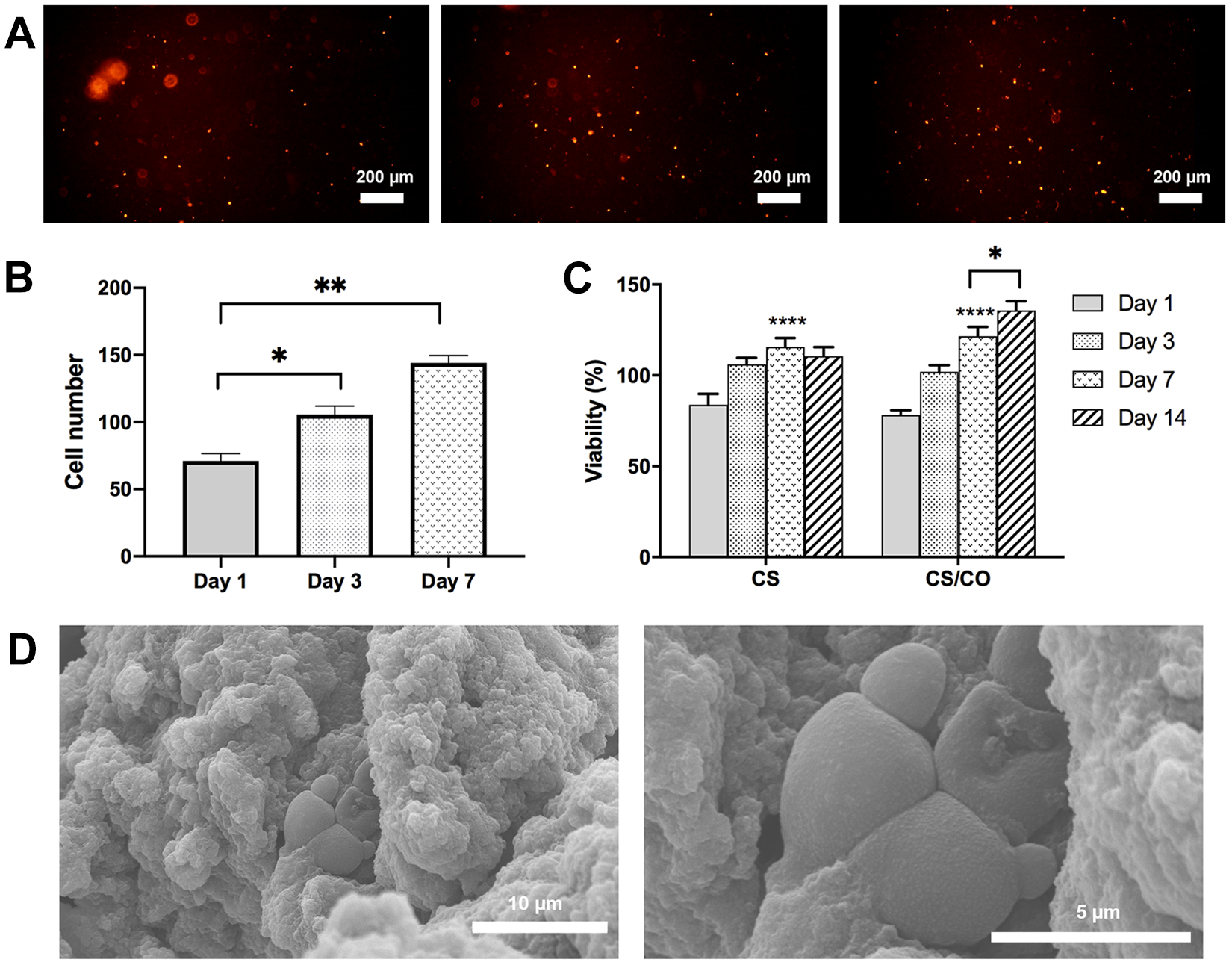
Viability and Proliferation Analyses of Cells in CS/CO Hydrogel
For viability and proliferation analysis of composite hydrogel, MSCs were encapsulated in CS/CO hydrogel. Dil staining of the encapsulated cells in the CS/CO hydrogel showed that the cells were proliferated over time (Fig. 4A). In addition, quantified results regarding assessment of proliferation showed that the number of DiI-positive encapsulated cells were significantly increased on days 3 and 7 compared to day 1 (*
Differentiation of Encapsulated Transduced Cells in CS/CO Hydrogel
3 days after transduction, transduced cells (MSCmiR-1/Myocd) were trypsinized and cultured in CS/CO hydrogel. Subsequently, qRT-PCR and ICC analysis were performed to evaluate the differentiation of encapsulated cells. As in the 2D culture, mRNA gene expression of
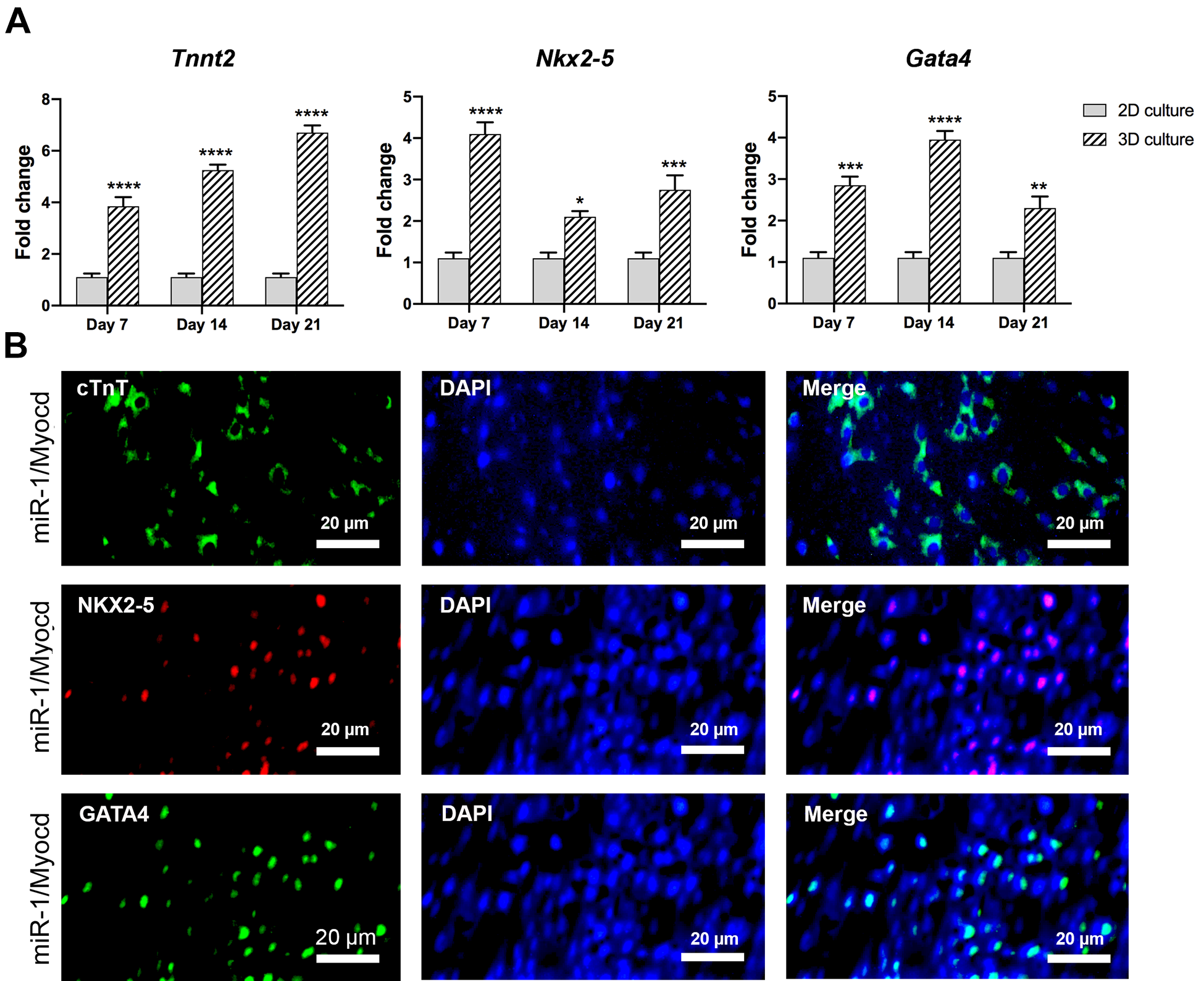
Effect of iCM/P on Cardiac Function
After MI induction (Fig. 6A), transplantation of patch was performed in different groups (Fig. 6B). Echocardiograph analysis was performed 30 days after patch transplantation in different groups (Fig. 6C). The increased EF and FS in both iCM/P and MSC/P groups indicated functional recovery after cell/patch transplantation in comparison with MI control group. Additionally, the increase in EF and FS was significantly higher in the iCM/P group compared to MSC/P group by *
Effect of iCM/P on Cardiac gene Expression in MI Zone
The expression levels of
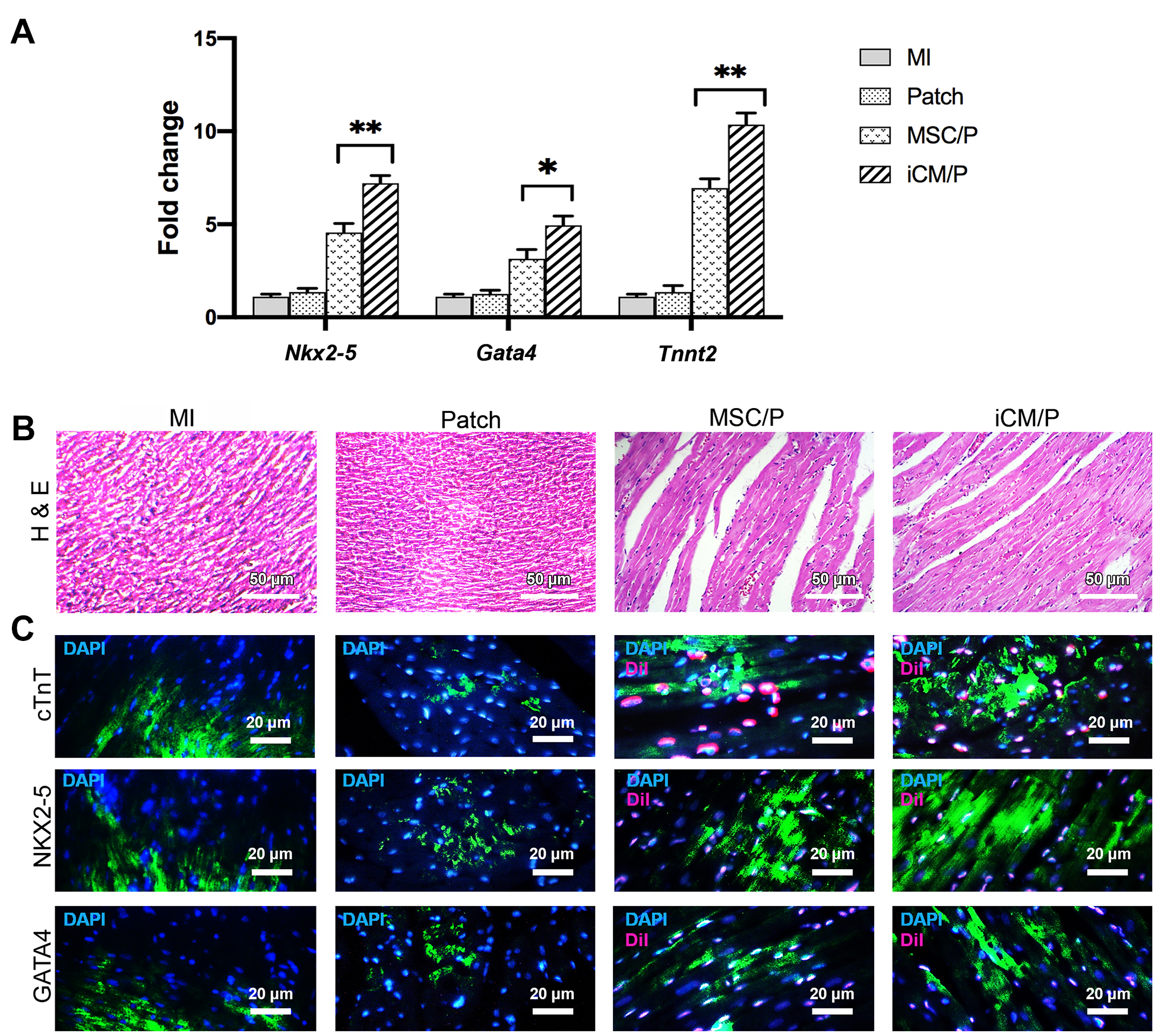
Histological and IHC Staining in MI Zones
For histological examination, H&E staining was performed for MI zone of patch transplantation-treated and MI groups. As shown in Fig. 7B, myocytes in iCM/P and MSC/P groups were arranged regularly and vascular structures and red blood cells were detectable. In contrast, no regularly and vascular structures were detected in MI zone of MI group. On day 30 post-transplantation, the patch-treated groups showed the grafted cells with DiI labeling in MI zone. In heart sections with DiI labeling in iCM/P and MSC/P groups, co-localization of DiI labeling and cTnT, NKX2.5, and GATA4 markers was observed. Expression of markers in Patch and MI groups was observed only in border of MI zones (Fig. 7C).
Discussion
The miRNAs are considered as regulators in bioprocesses, such as differentiation, proliferation, and development and they have important roles in cardiogenesis
38
. Among cardiac-specific miRNAs,
Most studies on cells function have been performed in 2D microenvironment, but recently 3D culture has been developed because of its advantages over 2D culture. In 3D cell culture, different cells interact with each other and with extracellular matrix. Whereas, in 2D cell culture, cells interact only with their surrounding cells 43,44 . In addition, mechanical forces between cell-cell and cell-environment in 3D culture provide a set of signals, regulating different functions of cells, such as proliferation, differentiation, and migration 45 . Moreover, structure and organization of cells in 3D culture change compared to a 2D culture, influencing function and cell signaling 46 . Therefore, it can be concluded that cell behaviors in 3D culture are different from 2D culture.
3D culture promotes an environment for better signaling between cells and their neighboring, and causes more realistic physiological, biomechanical, and biochemical properties, and less substrate stiffness than 2D environment, collectively increasing differentiation capability of MSCs into different cell lines. A combination of these conditions alters gene expression of MSCs and thus, activates different signaling pathways ultimately improving efficacy of MSCs differentiation
47
. Pineda et al., investigated myocardial differentiation of embedded mouse embryonic stem cells (M-ESCs) in collagen type I gels (GELs)
48
. They indicated that expression of
CS as a biocompatible polysaccharide is frequently used in tissue engineering due to its antioxidant activity, lack of immunogenicity, and bioactive properties 26 . Studies have shown that CS as a scaffold in combination with MSCs can improve heart function in MI models of rats 27,28 . There are several mechanisms for improving function of ischemic heart and reduction of infarct size for example, (1) CS, as a carrier of MSCs increases their survival and engraftment, which is related to antioxidant activity of CS and eliminating of reactive oxygen species (ROS), diminishing cell adhesion, and eventually improving MI environment 50 . (2) CS is involved in formation of neovasculature. In addition, MSCs in combination with CS play a synergistic role in ischemic myocardial angiogenesis because they have paracrine effect and release angiogenic factors like vascular endothelial growth factor (VEGF), basic fibroblast growth factor (b-FGF), and hepatocyte growth factor (HGF) 51 . (3) CS increases differentiation of MSCs into cardiac-like cells due to collagen synthesis 28 . CO is one of the main components of ECM playing an important role in process of cardiac maturation and differentiation of MSCs 30 . Therefore, heart function is improved by replacing cardiac differentiated cells in MI zone.
CO facilitates cell adhesion by integrin receptors, activating signaling pathways, which in turn regulate cell survival, proliferation, and differentiation
30
. Therefore, in combination with CS, efficiency of differentiation of MSCs into cardiac-like cells is improved. Moreover, due to similarity of CO to the pericardium and native ECM, and enhancement of tensile strength, composite hydrogel of CS and CO improves heart function in a MI model of rat
33
. The prepared integrated porous CS/CO hydrogel network provided a microenvironment for cell proliferation and differentiation by permeability of nutrients and biological molecules and development of signaling pathway among embedded cells in hydrogel
33
. Moreover, the presence of CO in the hydrogel increases compaction. As a result, CS/CO hydrogel is more compact and stiffer than pure CS
33
. Additionally, results of viability test and Dil staining showed that the composite CS/CO hydrogel promoted growth and proliferation of the encapsulated cells and had no cytotoxicity effect. In the current study, first mouse MSCs were co-transduced with
Previous studies have shown that transplantation of MSCs in MI zone does not effectively improve heart function due to high apoptosis rate of the transplanted cells in the infarct environment and low efficiency of their differentiating into cardiomyocytes and endothelial cells 48,52 . Therefore, recently, using different hydrogels has been considered for injection or transplantation of MSCs in the MI zone 53,54 . In the present report, for in vivo studies, after co-transduction of MSCs with miR-1 and Myocd lentiviruses, iCMs were encapsulated into CS/CO hydrogel followed by placed on PDMS for making patch. PDMS is a biocompatible and biodegradable substrate that has been shown in several studies to use as a substrate to promote the self-renewal and differentiation of stem cells into cardiomyocyte 58 . PDMS have also been used in several studies as a suitable substrate for cultivation 59 and implantation 60 .
After MI induction, patches were implanted to the MI zone in different groups including iCM/P, MSC/P, and cell-free/P groups. Echocardiography of rat heart tissue was performed for assessing heart function. The results confirmed that EF and FS were increased in iCM/P and MSC/P groups, indicating an improvement in the impaired heart function after transplantation of the encapsulated cells 55 . Moreover, cardiac markers, at the level of gene and protein, in iCM/P group significantly increased in comparison with control group.
As mentioned in previous studies 56,57 , MSCs transplantation in MI zone can partially improve heart function due to induction of angiogenesis, paracrine effects, and stimulating differentiation of the implanted MSCs into cardiomyocyte. In the iCM/P group, not all MSCs are differentiated into iCMs, and this group includes a heterogeneous population of differentiated iCMs and undifferentiated MSCs. Therefore, in the iCM/P group, both iCMs and MSCs are effective in improving heart function after MI induction. As a result, in the MSC/P group, a relative improvement in heart function is observed.
Conclusion
Our results revealed that co-induction of miR-1 and Myocd followed by culture in CS/CO hydrogel led to greater differentiation and maturation of MSCs. Additionally, heart function was improved after transplantation of encapsulated iCMs in MI model of rat. Therefore, our findings indicated importance of 3D culture in stem cell differentiation. So, in the future studies, progress in cardiac regeneration based on cell therapy can be achieved by improving differentiation conditions of MSCs and subsequently, obtaining mature cardiac cells.
Supplemental Material
Supplemental Material, sj-docx-1-cll-10.1177_09636897211048786 - Improvement of Heart Function After Transplantation of Encapsulated Stem Cells Induced with miR-1/Myocd in Myocardial Infarction Model of Rat
Supplemental Material, sj-docx-1-cll-10.1177_09636897211048786 for Improvement of Heart Function After Transplantation of Encapsulated Stem Cells Induced with miR-1/Myocd in Myocardial Infarction Model of Rat by Samaneh Khazaei, Masoud Soleimani, Seyed Hossein Ahmadi Tafti, Rouhollah Mehdinavaz Aghdam and Zohreh Hojati in Cell Transplantation
Footnotes
Acknowledgment
The authors would like to thank the Tehran Heart Center for helping us in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval to report this case was obtained from the Ethics Committee of the Tarbiat Modares University, Iran (IR.MODARES.REC.1398.125).
Statement of human and animal rights
All procedures in this study were conducted in accordance with the Ethics Committee of the Tarbiat Modares University, Iran (IR.MODARES.REC.1398.125) approved protocols.
Statement of informed consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Tarbiat Modares University and in part by the Isfahan University.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
